Abstract
Background/Objectives
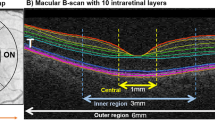
To identify longitudinal changes in each retinal layer thickness in diabetic retinopathy (DR) patients who underwent pan-retinal photocoagulation (PRP).
Subjects/Methods
The subjects were divided into three groups: type 2 diabetes patients without DR (DM group), those with DR (DR group), and those who underwent PRP ≥ 6 months earlier (PRP group). Following the baseline visit, patients underwent three additional assessments at 1-year intervals.
Results
In total, 297 eyes were included: 87, 124, and 76 in the DM, DR, and PRP groups, respectively. The baseline ganglion cell complex (GCC) thickness was 110.4 ± 13.4, 112.5 ± 13.2, and 116.1 ± 17.6 μm in the DM, DR, and PRP groups, respectively (P = 0.047). The baseline thickness of inner nuclear layer (P = 0.026) and outer plexiform layer (P = 0.002) differed significantly, which was significantly thicker in the PRP group. The photoreceptor layer and retinal pigment epithelium thicknesses differed significantly among the groups (both P < 0.001), and those in the PRP group were significantly thinner than in the other groups. In the PRP group, there were significant decreases in GCC and outer nuclear layer (ONL) thickness over time, while the other layers did not change significantly. The GCC (estimate = −0.15, P = 0.012) and ONL (estimate = −0.16, P = 0.019) thicknesses were significantly associated with changes in best-corrected visual acuity.
Conclusions
The thickness of each retinal layer of patients who underwent PRP changed differently over time, and these changes were significantly associated with changes in visual acuity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 18 print issues and online access
$259.00 per year
only $14.39 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
Data availability
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Solomon SD, Chew E, Duh EJ, Sobrin L, Sun JK, VanderBeek BL, et al. Diabetic retinopathy: a position statement by the American Diabetes Association. Diab Care. 2017;40:412.
Group DRSR. Photocoagulation treatment of proliferative diabetic retinopathy: clinical application of Diabetic Retinopathy Study (DRS) findings, DRS Report Number 8. Ophthalmology. 1981;88:583–600.
Network DRCR. Observational study of the development of diabetic macular edema following panretinal (scatter) photocoagulation given in 1 or 4 sittings. Arch Ophthalmol. 2009;127:132–40.
Shin JS, Lee YH. Changes in macular retinal layers and peripapillary nerve fiber layer thickness after 577-nm pattern scanning laser in patients with diabetic retinopathy. Korean J Ophthalmol. 2017;31:497.
Lim HB, Shin YI, Lee MW, Koo H, Lee WH, Kim JY. Ganglion cell–inner plexiform layer damage in diabetic patients: 3-year prospective, longitudinal, observational study. Sci Rep. 2020;10:1470.
Vujosevic S, Muraca A, Gatti V, Masoero L, Brambilla M, Cannillo B, et al. Peripapillary microvascular and neural changes in diabetes mellitus: an OCT-angiography study. Investig Ophthalmol Vis Sci. 2018;59:5074–81.
Lee M-W, Lee W-H, Ryu C-K, Kim T-Y, Lim H-B, Lee Y-H, et al. Effects of prolonged type 2 diabetes on the inner retinal layer and macular microvasculature: an optical coherence tomography angiography study. J Clin Med. 2020;9:1849.
Borrelli E, Barresi C, Feo A, Lari G, Grosso D, Querques L, et al. Imaging biomarkers and clinical factors associated with the rate of progressive inner and outer retinal thinning in patients with diabetic macular edema. Sci Rep. 2023;13:3224.
Li M, Tian M, Wang Y, Ma H, Zhou Y, Jiang X, et al. Updates on RPE cell damage in diabetic retinopathy. Mol Med Rep. 2023;28:1–8.
Shimura M, Yasuda K, Nakazawa T, Abe T, Shiono T, Iida T, et al. Panretinal photocoagulation induces pro-inflammatory cytokines and macular thickening in high-risk proliferative diabetic retinopathy. Graefe’s Arch Clin Exp Ophthalmol. 2009;247:1617–24.
Simó R, Stitt AW, Gardner TW. Neurodegeneration in diabetic retinopathy: does it really matter? Diabetologia. 2018;61:1902–12.
Scholl S, Kirchhof J, Augustin AJ. Pathophysiology of macular edema. Ophthalmologica. 2010;224:8–15.
Sidorczuk P, Obuchowska I, Konopinska J, Dmuchowska DA. Correlation between choroidal vascularity index and outer retina in patients with diabetic retinopathy. J Clin Med. 2022;11:3882.
Sim DA, Keane PA, Fung S, Karampelas M, Sadda SR, Fruttiger M, et al. Quantitative analysis of diabetic macular ischemia using optical coherence tomography. Investig Ophthalmol Vis Sci. 2014;55:417–23.
Paulus YM, Jain A, Gariano RF, Stanzel BV, Marmor M, Blumenkranz MS, et al. Healing of retinal photocoagulation lesions. Investig Ophthalmol Vis Sci. 2008;49:5540–5.
Zhang Z, Meng X, Wu Z, Zou W, Zhang J, Zhu D, et al. Changes in choroidal thickness after panretinal photocoagulation for diabetic retinopathy: a 12-week longitudinal study. Investig Ophthalmol Vis Sci. 2015;56:2631–8.
Mitsch C, Pemp B, Kriechbaum K, Bolz M, Scholda C, Schmidt-Erfurth U. Retinal morphometry changes measured with spectral domain-optical coherence tomography after pan-retinal photocoagulation in patients with proliferative diabetic retinopathy. Retina. 2016;36:1162–9.
Lascaratos G, Ji D, Wood JP, Osborne NN. Visible light affects mitochondrial function and induces neuronal death in retinal cell cultures. Vis Res. 2007;47:1191–201.
Sparrow JR, Zhou J, Ben-Shabat S, Vollmer H, Itagaki Y, Nakanishi K. Involvement of oxidative mechanisms in blue-light-induced damage to A2E-laden RPE. Investig Ophthalmol Vis Sci. 2002;43:1222–7.
Sandbach JM, Coscun PE, Grossniklaus HE, Kokoszka JE, