Abstract
Background/Objectives
Maintaining an adequate vitamin D status is challenging for part of the Dutch adults; therefore, use of vitamin D supplements is advised for certain subgroups. Recent studies on vitamin D status in the general Dutch population are lacking. This study aimed to investigate vitamin D status in a Dutch adult population and how it compared to twelve years earlier.
Subjects/Methods
Blood samples from participants of the Lifelines cohort collected in March 2023 were analysed for 25(OH)D₃ concentration (N = 1000). Log 25(OH)D₃ concentrations and proportion participants with a 25(OH)D₃ concentration <30 (18–69 yrs)/<50 nmol/L (≥70 yrs) were compared across gender and age groups (18–49, 50–69, and ≥70 yrs) using t-tests and ANOVA. Vitamin D status in March 2023 was compared to the status in March 2011 (N = 1221). Additionally, monthly variation in vitamin D status within 2011 was studied.
Results
Men had a significantly lower median 25(OH)D₃ concentration compared to women. 25(OH)D₃ concentration increased with age. Vitamin D deficiency was observed in 7% of women 50–69 yrs and in 26% of men and 13% of women ≥70 yrs. Compared to March 2011, 25(OH)D₃ concentration in March 2023 was significantly higher (median 45–49 vs. 54–75 nmol/L). Vitamin D status throughout 2011 showed highest deficiency levels in winter.
Conclusions
Vitamin D status improved between 2011 and 2023, but still up to a quarter of men and women in different age groups were vitamin D deficient at the end of the winter of 2023. Vitamin D status was highest in the subgroups with supplementation advice.
Similar content being viewed by others
Introduction
Vitamin D is essential for maintaining bone health and is increasingly recognized for its roles in muscle function, immune function, cardiovascular health, and the prevention of chronic diseases [1, 2]. Consequences of vitamin D deficiency include muscle cramps and weakness, rachitis in children, and osteomalacia in adults. In older adults, vitamin D deficiency is associated with reduced bone mass and increased risk of osteoporosis, as insufficient vitamin D impairs the absorption of calcium and phosphorus from the diet [3]. Only a limited number of foods are naturally rich sources of vitamin D, such as fatty fish [3]. In the Netherlands, important dietary sources of vitamin D, according to the most recent Dutch National Food Consumption Survey (DNFCS) 2019–2021, include dietary supplements, vitamin D-fortified fats and oils, and meat products [4]. The human body can also synthesize vitamin D in the skin through exposure to sunlight [3]. Given the latitude of the Netherlands (52°N), cutaneous vitamin D synthesis is impaired during the winter months (approximately November to March) due to insufficient UVB radiation under low solar angles [5,6,7,8].
Assuming insufficient sunlight exposure, the Health Council of the Netherlands (HCNL) set an adequate intake of 10 µg vitamin D per day for the general Dutch population aged 0–70 years [8, 9]. The Council states that, with sufficient sunlight exposure and a fair skin type, two-thirds of the vitamin D requirement is estimated to be covered through skin synthesis. This assumes higher synthesis in the summer, and almost none in winter. To limit the risk of bone fractures, for elderly ≥70 years the HCNL set an average requirement of 10 µg/day for vitamin D and an accompanying recommended daily allowance of 20 µg/day. For some specific subgroups in the Dutch population, it is difficult to have an adequate vitamin D supply; therefore, a vitamin D supplement is advised, e.g., for women 50–69 years (10 µg/day) and older adults ≥70 years (20 µg/day) [8, 9]. Results from the DNFCS have repeatedly shown that the Dutch population does not acquire enough vitamin D from the diet to comply with the advice of the HCNL, assuming insufficient sunlight exposure [10, 11]. However, the DNFCS is a poor predictor of vitamin D status because there is no information on participants’ sunlight exposure.
Studies on vitamin D status in the Dutch population are ≥10 years old and mostly focus on high-risk groups such as children [12], non-Western immigrants [13,14,15], pregnant women [16, 17], and older adults [18,19,20]. These studies found vitamin D deficiency in 1–41% of subgroups with deficiency cut-off values of <20–30 nmol/L [12,13,14,15,16,17, 20] and 36-51% with a cut-off value of <50 nmol/L [18,19,20]. Prevalence was lowest in young children [12]. More than a decade ago, a few studies measured vitamin D status in a general adult population, and deficiency ranged from 4 to 16% [21, 22]. Recent insight into vitamin D status for the general adult population in the Netherlands is lacking.
Our study aimed to investigate the current vitamin D status of a Dutch adult population at the end of winter (2023), when vitamin D status is expected to be the lowest, using data from the Lifelines cohort study. A further objective was to examine whether vitamin D status improved over time between 2011 and 2023. We hypothesized that vitamin D status may have improved over this time, while deficiency would still be present in part of the population. The last aim was to obtain insight into vitamin D status fluctuation over the year.
Subjects and methods
Study design
For this cross-sectional study, data from the Lifelines cohort were used. In brief, Lifelines is a multi-disciplinary prospective population-based cohort study examining in a unique three-generation design the health and health-related behaviours of 167,729 persons living in the North of the Netherlands. It employs a broad range of investigative procedures in assessing the biomedical, socio-demographic, behavioural, physical and psychological factors which contribute to the health and disease of the general population, with a special focus on multi-morbidity and complex genetics. Further details are described elsewhere [23].
Participant selection data 2023
Participants were eligible for this study if a blood sample was collected in March 2023 (N = 2328). Children <18 years old, participants with dark skin, and pregnant women were underrepresented in the participant pool of March 2023 and were therefore excluded from this study. From the remaining participants (N = 2232), a subset of 1000 was selected for vitamin D status analysis, based on a sample size calculation ensuring sufficient power to estimate both the serum vitamin D concentration and the proportion below the cut-off for deficiency (margin of error: 0.03; Z-score: 1.96). The sample population (N = 1000) was stratified into three equal age groups (either n = 333 or n = 334), based on the age groups from the Dutch vitamin D supplementation advice: 18–49 years, 50–69 years, and ≥70 years. These age groups will be referred to as the youngest-, middle-, and oldest age groups, respectively. To increase the representativeness of the Dutch population, participants within each stratum were randomly selected based on the conditional distribution of gender, age, and education level using data from Statistics Netherlands (CBS, 2023) [24].
Participant selection data 2011
For 2011, 25(OH)D₃ concentrations had already been measured and were available in the Lifelines database. These data were used to observe changes in vitamin D status over time. Data were available for 7515 participants, distributed across eight months of the year, after applying the same exclusion criteria as described earlier. Due to underrepresentation, older adults ≥70 years old were excluded, as well as the data from July, August, November, and December, due to few (n < 10) or no data points being available, which further reduced the number of participants to N = 7315. Participant distribution by month was as follows: January (n = 923), February (n = 1117), March (n = 1221), April (n = 1158), May (n = 1343), June (n = 548), September (n = 792), and October (n = 213). There was minimal overlap in participants between the March 2023 sample and the March 2011 sample (n = 10). Across all months of 2011 compared to March 2023, the participant overlap was limited to n = 68 (n ≤ 10 for each month).
Questionnaires
For the sample population of March 2023, background information including age, education level, and degree of urbanisation was obtained from the most recently completed questionnaire available (between 2019 and 2023), collected at five-year intervals. Participant age was defined as the age in March 2023. Body mass index (BMI) was calculated using objectively measured weight and height, and categorized into underweight/normal weight (≤25 kg/m²), overweight (>25–<30 kg/m²), and obese (≥30 kg/m²). Education level was divided into three categories: low (no education, primary education, lower/preparatory secondary vocational education, or junior general secondary education), middle (secondary vocational education, work-based learning pathway or senior general secondary education, pre-university secondary education), and high (higher vocational education or university education) [25]. Degree of urbanization was determined using the participant’s postal code and was categorized into three groups based on address density: extremely/strongly urbanised (>1500–≥2500 addresses/km²), moderately/hardly urbanised (500–1500 addresses/km²), and not urbanised (<500 addresses/km²) [26]. For the sample population from 2011, age was defined as the age in the month for which vitamin D status was available in 2011.
