Abstract
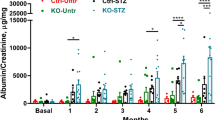
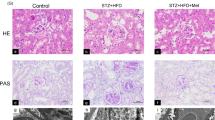
AMP-activated protein kinase (AMPK) has been postulated to be crucial in regulating various renal physiology and pathophysiology processes, including energy metabolism, ion and water transport, inflammation, and hypertrophy. However, the specific roles of AMPK in podocytes, cells critical for maintaining glomerular filtration, have not been fully explored using genetic model animals. In this study, we generated mice lacking both AMPK α1 and α2 catalytic subunits in glomerular podocytes (pmut). Our findings revealed that, surprisingly, AMPK is dispensable for normal podocyte function. These knockout mice could live as long as their wild-type littermates without showing any pathological alterations in their glomeruli or glomerular function at two years of age. However, under diabetic conditions, the diabetic pmut mice exhibited increased lipid and collagen accumulation and an elevated expression of mesenchymal proteins in their glomeruli. They also showed more significant albuminuria compared to control diabetic mice. Under high-glucose culture conditions, glomeruli isolated from pmut mice showed reduced expression of mitochondrial genes (e.g., Ndufv2) and increased leakage of mitochondrial components. Additionally, there was increased expression of genes associated with nucleotide-sensing and pro-inflammatory pathways (including mb21d2, IL-1β, and NF-κB). These observations suggest that while AMPK is not necessary for podocyte function in healthy kidneys, it is crucial for preventing glomerular fibrosis resulting from lipotoxicity and inflammation under diabetic conditions.
Similar content being viewed by others
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author (Swayam Prakash Srivastava; spsr@umich.edu) upon request.
References
Jeon SM. Regulation and function of AMPK in physiology and diseases. Exp Mol Med. 2016;48:e245.
Trefts E, Shaw RJ. AMPK: restoring metabolic homeostasis over space and time. Mol Cell. 2021;81:3677–90.
Steinberg GR, Hardie DG. New insights into the activation and function of the AMPK. Nat Rev Mol Cell Biol. 2023;24:255–72.
Oakhill JS, Steel R, Chen ZP, Scott JW, Ling N, Tam S, et al. AMPK is a direct adenylate charge-regulated protein kinase. Science. 2011;332:1433–35.
Xiao B, Sanders MJ, Underwood E, Heath R, Mayer FV, Carmena D, et al. Structure of mammalian AMPK and its regulation by ADP. Nature. 2011;472:230–33.
Zhang CS, Hawley SA, Zong Y, Li M, Wang Z, Gray A, et al. Fructose-1,6-bisphosphate and aldolase mediate glucose sensing by AMPK. Nature. 2017;548:112–6.
Zhang YL, Guo H, Zhang CS, Lin SY, Yin Z, Peng Y, et al. AMP as a low-energy charge signal autonomously initiates assembly of AXIN-AMPK-LKB1 complex for AMPK activation. Cell Metab. 2013;18:546–55.
Ruderman NB, Carling D, Prentki M, Cacicedo JM. AMPK, insulin resistance, and the metabolic syndrome. J Clin Investig. 2013;123:2764–72.
Cooper M, Warren AM. A promising outlook for diabetic kidney disease. Nat Rev Nephrol. 2019;15:68–70.
Gujarati NA, Frimpong BO, Zaidi M, Bronstein R, Revelo MP, Haley JD, et al. Podocyte-specific KLF6 primes proximal tubule CaMK1D signaling to attenuate diabetic kidney disease. Nat Commun. 2024;15:8038.
Zhao Y, Fan S, Zhu H, Zhao Q, Fang Z, Xu D, et al. Podocyte OTUD5 alleviates diabetic kidney disease through deubiquitinating TAK1 and reducing podocyte inflammation and injury. Nat Commun. 2024;15:5441.
Thomas MC, Brownlee M, Susztak K, Sharma K, Jandeleit-Dahm KA, Zoungas S, et al. Diabetic kidney disease. Nat Rev Dis Primers. 2015;1:15018.
Tuttle KR, Agarwal R, Alpers CE, Bakris GL, Brosius FC, Kolkhof P, et al. Molecular mechanisms and therapeutic targets for diabetic kidney disease. Kidney Int. 2022;102:248–60.
Kim MK. Treatment of diabetic kidney disease: current and future targets. Korean J Intern Med. 2017;32:622–30.
