Abstract
Medical devices are indispensable in modern medicine for saving patients with injuries or illnesses and improving their quality of life. The development of polymers that elicit minimal biological reactions upon contact with tissues or biological components is critical for the safe and effective use of these devices. 2-Methacryloyloxyethyl phosphorylcholine (MPC) polymers, invented in Japan, represent a milestone in bioinspired materials science because they have been designed to mimic the phosphorylcholine groups naturally present on cell membranes, endowing them with excellent biocompatibility and resistance to nonspecific protein adsorption. Diverse MPC polymer architectures have been synthesized to optimize functional expression and tailor surface properties. Moreover, molecular designs have enabled the modification of conventional polymers, metals, and ceramics, thereby expanding their utility in diverse biomedical applications. This review highlights the molecular design concept, interfacial properties, and current status of MPC polymers as highly reliable surface treatment materials and summarizes their broad implementation in medical devices.
Similar content being viewed by others
Molecular design of bioinspired 2-methacryloyloxyethyl phosphorylcholine polymers for obtaining biocompatible surfaces
Molecular assembly of phospholipid molecules as biointerfaces
Understanding and controlling intermolecular interactions are important for the development of biocompatible polymeric materials. From the standpoint of the interfaces that interact with biological systems, cell membranes constitute a critical model [1, 2]. A notable feature of the cell membrane is the asymmetric lipid distribution between the inner and outer leaflets. The outer leaflet is enriched in phospholipids containing phosphorylcholine (PC) groups, whereas the inner leaflet is predominantly composed of acidic lipids such as phosphatidylserine and phosphatidylethanolamine [3]. This asymmetry contributes to various biological functions, including signal transduction and selective molecular recognition. Nonspecific adsorption of extracellular molecules can disrupt cellular homeostasis and impair function. Thus, the outer membrane must exhibit selective interactions that suppress nonspecific binding.
PC group-bearing phospholipids possess zwitterionic head groups comprising phosphate esters and trimethylammonium moieties covalently linked via ethylene chains. The close spatial arrangement of these oppositely charged groups yields intramolecular ion pairing, neutralizing the net charge of the functional group. Because this zwitterionic character underlies the nonfouling and biocompatible properties of these lipids, phosphatidylcholine is widely used in drug delivery systems, including liposomes and lipid-based microspheres [4, 5]. These systems facilitate the dispersion of hydrophobic drugs in aqueous environments and enable their in vivo administration. In the 1970s and 1980s, the Ringsdorf and Chapman groups made significant efforts to improve the stability of phospholipid aggregates by synthesizing polymerizable phospholipids and optimizing polymerization techniques in combination with self-assembled membrane (SAM) preparation techniques [6, 7].
MPC as a new monomer for biocompatible polymers
Research on bioinspired polymers was initiated in 1987 by Ishihara and coworkers to obtain excellent biocompatibility on the basis of the chemical structures of phospholipid molecules [8]. Many studies have shown that the PC group is a candidate for weak interactions with biological systems [7, 9]. To enhance the physical stability and freedom of polymer preparation, a polymerizable group must be introduced into the molecule. Therefore, the target molecule was determined to be 2-methacryloyloxyethyl phosphorylcholine (MPC). The chemical structure of MPC is simple; the PC group is bound to a polymerizable methacrylate group with only two methylene groups, as shown in Fig. 1 [8, 10]. However, the synthesis of MPC is difficult because of its moisture sensitivity and deliquescence. Thus, it is necessary to prevent the inclusion of moisture in the synthesis process and establish a purification process in a completely dehydrated system. In the early stages of synthesis research, it took time to create a completely dehydrated solvent and devise an apparatus that prevented water from being mixed during the synthesis procedure. It was particularly difficult to devise a method to obtain pure trimethylamine from an aqueous solution, which was available as a reagent at that time. It took more than four months to complete the process by individually improving the series of operational steps. The product was recrystallized in a flask to obtain a white powder with a melting point (mp) of approximately 140 °C. This synthetic process was reported in the Polymer Journal in 1990 [8] and was applied for the industrialization of MPC in Japan by NOF Co. after a slight modification in 1999, supported by the Japan Science and Technology Agency (JST) [10].
A Synthesis of 2-methacryloyloxyethyl phosphorylcholine (MPC) polymers and MPC derivatives with various methods. ATRP: atom-transfer radical polymerization, RAFT polymerization: reversible addition-fragmentation chain-transfer polymerization. B Molecular simulation image of the poly(MPC) chain. Reprinted with permission from [77], copyright 2022 Taylor & Francis
The polymerization ability of MPC results in its homopolymer, poly(MPC), and copolymers with various vinyl monomers using a conventional radical polymerization procedure [8, 11]. In addition, advanced living radical polymerization techniques have been used to obtain well-defined MPC polymers [12,13,14,15,16,17,18]. The introduction of other monomer units into the MPC polymers alters its solubility. The MPC unit composition of the polymer is easily regulated by adjusting the monomer feed ratio. When MPC is copolymerized with various alkyl methacrylates such as n-butyl methacrylate (BMA) (forming poly(MPC-co-BMA [PMB])) or n-dodecyl methacrylate (DMA) (forming poly(MPC-co-DMA [PMD])), the polymerization proceeds smoothly, resulting in a statistically random sequence [11]. The solubility of MPC polymers in the solvent depends on the MPC unit composition, the hydrophobicity of the alkyl methacrylate unit, and the molecular weight of the polymer. For example, PMB with 30 mol% MPC, whose molecular weight is ≥500 kDa, enhances stability under biological conditions, preventing easy detachment from the substrate even when the solution casting process is applied. It remains stable in sterilized environments such as γ-rays, ethylene oxide gas, and autoclave sterilization, making it suitable for use in medical devices. This polymer is registered in the Master Files for Devices of the US Food and Drug Administration as Lipidure®-CM5206 (NOF Co) [10]. Conversely, when the molecular weight of the PMB is less than approximately 50 kDa, it is water soluble despite the MPC unit mole fraction being the same [19]. This is a unique property of the PC group; a small fraction of the MPC units (30 mol%) in the polymer can dissolve a large fraction of the hydrophobic BMA units (70 mol%).
Various MPC derivatives and MPC polymers with other functional monomer units have been synthesized and studied worldwide [20,21,22].
Another crucial MPC reaction involves Michael addition of thiol or amine compounds. In addition, the hydrosilylation reaction between the silane compounds and the methacrylate group in MPC proceeds well. These reactions allow for the introduction of PC groups into compounds with various functional groups, including silane couplings, diols, and primary amino groups [23,24,25].
Comments on misleading research reports in older MPC polymer studies
A research group at Tokyo Medical and Dental University first reported the synthesis of MPC and its polymers in 1978 in Kobunshi Ronbunshu, the official journal of the Society of Polymer Science, Japan [26]. They synthesized MPC by condensing 2-hydroxyethyl methacrylate (HEMA) with 2-bromoethyl phosphoryl dichloride, followed by quaternization with trimethylamine. Water was required to convert phosphoryl chloride to a PC group; however, its presence hindered crystallization and resulted in low yields. Owing to the high water absorption and deliquescent nature of MPC, the physical and biological properties of both MPC and its polymers are often misunderstood. For example, both MPC and poly(MPC) strongly induce hemolysis of red blood cells. Additionally, cell culture tests revealed that fibroblasts proliferated well on plates coated with poly(MPC-co-methyl methacrylate (MMA)), similar to those on uncoated controls. When poly(MPC-co-MMA) was exposed to whole blood, platelets adhered and became deformed [27]. These observations are inconsistent with the typical characteristics of MPC polymers.
Another report in Polymer Journal noted that poly(MPC) behaves like a polyelectrolyte, exhibiting increased viscosity upon dilution at low ionic strength [28]. This behavior, which is typically observed in polyelectrolytes, is typically attributed to the dissociation of counterions from the polymer chains at low concentrations, leading to changes in polymer conformation [29]. However, this interpretation conflicts with the known properties of poly(MPC), as such viscosity changes are unlikely because of the covalently bonded internal salt formed between the phosphate anion and the trimethylammonium cation. At that time, the reported melting point of MPC was 15 °C, suggesting that the MPC used in the experiment was of insufficient purity.
Today, reliable synthesis methods for MPC have been established, enabling the worldwide availability of high-purity MPC (with reagent-grade purity exceeding 97%). As a result, previously reported anomalous phenomena are no longer observed.
