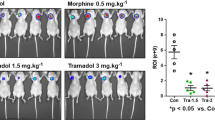
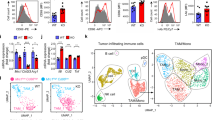
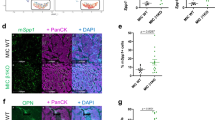
Abstract
The clinical approval of p110δ PI3K inhibitors raised hopes in treating aggressive tumours expressing high levels of non-mutated p110δ, however, the severe adverse effects that those inhibitors caused became a barrier to their clinical application. IOA-244 is the first-in-class, highly selective and non-ATP competitive p110δ PI3K inhibitor showing high selectivity and low toxicity in several preclinical models. Here we show that IOA-244, as a single agent treatment, blocks the progression of early phase breast tumours by attacking the survival of cancer cells and the polarisation of TAMs to a pro-tumourigenic phenotype leading to suppression of TAMs-expressed ATX. In established tumours, IOA-244 alone was insufficient to control the high levels of both M2-like macrophages and ATX, and while it reduced tumour progression, it did not completely block it. Full tumour control, however, was achieved when IOA-244 used in a combinatorial regimen with the PF-8380 ATX inhibitor. In agreement with the mouse model, the amount of CD163+/CD204+macrophages and ATX were much higher in grade III human breast carcinomas compared to grade I. Our work provides the first in vivo preclinical evidence showing that IOA-244 is a potential highly effective drug for breast cancer treatment and depending on the phase of the tumour can be used either as a single agent or as a combinatorial regimen.
Similar content being viewed by others
Data availability
The authors declare that the data supporting the findings of this study are available within the paper and/or are available upon request from the corresponding author. Full and uncropped western blots are shown in Supplementary Fig. 4.
References
Engelman JA. Targeting PI3K signalling in cancer: opportunities, challenges and limitations. Nat Rev Cancer. 2009;9:550–62.
Tzenaki N, Papakonstanti EA. p110delta PI3 kinase pathway: emerging roles in cancer. Front Oncol. 2013;3:40.
Xenou L, Papakonstanti EA. p110delta PI3K as a therapeutic target of solid tumours. Clin Sci. 2020;134:1377–97.
Vanhaesebroeck B, Whitehead MA, Pineiro R. Molecules in medicine mini-review: isoforms of PI3K in biology and disease. J Mol Med. 2016;94:5–11.
Scott J, Rees L, Gallimore A, Lauder SN. PI3K Isoform Immunotherapy for Solid Tumours. Curr Top Microbiol Immunol. 2022;436:369–92.
Yu M, et al. Development and safety of PI3K inhibitors in cancer. Arch Toxicol. 2023;97:635–50.
Wood LD, et al. The genomic landscapes of human breast and colorectal cancers. Science. 2007;318:1108–13.
Lucas CL, et al. Dominant-activating germline mutations in the gene encoding the PI(3)K catalytic subunit p110delta result in T cell senescence and human immunodeficiency. Nat Immunol. 2014;15:88–97.
Coulter TI, et al. Clinical spectrum and features of activated phosphoinositide 3-kinase delta syndrome: a large patient cohort study. J Allergy Clin Immunol. 2017;139:597–606.e594.
Rommel C. Taking PI3Kδ and PI3Kγ one step ahead: dual active PI3Kδ/γ inhibitors for the treatment of immune-mediated inflammatory diseases. Curr Top Microbiol Immunol. 2010;346:279–99.
Soond DR, et al. PI3K p110delta regulates T-cell cytokine production during primary and secondary immune responses in mice and humans. Blood. 2010;115:2203–13.
Castillo JJ, Furman M, Winer ES. CAL-101: a phosphatidylinositol-3-kinase p110-delta inhibitor for the treatment of lymphoid malignancies. Expert Opin Investig Drugs. 2012;21:15–22.
Furman RR, et al. Idelalisib and rituximab in relapsed chronic lymphocytic leukemia. N Engl J Med. 2014;370:997–1007.
Gopal AK, et al. PI3Kδ inhibition by idelalisib in patients with relapsed indolent lymphoma. N Engl J Med. 2014;370:1008–18.