Serum 25(OH)D₃ analysis
Serum samples from the 1000 selected participants in March 2023 were drawn from the Lifelines biobank. Serum 25(OH)D₃ concentration was measured at the Radboud Medical Center (Nijmegen, the Netherlands) using liquid chromatography-tandem mass spectrometry (LC-MS/MS) with the Waters Xevo TQ-XS Tandem MS and software version MassLynx V4.2. Internal controls with low and high levels of 25(OH)D₃ were added to each analysed batch of samples (low: CV = 4.4%, high: CV = 3.2%). The Radboud Medical Center participates in the external quality assurance (EQA) by the Dutch Foundation for Quality Assessment in Medical Laboratory Diagnostics (SKML). 25(OH)D₃ concentration from samples of 2011 was also measured by LC/MS-MS. More detailed information about the measurement of the 2011 samples is described elsewhere [21].
Indicators for high and low (deficiency) levels of vitamin D
The cut-off values for deficiency in this study align with the recommendations from the HCNL: serum 25(OH)D₃ concentration <50 nmol/L for older adults ≥70 years and <30 nmol/L for the rest of the population [8]. A serum 25(OH)D₃ concentration >200 nmol/L was used as an indication of high serum levels. This threshold aligns with the no observed adverse effect level (NOAEL) outlined in a prior revision of the upper level (UL) for vitamin D by the Scientific Committee on Food [27]. The latest revision of the UL for adults by the European Food Safety Authority (EFSA) was based on evidence of persistent hypercalciuria rather than a specific serum 25(OH)D concentration [28].
Statistical analysis
Statistical analyses were conducted using R (version 4.2.0) in RStudio (version 2023.12.0). Normality was checked visually with QQ plots and data were log-transformed when not normally distributed. For March 2023, baseline characteristics were compared between men and women using a Chi-square test for education level, BMI, and urbanization, and a Mann–Whitney U test for age. No weight factor was applied. Median (P25-P75), mean (SD), and proportion below the HCNL cut-off values or above the NOAEL of SCF were calculated for 25(OH)D₃ concentrations. Differences in log(25(OH)D₃ concentration) and proportions below cut-off values between age groups, gender, and educational level were tested using t-tests or ANOVA (including post-hoc tests where appropriate). Linear regression analysis was performed to check for associations with demographic factors. P-values were corrected for multiple testing using the Benjamini–Hochberg procedure to ensure the false discovery rate was ≤5% [29].
The participant selection from the 2011 population sample was stratified to ensure similar distributions of age, gender, and educational level compared to the 2023 sample. Median (P25–P75), mean (SD) and proportion <30, <50, or >200 nmol/L for 25(OH)D₃ concentrations in March 2011 were compared to those in March 2023 using a weighted t-test. Furthermore, a boxplot was created to observe monthly variation in status over the year 2011, and significance of ‘month’ as a variable was tested using ANOVA.
Results
Baseline characteristics of participants in March 2023, stratified by age group and gender, are presented in Table 1. Men were significantly more overweight compared to women, while women were more likely to be underweight or have normal weight.
Vitamin D status March 2023
Median (P25–P75), mean (SD), and percentage of participants below the cut-off value for deficiency are described in Table 2. Vitamin D concentration (25(OH)D₃ nmol/L) was significantly higher in women compared to men (see Supplementary Table 1 for p-values). Vitamin D concentration increased with age; median serum 25(OH)D₃ concentration for men ranged between 47 and 73 nmol/L for the youngest and oldest age groups, respectively; for women, this range was between 64 and 86 nmol/L. In the youngest and middle age groups, 7–10% of women and 13–15% of men were deficient. For the oldest age group, 13% of women and 26% of men were deficient. A 25(OH)D₃ concentration >200 nmol/L was seen for <10 participants (data not shown). Regression analysis showed that being older, being a woman, and having a lower BMI were significantly associated with a higher vitamin D status (Supplementary Table 2). When stratifying by BMI within age and sex subgroups, we observed lower vitamin D status in individuals with higher BMI in some, but not all, subgroups. However, these comparisons were limited by the small sample sizes in several of the subgroups (Table 1 and Supplementary Table 3).
