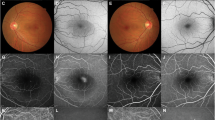
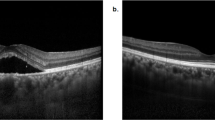
Abstract
The pathogenesis of central serous chorioretinopathy (CSCR), a pachychoroid disease, is poorly understood. While choroid hyperpermeability and retinal pigment epithelium dysfunction are cornerstones for developing CSCR, the mechanisms at the retinal, vascular, retinal pigment epithelium, and cellular level continue to be an enigma. A few preclinical studies and the development of small-sized, poorly controlled clinical trials have resulted in limited insight into the disease mechanism. Effective treatments for CSCR are still lacking as current trials have produced inconsistent results for functional and structural gains. Thus, critically evaluating the literature to explore disease mechanisms and provide an up-to-date understanding of pathophysiology can provide valuable information and avenues to new treatments. In this study, a comprehensive summary of the mechanistic insight into CSCR is presented while highlighting the shortcomings of current literature. The mechanism was divided into seven sub-categories including mechanical obstruction, inflammation, oxidative stress, paracrine factors, autonomic dysfunction, mineralocorticoid receptors activation, and medications. We implemented validated tools like the JBI and CAMARADES to objectively analyze the quality of both clinical and preclinical studies, respectively. Overall, our analysis of the literature showed that no single mechanism was populated with a large number of sufficiently sized and good-quality studies. However, compiling these studies gave hints not only to CSCR pathogenesis but also pachychoroid disease in general while providing suggestions for future exploration.
摘要
中心性浆液性脉络膜视网膜病变 (central serous chorioretinopathy, CSCR) 为肥厚性脉络膜疾病, 其发病机制尚不清楚。虽然脉络膜高通透性和视网膜色素上皮功能障碍是CSCR发生的基础, 但其在视网膜、血管、视网膜色素上皮和细胞水平上的机制仍是一个谜。一些临床前研究和小规模、控制不佳的临床试验的进展使我们对疾病机制的了解有限。由于目前的试验对CSCR的功能和结构获得的结果不一致, 因此仍然缺乏有效的治疗方法。因此, 批判性地评估文献以探索疾病机制, 并提供病理生理学方面的最新理解, 可为新的治疗方法提供有价值的信息和途径。在本研究中, 我们全面总结了CSCR的机制, 并强调了当前文献的不足之处。我们将其机制分为机械性阻塞、炎症、氧化应激、旁分泌因子、自主神经功能障碍、盐皮质激素受体激活和药物治疗等7个亚类。我们使用JBI和CAMARADES等验证工具分别客观分析临床和临床前研究的质量。总之, 我们对文献的分析表明, 缺乏大量足够大的、高质量的研究支持某个单一的机制。然而, 这些研究的汇总不仅为CSCR的发病机制提供了线索, 也提示了厚脉络疾病的一般发病机制, 同时为未来的探索提供了建议。
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
van Rijssen TJ, van Dijk EHC, Yzer S, Ohno-Matsui K, Keunen JEE, Schlingemann RO, et al. Central serous chorioretinopathy: towards an evidence-based treatment guideline. Prog Retin Eye Res. 2019;73:100770.
Dansingani KK, Balaratnasingam C, Naysan J, Freund KB. En face imaging of pachychoroid spectrum disorders with swept-source optical coherence tomography. Retina. 2016;36:499–516.
Liew G, Quin G, Gillies M, Fraser-Bell S. Central serous chorioretinopathy: a review of epidemiology and pathophysiology. Clin Exp Ophthalmol. 2013;41:201–14.
Daruich A, Matet A, Marchionno L, De Azevedo JD, Ambresin A, Mantel I, et al. Acute central serous chorioretinopathy: factors influencing episode duration. Retina. 2017;37:1905–15.
Daruich A, Matet A, Dirani A, Bousquet E, Zhao M, Farman N, et al. Central serous chorioretinopathy: recent findings and new physiopathology hypothesis. Prog Retin Eye Res. 2015;48:82–118.