Clayton E, et al. A crucial role for the p110delta subunit of phosphatidylinositol 3-kinase in B cell development and activation. J Exp Med. 2002;196:753–63.
Ikeda H, et al. PI3K/p110delta is a novel therapeutic target in multiple myeloma. Blood. 2010;116:1460–8.
Goulielmaki E, et al. Pharmacological inactivation of the PI3K p110delta prevents breast tumour progression by targeting cancer cells and macrophages. Cell Death Dis. 2018;9:678.
Tzenaki N, et al. High levels of p110delta PI3K expression in solid tumor cells suppress PTEN activity, generating cellular sensitivity to p110delta inhibitors through PTEN activation. FASEB J. 2012;26:2498–508.
Ko E, et al. PI3Kdelta is a therapeutic target in hepatocellular carcinoma. Hepatology. 2018;68:2285–300.
Tarantelli C, Argnani L, Zinzani PL, Bertoni F. PI3Kdelta inhibitors as immunomodulatory agents for the treatment of lymphoma patients. Cancers. 2021;13. https://doi.org/10.3390/cancers132155355535cancers13215535.
Borazanci E, et al. A phase Ib study of single-agent idelalisib followed by idelalisib in combination with chemotherapy in patients with metastatic pancreatic ductal adenocarcinoma. Oncologist. 2020;25:e1604–e1613.
Eschweiler S, et al. Intermittent PI3Kdelta inhibition sustains anti-tumour immunity and curbs irAEs. Nature. 2022;605:741–6.
Ahmad S, et al. Differential PI3Kdelta signaling in CD4(+) T-cell subsets enables selective targeting of T regulatory cells to enhance cancer immunotherapy. Cancer Res. 2017;77:1892–904.
Akhiani AA, et al. Idelalisib rescues natural killer cells from monocyte-induced immunosuppression by inhibiting NOX2-derived reactive oxygen species. Cancer Immunol Res. 2020;8:1532–41.
Markham A. Idelalisib: first global approval. Drugs. 2014;74:1701–7.
Yang Q, Modi P, Newcomb T, Queva C, Gandhi V. Idelalisib: First-in-class PI3K delta inhibitor for the treatment of chronic lymphocytic leukemia, small lymphocytic leukemia, and follicular lymphoma. Clin Cancer Res. 2015;21:1537–42. [pii].
Flinn IW, et al. The phase 3 DUO trial: duvelisib vs ofatumumab in relapsed and refractory CLL/SLL. Blood. 2018;132:2446–55. [pii]2018/850461 [pii].
Blair HA. Duvelisib: first global approval. Drugs. 2018;78:1847–53. [pii].
Deng C, et al. Silencing c-Myc translation as a therapeutic strategy through targeting PI3Kdelta and CK1epsilon in hematological malignancies. Blood. 2017;129:88–99. [pii]2016/731240 [pii].
Dhillon S, Keam SJ. Umbralisib: first approval. Drugs. 2021;81:857–66.
Sharman JP, et al. Final results of a randomized, phase III study of rituximab with or without idelalisib followed by open-label idelalisib in patients with relapsed chronic lymphocytic leukemia. J Clin Oncol. 2019;37:1391–402.
Meng D, et al. Development of PI3K inhibitors: advances in clinical trials and new strategies (Review). Pharm Res. 2021;173:105900.
Haselmayer P, et al. Characterization of novel PI3Kdelta inhibitors as potential therapeutics for SLE and Lupus nephritis in pre-clinical studies. Front Immunol. 2014;5:233.
Johnson Z, et al. 93P - Preclinical development of a novel, highly selective PI3Kδ inhibitor, IOA-244, for the treatment of solid malignancies. Ann Oncol. 2019;30:vii27.
Johnson Z, et al. IOA-244 is a Non-ATP-competitive, highly selective, tolerable PI3K delta inhibitor that targets solid tumors and breaks immune tolerance. Cancer Res Commun. 2023;3:576–91.
Tzenaki N, et al. A combined opposite targeting of p110delta PI3K and RhoA abrogates skin cancer. Commun Biol. 2024;7:26.
Benesch MGK, Tang X, Brindley DN. Autotaxin and breast cancer: towards overcoming treatment barriers and sequelae. Cancers. 2020;12. https://doi.org/10.3390/cancers12020374374cancers12020374.
