Abstract
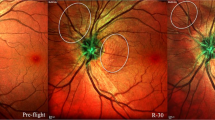
Interesting novel and somewhat perplexing physiologic and pathologic neuro-ocular findings have been documented in astronauts during and after long duration space flight (LDSF). These findings collectively have been termed the “space flight-associated neuro-ocular syndrome” (SANS). The National Aeronautics and Space Administration (NASA) in the United States has meticulously and prospectively documented the clinical, ultrasound, optical coherence tomography imaging, and radiographic findings of SANS including unilateral and bilateral optic disc edema, globe flattening, choroidal and retinal folds, hyperopic refractive error shifts, and nerve fiber layer infarcts (i.e., cotton wool spots). NASA and collaborating researchers continue to study SANS in preparation for future manned missions to space, including continued trips to the ISS, a return to the moon, or perhaps new voyages to the asteroid belt, or the planet, Mars.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Mader TH, Gibson CR, Pass AF, Kramer LA, Lee AG, Fogarty J, et al. Optic disc edema, globe flattening, choroidal folds, and hyperopic shifts observed in astronauts after long-duration space flight. Ophthalmology. 2011;118:2058–69.
Lee AG, Tarver WJ, Mader TH, Gibson CR, Hart SF, Otto CA. Neuro-ophthalmology of space flight. J Neuroophthalmol. 2016;36:85–91.
Mader TH, Gibson CR, Lee AG. Choroidal folds in astronauts. Invest Ophthalmol Vis Sci. 2016;57:592.
Mader TH, Gibson CR, Hart SF, Lee AG. Asymmetric papilledema in idiopathic intracranial hypertension. J Neuroophthalmol. 2016;36:111–2.
Mader TH, Gibson CR, Otto CA, Sargsyan AE, Miller NR, Subramanian PS, Hart SF, Lipsky W, Patel NB, Lee AG. Persistent asymmetric optic disc swelling after long-duration space flight: implications for pathogenesis. J Neuroophthalmol. 2017;37:133–39.
Sarraf D, Schwartz SD. Bilateral choroidal folds and optic neuropathy: a variant of the crowded disk syndrome? Ophthalmology. 2003;110:1047–52.
Sharma M, Volpe NJ, Patel T, Kimmel A. Intracranial hypertension associated with acquired hyperopia and choroidal folds. Retina. 1999;19:260–2.
Jacobson DM. Intracranial hypertension and the syndrome of acquired hyperopia with choroidal folds. J Neuroophthalmol. 1995;15:178–85.
Cassidy LM, Sanders MD. Choroidal folds and papilloedema. Br J Ophthalmol. 1999;83:1139–43.
Dailey RA, Mills RP, Stimac GK, et al. The natural history and CT appearance of acquired hyperopia with choroidal folds. Ophthalmology. 1986;93:1336–42.
Liu D, Kahn M. Measurement and relationship of subarachnoid pressure of the optic nerve to intracranial pressures in fresh cadavers. Am J Ophthalmol. 1993;116:548–56.
Tso MO, Hayreh SS. Optic disc edema in raised intracranial pressure. IV. Axoplasmic transport in experimental papilledema. Arch Ophthalmol. 1977;95:1458–62.
Killer HE, Jaggi GP, Flammer J, et al. The optic nerve: a new window into cerebrospinal fluid composition? Brain. 2006;129:1027–30.
Killer HE, Jaggi GP, Flammer J, et al. Cerebrospinal fluid dynamics between the intracranial and the subarachnoid space of the optic nerve. Is it Always bidirectional?. Brain. 2007;130:514–20.
Kelman SE, Sergott RC, Cioffi GA, et al. Modified optic nerve decompression in patients with functioning lumboperitoneal shunts and progressive visual loss. Ophthalmology. 1991;98:1449–53.
Killer HE, Jaggi GP, Miller NR. Papilledema revisited: is its pathophysiology really understood? Clin Exp Ophthalmol. 2009;37:444–7.
Iwasaki K, Levine BD, Zhang R, et al. Human cerebral autoregulation before, during and after spaceflight. J Physiol. 2007;579:799–810.
Frey MA, Mader TH, Bagian JP, et al. Cerebral blood velocity and other cardiovascular responses to 2 days of head-down tilt. J Appl Physiol. 1993;74:319–25.
Thornton WE, Hoffler GW, Rummel JA. Anthropometric changes and fluid shifts. In: Johnston R, Dietlein L, editors. Biomedical results from Skylab. Washington, DC: Scientific and Technical Information Office, NASA; 1977. http://lsda.jsc.nasa.gov/books/skylab//Ch32.htm.
Arbeille P, Fomina G, Roumy J, et al. Adaptation of the left heart, cerebral and femoral arteries, and jugular and femoral veins during short- and long-term head-down tilt and space flights. Eur J Appl Physiol. 2001;86:157–68.
Harris BA Jr, Billica RD, Bishop SL, et al. Physical examination during space flight. Mayo Clin Proc. 1997;72:301–8.
Herault S, Fomina G, Alferova I, et al. Cardiac, arterial and venous adaptation to weightlessness during 6-month MIR spaceflights with and without thigh cuffs (bracelets). Eur J Appl Physiol. 2000;81:384–90.
Davson H, Domer FR, Hollingsworth JR. The mechanism of drainage of the cerebrospinal fluid. Brain. 1973;96:329–36.
Andersson N, Malm J, Eklund A. Dependency of cerebrospinal fluid outflow resistance on intracranial pressure. J Neurosurg. 2008;109:918–22.
Alperin N, Lee SH, Mazda M, et al. Evidence for the importance of extracranial venous flow in patients with idiopathic intracranial hypertension (IIH). Acta Neurochir Suppl. 2005;95:129–32.
Kapoor KG, Katz SE, Grzybowski DM, Lubow M. Cerebrospinal fluid outflow: an evolving perspective. Brain Res Bull. 2008;77:327–34.
Oreskovic D, Klarica M. The formation of cerebrospinal fluid: nearly a hundred years of interpretations and misinterpretations. Brain Res Rev. 2010;64:241–62.
Giuseffi V, Wall M, Siegel PZ, Rojas PB. Symptoms and disease associations in idiopathic intracranial hypertension (pseudotumor cerebri): a case-control study. Neurology. 1991;41:239–44.
Johnston M, Zakharov A, Koh L, Armstrong D. Subarachnoid injection of Microfil reveals connections between cerebrospinal fluid and nasal lymphatics in the non-human primate. Neuropathol Appl Neurobiol. 2005;31:632–40.
Bershad EM, Urfy MZ, Calvillo E, Tang R, Cajavilca C, Lee AG, et al. Marked olfactory impairment in idiopathic intracranial hypertension. J Neurol Neurosurg Psychiatry. 2014;85:959–64. https://doi.org/10.1136/jnnp-2013-307232. [Epub ahead of print]
Mader TH, Taylor GR, Hunter N, et al. Intraocular pressure, retinal vascular, and visual acuity changes during 48h of 10 degrees head-down tilt. Aviat Space Environ Med. 1990;61:810–3.
Chiquet C, Custaud MA, Le Traon AP, et al. Changes in intraocular pressure during prolonged (7-day) head-down tilt bedrest. J Glaucoma. 2003;12:204–8.
Drozdova NT, Grishin EP. State of the visual analyzer during hypokinesia [in Russian]. Kosm Biol Med. 1972;6:46–9.
Mader TH, Gibson CR, Caputo M, et al. Intraocular pressure and retinal vascular changes during transient exposure to microgravity. Am J Ophthalmol. 1993;115:347–50.
Shinojima A, Iwasaki K, Aoki K, Ogawa Y, Yanagida R, Yuzawa M. Sufoveal choroidal thickness and foveal retinal thickness during head-down tilt. Aviat Space Environ Med. 2012;83:388–393.
Mader TH, Gibson CR, Otto CA, Sargsyan AE, Miller NR, Subramanian PS, Hart SF, Lipsky W, Patel NB, Lee AG. Why a one-way ticket to Mars may result in one-way directional glymphatic flow to the eye: response. J Neuroophthalmol. 2017;37:463–464.
Mader TH, Gibson CR, Lee AG. Optic nerve sheath distention as a protective mechanism against the visual impairment and intracranial pressure syndrome in astronauts. Invest Ophthalmol Vis Sci. 2017;58:4603–4604.
Lee AG, Mader TH, Gibson CR, Tarver W. Space flight-associated neuro-ocular syndrome. JAMA Ophthalmol. 2017;135:992–994.
Mader TH, Gibson CR, Lee AG, Patel NB, Hart SF, Pettit DR. Unilateral loss of spontaneous venous pulsations in an astronaut. J Neuroophthalmol. 2015;35:226–7.
BR Macias, Liu JH, Grande-Gutierrez N, Hargens AR. Intraocular and intracranial pressures during head-down tilt with lower body negative pressure. Aviat Space Environ Med. 2015;86:3–7.
Taibbi G, Ronita L, Cromwell RL, Kapil GK. The effect of microgravity on ocular structures and visual function: a review. Surv Ophthalmol. 2013;58:155–163.
Guiffre G, Distefano MG. Optical coherence tomography of chorioretinal and choroidal folds. Acta Ophthalmol Scand. 2007;85:333–6.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Lee, A.G., Mader, T.H., Gibson, C.R. et al. Space flight-associated neuro-ocular syndrome (SANS). Eye 32, 1164–1167 (2018). https://doi.org/10.1038/s41433-018-0070-y
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41433-018-0070-y
This article is cited by
-
Submarine medicine as an analog for spaceflight: a review of acute medical care
npj Microgravity (2026)
-
Non invasive monitoring for spaceflight associated neuro ocular syndrome: responding to a need for In flight methodologies
npj Microgravity (2025)
-
Brains in space: impact of microgravity and cosmic radiation on the CNS during space exploration
Nature Reviews Neuroscience (2025)
-
Influence of hydrostatic gradients and diurnal rhythm on cerebral and ocular blood flow
European Journal of Applied Physiology (2025)
-
Artificial gravity as a potential countermeasure for Spaceflight Associated Neuro-Ocular Syndrome
Eye (2024)