Imamura Y, Fujiwara T, Spaide RF. Fundus autofluorescence and visual acuity in central serous chorioretinopathy. Ophthalmology. 2011;118:700–5.
Gackle HC, Lang GE, Freissler KA, Lang GK. [central serous chorioretinopathy. Clinical, fluorescein angiography and demographic aspects]. Ophthalmologe. 1998;95:529–33.
Sahoo NK, Singh SR, Kammari P, Jonnadula GB, Das AV, Chhablani J. Prevalence and profile of central serous chorioretinopathy in an indian cohort. Nepal J Ophthalmol. 2019;11:5–10.
Mrejen S, Balaratnasingam C, Kaden TR, Bottini A, Dansingani K, Bhavsar KV, et al. Long-term visual outcomes and causes of vision loss in chronic central serous chorioretinopathy. Ophthalmology. 2019;126:576–88.
Sahoo NK, Singh SR, Rajendran A, Shukla D, Chhablani J. Masqueraders of central serous chorioretinopathy. Surv Ophthalmol. 2019;64:30–44.
Teussink MM, Breukink MB, van Grinsven MJ, Hoyng CB, Klevering BJ, Boon CJ, et al. Oct angiography compared to fluorescein and indocyanine green angiography in chronic central serous chorioretinopathy. Investig Ophthalmol Vis Sci. 2015;56:5229–37.
Chan SY, Wang Q, Wei WB, Jonas JB. Optical coherence tomographic angiography in central serous chorioretinopathy. Retina. 2016;36:2051–8.
Min JY, Lv Y, Yu S, Gong YY. Findings of oct-angiography compared to fluorescein and indocyanine green angiography in central serous chorioretinopathy. Lasers Surg Med. 2018;50:987–93.
Manayath GJ, Shah VS, Saravanan VR, Narendran V. Polypoidal choroidal vasculopathy associated with central serous chorioretinopathy: pachychoroid spectrum of diseases. Retina. 2018;38:1195–204.
Yang L, Jonas JB, Wei W. Optical coherence tomography-assisted enhanced depth imaging of central serous chorioretinopathy. Investig Ophthalmol Vis Sci. 2013;54:4659–65.
Yanagi Y. Pachychoroid disease: a new perspective on exudative maculopathy. Jpn J Ophthalmol. 2020;64:323–37.
Warrow DJ, Hoang QV, Freund KB. Pachychoroid pigment epitheliopathy. Retina. 2013;33:1659–72.
Castro-Correia J, Coutinho MF, Rosas V, Maia J. Long-term follow-up of central serous retinopathy in 150 patients. Doc Ophthalmol. 1992;81:379–86.
Mohabati D, van Rijssen TJ, van Dijk EH, Luyten GP, Missotten TO, Hoyng CB, et al. Clinical characteristics and long-term visual outcome of severe phenotypes of chronic central serous chorioretinopathy. Clin Ophthalmol. 2018;12:1061–70.
Takahashi A, Ooto S, Yamashiro K, Tamura H, Oishi A, Miyata M, et al. Pachychoroid geographic atrophy: clinical and genetic characteristics. Ophthalmol Retin. 2018;2:295–305.
Zhao M, Célérier I, Bousquet E, Jeanny JC, Jonet L, Savoldelli M, et al. Mineralocorticoid receptor is involved in rat and human ocular chorioretinopathy. J Clin Investig. 2012;122:2672–9.
Pang CE, Shah VP, Sarraf D, Freund KB. Ultra-widefield imaging with autofluorescence and indocyanine green angiography in central serous chorioretinopathy. Am J Ophthalmol. 2014;158:362–71e2.
Kishi S, Matsumoto H, Sonoda S, Hiroe T, Sakamoto T, Akiyama H. Geographic filling delay of the choriocapillaris in the region of dilated asymmetric vortex veins in central serous chorioretinopathy. PLoS ONE. 2018;13:e0206646.