If incorrect information had remained uncorrected, it could have hindered the progress of research that may have led to new medical device applications and improvements in patients’ quality of life. Therefore, scientific information must be continuously updated. In the biotechnology industry, new materials and reagents are constantly being developed, and innovative analytical methods and techniques are emerging. As such, it is essential to verify and validate new findings by comparing them with existing data. This process helps ensure that research contributes effectively to advancements in health care and the enhancement of patients’ “quality of life (QOL)”.
Control of interfacial physicochemical properties on the MPC polymer surface under aqueous conditions
Surface modification techniques using MPC polymers
Obtaining manufacturing approval from a national organization is extremely important in the development of medical devices. Thus, confirming the performance, therapeutic effect, and safety of medical devices, as well as the safety and stability of the materials, is necessary. From an engineering perspective, it is also necessary for medical devices to be easy to mold and assemble. An important strategy is to treat the surfaces of medical devices made from existing materials using biocompatible polymers [30, 31]. In other words, dividing the functions between the bulk and surface can enable the synthesis of a composite functional material that combines moldability, mechanical strength, and biocompatibility. Therefore, MPC polymers have been surface-treated on substrates with a proven track record in medical devices. Table 1 summarizes the surface treatment methods used for MPC polymers on various substrates [32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62]. Copolymerization with HEMA and a small amount of a crosslinker provides a hydrogel for soft contact lenses [32]. Additionally, some MPC polymers with hydrophobic monomer units are water insoluble but soluble in organic solvents and can be applied in the coating process [37,38,39,40,41,42,43,44]. Water-soluble and amphiphilic MPC polymers can adsorb onto natural tissues and retain moisture or exhibit antifouling properties [33,34,35,36]. Therefore, they are commonly used as tissue treatment solutions. The simplest surface treatment method involves coating a surface with a polymer solution. In this case, it is necessary to use a solvent that does not affect the substrate so that the polymer does not aggregate during the solvent evaporation process and the coating layer does not peel off easily, even in the wet state. As mentioned above, PMB and PMD have been used to coat conventional medical devices.
For medical devices that may be subjected to mechanical stress, such as friction and deformation, stabilizing the surface treatment layer through chemical reactions is effective [46,47,48,49]. Specifically, chemical bonds can form between the substrate and the surface-treated polymer, or the polymer can be crosslinked within the coating layer. With the molecular design of MPC polymers, photoreactive polymers can be obtained by introducing phenylazide groups into some of the PMB or surface-bonded polymers by introducing catechol groups. Photoreactive MPC polymers are used as antifouling agents in dental dentures [49].
After the surface-treated polymer is dissolved in the base polymer solution to obtain a homogeneous solution, the surface-treated polymer is concentrated near the surface by controlling the solvent evaporation [56]. By dissolving the poly(MPC) and polyethylene (PE) in a mixture of xylene and n-butanol, the solvent evaporation process can make a blend membrane composed of these polymers with good transparency. It is quite unusual because the solubility parameters of these two polymers differ significantly [57]. Also, when the MPC polymer and segmented polyurethane (SPU) dissolved in a mixed solvent of methylene chloride/ethanol and then spread on a glass substrate, the MPC polymer was concentrated on the glass surface side. It is due to the difference in the evaporation rate of the solvent and solubility of these polymers. This allowed for the MPC polymer to be concentrated on the inner surface by the solvent casting method using a glass rod, and a tube whose overall mechanical properties were dominated by SPU could be obtained [54]. To create a small-diameter artificial blood vessel, patency was examined in animal experiments, which revealed superior performance compared with that of the untreated SPU. Moreover, surface modification of cellulose acetate and polysulfone (PSf) is possible through polymer blending [58, 59].
Surface grafting can maximize the surface density of MPC polymers [52, 53]. Specifically, two types of reaction patterns are considered: “graft from,” where the polymerization reaction proceeds from the substrate surface, and “graft to,” where reactive functional groups are introduced near the ends of the MPC polymer and react with the surface. Polymer-grafted surfaces can be fabricated by adsorbing benzophenone onto a polyethylene surface in an aqueous monomer solution and irradiating it with light [49]. This method is particularly effective when polyethylene does not contain reactive functional groups. UHMWPE particles grafted with poly(MPC) were used in liners to form the sliding surfaces of artificial hip joints [63]. In addition, the hydrophilicity and antifouling properties of silicone hydrogel surfaces have been improved by surface treatment with reactive MPC polymers to obtain enhanced contact lens materials [61]. The grafting of these MPC polymers was applied as a surface treatment for medical devices, as described below.
To clarify the antifouling properties of MPC polymers, many researchers have fabricated high-density polymer brush surfaces using surface-initiated living radical polymerization reactions and elucidated the mechanisms by which these surfaces inhibit biological responses such as protein adsorption and cell adhesion [64,65,66,67]. Furthermore, research has been conducted on the process by which the MPC polymer naturally and firmly coats the surface during coagulation when the polymer solution phase comes into contact with an aqueous MPC polymer solution, utilizing the formation of aggregates of water-soluble, amphiphilic MPC polymers in water [58,59,60].
Unique hydration states of the MPC polymers for preventing biological reactions
The excellent hydrophilicity of MPC polymers has attracted considerable attention in the study of interfacial phenomena [10, 20, 30, 63]. Surface modification using solvent-cast MPC polymers, especially copolymers containing hydrophobic alkyl methacrylate units, is affected by several factors, including the type of substrate, polymer concentration, and solvent evaporation time. In MPC polymers with hydrophobic side chains, the hydrophobic groups preferentially orient and concentrate at the air interface during solvent evaporation. When hydrophilic substrates such as glass are used, MPC units tend to accumulate at the substrate interface, whereas hydrophobic alkyl groups concentrate at the air interface, resulting in an increased water contact angle. However, once fully hydrated, the inherent hydrophilicity of the MPC units reduces the water contact angle to less than 20°, resulting in a highly hydrophilic surface. This behavior can be tuned by varying the structure of the hydrophobic groups. For example, incorporating rigid hydrophobic units such as naphthalene groups restricts the mobility and orientation of the polymer chains, allowing for the MPC units to remain exposed to the surface even during solvent evaporation, significantly decreasing the time required for hydration and equilibration [68]. Importantly, the surface properties of the substrate must also be considered to achieve stable deposition of the MPC polymer layer. Once solvent-cast polymer layers reach hydration equilibrium, they exhibit stable interfacial properties, as discussed below. In contrast, grafting MPC polymers to substrates enhances chain mobility, facilitating equilibration and further shortening hydration times. For example, poly(MPC) brush layers formed on surfaces exhibit water contact angles of less than 5° [69], demonstrating the high hydrophilicity of the MPC units. Furthermore, such surfaces prohibit air bubble and oil droplet attachment in aqueous environments. The contact angles of hexadecane and silicone oil on poly(MPC) brush surfaces exceed 170°.
Furthermore, the surface ζ potential of a substrate treated with an MPC polymer has a very small negative value, almost 0 mV, regardless of the substrate. This is a major characteristic not observed in other hydrophilic polymers. In other words, in the MPC polymer, even if the PC groups have negative and positive charges, their effects are shielded from each other. This is due to the zwitterionic structure, in which the charges in the molecule are exactly balanced such that they cancel each other. These interfacial properties provide strong evidence for the hypothesis that MPC polymers bearing PC groups maintain small interactions with nearby water molecules, which has been discussed in the literature [70,71,72,73,74] as a unique hydration state of MPC polymers (see Supporting Information).
Whitesides and colleagues proposed a key hypothesis regarding the characteristics of functional groups on a material surface that can prevent protein adsorption [75, 76]. Protein adsorption was observed using SAMs with different terminal functional groups; the following four characteristics were found to be associated with surfaces that resist protein adsorption: (1) hydrophilicity; (2) neutral charge; (3) the presence of hydrogen bond acceptors; and (4) the absence of hydrogen bond donors. According to these findings, the behavior of surrounding water molecules significantly affects protein adsorption at the substrate–water interface. In other words, the intermolecular interactions mediated by water molecules, such as hydrogen bonds, as well as electrostatic and hydrophobic interactions, make significant contributions. The chemical properties of the PC group fully align with Whiteside’s hypothesis, making it a potential candidate for the suppression of protein interactions.
Applications of MPC polymers in medical devices
The fundamental properties of MPC polymers and their biological functionalities have been reported [10, 77,78,79,80]. These properties can be easily and systematically controlled using conventional polymerization techniques. Polymer molecules have numerous functions, resulting in their increased use in biomedical and clinical applications. Table 2 presents representative examples of medical devices approved and treated with MPC polymers [74]. Moreover, new medical devices employing MPC polymers have been developed [41, 81]. These medical devices utilize the unique properties expressed at interfaces treated with MPC polymers to improve biocompatibility, achieve high lubricity, and sustain long-term therapeutic effects, thereby improving patients’ QOL.