Tang X, Benesch MGK, Brindley DN. Role of the autotaxin-lysophosphatidate axis in the development of resistance to cancer therapy. Biochim Biophys Acta Mol Cell Biol Lipids. 2020;1865:158716.
Tang X, et al. Inhibition of autotaxin with GLPG1690 increases the efficacy of radiotherapy and chemotherapy in a mouse model of breast cancer. Mol Cancer Ther. 2020;19:63–74.
Brindley DN, Tang X, Meng G, Benesch MGK. Role of adipose tissue-derived autotaxin, lysophosphatidate signaling, and inflammation in the progression and treatment of breast cancer. Int J Mol Sci. 2020;21. https://doi.org/10.3390/ijms211659385938ijms21165938.
Perrakis A, Moolenaar WH. Autotaxin: structure-function and signaling. J Lipid Res. 2014;55:1010–8. [pii]r046391 [pii].
Houben AJ, Moolenaar WH. Autotaxin and LPA receptor signaling in cancer. Cancer Metastasis Rev. 2011;30:557–65.
Oda SK, et al. Lysophosphatidic acid inhibits CD8 T cell activation and control of tumor progression. Cancer Immunol Res. 2013;1:245–55.
Umezu-Goto M, et al. Autotaxin has lysophospholipase D activity leading to tumor cell growth and motility by lysophosphatidic acid production. J Cell Biol. 2002;158:227–33.
Gaetano CG, et al. Inhibition of autotaxin production or activity blocks lysophosphatidylcholine-induced migration of human breast cancer and melanoma cells. Mol Carcinog. 2009;48:801–9.
Benesch MG, et al. Inhibition of autotaxin delays breast tumor growth and lung metastasis in mice. FASEB J. 2014;28:2655–66. [pii].
Larionova I, et al. Tumor-associated macrophages in human breast, colorectal, lung, ovarian and prostate cancers. Front Oncol. 2020;10:566511.
Aiello S, Casiraghi F. Lysophosphatidic acid: promoter of cancer progression and of tumor microenvironment development. a promising target for anticancer therapies? Cells.2021;10. https://doi.org/10.3390/cells100613901390cells10061390.
Benesch MG, Ko YM, McMullen TP, Brindley DN. Autotaxin in the crosshairs: taking aim at cancer and other inflammatory conditions. FEBS Lett. 2014;588:2712–27.
Samadi N, et al. Regulation of lysophosphatidate signaling by autotaxin and lipid phosphate phosphatases with respect to tumor progression, angiogenesis, metastasis and chemo-resistance. Biochimie. 2011;93:61–70.
Chan FK, Moriwaki K, De Rosa MJ. Detection of necrosis by release of lactate dehydrogenase activity. Methods Mol Biol. 2013;979:65–70.
Sopik V, Narod SA. The relationship between tumour size, nodal status and distant metastases: on the origins of breast cancer. Breast Cancer Res Treat. 2018;170:647–56.
Malekian S, et al. Expression of diverse angiogenesis factor in different stages of the 4T1 tumor as a mouse model of triple-negative breast cancer. Adv Pharm Bull. 2020;10:323–8.
Koscielny S, et al. Breast cancer: relationship between the size of the primary tumour and the probability of metastatic dissemination. Br J Cancer. 1984;49:709–15.
Carter CL, Allen C, Henson DE. Relation of tumor size, lymph node status, and survival in 24,740 breast cancer cases. Cancer. 1989;63:181–7.
Olivotto IA, et al. Prediction of axillary lymph node involvement of women with invasive breast carcinoma: a multivariate analysis. Cancer. 1998;83:948–55.
Emami Nejad A, et al. The role of hypoxia in the tumor microenvironment and development of cancer stem cell: a novel approach to developing treatment. Cancer Cell Int. 2021;21:62.
You L, et al. The role of hypoxia-inducible factor 1 in tumor immune evasion. Med Res Rev. 2021;41:1622–43.
Dey S, et al. Critical pathways of oral squamous cell carcinoma: molecular biomarker and therapeutic intervention. Med Oncol. 2022;39:30.