Terao N, Koizumi H, Kojima K, Yamagishi T, Nagata K, Kitazawa K, et al. Association of upregulated angiogenic cytokines with choroidal abnormalities in chronic central serous chorioretinopathy. Investig Ophthalmol Vis Sci. 2018;59:5924–31.
Jung SH, Kim KA, Sohn SW, Yang SJ. Cytokine levels of the aqueous humour in central serous chorioretinopathy. Clin Exp Optom. 2014;97:264–9.
Erol MK, Balkarli A, Yucel O, Akar Y, Dogan B, Suren E. Neutrophil/lymphocyte ratio and mean platelet volume in central serous chorioretinopathy. Ther Clin Risk Manag. 2017;13:945–50.
Ma LL, Wang YY, Yang ZH, Huang D, Weng H, Zeng XT. Methodological quality (risk of bias) assessment tools for primary and secondary medical studies: What are they and which is better? Mil Med Res. 2020;7:1–11.
Institute JB. Critical appraisal tools. Joanna Briggs Institute, The University of Adelaide. 2020. https://jbi.global/critical-appraisal-tools.
Sena E, van der Worp HB, Howells D, Macleod M. How can we improve the pre-clinical development of drugs for stroke? Trends Neurosci. 2007;30:433–9.
Hiroe T, Kishi S. Dilatation of asymmetric vortex vein in central serous chorioretinopathy. Ophthalmol Retin. 2018;2:152–61.
Imanaga N, Terao N, Nakamine S, Tamashiro T, Wakugawa S, Sawaguchi K, et al. Scleral thickness in central serous chorioretinopathy. Ophthalmol Retina. 2021;5:285–91.
Venkatesh P, Chawla R, Tripathy K, Singh HI, Bypareddy R. Scleral resection in chronic central serous chorioretinopathy complicated by exudative retinal detachment. Eye Vis. 2016;3:23.
Verma S, Kumar V, Azad S, Bhayana AA, Surve A, Kumar S, et al. Focal choroidal excavation: review of literature. Br J Ophthalmol. 2021;105:1043–8.
Chung CY, Li SH, Li KKW. Focal choroidal excavation—morphological features and clinical correlation. Eye. 2017;31:1373–9.
Obata R, Takahashi H, Ueta T, Yuda K, Kure K, Yanagi Y. Tomographic and angiographic characteristics of eyes with macular focal choroidal excavation. Retina. 2013;33:1201–10.
Yang HS, Kang TG, Park H, Heo JS, Park J, Lee KS, et al. Quantitative evaluation of choriocapillaris using optical coherence tomography and optical coherence tomography angiography in patients with central serous chorioretinopathy after half-dose photodynamic therapy. PLoS ONE. 2020;15:e0227718.
Chan WM, Lam DS, Lai TY, Tam BS, Liu DT, Chan CK. Choroidal vascular remodelling in central serous chorioretinopathy after indocyanine green guided photodynamic therapy with verteporfin: a novel treatment at the primary disease level. Br J Ophthalmol. 2003;87:1453–8.
Sirakaya E, Duru Z, Kucuk B, Duru N. Monocyte to high-density lipoprotein and neutrophil-to-lymphocyte ratios in patients with acute central serous chorioretinopathy. Indian J Ophthalmol. 2020;68:854–8.
Mazzolani F, Togni S. Oral administration of a curcumin-phospholipid delivery system for the treatment of central serous chorioretinopathy: a 12-month follow-up study. Clin Ophthalmol. 2013;7:939–45.
Khan NA, Khan AA, Khan A, Khan A, Memon JI, Shaikh M. Treatment of central serous chorioretinopathy (csc) using diclofenac through different routes of administration, a comparative study. Adv Ophthalmol Vis Syst. 2017;6:101–5.
Bahadorani S, Maclean K, Wannamaker K, Chu ER, Gresores N, Sohn JH, et al. Treatment of central serous chorioretinopathy with topical nsaids. Clin Ophthalmol. 2019;13:1543–8.