Comparison to vitamin D status in March 2011
Similar to results from March 2023, vitamin D status in March 2011 increased with age (Table 2). Vitamin D deficiency was observed among 12–23% of men and 10–15% of women. There were no participants with a concentration >200 nmol/L. For men in the youngest age group, and for both the youngest and middle age groups in women, the 25(OH)D₃ concentration in March 2023 was significantly higher than in March 2011 (Table 2 and Supplementary Table 4). Interquartile range (IQR) was wider for the concentrations in 2023 than in 2011, indicating greater inter-individual variation in 2023. Despite significant differences in the proportion of deficient men and women between 2023 and 2011, no significant differences were found within age categories by gender. Similar patterns to those seen in March 2023 were observed when stratifying for BMI, as presented in Supplementary Table 3.
Monthly vitamin D status in 2011
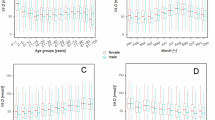
Data from 2011 showed that vitamin D status fluctuated over the months (see Fig. 1). Month was significantly associated with 25(OH)D₃ status, with highest concentrations observed in summer and lowest concentrations in winter. The proportion of participants in the youngest and middle age groups with a status below the cut-off for deficiency decreased from 12–17% in March to 5–7% in April, and few participants (0–5%) were deficient between May and October (Supplementary Table 5).
A horizontal line indicates the cut-off for deficiency (30 nmol/L) [8]. Each pair of boxplots represents the distribution of vitamin D levels for men (light grey, left) and women (dark grey with stripes, right) for the given month.
Discussion
This study provides recent insights into the vitamin D status of a population from the north of the Netherlands at the end of winter and demonstrates that up to a quarter was vitamin D deficient. Due to the latitude of the Netherlands, there is seasonal fluctuation in vitamin D status, which is observed in our 2011 data. Consequently, vitamin D deficiency also shows a seasonal fluctuation, as shown in several other publications [15, 18, 21, 30], suggesting a temporary deficiency. It is well known that vitamin D deficiency results in decreased bone health [1, 31,32,33] and there is research suggesting that higher serum 25(OH)D concentrations are related to lower risks for diseases such as colorectal cancer and type 2 diabetes [34, 35]. In general, these studies refer to long-term or chronic vitamin D deficiency. Therefore, it remains uncertain whether temporary (seasonal) vitamin D deficiency in winter will result in health complications. Similar to Hansen et al. [36], we recommend studying the health effects of periodic short-term vitamin D deficiency [36].
Our study showed an increase in vitamin D status from March 2011 to March 2023, which is consistent with earlier findings in other subgroups of the Dutch population [16, 22, 37]. Unfortunately, the Lifelines cohort did not collect data on possible explanatory factors for differences in vitamin D status, such as vitamin D supplement use, sunlight exposure, and dietary intake. Our data showed that the IQR of vitamin D status was larger in March 2023 compared to March 2011, especially at the upper end and among older adults. For some, this increase might be due to supplement intake, according to age-specific supplementation advice, but this requires further investigation. The COVID-19 pandemic brought increased attention to the possible health benefits of dietary supplements, including vitamin D [38]. This was reflected by a higher use of the supplements by the Dutch population [39], which may have persisted beyond the pandemic. A higher supplement use in women might explain their higher vitamin D status [39]. Regarding sunlight exposure, UV-environmentally available dose from the summer prior- up to the moment of measurement was similar in 2011 and 2023 (A. van Dijk (RIVM), personal communication, 28 November 2024).
Due to differences in latitude, sampling period, cut-off values, and age groups, our results are difficult to compare with studies from other European countries. Higher, lower, as well as similar proportions of vitamin D deficiency were observed in these countries [36, 40,41,42]. It remains unclear what factors explain the differences and whether these factors could be adapted in the Netherlands to ensure adequate vitamin D status throughout the year. Furthermore, the cut-off value for deficiency for individuals <70 years, set by the HCNL and used in this study, is lower compared to those set by EFSA, Institute of Medicine, Nordic Council of Ministers, and the nutrition societies of Germany, Austria, and Switzerland. Logically, if higher cut-off values had been used in this study, this would have resulted in higher percentages of deficiency. However, the HCNL concluded that concrete health results for this higher cut-off value for individuals <70 years were absent and therefore did not implement a higher cut-off value in the Netherlands [8, 9, 43,44,45,46].