Dieter BP, Alicic RZ, Meek RL, Anderberg RJ, Cooney SK, Tuttle KR. Novel therapies for diabetic kidney disease: storied past and forward paths. Diab Spectr. 2015;28:167–74.
Pichler R, Afkarian M, Dieter BP, Tuttle KR. Immunity and inflammation in diabetic kidney disease: translating mechanisms to biomarkers and treatment targets. Am J Physiol Ren Physiol. 2017;312:F716–31.
Bonnet F, Cooper ME, Kopp L, Fouque D, Candido R. A review of the latest real-world evidence studies in diabetic kidney disease: What have we learned about clinical practice and the clinical effectiveness of interventions? Diab Obes Metab. 2024;26:55–65.
Glastras SJ, Pollock CA. Targeted identification of risk and treatment of diabetic kidney disease. Nat Rev Nephrol. 2024;20:75–6.
Kanasaki K, Nangaku M, Ueki K. DKD as the kidney disease relevant to individuals with diabetes. Diabetol Int. 2024;15:673–6.
Kanasaki K, Ueki K, Nangaku M. Diabetic kidney disease: the kidney disease relevant to individuals with diabetes. Clin Exp Nephrol. 2024;28:1213–20.
Srivastava SP, Kanasaki K, Goodwin JE. Editorial: combating diabetes and diabetic kidney disease. Front Pharmacol. 2021;12:716029.
Srivastava SP, Kanasaki K. Editorial: receptor biology and cell signaling in diabetes. Front Pharmacol. 2022;13:864117.
Szrejder M, Rachubik P, Rogacka D, Audzeyenka I, Rychlowski M, Kreft E, et al. Metformin reduces TRPC6 expression through AMPK activation and modulates cytoskeleton dynamics in podocytes under diabetic conditions. Biochim Biophys Acta Mol Basis Dis. 2020;1866:165610.
Lu J, Chen PP, Zhang JX, Li XQ, Wang GH, Yuan BY, et al. GPR43 deficiency protects against podocyte insulin resistance in diabetic nephropathy through the restoration of AMPKalpha activity. Theranostics. 2021;11:4728–42.
Li X, Li Q, Jiang X, Song S, Zou W, Yang Q, et al. Inhibition of SGLT2 protects podocytes in diabetic kidney disease by rebalancing mitochondria-associated endoplasmic reticulum membranes. Cell Commun Signal. 2024;22:534.
Lehtonen S. Metformin protects against podocyte injury in diabetic kidney disease. Pharmaceuticals. 2020;13:452
Han YC, Tang SQ, Liu YT, Li AM, Zhan M, Yang M, et al. AMPK agonist alleviate renal tubulointerstitial fibrosis via activating mitophagy in high-fat and streptozotocin-induced diabetic mice. Cell Death Dis. 2021;12:925.
Salatto CT, Miller RA, Cameron KO, Cokorinos E, Reyes A, Ward J, et al. Selective activation of AMPK beta1-containing isoforms improves kidney function in a rat model of diabetic nephropathy. J Pharmacol Exp Ther. 2017;361:303–11.
Zhou X, Muise ES, Haimbach R, Sebhat IK, Zhu Y, Liu F, et al. PAN-AMPK activation improves renal function in a rat model of progressive diabetic nephropathy. J Pharmacol Exp Ther. 2019;371:45–55.
Agur T, Steinmetz T, Goldman S, Zingerman B, Bielopolski D, Nesher E, et al. The impact of metformin on kidney disease progression and mortality in diabetic patients using SGLT2 inhibitors: a real-world cohort study. Cardiovasc Diabetol. 2025;24:97.
Agranovich AL, Anderson GH, Manji M, Acker BD, Macdonald WC, Threlfall WJ. Carcinoid tumour of the gastrointestinal tract: prognostic factors and disease outcome. J Surg Oncol. 1991;47:45–52.
Rogacka D, Audzeyenka I, Piwkowska A. Regulation of podocytes function by AMP-activated protein kinase. Arch Biochem Biophys. 2020;692:108541.
Rogacka D, Piwkowska A, Audzeyenka I, Angielski S, Jankowski M. Involvement of the AMPK-PTEN pathway in insulin resistance induced by high glucose in cultured rat podocytes. Int J Biochem Cell Biol. 2014;51:120–30.