Dursun A, Toker MI, Ozec AV, Bozali E, Kirboga K, Dursun FG, et al. Relationship between mean platelet volume and central serous chorioretinopathy. Int Ophthalmol. 2017;37:119–24.
Caccavale A, Romanazzi F, Imparato M, Negri A, Morano A, Ferentini F. Low-dose aspirin as treatment for central serous chorioretinopathy. Clin Ophthalmol. 2010;4:899–903.
Lim JW, Kim MU, Shin MC. Aqueous humor and plasma levels of vascular endothelial growth factor and interleukin-8 in patients with central serous chorioretinopathy. Retina. 2010;30:1465–71.
Shin MC, Lim JW. Concentration of cytokines in the aqueous humor of patients with central serous chorioretinopathy. Retina. 2011;31:1937–43.
Sakai T, Tsuneoka H. Reduced blood serotonin levels in chronic central serous chorioretinopathy. Ophthalmol Retina. 2017;1:145–8.
Kimura T, Araki T, Komuku Y, Iwami H, Gomi F. Central serous chorioretinopathy and blood serotonin concentrations. J Clin Med. 2021;10:558.
Matet A, Jaworski T, Bousquet E, Canonica J, Gobeaux C, Daruich A, et al. Lipocalin 2 as a potential systemic biomarker for central serous chorioretinopathy. Sci Rep. 2020;10:20175.
Conde J, Lazzaro V, Scotece M, Abella V, Villar R, López V, et al. Corticoids synergize with il-1 in the induction of lcn2. Osteoarthr Cartil. 2017;25:1172–8.
Ji S, Wei Y, Chen J, Tang S. Clinical efficacy of anti-vegf medications for central serous chorioretinopathy: a meta-analysis. Int J Clin Pharm. 2017;39:514–21.
Bae SH, Heo J, Kim C, Kim TW, Shin JY, Lee JY, et al. Low-fluence photodynamic therapy versus ranibizumab for chronic central serous chorioretinopathy: One-year results of a randomized trial. Ophthalmology. 2014;121:558–65.
Chung YR, Kim JW, Song JH, Park A, Kim MH. Twelve-month efficacy of intravitreal bevacizumab injection for chronic, atypical, or recurrent central serous chorioretinopathy. Retina. 2019;39:134–42.
Lim JW, Ryu SJ, Shin MC. The effect of intravitreal bevacizumab in patients with acute central serous chorioretinopathy. Korean J Ophthalmol. 2010;24:155–8.
Karska-Basta I, Pociej-Marciak W, Chrzaszcz M, Kubicka-Trzaska A, Debicka-Kumela M, Gawecki M, et al. Imbalance in the levels of angiogenic factors in patients with acute and chronic central serous chorioretinopathy. J Clin Med 2021;10:1087.
Shiragami C, Takasago Y, Osaka R, Kobayashi M, Ono A, Yamashita A, et al. Clinical features of central serous chorioretinopathy with type 1 choroidal neovascularization. Am J Ophthalmol. 2018;193:80–6.
Loo RH, Scott IU, Flynn HW Jr, Gass JD, Murray TG, Lewis ML, et al. Factors associated with reduced visual acuity during long-term follow-up of patients with idiopathic central serous chorioretinopathy. Retina. 2002;22:19–24.
Bousquet E, Bonnin S, Mrejen S, Krivosic V, Tadayoni R, Gaudric A. Optical coherence tomography angiography of flat irregular pigment epithelium detachment in chronic central serous chorioretinopathy. Retina. 2018;38:629–38.
Cheung CMG, Lee WK, Koizumi H, Dansingani K, Lai TYY, Freund KB. Pachychoroid disease. Eye. 2019;33:14–33.
Kitaya N, Nagaoka T, Hikichi T, Sugawara R, Fukui K, Ishiko S, et al. Features of abnormal choroidal circulation in central serous chorioretinopathy. Br J Ophthalmol. 2003;87:709–12.