Our study provides data on vitamin D status in a Dutch adult population but is not nationally representative, as the Lifelines cohort only includes residents from the northern Netherlands. To enhance representativeness, the study population was stratified by educational attainment to align with the demographic distribution of the Dutch population. Furthermore, the latest DNFCS reported no significant regional differences in the consumption of vitamin D-rich foods such as dairy, meat (including substitutes), fish, and eggs, except for fats and oils [4]. Similarly, our study found no significant differences in vitamin D status between urban and non-urban areas, suggesting that vitamin D status may be generalisable across these geographical settings. However, regional differences in sunlight exposure and supplement use remain unknown. Another topic with regard to representativeness is that more than half of the study participants were overweight or obese. This percentage is higher compared to the national average (65% vs. 50%) [47]. Multiple studies have demonstrated that a higher BMI and fat percentage are associated with lower serum 25(OH)D concentrations [48, 49]. In our study, we did not see a consistent pattern of lower vitamin D status with higher BMI category across all BMI-stratified subgroups, so whether vitamin D deficiency may be overestimated in this population remains speculative. Suggested underlying mechanisms for a lower vitamin D status are e.g., volumetric dilution or vitamin D sequestration in adipose tissue, but it is unclear whether these result in health risks linked to deficiency [50, 51].
Strengths of this study are the large sample size and the provision of recent insights into vitamin D status of a Dutch adult population, which also includes two subgroups with a vitamin D supplementation advice based on their age: women 50–69 years and elderly ≥70 years. Other important subgroups i.e., dark-skinned individuals, pregnant women, and children, were outside the scope of this research paper. Given previous findings of high deficiency rates in pregnant women and dark-skinned individuals, evaluating vitamin D status and adherence to supplementation advice in these groups should therefore be prioritized [16, 17]. A limitation of this study was the absence of data on supplement use, dietary vitamin D intake, and sunlight exposure, as missing this information limits our ability to fully contextualize higher vitamin D status over time. A second limitation was that vitamin D status was measured as cholecalciferol (vitamin D₃; 25(OH)D₃) concentration. The preferred marker for measuring vitamin D status is 25(OH)D (sum of ergocalciferol (vitamin D₂) and cholecalciferol [1, 52]), though the majority of vitamin D obtained is present as cholecalciferol, and thus 25(OH)D₃ concentration provides a good indication of total vitamin D status [33].
To conclude, up to a quarter of men and women in different age groups from this Dutch adult population had vitamin D deficiency at the end of winter 2023, and vitamin D status improved compared to the end of winter 2011. Vitamin D status was highest in the subgroups with a supplementation advice. However, given the higher cut-off values for elderly, proportion of deficiency was also highest in this group. Although our study highlights the risk for vitamin D deficiency, the associated factors remain unclear. We recommend studying vitamin D status in high-risk groups, and factors explaining the status, e.g., adherence to supplementation advice, exposure to sunlight, and dietary intake, as well as health implications of recurring short-term vitamin D deficiencies. This will help to improve current policy and provide perspective for public health strategies.
Data availability
Data described in the manuscript will not be made available. The authors do not have the authority to share the data because it is owned by Lifelines. Other researchers can apply to obtain access to Lifelines data through www.lifelines.nl.
References
Holick MF. Resurrection of vitamin D deficiency and rickets. J Clin Investig. 2006;116:2062–72.
Bouillon R, Marcocci C, Carmeliet G, Bikle D, White JH, Dawson-Hughes B, et al. Skeletal and extraskeletal actions of vitamin D: current evidence and outstanding questions. Endocr Rev. 2019;40:1109–51.
Holick MF. Vitamin D deficiency. N Engl J Med. 2007;357:266–81.
Sanderman-Nawijn E, Brants H, Dinnissen C, Ocké M, Van Rossum C. Energy and nutrient intake in the Netherlands: Results of the Dutch National Food Consumption Survey 2019-2021. Bilthoven: National Institute for Public Health and the Environment; 2024. Report No.: 2024-0071.
Holick MF. Sunlight and vitamin D for bone health and prevention of autoimmune diseases, cancers, and cardiovascular disease. Am J Clin Nutr. 2004;80:1678s–88s.
Spiro A, Buttriss JL. Vitamin D: an overview of vitamin D status and intake in Europe. Nutr Bull. 2014;39:322–50.