McGee SL, Howlett KF, Starkie RL, Cameron-Smith D, Kemp BE, Hargreaves M. Exercise increases nuclear AMPK alpha2 in human skeletal muscle. Diabetes. 2003;52:926–8.
Torsoni MA, Borges BC, Cote JL, Allen SJ, Mahany E, Garcia-Galiano D, et al. AMPKalpha2 in Kiss1 neurons is required for reproductive adaptations to acute metabolic challenges in adult female mice. Endocrinology. 2016;157:4803–16.
Egan DF, Shackelford DB, Mihaylova MM, Gelino S, Kohnz RA, Mair W, et al. Phosphorylation of ULK1 (hATG1) by AMP-activated protein kinase connects energy sensing to mitophagy. Science. 2011;331:456–61.
Park JM, Lee DH, Kim DH. Redefining the role of AMPK in autophagy and the energy stress response. Nat Commun. 2023;14:2994.
Jia J, Bissa B, Brecht L, Allers L, Choi SW, Gu Y, et al. AMPK, a regulator of metabolism and autophagy, is activated by lysosomal damage via a novel galectin-directed ubiquitin signal transduction system. Mol Cell. 2020;77:951–69.e959.
Hartleben B, Godel M, Meyer-Schwesinger C, Liu S, Ulrich T, Kobler S, et al. Autophagy influences glomerular disease susceptibility and maintains podocyte homeostasis in aging mice. J Clin Investig. 2010;120:1084–96.
Borges CM, Fujihara CK, Malheiros D, de Avila VF, Formigari GP, Lopes de Faria JB. Metformin arrests the progression of established kidney disease in the subtotal nephrectomy model of chronic kidney disease. Am J Physiol Ren Physiol. 2020;318:F1229–36.
Wang F, Sun H, Zuo B, Shi K, Zhang X, Zhang C, et al. Metformin attenuates renal tubulointerstitial fibrosis via upgrading autophagy in the early stage of diabetic nephropathy. Sci Rep. 2021;11:16362.
Takiar V, Nishio S, Seo-Mayer P, King JD Jr, Li H, Zhang L, et al. Activating AMP-activated protein kinase (AMPK) slows renal cystogenesis. Proc Natl Acad Sci USA. 2011;108:2462–7.
Qi B, Chen Y, Lan Y, Lou Y, Cui P, Yuan R, et al. Metformin attenuates O-GlcNAc modification to improve renal function via AMPK/mTOR signaling in diabetic nephropathy. J Biol Chem. 2025;301:110909.
Srivastava SP, Li J, Takagaki Y, Kitada M, Goodwin JE, Kanasaki K, et al. Endothelial SIRT3 regulates myofibroblast metabolic shifts in diabetic kidneys. iScience. 2021;24:102390.
Kang HM, Ahn SH, Choi P, Ko YA, Han SH, Chinga F, et al. Defective fatty acid oxidation in renal tubular epithelial cells has a key role in kidney fibrosis development. Nat Med. 2015;21:37–46.
Zhou HL, Zhang R, Anand P, Stomberski CT, Qian Z, Hausladen A, et al. Metabolic reprogramming by the S-nitroso-CoA reductase system protects against kidney injury. Nature. 2019;565:96–100.
Schoors S, Bruning U, Missiaen R, Queiroz KC, Borgers G, Elia I, et al. Fatty acid carbon is essential for dNTP synthesis in endothelial cells. Nature. 2015;520:192–7.
Tian Z, Liang M. Renal metabolism and hypertension. Nat Commun. 2021;12:963.
Huang H, Parikh SM. Skimming the fat in diabetic kidney disease: KIM-1 and tubular fatty acid uptake. Kidney Int. 2021;100:969–72.
Ralto KM, Rhee EP, Parikh SM. NAD(+) homeostasis in renal health and disease. Nat Rev Nephrol. 2020;16:99–111.
Tran MT, Zsengeller ZK, Berg AH, Khankin EV, Bhasin MK, Kim W, et al. PGC1alpha drives NAD biosynthesis linking oxidative metabolism to renal protection. Nature. 2016;531:528–32.
Srivastava SP, Kanasaki K, Goodwin JE. Loss of mitochondrial control impacts renal health. Front Pharmacol. 2020;11:543973.
Srivastava SP, Shi S, Koya D, Kanasaki K. Lipid mediators in diabetic nephropathy. Fibrogenes Tissue Repair. 2014;7:12.