Li Q, Li Z, Luo T, Shi H. Targeting the PI3K/AKT/mTOR and RAF/MEK/ERK pathways for cancer therapy. Mol Biomed. 2022;3:47.
Xue C, et al. Epidermal growth factor receptor overexpression results in increased tumor cell motility in vivo coordinately with enhanced intravasation and metastasis. Cancer Res. 2006;66:192–7.
Gilles C, et al. Transactivation of vimentin by β-catenin in human breast cancer cells. Cancer Res. 2003;63:2658–64.
Korsching E, et al. The origin of vimentin expression in invasive breast cancer: epithelial–mesenchymal transition, myoepithelial histogenesis or histogenesis from progenitor cells with bilinear differentiation potential?. J Pathol. 2005;206:451–7.
Condeelis J, Pollard JW. Macrophages: obligate partners for tumor cell migration, invasion, and metastasis. Cell. 2006;124:263–6.
Bingle L, Brown NJ, Lewis CE. The role of tumour-associated macrophages in tumour progression: implications for new anticancer therapies. J Pathol. 2002;196:254–65.
Ostuni R, Kratochvill F, Murray PJ, Natoli G. Macrophages and cancer: from mechanisms to therapeutic implications. Trends Immunol. 2015;36:229–39.
Mantovani A, Allavena P. The interaction of anticancer therapies with tumor-associated macrophages. J Exp Med. 2015;212:435–45.
Reinartz S, et al. Cell type-selective pathways and clinical associations of lysophosphatidic acid biosynthesis and signaling in the ovarian cancer microenvironment. Mol Oncol. 2019;13:185–201.
Sadhu C, Masinovsky B, Dick K, Sowell CG, Staunton DE. Essential role of phosphoinositide 3-kinase d in neutrophil directional movement. J Immunol. 2003;170:2647–54.
Kim T, et al. TRIB1 regulates tumor growth via controlling tumor-associated macrophage phenotypes and is associated with breast cancer survival and treatment response. Theranostics. 2022;12:3584–600.
Somasundaram V, et al. Inducible nitric oxide synthase-derived extracellular nitric oxide flux regulates proinflammatory responses at the single cell level. Redox Biol. 2020;28:101354.
Ghalavand M, Moradi-Chaleshtori M, Dorostkar R, Mohammadi-Yeganeh S, Hashemi SM. Exosomes derived from rapamycin-treated 4T1 breast cancer cells induced polarization of macrophages to M1 phenotype. Biotechnol Appl Biochem. 2023;70:1754–71.
Deken MA, et al. Characterization and translational development of IOA-289, a novel autotaxin inhibitor for the treatment of solid tumors. Immunooncol Technol. 2023;18:100384.
Gierse J, et al. A novel autotaxin inhibitor reduces lysophosphatidic acid levels in plasma and the site of inflammation. J Pharm Exp Ther. 2010;334:310–7.
Katsifa A, et al. The bulk of autotaxin activity is dispensable for adult mouse life. PLoS ONE. 2015;10:e0143083. https://doi.org/10.1371/journal.pone.0143083e0143083PONE-D-15-30701.
Cao P, et al. Autocrine lysophosphatidic acid signaling activates beta-catenin and promotes lung allograft fibrosis. J Clin Invest. 2017;127:1517–30.
Tang X, Wang X, Zhao YY, Curtis JM, Brindley DN. Doxycycline attenuates breast cancer-related inflammation by decreasing plasma lysophosphatidate concentrations and inhibiting NF-kappaB activation. Mol Cancer. 2017;16:36.
Chaintreuil P, et al. The generation, activation, and polarization of monocyte-derived macrophages in human malignancies. Front Immunol. 2023;14:1178337.
Amer HT, Stein U, El Tayebi HM. The monocyte, a maestro in the tumor microenvironment (TME) of breast cancer. Cancers. 2022;14. https://doi.org/10.3390/cancers142154605460cancers14215460.
Biswas SK, Mantovani A. Macrophage plasticity and interaction with lymphocyte subsets: cancer as a paradigm. Nat Immunol. 2010;11:889–96.
Davies LC, Jenkins SJ, Allen JE, Taylor PR. Tissue-resident macrophages. Nat Immunol. 2013;14:986–95.