Webb AR, Kline L, Holick MF. Influence of season and latitude on the cutaneous synthesis of vitamin D3: exposure to winter sunlight in Boston and Edmonton will not promote vitamin D3 synthesis in human skin. J Clin Endocrinol Metab. 1988;67:373–8.
Health Council of the Netherlands. Evaluation of the dietary reference intakes for vitamin D [In Dutch: evaluatie van de voedingsnormen voor vitamine D]. The Hague: Health Council of the Netherlands; 2012. Contract No.: 2012/15.
Health Council of the Netherlands. Dietary reference values for vitamins and minerals for adults [In Dutch: voedingsnormen voor vitamines en mineralen voor volwassenen]. The Hague: Health Council of the Netherlands; 2018. Report No.: 2018/19.
Van Rossum C, Buurma-Rethans E, Dinnissen C, Beukers M, Brants H, Ocké M The diet of the Dutch: Results of the Dutch national food consumption survey 2012-2016. Bilthoven: National Institute for Public Health and the Environment; 2020.
Van Rossum C, Fransen H, Verkaik-Kloosterman J, Buurma-Rethans E, Ocké M. Dutch National Food Consumption Survey 2007-2010: diet of children and adults aged 7 to 69 years. Bilthoven: National Institute for Public Health and the Environment; 2011. Report No.: 350050006/2011.
Hoevenaar-Blom MP, Wielders JPM, Groeneveld H, de Leeuw E, Schmits RJH, Pepermans C, et al. Prevalence and determinants of vitamin D deficiency in infants and toddlers in the Netherlands: a pilot study. Ann Clin Biochem. 2019;56:613–8.
Man PW, Lin W, van der Meer IM, Heijboer AC, Wolterbeek R, Numans ME, et al. Vitamin D status in the Chinese population in the Netherlands: The DRAGON study. J Steroid Biochem Mol Biol. 2016;164:194–8.
van der Meer IM, Boeke AJ, Lips P, Grootjans-Geerts I, Wuister JD, Devillé WL, et al. Fatty fish and supplements are the greatest modifiable contributors to the serum 25-hydroxyvitamin D concentration in a multiethnic population. Clin Endocrinol. 2008;68:466–72.
Verkaik-Kloosterman J, Van Valkengoed I, de Boer E, Nicolaou M, van der AD. Nutritional status of Asian and African Surinamese and ethnic Dutch in the Netherlands – The SUNSET study [In Dutch: voedingsstatus van Hindoestaanse en Creoolse Surinamers en autochtone Nederlanders in Nederland: Het SUNSET-onderzoek]. Bilthoven: National Institute for Public Health & the Environment; 2011.
Brouwer-Brolsma EM, Vrijkotte TGM, Feskens EJM. Maternal vitamin D concentrations are associated with faster childhood reaction time and response speed, but not with motor fluency and flexibility, at the age of 5-6 years: the Amsterdam Born Children and their Development (ABCD) Study. Br J Nutr. 2018;120:345–52.
van Zwol - Janssens C, Trasande L, Asimakopoulos AG, Martinez-Moral M-P, Kannan K, Philips EM, et al. Fetal exposure to bisphenols and phthalates and childhood bone mass: a population-based prospective cohort study. Environ Res. 2020;186:109602.
Brouwer-Brolsma EM, Vaes AMM, van der Zwaluw NL, van Wijngaarden JP, Swart KMA, Ham AC, et al. Relative importance of summer sun exposure, vitamin D intake, and genes to vitamin D status in Dutch older adults: The B-PROOF study. J Steroid Biochem Mol Biol. 2016;164:168–76.
Merlijn T, Swart KMA, Lips P, Heymans MW, Sohl E, Van Schoor NM, et al. Prediction of insufficient serum vitamin D status in older women: a validated model. Osteoporos Int. 2018;29:1539–47.
Janssen HCJP, Emmelot-Vonk MH, Verhaar HJJ, van der Schouw YT. Determinants of vitamin D status in healthy men and women aged 40–80 years. Maturitas. 2013;74:79–83.
Meems LMG, de Borst MH, Postma DS, Vonk JM, Kremer HPH, Schuttelaar M-LA, et al. Low levels of vitamin D are associated with multimorbidity: Results from the LifeLines Cohort Study. Ann Med. 2015;47:474–81.