Srivastava SP, Goodwin JE. Loss of endothelial glucocorticoid receptor accelerates organ fibrosis in db/db mice. Am J Physiol Renal Physiol. 2023;325:F519–F526.
Li J, Liu H, Srivastava SP, Hu Q, Gao R, Li S, et al. Endothelial FGFR1 (Fibroblast Growth Factor Receptor 1) deficiency contributes differential fibrogenic effects in kidney and heart of diabetic mice. Hypertension. 2020;76:1935–44.
Shi S, Srivastava SP, Kanasaki M, He J, Kitada M, Nagai T, et al. Interactions of DPP-4 and integrin beta1 influences endothelial-to-mesenchymal transition. Kidney Int. 2015;88:479–89.
Zhou H, Mehta S, Srivastava SP, Grabinska K, Zhang X, Wong C, et al. Endothelial cell-glucocorticoid receptor interactions and regulation of Wnt signaling. JCI Insight. 2020;5:e131384.
Srivastava SP, Hedayat AF, Kanasaki K, Goodwin JE. microRNA crosstalk influences epithelial-to-mesenchymal, endothelial-to-mesenchymal, and macrophage-to-mesenchymal transitions in the kidney. Front Pharmacol. 2019;10:904.
Li J, Shi S, Srivastava SP, Kitada M, Nagai T, Nitta K, et al. FGFR1 is critical for the anti-endothelial mesenchymal transition effect of N-acetyl-seryl-aspartyl-lysyl-proline via induction of the MAP4K4 pathway. Cell Death Dis. 2017;8:e2965.
Srivastava SP, Koya D, Kanasaki K. MicroRNAs in kidney fibrosis and diabetic nephropathy: roles on EMT and EndMT. Biomed Res Int. 2013;2013:125469.
Srivastava SP, Goodwin JE, Tripathi P, Kanasaki K, Koya D. Interactions among long non-coding RNAs and microRNAs influence disease phenotype in diabetes and diabetic kidney disease. Int J Mol Sci. 2021;22:6027.
Lovisa S, Kalluri R. Fatty acid oxidation regulates the activation of endothelial-to-mesenchymal transition. Trends Mol Med. 2018;24:432–4.
Srivastava SP, Zhou H, Shenoi R, Morris M, Lainez-Mas B, Goedeke L, et al. Renal Angptl4 is a key fibrogenic molecule in progressive diabetic kidney disease. Sci Adv. 2024;10:eadn6068.
Wang X, Chang HC, Gu X, Han W, Mao S, Lu L, et al. Renal lipid accumulation and aging linked to tubular cells injury via ANGPTL4. Mech Ageing Dev. 2024;219:111932.
Chung KW, Dhillon P, Huang S, Sheng X, Shrestha R, Qiu C, et al. Mitochondrial damage and activation of the sting pathway lead to renal inflammation and fibrosis. Cell Metab. 2019;30:784–99.e785.
Decout A, Katz JD, Venkatraman S, Ablasser A. The cGAS-STING pathway as a therapeutic target in inflammatory diseases. Nat Rev Immunol. 2021;21:548–69.
Chen M, Yu S, van der Sluis T, Zwager MC, Schroder CP, van der Vegt B, et al. cGAS-STING pathway expression correlates with genomic instability and immune cell infiltration in breast cancer. NPJ Breast Cancer. 2024;10:1.
Nunes JRC, O’Dwyer C, Ghorbani P, Smith TKT, Chauhan S, Robert-Gostlin V, et al. Myeloid AMPK signaling restricts fibrosis but is not required for metformin improvements during CDAHFD-induced NASH in mice. J Lipid Res. 2024;65:100564.
Wei X, Long M, Fan Z, Hou Y, Yang L, Du Y. Ulinastatin attenuates renal fibrosis by regulating AMPK/HIF-1alpha signaling pathway-mediated glycolysis. Sci Rep. 2024;14:28032.
Wang Y, Jia L, Hu Z, Entman ML, Mitch WE, Wang Y. AMP-activated protein kinase/myocardin-related transcription factor—a signaling regulates fibroblast activation and renal fibrosis. Kidney Int. 2018;93:81–94.
Yanagi T, Kikuchi H, Takeuchi K, Susa K, Mori T, Chiga M, et al. ULK1-regulated AMP sensing by AMPK and its application for the treatment of chronic kidney disease. Kidney Int. 2024;106:887–906.