Blood-contacting devices
Providing a surface that inhibits blood coagulation is important for medical devices that come in contact with blood. Anticoagulant drug therapies, such as heparin, have become extremely successful. Thus, antithrombogenic function is not as important for extracorporeal circulation system medical devices, including artificial kidneys and lungs, used at the bedside. On the other hand, these medical devices require the transport of substances through a membrane; a problem arises when the decrease in solute permeability caused by the adhesion of blood components to the hollow fiber membrane surface reduces the therapeutic effect. Efforts have been made to improve the function of artificial lungs by coating them with MPC polymers. PMD can be easily coated on the membrane surface after the artificial lung is assembled, allowing for surface modification [82]. PMD treatment suppresses platelet adhesion and activation.
When anticoagulants cannot be administered continuously, blood compatibility on the surface becomes even more crucial. In general, aspirin is orally administered to suppress platelet activation. This allows for patients to be free from bed restraints and is expected to significantly enhance their QOL. The development of artificial hearts has a long history dating back to the 1950s. The first artificial heart implanted in patients in clinical practice was of the pulsatile-flow type, with an air-driven SPU diaphragm and discharge pulsating flow similar to a living heart. However, it did not work for a long period owing to mechanical movement and material issues. This type of artificial heart is currently used as a pump to maintain blood circulation outside the body.
Instead of the diaphragm-driven artificial heart, a continuous flow left ventricular assist system has been developed. The rotating disk-shaped propeller creates blood flow, with a rate controlled by a motor. In this process, metals are used, particularly lightweight titanium (Ti)-based materials (Fig. 2A) [83]. Ti-based materials are relatively resistant to blood clotting when polished to a mirror finish. Additionally, patients with artificial heart implants take antiplatelet drugs to suppress blood clotting. However, microthrombi may form depending on the state of blood flow and reactions with the surface, potentially blocking blood vessels, particularly in the brain. To avoid this risk, the inner and outer surfaces of the pump, as well as the cannula connecting the pump made by SPU to the living heart, can be coated with the MPC polymer [37, 83]. High-molecular-weight PMB (500 kDa or more) adheres to the Ti substrate via van der Waals forces, forming a stable coating layer even in aqueous environments, including through polymer chain entanglement. On the other hand, even with the same MPC unit fraction, PMB with a molecular weight of approximately 100 kDa exhibits good adhesion in static environments. However, when it is subjected to fluid flow (3–4 L/min, as in an artificial heart), shear forces tend to cause defects in the coating layer. These findings demonstrate that molecular weight plays a crucial role in this application. As shown in Fig. 2B, the adhesion of platelets to the Ti substrate was significantly reduced by the MPC polymer coating. In vivo animal experiments were repeated in the US and Japan, confirming that the device could function for more than 800 days without anticoagulation therapy [84]. The pump interior after implantation for 35 days in a cow without anticoagulation therapy is shown in Fig. 2C. The surfaces of both parts of the pump were clean, with no observed thrombi. After these in vitro and in vivo animal experiments were performed, clinical trials began in 2005; in 2011, it became registered and available for clinical use as Japan’s first implantable artificial heart (EVAHEART®). Patients who have had this artificial heart implanted have been able to return to society. According to the literature, there were more than 207 cases in Japan by October 2021 and more than 15 cases in China, with a function duration of up to 10 years [85, 86]. Although its main purpose is to support blood circulation in patients with dilated cardiomyopathy, the use of EVAHEART® has enabled patients to return from hospital care back to their social activities.
A Illustration of the total setup of the implantable artificial heart (EVAHEART®). Reprinted with permission from [83], copyright 2019 Wiley. B SEM image of platelet adhesion on the original Ti substrate and 2-methacryloyloxyethyl phosphorylcholine polymer-coated Ti substrate. C Photograph of the pump interior after 35 days of implantation in a cow without anticoagulant therapy. Reprinted with permission from [37], copyright 2003 Wiley
Other blood-contacting medical devices with surface treatment using MPC polymers or MPC derivatives are in clinical use, including coronary artery stents implanted in the body for long periods and stents for treating cerebral aneurysms as flow diverter devices [48, 87,88,89].
Orthopedic devices
In particular, a decline in motor function due to joint disease significantly reduces a patient’s QOL and affects the maintenance of health thereafter. Thus, a fundamental treatment involves replacing the entire joint with an artificial joint. Artificial joints are medical devices that have not changed significantly in shape or material over the past 60 years. They form a sliding surface between a polymer liner and a metal or ceramic femoral head.
The loosening of artificial hip joints is the greatest problem with artificial hip joint replacement. This occurs because the wear on the sliding surface of the artificial joint activates the surrounding cells and dissolves the bone. Therefore, to simultaneously improve the lubrication properties and biocompatibility of the surface, a method for the photoinduced graft polymerization of an MPC onto the surface of PE was developed using benzophenone chemistry in 1999 [90]. This process was applied to treat a slightly crosslinked UHMPE (CLPE) liner of an artificial hip joint [50, 91]. The treatment thickness ranged from approximately 100–150 nm (Fig. 3A) [91]. During the photochemical grafting process, the molecular weight of the resulting poly(MPC) graft chains can be controlled by adjusting the monomer concentration. In addition, the density of the grafted polymer chains can be tuned by varying the irradiation time. In our fundamental studies, a systematic investigation of these parameters and their effects on surface wettability revealed that stable lubricating properties are achieved when the graft layer thickness reaches at least 100 nm.
A Chemical structure of poly(2-methacryloyloxyethyl phosphorylcholine [MPC])-grafted CLPE and corresponding FE-TEM image of the interface. Reprinted with permission from [77], copyright 2022 Taylor & Francis. B Mechanical wear test results of conventional CLPE and poly(MPC)-grafted CLPE. Reprinted with permission from [92], copyright 2014 Wiley. C Relationship between the implantation period and the Japanese Orthopedic Association (JOA) hip score, which represents patient QOL
The hip joint of a living body is subjected to loads that are several times its body weight from various directions during the walking cycle. To observe the wear resistance effect in an environment closer to that in the body, a mechanical hip joint simulator test under a 280 kgf load of 20 million consecutive cycles (equivalent to approximately 20 years of normal life) was performed using a Co-Cr-Mo alloy femoral head (Fig. 3B) [91]. The original CLPE surface was hydrophobic; however, by treating the surface with a hydrophilic poly(MPC) grafting layer, a water-lubrication mechanism was generated on the sliding surface of the joint. The friction torque of the sliding surface of the poly(MPC)-grafted CLPE joint was reduced to approximately 1/10 that of the untreated CLPE. When the weight change of the liner was measured over time, the effect of poly(MPC) grafting on the amount of wear was evident, being 1/40 that of untreated UHMWPE and 1/4 that of CLPE. Analysis of the sliding surface after the test using an X-ray photoelectron spectrometer and a field-emission transmission electron microscope (FE-TEM) indicated the presence of phosphorus and nitrogen atoms in the MPC unit. By optimizing the poly(MPC) grafting procedure, significant wear resistance was observed even after more than 70 million continuous cycles of walking load, and the treatment effect was maintained [92]. Analysis of the CLPE wear debris collected from the medium during the mechanical simulation tests revealed that the number of wear debris particles from the poly(MPC)-grafted CLPE was less than 1/100 that of the untreated CLPE. Furthermore, biological experiments using macrophage suspensions demonstrated that untreated polymer microparticles significantly increased cytokine and prostaglandin levels and induced osteoclast differentiation. These results suggest that even when wear debris is generated from poly(MPC)-grafted CLPE, it is unlikely to induce subsequent biological responses. These basic research studies have led to the development of long-lasting artificial joints that dramatically extend the lifespan of hip implants. It has also enabled the creation of a groundbreaking new technology named “Aquala® Technology (Kyocera, Co., Japan),” which has expanded treatment options, such as the use of artificial hip joint surgery in younger patients for whom surgical intervention was previously considered challenging. After clinical trials involving medical engineering and industry-academia collaboration, the product was approved for manufacturing in April 2011 and introduced into clinical practice in October of the same year. As of September 2025, approximately 110,000 artificial hip joint replacement surgeries have been performed using poly(MPC)-grafted CLPE liners. A recent 15-year follow-up study revealed that the Japanese Orthopedic Association (JOA) hip score, an indicator of patient QOL, increased from approximately 43/100 before surgery to more than 92/100 at 1 year after surgery and remained at a value of greater than 94/100 for the next 15 years (Fig. 3C) [93].