Milder I, Hendriksen M, van der AD. NL de Maat study: insufficient vitamin D status during the winter [In Dutch: NL de Maat studie: Te lage vitamine D-status in winter]. Voeding NU. 2014:8–10.
Scholtens S, Smidt N, Swertz MA, Bakker SJ, Dotinga A, Vonk JM, et al. Cohort Profile: LifeLines, a three-generation cohort study and biobank. Int J Epidemiol. 2014;44:1172–80.
Anomymous. Population; highest level of education and direction of education [in Dutch: bevolking; hoogstbehaald onderwijsniveau en onderwijsrichting]: Centraal Bureau voor Statistiek (CBS). Available from: https://opendata.cbs.nl/#/CBS/nl/dataset/85313NED/table. Accessed 2 Sept 2024.
Anonymous. Educational Attainment: Lifelines Wiki. Available from: https://wiki.lifelines.nl/doku.php?id=educational_attainment. Accessed 9 Aug 2024.
Anonymous. Urbanisation (of an area) [In Dutch: stedelijkheid (van een gebied)]: Centraal Bureau voor Statistiek (CBS). Available from: https://www.cbs.nl/nl-nl/onze-diensten/methoden/begrippen/stedelijkheid--van-een-gebied--#:~:text=%2D%20sterk%20stedelijk%3A%20gemiddelde%20oad%20van,dan%20500%20adressen%20per%20km2. Accessed 9 Aug 2024.
Scientific Committee on Food, Scientific Panel on Dietetic Products Nutrition and Allergies. Tolerable upper intake levels for vitamins and minerals. European Food Safety Authority; 2006.
EFSA Panel on Nutrition Novel Foods and Food Allergens (NDA), Turck D, Bohn T, Castenmiller J, de Henauw S, Hirsch-Ernst K-I, et al. Scientific opinion on the tolerable upper intake level for vitamin D, including the derivation of a conversion factor for calcidiol monohydrate. EFSA J. 2023;21:e08145.
Benjamini Y, Hochberg Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc Ser B. 1995;57:289–300.
Boonman-de Winter LJ, Albersen A, Mohrmann K, Bakx-van Baal CM, Meijer Timmerman Thijssen DW, Bressers JP. [High prevalence of vitamin D deficiency in the south-west Netherlands]. Ned Tijdschr Geneeskd. 2015;159:A8167.
Compston J, Vedi S, Croucher P. Low prevalence of osteomalacia in elderly patients with hip fracture. Age Ageing. 1991;20:132–4.
Bhan A, Rao AD, Rao DS. Osteomalacia as a result of vitamin D deficiency. Rheum Dis Clin. 2012;38:81–91.
Holick MF. Vitamin D: a d-lightful solution for health. J Invest Med. 2011;59:872–80.
Theodoratou E, Tzoulaki I, Zgaga L, Ioannidis JP. Vitamin D and multiple health outcomes: umbrella review of systematic reviews and meta-analyses of observational studies and randomised trials. BMJ. 2014;348:g2035.
Liu D, Meng X, Tian Q, Cao W, Fan X, Wu L, et al. Vitamin D and multiple health outcomes: an umbrella review of observational studies, randomized controlled trials, and mendelian randomization studies. Adv Nutr. 2022;13:1044–62.
Hansen L, Tjønneland A, Køster B, Brot C, Andersen R, Cohen AS, et al. Vitamin D status and seasonal variation among Danish children and adults: a descriptive study. Nutrients. 2018;10:1801.
Visser M, Deeg DJH, Puts MTE, Seidell JC, Lips P. Low serum concentrations of 25-hydroxyvitamin D in older persons and the risk of nursing home admission. Am J Clin Nutr. 2006;84:616–22.
Health Council of the Netherlands. Vitamin D and the prevention of COVID-19 and acute respiratory infections [In Dutch: vitamine D en de preventie van COVID-19 en acute luchtweginfecties]. The Hague: Health Council of the Netherlands; 2021.
Van Rossum C, Sanderman-Nawijn E, Brants H, Dinnissen C, Jansen-van der Vliet M, Beukers M, et al. The diet of the Dutch Results of the Dutch National Food Consumption Survey 2019-2021 on food consumption and evaluation with dietary guidelines. Bilthoven: National Institute for Public Health and the Environment; 2023.