Podkowinski D, Foessl B, de Sisternes L, Beka S, Mursch-Edlmayr AS, Strauss RW, et al. Early alterations in retinal microvasculature on swept-source optical coherence tomography angiography in acute central serous chorioretinopathy. Sci Rep. 2021;11:3129.
Saito M, Saito W, Hirooka K, Hashimoto Y, Mori S, Noda K, et al. Pulse waveform changes in macular choroidal hemodynamics with regression of acute central serous chorioretinopathy. Invest Ophthalmol Vis Sci. 2015;56:6515–22.
Baek J, Kook L, Lee WK. Choriocapillaris flow impairments in association with pachyvessel in early stages of pachychoroid. Sci Rep. 2019;9:5565.
Schworm B, Luft N, Keidel LF, Hagenau F, Kern C, Herold T, et al. Response of neovascular central serous chorioretinopathy to an extended upload of anti-vegf agents. Graefes Arch Clin Exp Ophthalmol. 2020;258:1013–21.
Peiretti E, Caminiti G, Serra R, Querques L, Pertile R, Querques G. Anti-vascular endothelial growth factor therapy versus photodynamic therapy in the treatment of choroidal neovascularization secondary to central serous chorioretinopathy. Retina. 2018;38:1526–32.
Hata M, Yamashiro K, Ooto S, Oishi A, Tamura H, Miyata M, et al. Intraocular vascular endothelial growth factor levels in pachychoroid neovasculopathy and neovascular age-related macular degeneration. Investig Ophthalmol Vis Sci. 2017;58:292–8.
Terao N, Koizumi H, Kojima K, Yamagishi T, Yamamoto Y, Yoshii K, et al. Distinct aqueous humour cytokine profiles of patients with pachychoroid neovasculopathy and neovascular age-related macular degeneration. Sci Rep. 2018;8:10520.
Matsumoto H, Hiroe T, Morimoto M, Mimura K, Ito A, Akiyama H. Efficacy of treat-and-extend regimen with aflibercept for pachychoroid neovasculopathy and type 1 neovascular age-related macular degeneration. Jpn J Ophthalmol. 2018;62:144–50.
Kunikata H, Sato R, Nishiguchi KM, Nakazawa T. Systemic oxidative stress level in patients with central serous chorioretinopathy. Graefes Arch Clin Exp Ophthalmol. 2020;258:1575–7.
Türkcü FM, Yüksel H, Yüksel H, Sahin A, Cinar Y, Cingü AK, et al. Serum dehydroepiandrosterone sulphate, total antioxidant capacity, and total oxidant status in central serous chorioretinopathy. Graefes Arch Clin Exp Ophthalmol. 2014;252:17–21.
Turkoglu EB, Dikci S, Çelik E, Erel Ö, Neselioglu S, Alışık M, et al. Thiol/disulfide homeostasis in patients with central serous chorioretinopathy. Curr Eye Res. 2016;41:1489–91.
Altinkaynak H, Kurkcuoglu PZ, Caglayan M, Yorgun MA, Yuksel N, Kosekahya P, et al. A novel marker in acute central serous chorioretinopathy: Thiol/disulfide homeostasis. Int Ophthalmol. 2018;38:175–81.
Yavaş GF, Küsbeci T, Kaşikci M, Günay E, Doğan M, Unlü M, et al. Obstructive sleep apnea in patients with central serous chorioretinopathy. Curr Eye Res. 2014;39:88–92.
Spiers JG, Chen HJ, Sernia C, Lavidis NA. Activation of the hypothalamic-pituitary-adrenal stress axis induces cellular oxidative stress. Front Neurosci. 2014;8:456.
Vijayvergiya R, Vadivelu R. Role of helicobacter pylori infection in pathogenesis of atherosclerosis. World J Cardiol. 2015;7:134–43.
Ratanasukon M, Bhurayanontachai P, Jirarattanasopa P. High-dose antioxidants for central serous chorioretinopathy; the randomized placebo-controlled study. BMC Ophthalmol. 2012;12:20.