Recently, vitamin E has been mixed with UHMWPE to prevent oxidation reactions caused by residual radicals generated during the radiation sterilization process, and MPC has been graft-polymerized onto this substrate, making it suitable for use in artificial hip joints as a more stable linear material [94,95,96].
Ophthalmic devices
In ophthalmology, various polymer materials are used in medical devices. In particular, the use of contact lenses for vision correction has also significantly improved, providing good safety and comfort, even for long-term wear. Various hydrophilic monomers have been used for copolymerization with HEMA to make hydrogel soft contact lenses. MPC has also been used as one of the components worldwide for more than 25 years under the contact lens material Omafilcon A (United States Adopted Name: USAN) [34], which is referred to in reviews of the evolution of contact lens materials [97,98,99].
Required properties for long-wear contact lenses include oxygen permeability and water content. In addition to ensuring the mechanical properties of the lens material, controlling the adhesion of the biological components to the lens surface is a key challenge. Furthermore, it is essential to consider the mechanical properties (elastic modulus and friction coefficient) of the lens material and its ability to withstand corneal tissue damage. To meet these stringent requirements, a technology for grafting hydrophilic polymers onto the surface of silicone hydrogel contact lenses has been developed [53, 100]. MPC polymers with reactive functional groups at the terminal region of the polymer chain were used for surface treatment of the silicone hydrogel substrate by the “grafting-to” procedure [53]. Because a chemical bond must form between the reactive functional groups introduced into the MPC polymer and the substrate, the molecular weight of the MPC polymer is a critical parameter for establishing an effective graft layer. In particular, higher molecular weights generally yield lower graft layer densities, whereas lower molecular weights result in higher densities. However, excessive density can alter the thickness of the thin water layer, which is essential for properties such as lubricity and strain resistance, ultimately decreasing these performance characteristics. Consequently, the molecular structure of the MPC polymer should be determined by carefully optimizing these factors.
The MPC polymer chains are uniformly and covalently bonded to the surface of the silicone hydrogel substrate, forming a grafted layer. This procedure is named “Celligent® Technology” (Alcon Inc., Switzerland) [100]. Because only the surface serves as the active site during this reaction, the mechanical strength and optical transparency of the silicone hydrogel material remain unchanged before and after the reaction. Environmental SEM was used to confirm the formation of a hydrated MPC polymer layer on the surface of the silicone hydrogel [53]. Observations of the near-surface area using atomic force microscopy and an FE-TEM revealed that the MPC polymer layer on the surface of the silicone hydrogel material was approximately 200-nm thick and uniformly covered the surface (Fig. 4A). Although untreated silicone hydrogel materials have a high elastic modulus near the surface (350 kPa), surface modification with the MPC polymer reduced the elastic modulus to approximately 40 kPa, approximately one-ninth the original value [101]. This value was comparable to the elastic modulus of the human cornea. The surface friction coefficient of the MPC polymer-modified silicone hydrogel material was approximately 0.05, a small value comparable to that of the surface of the living cornea [102]. These phenomena are attributed to the high mobility of water in the MPC polymer graft layer. Specifically, grafting the surface with the MPC polymer to mimic the corneal surface reduced the elastic modulus and friction near the contact lens surface, achieving values comparable to those of living tissue. This significantly reduces the impact of contact lenses on the cornea and eyelids during wear, improves comfort, and protects living tissues. This silicone hydrogel material is called Lehfilcon A (USAN), with an MPC polymer graft layer on its surface, and is used in contact lenses that can be worn for 14 days in Japan (Total 14TM, Alcon Japan) and 30 days overseas (Total 30TM, Alcon) [98].
A FE-TEM image of the interface between the silicone hydrogel substrate and the grafted 2-methacryloyloxyethyl phosphorylcholine (MPC) polymer layer. Reprinted with permission from [101], copyright 2021 American Chemical Society. B Optical view of conventional and MPC polymer-grafted silicone hydrogel contact lenses stained with Sudan Black B. Reprinted with permission from [103], copyright 2025 American Chemical Society. C Lipid adsorption and absorption of conventional and MPC polymer-grafted silicone hydrogel contact lenses. Reprinted with permission from [99], copyright 2023 American Chemical Society
One of the characteristics required for extended-wear contact lenses is strain resistance. They must effectively resist the adsorption of tear components, such as proteins and lipids, and allow for easy removal of contaminants such as cosmetic residues and airborne particles through simple daily cleaning. Lipid adsorption affects the long-term biocompatibility of silicone hydrogel lenses as the contact lens wear time increases and as the amount of adsorption from the tear fluid increases. Initial lipid absorption is important for promoting contact lens lubrication. However, as the number of blinking friction cycles increases, the unsaturated fatty acid moieties in the adsorbed lipids become insoluble. These hydrophobic molecules displace water molecules hydrated with hydrophilic functional groups on the contact lens surface, reducing the water wettability and increasing the friction coefficient of the lens surface. Therefore, reducing lipid absorption is a key requirement for the development of contact lens materials that can be worn over extended periods of time.
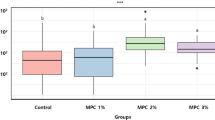
The lipid adsorption state of the silicone hydrogel contact lenses immersed in a lipid solution is shown in Fig. 4B [99, 103]. Cholesterol esters and triglyceride lipids were detected using fluorescence staining. In silicone hydrogel materials, lipids adsorb on the surface and diffuse into the interior of the material. This is because the silicone phase present within the silicone hydrogel substrate, which increases the oxygen permeability, is exposed on the surface, providing a pathway for lipid adsorption and diffusion. In conventional lenses, strong fluorescence associated with lipid absorption is observed on the lens surface. Observation of the lens cross-section revealed fluorescence within the material, indicating that the lipids had diffused into the interior. Conversely, in the case of Lehfilcon A, fluorescence associated with lipid absorption was observed on the surface or interior of the lens. The antibacterial adhesion resistance observed on the surface of Lehfilcon A is another important feature of long-wear contact lenses [104, 105]. Furthermore, the surface removes dirt such as cosmetic contaminants [106], pollen, and fine dust [107]. The staining evaluated using a hydrophobic dye (Sudan Black B) on a silicone hydrogel contact lens is shown in Fig. 4C [103]. The dye remained on the surface of the conventional silicone hydrogel contact lens, whereas the surface of the Lehfilcon A silicone hydrogel contact lens was clean.
Concluding remarks and future perspective
On the basis of the research findings on MPC polymers, numerous studies have examined related zwitterionic polymers and their biomedical applications [31, 76, 77, 80, 108,109,110,111]. Representative examples include polymers bearing sulfobetaine groups (sulfobetaine methacrylate, SBMA: 2-(N-3-sulfopropyl-N,N-dimethylamnnmonium)ethyl methacrylate) or carboxybetaine groups (carboxybetaine methacrylate, CBMA: 3-((2-(methacryloyloxy)ethyl)dimethylammonio)propanoate), whose monomers are commercially available as reagents. These polymers possess both positive and negative charges within a single molecule and are expected to exhibit properties similar to those of phosphorylcholine groups; however, their specific characteristics vary with the functional group and the structure of the substituent. For example, sulfobetaine compounds display very low water solubility [112], which is attributed to strong intermolecular electrostatic interactions arising from the independent action of the sulfonate anion and trimethylammonium cation. In contrast, their solubility increases markedly in aqueous NaCl solutions, where counterion shielding mitigates electrostatic interactions. Thus, SBMA-based polymers show good water solubility under high-ionic-strength conditions such as those in physiological environments. The possibility of ionic bond formation between sulfobetaine groups has also been noted. Because these compounds may be present in the body for extended periods, further investigation is warranted to clarify potential issues such as precipitation or adsorption induced by multivalent ions. Nevertheless, SBMA polymers have been applied as surface coatings for leukocyte separation filters used in blood product preparation [113], and their use in blood-contacting devices [114] is under active study. Carboxybetaine compounds, by contrast, possess high water solubility and are retained even after polymerization. Like phosphorylcholine groups, the zwitterionic structure of a weak acid combined with a strong base maintains the internal ionic balance and promotes extensive hydration [115]. Although medical devices incorporating CBMA polymers are not yet clinically available, their application to blood separation filters [116] and extracorporeal blood circuits is being explored [117]. However, reports of immunogenicity indicate that a more detailed evaluation is needed before their use as soluble polymers or modifiers on liposomes [118]. In addition, polymers containing a choline phosphate group, in which the anionic and cationic groups of the phosphorylcholine moiety are reversed, have been synthesized and characterized [119, 120]. Unlike poly(MPC), polymers containing choline phosphate groups are significantly affected by external ionic strength, resulting in the stretching of polymer chains. Comparative studies of their structural properties with those of SBMA and CBMA polymers have been conducted, but concerns remain regarding their potential for strong interactions with red blood cell membranes [121].