Souberbielle JC, Massart C, Brailly-Tabard S, Cavalier E, Chanson P. Prevalence and determinants of vitamin D deficiency in healthy French adults: the VARIETE study. Endocrine. 2016;53:543–50.
Cashman KD, Dowling KG, Škrabáková Z, Gonzalez-Gross M, Valtueña J, De Henauw S, et al. Vitamin D deficiency in Europe: pandemic?. Am J Clin Nutr. 2016;103:1033–44.
Kerlikowsky F, Schuchardt JP, Hahn A. Folate, vitamin B12 and vitamin D status in healthy and active home-dwelling people over 70 years. BMC Geriatr. 2023;23:673.
European Food Satefy Authority (EFSA). Scientific opinion on dietary reference values for vitamin D. EFSA J. 2016;14:4547.
Nordic Council of Ministers (NCM). Nordic Nutrition Recommendations 2012 Integrating nutrition and physical activity. Copenhagen: NCM; 2014.
Institute of Medicine (IOM). Dietary Reference Intakes for calcium and vitamin D. 2011.
Nutrition Societies in Germany and Austria and Switzerland (D-A-CH). Referenzwerte für die Nährstoffzufuhr. Germany: Neustadt an der Weinstraße; 2013.
Overweight | Adults [In Dutch: Overgewicht | Volwassenen] Bilthoven: National Institute for Public Health & the Environment. Available from: https://www.vzinfo.nl/overgewicht/volwassenen#:~:text=Helft%20van%20de%20volwassen%20Nederlanders,%E2%89%A5%2025%20kg%2Fm2%20. Accessed 19 Nov 2024.
Ekwaru JP, Zwicker JD, Holick MF, Giovannucci E, Veugelers PJ. The importance of body weight for the dose response relationship of oral vitamin D supplementation and serum 25-hydroxyvitamin D in healthy volunteers. PLoS One. 2014;9:e111265.
Parikh SJ, Edelman M, Uwaifo GI, Freedman RJ, Semega-Janneh M, Reynolds J, et al. The relationship between obesity and serum 1, 25-dihydroxy vitamin D concentrations in healthy adults. J Clin Endocrinol Metab. 2004;89:1196–9.
Pourshahidi LK. Vitamin D and obesity: current perspectives and future directions. Proc Nutr Soc. 2015;74:115–24.
Prasad P, Kochhar A. Interplay of vitamin D and metabolic syndrome: a review. Diab Metab Syndrome Clin Res Rev. 2016;10:105–12.
Holick MF. High prevalence of vitamin D inadequacy and implications for health. Mayo Clin Proc. 2006;81:353–73.
Acknowledgements
The authors would like to thank Dr. Jeroen Rodenberg (National Institute for Public Health and the Environment (RIVM)) for his help with the statistical analysis.
Funding
This study was funded by the Dutch Ministry of Health, Welfare and Sport. The Dutch Ministry of Health, Welfare and Sports had no role in the design, analysis, interpretation, or writing of this article. The Lifelines initiative has been made possible by subsidy from the Dutch Ministry of Health, Welfare and Sport, the Dutch Ministry of Economic Affairs, the University Medical Center Groningen (UMCG), Groningen University and the Provinces in the North of the Netherlands (Drenthe, Friesland, Groningen).
Author information
Authors and Affiliations
Contributions
MRV, NK, CSD, and MHdJ developed the research plan; MRV performed the statistical analysis and wrote the draft manuscript; JVK, CD, MO, and MHdJ reviewed draft versions of the manuscript. All authors reviewed and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
Ethical review and approval were waived for this study. The Lifelines cohort study was approved by the Medical Ethics Committee of the University Medical Center Groningen, the Netherlands (2007/152). All methods were performed in accordance with the relevant guidelines and regulations. All participants signed informed consent upon inclusion in the cohort [23].
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Verbakel, M.R., Verkaik-Kloosterman, J., Dinnissen, C.S. et al. Vitamin D status of adults in the North of the Netherlands: cross-sectional results from the Lifelines cohort study. Eur J Clin Nutr 79, 1114–1120 (2025). https://doi.org/10.1038/s41430-025-01667-1
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41430-025-01667-1