McDougal DH, Gamlin PD. Autonomic control of the eye. Compr Physiol. 2015;5:439–73.
Michael JC, Pak J, Pulido J, de Venecia G. Central serous chorioretinopathy associated with administration of sympathomimetic agents. Am J Ophthalmol. 2003;136:182–5.
Tewari HK, Gadia R, Kumar D, Venkatesh P, Garg SP. Sympathetic-parasympathetic activity and reactivity in central serous chorioretinopathy: A case-control study. Invest Ophthalmol Vis Sci. 2006;47:3474–8.
Pierce KK, Lane RG. Central serous chorioretinopathy associated with the use of ephedra. Retin Cases Brief Rep. 2009;3:376–8.
Chen LC, Ma JW, Shieh PC, Horng CT. Oral treatment of central serous chorioretinopathy patients using propranolol tablets. Pharmaceuticals. 2020;13:336.
Chrapek O, Jirkova B, Kandrnal V, Rehak J, Sin M. Treatment of central serous chorioretinopathy with beta-blocker metipranolol. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub. 2015;159:120–3.
Kianersi F, Fesharaki F. Effects of propranolol in patients with central serous chorioretinopathy. J Res Med Sci. 2008;13:103–7.
Sibayan SA, Kobuch K, Spiegel D, Eckert E, Leser R, Monzer J, et al. Epinephrine, but not dexamethasone, induces apoptosis in retinal pigment epithelium cells in vitro: possible implications on the pathogenesis of central serous chorioretinopathy. Graefes Arch Clin Exp Ophthalmol. 2000;238:515–9.
Ullian ME. The role of corticosteriods in the regulation of vascular tone. Cardiovasc Res. 1999;41:55–64.
Scarinci F, Ghiciuc CM, Patacchioli FR, Palmery M, Parravano M. Investigating the hypothesis of stress system dysregulation as a risk factor for central serous chorioretinopathy: a literature mini-review. Curr Eye Res. 2019;44:583–9.
Liu B, Deng T, Zhang J. Risk factors for central serous chorioretinopathy: a systematic review and meta-analysis. Retina. 2016;36:9–19.
Jain AK, Kaines A, Schwartz S. Bilateral central serous chorioretinopathy resolving rapidly with treatment for obstructive sleep apnea. Graefes Arch Clin Exp Ophthalmol. 2010;248:1037–9.
Schellevis RL, Altay L, Kalisingh A, Mulders TWF, Sitnilska V, Hoyng CB, et al. Elevated steroid hormone levels in active chronic central serous chorioretinopathy. Investig Ophthalmol Vis Sci. 2019;60:3407–13.
van Haalen FM, van Dijk EHC, Andela CD, Dijkman G, Biermasz NR, Pereira AM, et al. Maladaptive personality traits, psychological morbidity and coping strategies in chronic central serous chorioretinopathy. Acta Ophthalmol. 2019;97:e572–e9.
Liang ZQ, Huang LZ, Qu JF, Zhao MW. Association between endogenous cortisol level and the risk of central serous chorioretinopathy: a meta-analysis. Int J Ophthalmol. 2018;11:296–300.
Arndt C, Sari A, Ferre M, Parrat E, Courtas D, De Seze J, et al. Electrophysiological effects of corticosteroids on the retinal pigment epithelium. Investig Ophthalmol Vis Sci. 2001;42:472–5.
Zhao M, Valamanesh F, Celerier I, Savoldelli M, Jonet L, Jeanny JC, et al. The neuroretina is a novel mineralocorticoid target: Aldosterone up-regulates ion and water channels in muller glial cells. FASEB J. 2010;24:3405–15.
Caccavale A, Romanazzi F, Imparato M, Negri A, Morano A, Ferentini F. Central serous chorioretinopathy: a pathogenetic model. Clin Ophthalmol. 2011;5:239–43.
Sogutlu Sari E, Yazici A, Eser B, Erol MK, Kilic A, Ermis SS, et al. The prevalence of 4g/5g polymorphism of plasminogen activator inhibitor-1 (pai-1) gene in central serous chorioretinopathy and its association with plasma pai-1 levels. Cutan Ocul Toxicol. 2014;33:270–4.