The temporal trends in publications retrieved from the Web of Science database using the search terms “phosphorylcholine and polymer” and “zwitterion and polymer” are shown in Fig. 5. Research on MPC polymers has been active since 2000, when MPC first became readily available as a reagent. This body of work has expanded globally to encompass polymer synthesis, the development of novel polymer architectures and interfaces, their characterization, and investigations into their medical performance as well as new medical, pharmaceutical, and biomedical applications. Inspired by studies on polymers bearing phosphorylcholine groups, research on zwitterionic polymers has increased markedly since 2007. Beyond polymers containing sulfobetaine or carboxybetaine groups, increasing attention has been given to newly designed zwitterionic polymers that feature a well-balanced distribution of positively and negatively charged functional groups within each polymer molecule.
Annual trend in the number of papers containing the keywords “phosphorylcholine and polymer” and “zwitterion and polymer” in the Web of Science Database
In addition to their medical functionality, research on strain [122,123,124] and frost resistance [125, 126], as well as ionic conductivity and other electrical properties [127,128,129,130,131,132], has become a new field that exploits the characteristics of MPC polymers. Research using MPC has expanded because of many factors, including the fact that MPC is a relatively easy compound to handle, polymerization is simple, raw materials are steadily being supplied, and MPC has become recognized as a common monomer. Further research will likely attract wide interest and continue the advancement of exploration in this field.
References
Fang RH, Jiang Y, Fang JC, Zhang L. Cell membrane-derived nanomaterials for biomedical applications. Biomaterials. 2017;128:69–83.
Zhao J, Ruan J, Lv G, Shan Q, Fan Z, Wang H, et al. Cell membrane-based biomimetic nanosystems for advanced drug delivery in cancer therapy: a comprehensive review. Colloids Surf B Biointerfaces. 2022;215:112503.
Miyata Y, Segawa K. Protocol to analyze lipid asymmetry in the plasma membrane. STAR Protoc. 2022;3:101870.
Weinstein JN, Leserman LD. Liposomes as drug carriers in cancer chemotherapy. Pharm Ther. 1984;24:207–33.
Yamaguchi T, Mizushima Y. Lipid microspheres for drug delivery from the pharmaceutical viewpoint. Crit Rev Ther Drug Carr Syst. 1994;11:215–29.
Hub HH, Hupfer B, Koch H, Ringsdorf H. Polymerizable phospholipid analogues—New stable biomembrane and cell models. Angew Chem Int Ed Engl. 1980;19:938–40.
Hayward JA, Johnston DS, Chapman D. Polymeric phospholipids as new biomaterials. Ann N Y Acad Sci. 1985;446:267–81.
Ishihara K, Ueda T, Nakabayashi N. Preparation of phospholipid polymers and their properties as polymer hydrogel membranes. Polym J. 1990;23:355–60.
Bonte F, Hsu MJ, Papp A, Wu K, Regen SL, Juliano RL. Interactions of polymerizable phosphatidylcholine vesicles with blood components: relevance to biocompatibility. Biochim Biophys Acta. 1987;900:1–9.
Ishihara K. Revolutionary advances in 2-methacryloyloxyethyl phosphorylcholine polymers as biomaterials. J Biomed Mater Res A. 2019;107:933–43.
Ueda T, Oshida H, Kurita K, Ishihara K, Nakabayashi N. Preparation of 2-methacryloyloxyethyl phosphorylcholine copolymers with alkyl methacrylates and their blood compatibility. Polym J. 2002;24:1259–69.
Lobb EJ, Ma I, Billingham NC, Armes SP, Lewis AL. Facile synthesis of well-defined, biocompatible phosphorylcholine-based methacrylate copolymers via atom transfer radical polymerization at 20 °C. J Am Chem Soc. 2001;123:7913–14.
Ma I, Lobb EJ, Billingham NC, Armes SP, Lewis AL, Lloyd AW, et al. Synthesis of biocompatible polymers. 1. Homopolymerization of 2-methacryloyloxyethyl phosphorylcholine via ATRP in protic solvents: an optimization study. Macromolecules. 2002;35:9306–14.
Adibnia V, Olszewski M, De Crescenzo G, Matyjaszewski K, Banquy X. Superlubricity of zwitterionic bottlebrush polymers in the presence of multivalent ions. J Am Chem Soc. 2020;142:14843–47.
Chantasirichot S, Inoue Y, Ishihara K. Photoinduced atom transfer radical polymerization in a polar solvent to synthesize a water-soluble poly(2-methacryloyloxyethyl phosphorylcholine) and its block-type copolymers. Polymer. 2015;61:55–60.
Yusa SI, Fukuda K, Yamamoto T, Ishihara K, Morishima Y. Synthesis of well-defined amphiphilic block copolymers having phospholipid polymer sequences as a novel biocompatible polymer micelle reagent. Biomacromolecules. 2005;6:663–70.
Inoue Y, Watanabe J, Takai M, Yusa SI, Ishihara K. Synthesis of sequence-controlled copolymers from extremely polar and apolar monomers by living radical polymerization and their phase-separated structures. J Polym Sci Part A: Polym Chem. 2005;43:6073–83.
Bhuchar N, Deng Z, Ishihara K, Narain R. Detailed study of the reversible addition-fragmentation chain transfer polymerization and co-polymerization of 2-methacryloyloxyethyl phosphorylcholine. Polym Chem. 2011;2:632–39.
Ishihara K, Iwasaki Y, Nakabayashi N. Polymeric lipid nanosphere consisting of water soluble poly(2-methacryloyloxyethyl phosphorylcholine-co-n-butyl methacrylate). Polym J. 1999;31:1231–6.
Monge S, Canniccioni B, Graillot A, Robin JJ. Phosphorus-containing polymers: a great opportunity for the biomedical field. Biomacromolecules. 2011;12:1973–82.
Iwasaki Y. Photoassisted Surface modification with zwitterionic phosphorylcholine polymers for the fabrication of ideal biointerfaces. Langmuir. 2023;39:15417–30.
Seetasang S, Xu Y. Recent progress and perspectives in applications of 2-methacryloyloxyethyl phosphorylcholine polymers in biodevices at small scales. J Mater Chem B. 2022;10:2323–37.
Matsuno R, Takami K, Ishihara K. Simple synthesis of a library of zwitterionic surfactants via Michael-type addition of methacrylate and alkane thiol compounds. Langmuir. 2010;26:13028–32.
Takami K, Matsuno R, Ishihara K. Synthesis of polyurethanes by polyaddition using diol compounds with methacrylate-derived functional groups. Polymer. 2011;52:5445–51.
Ye SH, Jang YS, Yun YH, Shankarraman V, Woolley JR, Hong Y, et al. Surface modification of a biodegradable magnesium alloy with phosphorylcholine (PC) and sulfobetaine (SB) functional macromolecules for reduced thrombogenicity and acute corrosion resistance. Langmuir. 2013;29:8320–7.
Kadoma Y, Nakabayashi N, Masuhara E, Yamauchi J. Synthesis and hemolysis test of the polymer containing phosphorylcholine groups. Kobunshi Ronbunshu. 1978;35:423 (Japanese Journal of Polymer Science and Technology)(in Japanese).
Fukushima S, Kadoma Y, Nakabayashi N. Interaction between polymer containing phosphorylcholine group and cells. Kobunshi Ronbunshu. 1983;40:785–93. (Japanese Journal of Polymer Science and Technology)(in Japanese).
Nakaya T, Toyoda H, Imoto M. Polymeric phospholipid analogues XIII. Synthesis and properties of vinyl polymers containing phosphatidyl choline groups. Polym J. 1986;18:881–5.
Yang H, Zheng Q, Cheng R. New insight into “polyelectrolyte effect. Colloids Surf A Physicochem Eng Asp. 2012;407:1–8.
Ratner BD. Surface modification of polymers: chemical, biological and surface analytical challenges. Biosens Bioelectron. 1995;10:797–804.