Kanasaki M, Srivastava SP, Yang F, Xu L, Kudoh S, Kitada M, et al. Deficiency in catechol-O-methyltransferase is linked to a disruption of glucose homeostasis in mice. Sci Rep. 2017;7:7927.
Jiang P, Ren L, Zhi L, Yu Z, Lv F, Xu F, et al. Negative regulation of AMPK signaling by high glucose via E3 ubiquitin ligase MG53. Mol Cell. 2021;81:629–37.e625.
Lee JY, Lee M, Lee JY, Bae J, Shin E, Lee YH, et al. Ipragliflozin, an SGLT2 inhibitor, ameliorates high-fat diet-induced metabolic changes by upregulating energy expenditure through activation of the AMPK/ SIRT1 pathway. Diab Metab J. 2021;45:921–32.
Koh ES, Lim JH, Kim MY, Chung S, Shin SJ, Choi BS, et al. Anthocyanin-rich Seoritae extract ameliorates renal lipotoxicity via activation of AMP-activated protein kinase in diabetic mice. J Transl Med. 2015;13:203.
Ye K, Zhao Y, Huang W, Zhu Y. Sodium butyrate improves renal injury in diabetic nephropathy through AMPK/SIRT1/PGC-1alpha signaling pathway. Sci Rep. 2024;14:17867.
Zhang C, Xiong Y, Luo Y, Liu K, Tong Q, Song Y, et al. Morroniside ameliorates high-fat and high-fructose-driven chronic kidney disease by motivating AMPK-TFEB signal activation to accelerate lipophagy and inhibiting inflammatory response. J Agric Food Chem. 2025;73:6158–72.
Liu X, Xu C, Xu L, Li X, Sun H, Xue M, et al. Empagliflozin improves diabetic renal tubular injury by alleviating mitochondrial fission via AMPK/SP1/PGAM5 pathway. Metabolism. 2020;111:154334.
Kim MY, Lim JH, Youn HH, Hong YA, Yang KS, Park HS, et al. Resveratrol prevents renal lipotoxicity and inhibits mesangial cell glucotoxicity in a manner dependent on the AMPK-SIRT1-PGC1alpha axis in db/db mice. Diabetologia. 2013;56:204–17.
Hong YA, Lim JH, Kim MY, Kim TW, Kim Y, Yang KS, et al. Fenofibrate improves renal lipotoxicity through activation of AMPK-PGC-1alpha in db/db mice. PLoS ONE. 2014;9:e96147.
Godel M, Hartleben B, Herbach N, Liu S, Zschiedrich S, Lu S, et al. Role of mTOR in podocyte function and diabetic nephropathy in humans and mice. J Clin Invest. 2011;121:2197–209.
Inoki K, Mori H, Wang J, Suzuki T, Hong S, Yoshida S, et al. mTORC1 activation in podocytes is a critical step in the development of diabetic nephropathy in mice. J Clin Invest. 2011;121:2181–96.
Puelles VG, van der Wolde JW, Wanner N, Scheppach MW, Cullen-McEwen LA, Bork T et al. mTOR-mediated podocyte hypertrophy regulates glomerular integrity in mice and humans. JCI Insight. 2019;4:e99271.
Inoki K, Ouyang H, Zhu T, Lindvall C, Wang Y, Zhang X, et al. TSC2 integrates Wnt and energy signals via a coordinated phosphorylation by AMPK and GSK3 to regulate cell growth. Cell. 2006;126:955–68.
Centers for Disease C. Progress toward achieving the 1990 high blood pressure objectives. Morb Mortal Wkly Rep. 1990;39:704–07.
Kim J, Kundu M, Viollet B, Guan KL. AMPK and mTOR regulate autophagy through direct phosphorylation of Ulk1. Nat Cell Biol. 2011;13:132–41.
Iwata W, Unoki-Kubota H, Kato H, Shimizu A, Matsumoto M, Imasawa T, et al. Podocyte-specific deletion of tubular sclerosis complex 2 promotes focal segmental glomerulosclerosis and progressive renal failure. PLoS ONE. 2020;15:e0229397.
Bork T, Liang W, Yamahara K, Lee P, Tian Z, Liu S, et al. Podocytes maintain high basal levels of autophagy independent of mTOR signaling. Autophagy. 2020;16:1932–48.