Ojalvo LS, King W, Cox D, Pollard JW. High-density gene expression analysis of tumor-associated macrophages from mouse mammary tumors. Am J Pathol. 2009;174:1048–64.
Lin EY, et al. Macrophages regulate the angiogenic switch in a mouse model of breast cancer. Cancer Res. 2006;66:11238–46.
Qian BZ, Pollard JW. Macrophage diversity enhances tumor progression and metastasis. Cell. 2010;141:39–51.
Jayasingam SD, et al. Evaluating the polarization of tumor-associated macrophages into M1 and M2 phenotypes in human cancer tissue: technicalities and challenges in routine clinical practice. Front Oncol. 2019;9:1512.
Benner B, et al. Generation of monocyte-derived tumor-associated macrophages using tumor-conditioned media provides a novel method to study tumor-associated macrophages in vitro. J Immunother Cancer. 2019;7:140.
Li M, Jiang P, Wei S, Wang J, Li C. The role of macrophages-mediated communications among cell compositions of tumor microenvironment in cancer progression. Front Immunol. 2023;14:1113312.
Obeid E, Nanda R, Fu YX, Olopade OI. The role of tumor-associated macrophages in breast cancer progression (review). Int J Oncol. 2013;43:5–12.
Komohara Y, et al. Involvement of protumor macrophages in breast cancer progression and characterization of macrophage phenotypes. Cancer Sci. 2023;114:2220–9.
Zhou L, et al. LincRNA-p21 knockdown reversed tumor-associated macrophages function by promoting MDM2 to antagonize* p53 activation and alleviate breast cancer development. Cancer Immunol Immunother. 2020;69:835–46.
Toor SM, et al. Myeloid cells in circulation and tumor microenvironment of breast cancer patients. Cancer Immunol Immunother. 2017;66:753–64.
Lee SC et al. Regulation of tumor immunity by lysophosphatidic acid. Cancers. 2020;12. https://doi.org/10.3390/cancers120512021202cancers12051202.
Cha YJ, Koo JS. Expression of autotaxin(-)lysophosphatidate signaling-related proteins in breast cancer with adipose stroma. Int J Mol Sci. 2019;20. https://doi.org/10.3390/ijms200921022102ijms20092102.
Tang X, Morris AJ, Deken MA, Brindley DN. Autotaxin Inhibition with IOA-289 decreases breast tumor growth in mice, whereas knockout of autotaxin in adipocytes does not. Cancers. 2023;5. https://doi.org/10.3390/cancers151129372937cancers15112937.
Papakonstanti EA, Ridley AJ, Vanhaesebroeck B. The p110delta isoform of PI 3-kinase negatively controls RhoA and PTEN. EMBO J. 2007;26:3050–61.
Weisser SB, et al. Arginase activity in alternatively activated macrophages protects PI3Kp110δ-deficient mice from dextran sodium sulfate-induced intestinal inflammation. Eur J Immunol. 2014;44:3353–67.
Porcheray F, et al. Macrophage activation switching: an asset for the resolution of inflammation. Clin Exp Immunol. 2005;142:481–9.
Underhill DM, Bassetti M, Rudensky A, Aderem A. Dynamic interactions of macrophages with T cells during antigen presentation. J Exp Med. 1999;190:1909–14.
Liu G, et al. Phenotypic and functional switch of macrophages induced by regulatory CD4+CD25+ T cells in mice. Immunol Cell Biol. 2011;89:130–42.
Hu X, et al. Induction of M2-like macrophages in recipient NOD-scid mice by allogeneic donor CD4(+)CD25(+) regulatory T cells. Cell Mol Immunol. 2012;9:464–72.
Liu C, et al. Treg cells promote the SREBP1-dependent metabolic fitness of tumor-promoting macrophages via repression of CD8(+) T cell-derived interferon-gamma. Immunity. 2019;51:381–97 e386.
Ali K, et al. Inactivation of PI(3)K p110delta breaks regulatory T-cell-mediated immune tolerance to cancer. Nature. 2014;510:407–11.
Cevey AC, et al. Benznidazole anti-inflammatory effects in murine cardiomyocytes and macrophages are mediated by class I PI3Kdelta. Front Immunol. 2021;12:782891.