Schubert C, Pryds A, Zeng S, Xie Y, Freund KB, Spaide RF, et al. Cadherin 5 is regulated by corticosteroids and associated with central serous chorioretinopathy. Hum Mutat. 2014;35:859–67.
Lotery A, Sivaprasad S, O'connell A, Harris RA, Culliford L, Ellis L, et al. Eplerenone for chronic central serous chorioretinopathy in patients with active, previously untreated disease for more than 4 months (vici): a randomised, double-blind, placebo-controlled trial. Lancet. 2020;395:294–303.
van Dijk EHC, Schellevis RL, van Bergen M, Breukink MB, Altay L, Scholz P, et al. Association of a haplotype in the nr3c2 gene, encoding the mineralocorticoid receptor, with chronic central serous chorioretinopathy. JAMA Ophthalmol. 2017;135:446–51.
Bousquet E, Dhundass M, Lejoyeux R, Shinojima A, Krivosic V, Mrejen S, et al. Predictive factors of response to mineralocorticoid receptor antagonists in nonresolving central serous chorioretinopathy. Am J Ophthalmol. 2019;198:80–7.
Pichi F, Carrai P, Ciardella A, Behar-Cohen F, Nucci P. Central Serous Chorioretinopathy Study G. Comparison of two mineralcorticosteroids receptor antagonists for the treatment of central serous chorioretinopathy. Int Ophthalmol. 2017;37:1115–25.
Gordon-Bennett P, Rimmer T. Central serous chorioretinopathy following oral tadalafil. Eye. 2012;26:168–9.
Fraunfelder FW, Fraunfelder FT. Central serous chorioretinopathy associated with sildenafil. Retina. 2008;28:606–9.
Jain M, Nevin RL, Ahmed I. Mefloquine-associated dizziness, diplopia, and central serous chorioretinopathy: a case report. J Med Case Rep. 2016;10:305.
Jain M. Quetiapine associated central serous chorioretinopathy: implicit role of serotonin and dopamine pathways. Indian J Ophthalmol. 2019;67:292–4.
Yuan Z, Hein TW, Rosa RH Jr., Kuo L. Sildenafil (viagra) evokes retinal arteriolar dilation: dual pathways via nos activation and phosphodiesterase inhibition. Investig Ophthalmol Vis Sci. 2008;49:720–5.
Breazzano MP, Coleman DJ, Chen RWS, Chang S, Daly S, Tsang SH. Prospective impact of sildenafil on chronic central serous chorioretinopathy: Pisces trial. Ophthalmol Retin. 2020;4:1119–23.
Schellevis RL, van Dijk EHC, Breukink MB, Altay L, Bakker B, Koeleman BPC, et al. Role of the complement system in chronic central serous chorioretinopathy: a genome-wide association study. JAMA Ophthalmol. 2018;136:1128–36.
de Jong EK, Breukink MB, Schellevis RL, Bakker B, Mohr JK, Fauser S, et al. Chronic central serous chorioretinopathy is associated with genetic variants implicated in age-related macular degeneration. Ophthalmology. 2015;122:562–70.
Ferreira VP, Pangburn MK, Cortes C. Complement control protein factor h: the good, the bad, and the inadequate. Mol Immunol. 2010;47:2187–97.
Kowalczuk L, Matet A, Dor M, Bararpour N, Daruich A, Dirani A, et al. Proteome and metabolome of subretinal fluid in central serous chorioretinopathy and rhegmatogenous retinal detachment: a pilot case study. Transl Vis Sci Technol. 2018;7:3.
Miki A, Kondo N, Yanagisawa S, Bessho H, Honda S, Negi A. Common variants in the complement factor h gene confer genetic susceptibility to central serous chorioretinopathy. Ophthalmology. 2014;121:1067–72.