Dugan LL, You YH, Ali SS, Diamond-Stanic M, Miyamoto S, DeCleves AE, et al. AMPK dysregulation promotes diabetes-related reduction of superoxide and mitochondrial function. J Clin Invest. 2013;123:4888–99.
Banu K, Lin Q, Basgen JM, Planoutene M, Wei C, Reghuvaran AC, et al. AMPK mediates regulation of glomerular volume and podocyte survival. JCI Insight. 2021;6:e150004.
Formigari GP, Datilo MN, Vareda B, Bonfante ILP, Cavaglieri CR, Lopes de Faria JM, et al. Renal protection induced by physical exercise may be mediated by the irisin/AMPK axis in diabetic nephropathy. Sci Rep. 2022;12:9062.
Lee HJ, Min L, Gao J, Matta S, Drel V, Saliba A, et al. Female protection against diabetic kidney disease is regulated by kidney-specific AMPK activity. Diabetes. 2024;73:1167–77.
Decleves A-E, Zolkipli Z, Satriano J, et al. Regulation of lipid accumulation by AMK-activated kinase in high-fat diet-induced kidney injury. Kidney Int. 2014;85:611–23.
Kim D, Lee JE, Jung YJ, Lee AS, Lee S, Park SK, et al. Metformin decreases high-fat diet-induced renal injury by regulating the expression of adipokines and the renal AMP-activated protein kinase/acetyl-CoA carboxylase pathway in mice. Int J Mol Med. 2013;32:1293–1302.
Rodriguez C, Sanchez A, Saenz-Medina J, Munoz M, Hernandez M, Lopez M, et al. Activation of AMP kinase ameliorates kidney vascular dysfunction, oxidative stress and inflammation in rodent models of obesity. Br J Pharmacol. 2021;178:4085–103.
Lee HJ, Mariappan MM, Norton L, Bakewell T, Feliers D, Oh SB et al. Proximal tubular epithelial insulin receptor mediates high-fat diet-induced kidney injury. JCI Insight. 2021;6:e143619.
Wu L, Liu C, Chang DY, Zhan R, Zhao M, Man Lam S, et al. The attenuation of diabetic nephropathy by annexin A1 via regulation of lipid metabolism through the AMPK/PPARalpha/CPT1b pathway. Diabetes. 2021;70:2192–203.
Srivastava SP, Li J, Kitada M, Fujita H, Yamada Y, Goodwin JE, et al. SIRT3 deficiency leads to induction of abnormal glycolysis in diabetic kidney with fibrosis. Cell Death Dis. 2018;9:997.
Li J, Liu H, Takagi S, Nitta K, Kitada M, Srivastava SP et al. Renal protective effects of empagliflozin via inhibition of EMT and aberrant glycolysis in proximal tubules. JCI Insight. 2020;5:e129034.
Wang Y, Li H, Jiang S, Fu D, Lu X, Lu M, et al. The glycolytic enzyme PFKFB3 drives kidney fibrosis through promoting histone lactylation-mediated NF-kappaB family activation. Kidney Int. 2024;106:226–40.
Zhao B, Xu Y, Chen Y, Cai Y, Gong Z, Li D, et al. Activation of TRPV4 by lactate as a critical mediator of renal fibrosis in spontaneously hypertensive rats after moderate- and high-intensity exercise. Front Physiol. 2022;13:927078.
Zhang X, Chen J, Lin R, Huang Y, Wang Z, Xu S, et al. Lactate drives epithelial-mesenchymal transition in diabetic kidney disease via the H3K14la/KLF5 pathway. Redox Biol. 2024;75:103246.
Shen Y, Jiang L, Wen P, Ye Y, Zhang Y, Ding H, et al. Tubule-derived lactate is required for fibroblast activation in acute kidney injury. Am J Physiol Ren Physiol. 2020;318:F689–701.
Hu S, Hang X, Wei Y, Wang H, Zhang L, Zhao L. Crosstalk among podocytes, glomerular endothelial cells and mesangial cells in diabetic kidney disease: an updated review. Cell Commun Signal. 2024;22:136.
Clark AR, Marshall J, Zhou Y, Montesinos MS, Chen H, Nguyen L, et al. Single-cell transcriptomics reveal disrupted kidney filter cell-cell interactions after early and selective podocyte injury. Am J Pathol. 2022;192:281–94.