Amoako K, Ukita R, Cook KE. Antifouling zwitterionic polymer coatings for blood-bearing medical devices. Langmuir. 2025;41:2994–3006.
Iwashita H, Itokawa T, Suzuki T, Okajima Y, Kakisu K, Hori Y. Evaluation of in vitro wettability of soft contact lenses using tear supplements. Eye Contact Lens. 2021;47:244–8.
Fujiwara N, Yumoto H, Miyamoto K, Hirota K, Nakae H, Tanaka S, et al. 2-Methacryloyloxyethyl phosphorylcholine(MPC)-polymer suppresses an increase of oral bacteria: a single-blind, crossover clinical trial. Clin Oral Investig. 2019;23:739–6.
Ayaki M, Iwasawa A, Niwano Y. Cytotoxicity assays of new artificial tears containing 2-methacryloyloxyethyl phosphorylcholine polymer for ocular surface cells. Jpn J Ophthalmol. 2011;55:541–6.
Kanekura T, Nagata Y, Miyoshi H, Ishihara K, Nakabayashi N, Kanzaki T. Beneficial effects of synthetic phospholipid polymer, poly(2-methacryloyloxyethyl phosphorylcholine-co-n-butyl methacrylate), on stratum corneum function. Clin Exp Dermatol. 2002;27:230–4.
Cho Lee AR, Moon H, Ishihara K. Stabilization of lipid lamellar bilayer structure of stratum corneum modulated by poly(2-methacryloyloxyethyl phosphorylcholine) in relation to skin hydration and skin protection. Tissue Eng Regen Med. 2021;18:953–62.
Kihara S, Yamazaki K, Litwak KN, Litwak P, Kameneva MV, Ushiyama H, et al. In vivo evaluation of a MPC polymer coated continuous flow left ventricular assist system. Artif Organs. 2003;27:188–92.
Fujii K, Matsumoto HN, Koyama Y, Iwasaki Y, Ishihara K, Takakuda K. Prevention of biofilm formation with a coating of 2-methacryloyloxyethyl phosphorylcholine polymer. J Vet Med Sci. 2008;70:167–73.
Kaneko T, Saito T, Shobuike T, Miyamoto H, Matsuda J, Fukazawa K, et al. 2-Methacryloyloxyethyl phosphorylcholine polymer coating inhibits bacterial adhesion and biofilm formation on a suture: An in vitro and in vivo study. Biomed Res Int. 2020;2020:5639651.
Pappalardo F, Della Valle P, Crescenzi G, Corno C, Franco A, Torracca L, et al. Phosphorylcholine coating may limit thrombin formation during high-risk cardiac surgery: a randomized controlled trial. Ann Thorac Surg. 2006;81:886–91.
Iida Y, Hongo K, Onoda T, Kita Y, Ishihara Y, Takabayashi N, et al. Use of catheter with 2-methacryloyloxyethyl phosphorylcholine polymer coating is associated with long-term availability of central venous port. Sci Rep. 2021;11:5385.
Nishida K, Sakakida M, Ichinose K, Uemura T, Uehara M, Kajiwara K, et al. Development of a ferrocene-mediated needle-type glucose sensor covered with newly designed biocompatible membrane, 2-methacryloyloxyethyl phosphorylcholine-co-n-butyl methacrylate. Med Prog Technol. 1995;21:91–103.
Ranucci M, Isgrò G, Soro G, Canziani A, Menicanti L, Frigiola A. Reduced systemic heparin dose with phosphorylcholine coated closed circuit in coronary operations. Int J Artif Organs. 2004;27:311–9.
Marguerite S, Levy F, Quessard A, Dupeyron JP, Gros C, Steib A. Impact of a phosphorylcholine-coated cardiac bypass circuit on blood loss and platelet function: a prospective, randomized study. J Extra Corpor Technol. 2012;44:5–9.
Ishihara K, Fukumoto K, Miyazaki H, Nakabayashi N. Improvement of hemocompatibility on a cellulose dialysis membrane with a novel biomedical polymer having a phospholipid polar group. Artif Organs. 1994;18:559–64.
Whelan DM, van der Giessen WJ, Krabbendam SC, van Vliet EA, Verdouw PD, Serruys PW, et al. Biocompatibility of phosphorylcholine coated stents in normal porcine coronary arteries. Heart. 2000;83:338–45.
Collingwood R, Gibson L, Sedlik S, Virmani R, Carter AJ. Stent-based delivery of ABT-578 via a phosphorylcholine surface coating reduces neointimal formation in the porcine coronary model. Catheter Cardiovasc Inter. 2005;65:227–32.
Hagen MW, Girdhar G, Wainwright J, Hinds MT. Thrombogenicity of flow diverters in an ex vivo shunt model: effect of phosphorylcholine surface modification. J Neurointerv Surg. 2017;9:1006–11.
Ikeya K, Iwasa F, Inoue Y, Fukunishi M, Takahashi N, Ishihara K, et al. Inhibition of denture plaque deposition on complete dentures by 2-methacryloyloxyethyl phosphorylcholine polymer coating: A clinical study. J Prosthet Dent. 2018;119:67–74.
Moro T, Takatori Y, Ishihara K, Konno T, Takigawa Y, Matsushita T, et al. Surface grafting of artificial joints with a biocompatible polymer for preventing periprosthetic osteolysis. Nat Mater. 2004;3:829–36.
Ishihara K, Nakabayashi N, Fukumoto K, Aoki J. Improvement of blood compatibility on cellulose dialysis membrane. I. Grafting of 2-methacryloyloxyethyl phosphorylcholine on to a cellulose membrane surface. Biomaterials. 1992;13:145–9.
Kyomoto M, Ishihara K. Self-initiated surface graft polymerization of 2-methacryloyloxyethyl phosphorylcholine on poly(ether ether ketone) by photoirradiation. ACS Appl Mater Interfaces. 2009;1:537–42.
Shi X, Cantu-Crouch D, Sharma V, Pruitt J, Yao G, Fukazawa K, et al. Surface characterization of a silicone hydrogel contact lens having bioinspired 2-methacryloyloxyethyl phosphorylcholine polymer layer in hydrated state. Colloids Surf B Biointerfaces. 2021;199:111539.
Yoneyama T, Ishihara K, Nakabayashi N, Ito M, Mishima Y. Short-term in vivo evaluation of small-diameter vascular prosthesis composed of segmented poly(etherurethane)/2-methacryloyloxyethyl phosphorylcholine polymer blend. J Biomed Mater Res. 1998;43:15–20.
Hasegawa T, Iwasaki Y, Ishihara K. Preparation and performance of protein-adsorption-resistant asymmetric porous membrane composed of polysulfone/phospholipid polymer blend. Biomaterials. 2001;22:243–51.
Ueda H, Watanabe J, Konno T, Takai M, Saito A, Ishihara K. Asymmetrically functional surface properties on biocompatible phospholipid polymer membrane for bioartificial kidney. J Biomed Mater Res A. 2006;77:19–27.
Ishihara K, Nishiuchi D, Watanabe J, Iwasaki Y. Polyethylene/phospholipid polymer alloy as an alternative to poly(vinylchloride)-based materials. Biomaterials. 2004;25:1115–22.
Iwasaki Y, Nakabayashi N, Ishihara K. In vitro and ex vivo blood compatibility study of 2-methacryloyloxyethyl phosphorylcholine (MPC) copolymer-coated hemodialysis hollow fibers. J Artif Organs. 2003;6:260–6.
Ye SH, Watanabe J, Takai M, Iwasaki Y, Ishihara K. In situ Modification on Cellulose Acetate Hollow Fiber Membrane Modified Phospholipid Polymer for Biomedical Application. J Membr Sci. 2005;249:133–45.
Konno T, Ito T, Takai M, Ishihara K. Enzymatic photochemical sensing using luciferase-immobilized polymer nanoparticles covered with artificial cell membrane. J Biomater Sci Polym Ed. 2006;17:1347–57.
Shimizu T, Goda T, Minoura N, Takai M, Ishihara K. Super-hydrophilic silicone hydrogels with interpenetrating poly(2-methacryloyloxyethyl phosphorylcholine) networks. Biomaterials. 2010;31:3274–80.
Iwasaki Y, Aiba Y, Morimoto N, Nakabayashi N, Ishihara K. Semi-interpenetrating polymer networks composed of biocompatible phospholipid polymer and segmented polyurethane. J Biomed Mater Res. 2000;52:701–8.