Dorner GT, Garhöfer G, Huemer KH, Golestani E, Zawinka C, Schmetterer L, et al. Effects of adrenomedullin on ocular hemodynamic parameters in the choroid and the ophthalmic artery. Investig Ophthalmol Vis Sci. 2003;44:3947–51.
Breukink MB, Schellevis RL, Boon CJ, Fauser S, Hoyng CB, den Hollander AI, et al. Genomic copy number variations of the complement component c4b gene are associated with chronic central serous chorioretinopathy. Investig Ophthalmol Vis Sci. 2015;56:5608–13.
Hosoda Y, Miyake M, Schellevis RL, Boon CJF, Hoyng CB, Miki A, et al. Genome-wide association analyses identify two susceptibility loci for pachychoroid disease central serous chorioretinopathy. Commun Biol. 2019;2:468.
Yoshioka H, Katsume Y, Akune H. Experimental central serous chorioretinopathy in monkey eyes: fluorescein angiographic findings. Ophthalmologica. 1982;185:168–78.
Bennis A, Gorgels TGMF, Ten Brink JB, van der Spek PJ, Bossers K, Heine VM, et al. Comparison of mouse and human retinal pigment epithelium gene expression profiles: potential implications for age-related macular degeneration. PLoS ONE. 2015;10:e0141597.
Park HK, Jo W, Choi HJ, Kim B, Lee G, Seo J, et al. Usefulness of optical coherence tomography to detect central serous chorioretinopathy in monkeys. J Appl Toxicol. 2015;35:199–204.
Matsumoto H, Mukai R, Hoshino J, Oda M, Matsuzaki T, Ishizaki Y, et al. Choroidal congestion mouse model: could it serve as a pachychoroid model? PLoS ONE. 2021;16:e0246115.
Negi A, Marmor MF. Experimental serous retinal detachment and focal pigment epithelial damage. Arch Ophthalmol. 1984;102:445–9.
Matsumoto H, Miller JW, Vavvas DG. Retinal detachment model in rodents by subretinal injection of sodium hyaluronate. J Vis Exp. 2013:50660.
Ji X, Liu Y, Hurd R, Wang J, Fitzmaurice B, Nishina PM, et al. Retinal pigment epithelium atrophy 1 (rpea1): a new mouse model with retinal detachment caused by a disruption of protein kinase c, theta. Investig Ophthalmol Vis Sci. 2016;57:877–88.
Luna G, Lewis GP, Linberg KA, Chang B, Hu Q, Munson PJ, et al. Anatomical and gene expression changes in the retinal pigmented epithelium atrophy 1 (rpea1) mouse: a potential model of serous retinal detachment. Investig Ophthalmol Vis Sci. 2016;57:4641–54.
Author information
Authors and Affiliations
Contributions
PK, AG and MSB were responsible for designing the study protocol. PK and AG were responsible for screening studies, data acquisition, analysis, organizing tables/figures and drafting the manuscript. MSB, CG, RK, and SGC were involved in editing the manuscript, helping revise the protocol and providing feedback. The final version was of the manuscript was approved by all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Kanda, P., Gupta, A., Gottlieb, C. et al. Pathophysiology of central serous chorioretinopathy: a literature review with quality assessment. Eye 36, 941–962 (2022). https://doi.org/10.1038/s41433-021-01808-3
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41433-021-01808-3
This article is cited by
-
Efficacy of subliminal laser in chronic central serous chorioretinopathy: A 2.5-Year retrospective study
Lasers in Medical Science (2025)
-
Rare genetic variation in PTPRB is associated with central serous chorioretinopathy, varicose veins and glaucoma
Nature Communications (2025)
-
Exacerbation of central serous chorioretinopathy during trauma-confronting psychotherapy– a case report
BMC Psychiatry (2024)
-
Central serous chorioretinopathy with a golden sheen
Eye (2024)
-
Comparisons of choroidal blood flow velocity between initial-onset acute uveitis associated with Vogt–Koyanagi–Harada disease and acute central serous chorioretinopathy
Eye (2024)