Greka A, Mundel P. Cell biology and pathology of podocytes. Annu Rev Physiol. 2012;74:299–323.
Srivastava SP, Zhou H, Setia O, Dardik A, Fernandez-Hernando C, Goodwin J. Podocyte glucocorticoid receptors are essential for glomerular endothelial cell homeostasis in diabetes mellitus. J Am Heart Assoc. 2021;10:e019437.
Fu Y, Wang Y, Liu Y, Tang C, Cai J, Chen G, et al. p53/sirtuin 1/NF-kappaB signaling axis in chronic inflammation and maladaptive kidney repair after cisplatin nephrotoxicity. Front Immunol. 2022;13:925738.
Nam BY, Jhee JH, Park J, Kim S, Kim G, Park JT, et al. PGC-1alpha inhibits the NLRP3 inflammasome via preserving mitochondrial viability to protect kidney fibrosis. Cell Death Dis. 2022;13:31.
Nakada D, Saunders TL, Morrison SJ. Lkb1 regulates cell cycle and energy metabolism in haematopoietic stem cells. Nature. 2010;468:653–58.
Srivastava SP, Zhou H, Setia O, Liu B, Kanasaki K, Koya D, et al. Loss of endothelial glucocorticoid receptor accelerates diabetic nephropathy. Nat Commun. 2021;12:2368.
Srivastava SP, Goodwin JE, Kanasaki K, Koya D. Metabolic reprogramming by N-acetyl-seryl-aspartyl-lysyl-proline protects against diabetic kidney disease. Br J Pharmacol. 2020;177:3691–711.
Srivastava SP, Goodwin JE, Kanasaki K, Koya D. Inhibition of angiotensin-converting enzyme ameliorates renal fibrosis by mitigating DPP-4 level and restoring antifibrotic microRNAs. Genes. 2020;11:211.
Kanasaki K, Shi S, Kanasaki M, He J, Nagai T, Nakamura Y, et al. Linagliptin-mediated DPP-4 inhibition ameliorates kidney fibrosis in streptozotocin-induced diabetic mice by inhibiting endothelial-to-mesenchymal transition in a therapeutic regimen. Diabetes. 2014;63:2120–31.
Srivastava SP, Zhou H, Shenoi R, Morris M, Lainez-Mas B, Takagaki Y, et al. Metabolic reprogramming by endothelial ANGPTL4 depletion protects against diabetic kidney disease. Preprint at https://doi.org/10.1101/2025.05.08.652142 2025.
Wang H, Sheng J, He H, Chen X, Li J, Tan R, et al. A simple and highly purified method for isolation of glomeruli from the mouse kidney. Am J Physiol Ren Physiol. 2019;317:F1217–23.
Moeller MJ, Sanden SK, Soofi A, Wiggins RC, Holzman LB. Podocyte-specific expression of cre recombinase in transgenic mice. Genesis. 2003;35:39–42.
Guzman J, Jauregui AN, Merscher-Gomez S, Maiguel D, Muresan C, Mitrofanova A, et al. Podocyte-specific GLUT4-deficient mice have fewer and larger podocytes and are protected from diabetic nephropathy. Diabetes. 2014;63:701–14.
Funding
This study was supported by grants from the NIH (DK124709 and GM145631).
Author information
Authors and Affiliations
Contributions
SPS and KI designed, conceptualized, performed in vivo experiments, immunostaining, biochemical assays, qPCR and wrote the paper. OKG, AK, AT, JW, EO, RN, AA, AJ, MH, MSB, KK, and DK also performed immunostaining experiments and quantifications. VS contributed to statistical analyses. SY developed the image analysis program. OK performed western blotting. NI and AU maintained mouse colonies and provided experimental mice, and SH provided experimental methods.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
All experimental methods were performed in accordance with the University guidelines and regulations. All animal experiments were approved by the Institutional Animal Care and Use Committee (IACUC) at the University of Michigan (#PRO00011945) and Kanazawa Medical University animal protocols (#2014-89, #2013-114, and #2014-101).
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Srivastava, S.P., Kopasz-Gemmen, O., Kunamneni, A. et al. AMPK is dispensable for physiological podocyte and glomerular functions but prevents glomerular fibrosis in experimental diabetes. Cell Death Discov. (2026). https://doi.org/10.1038/s41420-026-03078-y
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41420-026-03078-y