Ishihara K. Highly lubricated polymer interfaces for advanced artificial hip joint through biomimetic design. Polym J. 2015;47:585–97.
Iwata R, Suk-In P, Hoven VP, Takahara A, Akiyoshi K, Iwasaki Y. Control of nanobiointerfaces generated from well-defined biomimetic polymer brushes for protein and cell manipulations. Biomacromolecules. 2004;5:2308–14.
Feng W, Zhu S, Ishihara K, Brash JL. Adsorption of fibrinogen and lysozyme on silicon grafted with poly(2-methacryloyloxyethyl phosphorylcholine) via surface-initiated atom transfer radical polymerization. Langmuir. 2005;21:5980–7.
Zhang Z, Morse AJ, Armes SP, Lewis AL, Geoghegan M, Leggett GJ. Effect of brush thickness and solvent composition on the friction force response of poly(2-(methacryloyloxy)ethylphosphorylcholine) brushes. Langmuir. 2011;27:2514–21.
Jiang Y, Su Y, Zhao L, Meng F, Wang Q, Ding C, et al. Preparation and antifouling properties of 2-(meth-acryloyloxy)ethyl cholinephosphate based polymers modified surface with different molecular architectures by ATRP. Colloids Surf B Biointerfaces. 2017;156:87–94.
Futamura K, Matsuno R, Konno T, Takai M, Ishihara K. Rapid development of hydrophilicity and protein adsorption resistance by polymer surfaces bearing phosphorylcholine and naphthalene groups. Langmuir. 2008;24:10340–4. 16.
Kobayashi M, Terayama Y, Hosaka N, Kaido M, Suzuki A, Yamada N, et al. Friction behavior of high-density poly(2-methacryloyloxyethyl phosphorylcholine) brush in aqueous media. Soft Matter. 2007;3:740–6.
Ishihara K, Mu M, Konno T, Inoue Y, Fukazawa K. The unique hydration state of poly(2-methacryloyloxyethyl phosphorylcholine). J Biomater Sci Polym Ed. 2017;28:884–99.
Kitano H, Imai M, Mori T, Gemmei-Ide M, Yokoyama Y, Ishihara K. Structure of water in the vicinity of phospholipid analogue copolymers as studied by vibrational spectroscopy. Langmuir. 2003;19:10260–6.
Kitano H. Characterization of polymer materials based on structure analyses of vicinal water. Polym J. 2016;48:15–24.
Shiomoto S, Inoue K, Higuchi H, Nishimura SN, Takaba H, Tanaka M, et al. Characterization of hydration water bound to choline phosphate-containing polymers. Biomacromolecules. 2022;23:2999–3008.
Ishihara K. Biomimetic polymers with phosphorylcholine groups as biomaterials for medical devices. Proc Jpn Acad, Ser B. 2024;100:579–606.
Holmlin RE, Chen X, Chapman RG, Takayama S, Whitesides GM. Zwitterionic SAMs that resist nonspecific adsorption of protein from aqueous buffer. Langmuir. 2001;17:2841–50.
Ostuni E, Chapman RG, Holmlin RE, Takayama S, Whitesides GM. A survey of structure−property relationships of surfaces that resist the adsorption of protein. Langmuir. 2001;17:5605–20.
Ishihara K. Biomimetic materials based on zwitterionic polymers toward human-friendly medical devices. Sci Technol Adv Mater. 2022;23:498–524.
Ishihara K, Nomura H, Mihara T, Kurita K, Iwasaki Y, Nakabayashi N. Why do phospholipid polymers reduce protein adsorption?. J Biomed Mater Res. 1998;39:323–30.
Ishihara K, Ziats NP, Tierney BP, Nakabayashi N, Anderson JM. Protein adsorption from human plasma is reduced on phospholipid polymers. J Biomed Mater Res. 1991;25:1397–407.
Ishihara K. Blood-compatible surfaces with phosphorylcholine-based polymers for cardiovascular medical devices. Langmuir. 2019;35:1778–87.
Çelebioğlu EC, Blevrakis E, Yilmaz M, Doğan İ, Erkent FD, Kortun Ş, et al. Efficacy and performance of the new pipeline vantage flow diverter stent with shield technology: Short-term results of a single-center experience. Sci Prog. 2025;108:368504251349714.
Campbell EJ, O’Byrne V, Stratford PW, Quirk I, Vick TA, Wiles MC, et al. Biocompatible surfaces using methacryloylphosphorylcholine laurylmethacrylate copolymer. ASAIO J. 1994;40:M853–7.
Snyder TA, Tsukui H, Kihara S, Akimoto T, Litwak KN, Kameneva MV, et al. Preclinical biocompatibility assessment of the EVAHEART ventricular assist device: coating comparison and platelet activation. J Biomed Mater Res A. 2007;81:85–92.
Yamabe T. Artificial organs with nano-technology and development of the new diagnosis tool. Ann NanoBME. 2009;2:1–10.
https://www.evaheart-usa.com/clinical-trial?utm_source=chatgpt.com
Chen HB, Wang XQ, Du J, Shi J, Ji BY, Shi L, et al. Long-term outcome of EVAHEART I implantable ventricular assist device for the treatment of end stage heart failure: clinical 3-year follow-up results of 15 cases. Zhonghua Xin Xue Guan Bing Za Zhi. Chin J Cardiovascular Dis). 2023;51:393–99. (in Chinese).
Lewis AL, Stratford PW. A review on phosphorylcholine-coated stents. J Long Term Eff Med Implants. 2017;27:233–52.
Song PS, Hahn JY, Kim DI, Song YB, Choi SH, Choi JH, et al. Duration of clopidogrel-based dual antiplatelet therapy and clinical outcomes after endeavor sprint zotarolimus eluting stent implantation in patients presenting with acute coronary syndrome. Eur J Intern Med. 2015;26:521–7.
Caroff J, Tamura T, King RM. Phosphorylcholine surface modified flow diverter associated with reduced intimal hyperplasia. J Neurointerv Surg. 2018;10:1097–101.
Ishihara K, Iwasaki Y, Ebihara S, Shindo Y, Nakabayashi N. Photoinduced graft polymerization of 2-methacryloyloxyethyl phosphorylcholine on polyethylene membrane surface for obtaining blood cell adhesion resistance. Colloids Surf B Biointerfaces. 2000;18:325–35.
Kyomoto M, Moro T, Saiga K, Hashimoto M, Ito H, Kawaguchi H, et al. Biomimetic hydration lubrication with various polyelectrolyte layers on cross-linked polyethylene orthopedic bearing materials. Biomaterials. 2012;33:4451–9.
Moro T, Takatori Y, Kyomoto M, Ishihara K, Hashimoto M, Ito H, et al. Long-term hip simulator testing of the artificial hip joint bearing surface grafted with biocompatible phospholipid polymer. J Orthop Res. 2014;32:369–76.
Moro T, Takatori Y, Tanaka S, Ishihara K, Oda H, Kim YT, et al. Clinical safety and wear resistance of the phospholipid polymer-grafted highly cross-linked polyethylene liner. J Orthop Res. 2017;35:2007–16.
Kyomoto M, Moro T, Yamane S, Watanabe K, Hashimoto M, Takatori Y, et al. Poly(2-methacryloyloxyethyl phosphorylcholine) grafting and vitamin E blending for high wear resistance and oxidative stability of orthopedic bearings. Biomaterials. 2014;35:6677–86.
Kyomoto M, Moro T, Yamane S, Takatori Y, Tanaka S, Ishihara K. A hydrated phospholipid polymer-grafted layer prevents lipid-related oxidative degradation of cross-linked polyethylene. Biomaterials. 2017;112:122–32.
Kyomoto M, Moro T, Yamane S, Watanabe K, Hashimoto M, Tanaka S, et al. A phospholipid polymer graft layer affords high resistance for wear and oxidation under load bearing conditions. J Mech Behav Biomed Mater. 2018;79:203–12.
Goda T, Ishihara K. Soft contact lens biomaterials from bioinspired phospholipid polymers. Expert Rev Med Devices. 2006;3:167–74.
Walsh K, Jones LW, Morgan P, Papas EB, Sulley A. Topical review: Twenty-five years of silicone hydrogel soft contact lenses. Optom Vis Sci. 2025;102:361–74.
Ishihara K, Shi X, Fukazawa K, Yamaoka T, Yao G, Wu JY. Biomimetic-engineered silicone hydrogel contact lens materials. ACS Appl Bio Mater. 2023;6:3600–16.