Weisser SB, et al. Arginase activity in alternatively activated macrophages protects PI3Kp110delta deficient mice from dextran sodium sulfate-induced intestinal inflammation. Eur J Immunol. 2014;44:3353–67.
Shen M, Wang J, Ren X. New insights into tumor-infiltrating B lymphocytes in breast cancer: clinical impacts and regulatory mechanisms. Front Immunol. 2018;9:470.
Wong SC, et al. Macrophage polarization to a unique phenotype driven by B cells. Eur J Immunol. 2010;40:2296–307.
Yanaba K, et al. A regulatory B cell subset with a unique CD1dhiCD5+ phenotype controls T cell-dependent inflammatory responses. Immunity. 2008;28:639–50.
Andreu P, et al. FcRgamma activation regulates inflammation-associated squamous carcinogenesis. Cancer Cell. 2010;17:121–34.
Sallusto F, Baggiolini M. Chemokines and leukocyte traffic. Nat Immunol. 2008;9:949–52.
Liu RX, et al. Chemokine (C-X-C motif) receptor 3-positive B cells link interleukin-17 inflammation to protumorigenic macrophage polarization in human hepatocellular carcinoma. Hepatology. 2015;62:1779–90.
Gao X, et al. Upregulation of HMGB1 in tumor-associated macrophages induced by tumor cell-derived lactate further promotes colorectal cancer progression. J Transl Med. 2023;21:53.
Sun L, Han X, Egeblad M. Isolation of mouse mammary carcinoma-derived macrophages and cancer cells for co-culture assays. STAR Protoc. 2022;3:101833.
Jiang S, Zhou Z, Sun Y, Zhang T, Sun L. Coral gasdermin triggers pyroptosis. Sci Immunol. 2020;5:eabd2591.
Tomayko MM, Reynolds CP. Determination of subcutaneous tumor size in athymic (nude) mice. Cancer Chemother Pharm. 1989;24:148–54.
Euhus DM, Hudd C, LaRegina MC, Johnson FE. Tumor measurement in the nude mouse. J Surg Oncol. 1986;31:229–34.
Gao M, et al. Direct therapeutic targeting of immune checkpoint PD-1 in pancreatic cancer. Br J Cancer. 2019;120:88–96.
Robertson D, Savage K, Reis-Filho J, Isacke C. Multiple immunofluorescence labelling of formalin-fixed paraffin-embedded (FFPE) tissue. BMC Cell Biol. 2008;9:13.
Nguyen D. Quantifying chromogen intensity in immunohistochemistry via reciprocal intensity. Protocol Exchange-Nat. 2013. https://doi.org/10.1038/protex.2013.097
Acknowledgements
We are grateful to Giusy Di Conza (iOnctura) and Laurence Neff (iOnctura) for the critical reading of the manuscript. We are also truly grateful to Stavros Topouzis (Pharmacology Department, University of Patras) for manuscript editing. This research was supported by iOnctura (Project Number: 10179) and by the Hellenic Foundation for Research and Innovation (H.F.R.I.) under the “First Call for H.F.R.I. Research Projects to support Faculty members and Researchers and the procurement of high-cost research equipment grant” (Project Number: 3405).
Author information
Authors and Affiliations
Contributions
EG and AT performed in vitro experiments and experiments with mice and data analyses and drafted the manuscript. LX performed in vitro experiments on cells and mice blood. MT performed histology and interpreted histopathology and immunohistochemistry. Clinical samples and clinical information were obtained from EDB. ZJ, KNS, and EAP conceived the project. EAP designed and analysed the experiments, supervised the research and wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The following authors are or have been employed by and/or share holders of iOnctura: KNS and ZJ. EAP have received research funding from iOnctura. EG and AT have received fees for providing services to this work. The other authors declare no conflict of interest relating to this work. EAP, EG and AT have no additional financial interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Goulielmaki, E., Tsapara, A., Xenou, L. et al. IOA-244, a novel p110δ PI3K inhibitor, blocks breast tumour progression on either mono- or combined-therapy. Cell Death Discov. (2026). https://doi.org/10.1038/s41420-026-03073-3
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41420-026-03073-3