Capote-Puente R, Sánchez-González JM, Sánchez-González MC, Bautista-Llamas MJ. Evaluation of Celligent® biomimetic water gradient contact lens effects on ocular surface and subjective symptoms. Diagnostics (Basel). 2023;13:1258.
Shi X, Sharma V, Cantu-Crouch D, Yao G, Fukazawa K, Ishihara K, et al. Nanoscaled morphology and mechanical properties of a biomimetic polymer surface on a silicone hydrogel contact lens. Langmuir. 2021;37:13961–7.
Sharma V, Shi XC, Yao G, Zheng Y, Spencer ND, Wu JY. Fluid confinement within a branched polymer structure enhances tribological performance of a poly(2-methacryloyloxyethyl phosphorylcholine)-surface-modified contact lens. R Soc Open Sci. 2024;11:240957.
Liang S, Zheng Y, Sharma V, Shows A, Dunbar DC, Shi X, et al. Surface and antifouling properties of a biomimetic reusable contact lens material. ACS Omega. 2025;10:19697–704.
Ishihara K, Fukazawa K, Sharma V, Liang S, Shows A, Dunbar DC, et al. Antifouling silicone hydrogel contact lenses with a bioinspired 2-methacryloyloxyethyl phosphorylcholine polymer surface. ACS Omega. 2021;6:7058–67.
Harris V, Pifer R, Shannon P, Crary M. Comparative evaluation of pseudomonas aeruginosa adhesion to a poly-(2-methacryloyloxyethyl phosphorylcholine)-modified silicone hydrogel contact lens. Vis (Basel). 2023;7:27.
Mimura T, Nakagomi R. Comparison of non-water proof mascara adhesion on the surface of different two-week frequent replacement silicone hydrogel contact lenses. Clin Optom (Auckl). 2025;17:73–82.
Mimura T, Nakagomi R, Fujishima H. Comparison of asian dust adhesion on the urface of different reusable silicone hydrogel contact lenses. Int Ophthalmol. 2025;45:226.
Sin MC, Chen SH, Chang Y. Hemocompatibility of zwitterionic interfaces and membranes. Polym J. 2014;46:436–43.
Hiranphinyophat S, Iwasaki Y. Controlled biointerfaces with biomimetic phosphorus-containing polymers. Sci Technol Adv Mater. 2021;22:301–16.
Moayedi S, Xia W, Lundergan L, Yuan H, Xu J. Zwitterionic polymers for biomedical applications: Antimicrobial and antifouling strategies toward implantable medical devices and drug delivery. Langmuir. 2024;40:23125–45.
Lv W, Wang Y, Fu H, Liang Z, Huang B, Jiang R, et al. Recent advances of multifunctional zwitterionic polymers for biomedical application. Acta Biomater. 2024;181:19–45.
Ahmed ST, Madinya JJ, Leckband DE. Ionic strength dependent forces between end-grafted Poly (sulfobetaine) films and mica. J Colloid Interface Sci. 2022;606:298–306.
Lien C-C, Chen P-J, Venault A, Tang S-H, Fu Y, Dizon GV, et al. A zwitterionic interpenetrating network for improving the blood compatibility of polypropylene membranes applied to leukodepletion. J Membr Sci. 2019;584:148–60.
Ye SH, Orizondo RA, De BN, Kim S, Frankowski BJ, Federspiel WJ, et al. Epoxy silane sulfobetaine block copolymers for simple, aqueous thromboresistant coating on ambulatory assist lung devices. J Biomed Mater Res A. 2024;112:99–109.
Xiang Y, Xu RG, Leng Y. Molecular understanding of ion effect on polyzwitterion conformation in an aqueous environment. Langmuir. 2020;36:7648–57.
Venault A, Ye CC, Lin YC, Tsai CW, Jhong JF, Ruaan RC, et al. Zwitterionic fibrous polypropylene assembled with amphiphatic carboxybetaine copolymers for hemocompatible blood filtration. Acta Biomater. 2016;40:130–41.
Lin X, Wu K, Zhou Q, Jain P, Boit MO, Li B, et al. Photoreactive carboxybetaine copolymers impart biocompatibility and inhibit plasticizer leaching on polyvinyl chloride. ACS Appl Mater Interfaces. 2020;12:41026–37.
Ryujin T, Shimizu T, Miyahara R, Asai D, Shimazui R, Yoshikawa T, et al. Blood retention and antigenicity of polycarboxybetaine-modified liposomes. Int J Pharm. 2020;586:119521.
Hu G, Emrick T. Functional choline phosphate polymers. J Am Chem Soc. 2020;138:1828–31.
Mukai M, Ihara D, Chu CW, Cheng CH, Takahara A. Synthesis and hydration behavior of a hydrolysis-resistant quasi-choline phosphate zwitterionic polymer. Biomacromolecules. 2020;21:2125–31.
Yu X, Yang X, Horte S, Kizhakkedathu JN, Brooks DE. A thermoreversible poly(choline phosphate) based universal biomembrane adhesive. Macromol Biosci. 2014;14:334–9.
Yao Y, Dang X, Qiao X, Li R, Chen J, Huang Z, et al. Crosslinked biomimetic coating modified stainless-steel-mesh enables completely self-cleaning separation of crude oil/water mixtures. Water Res. 2022;224:119052.
He K, Duan H, Chen GY. Cleaning of oil fouling with water enabled by zwitterionic polyelectrolyte coatings: Overcoming the imperative challenge of oil-water separation membranes. ACS Nano. 2015;9:9188–98.
Niu J, Wang H, Chen J. Bio-inspired zwitterionic copolymers for antifouling surface and oil-water separation. Colloids Surf A Physicochemical Eng Asp. 2021;626:127016.
Liu Q, Locklin J. Transparent grafted zwitterionic copolymer coatings that exhibit both antifogging and self-cleaning properties. ACS Omega. 2018;3:17743–50.
Ma MQ, Zhang C, Chen TT, Yang J, Wang JJ, Ji J, et al. Bioinspired polydopamine/polyzwitterion coatings for underwater anti-oil and -freezing surfaces. Langmuir. 2019;35:1895–901.
Taylor ME, Panzer MJ. Fully-zwitterionic polymer-supported ionogel electrolytes featuring a hydrophobic ionic liquid. J Phys Chem. 2018;122:8469–76.
Yoshizawa-Fujita M, Ohno H. Applications of zwitterions and zwitterionic polymers for Li-ion batteries. Chem Rec. 2023;23:e202200287.
Tadesse MY, Zhang Z, Marioni N, Zofchak ES, Duncan TJ, Ganesan V. Mechanisms of ion transport in lithium salt-doped zwitterionic polymer-supported ionic liquid electrolytes. J Chem Phys. 2024;160:024905.
Alsaedi MK, Tadesse MY, Ganesan V. Zwitterionic polymer ionogel electrolytes supported by coulombic cross-links: Impacts of alkali metal cation identity. J Phys Chem B. 2024;128:3273–81.
Kim H, Hight-Huf N, Kang JH, Bisnoff P, Sundararajan S, Thompson T, et al. Polymer zwitterions for stabilization of CsPbBr3 perovskite nanoparticles and nanocomposite films. Angew Chem Int Ed Engl. 2020;59:10802–6.
Zhang L, Gao J, You Z. A multifunctional phosphorylcholine-based polymer reduces energy loss for efficient perovskite solar cells. J Mater Chem C. 2022;10:16781–8.
Acknowledgements
The author would like to thank the Guest Editor of this special issue, Prof. Chie Kojima, Institute of Science Tokyo, for the invitation to contribute to the manuscript. The author deeply appreciates Dr. Kyoko Fukazawa of Alcon Research, LLC, for reviewing the draft manuscript and providing significant suggestions. Additionally, the author thanks Prof. Yasuhiko Iwasaki, Kansai University; Dr. Kenji Yamazaki, Hokkaido Cardiovascular Hospital; Prof. Toru Moro, The University of Tokyo; and Dr. Masayuki Kyomoto, Kyocera Co., for providing important information used in this review manuscript. The author would also like to thank all the staff, students, and collaborators who supported the execution of this research.
Author information
Authors and Affiliations
Contributions
KI wrote the manuscript alone.
Corresponding author
Ethics declarations
Conflict of interest
The author declares no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ishihara, K. Invention and global impact of bioinspired 2-methacryloyloxyethyl phosphorylcholine polymers: molecular design, functions, and implementation in medical devices. Polym J 58, 303–317 (2026). https://doi.org/10.1038/s41428-025-01109-6
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41428-025-01109-6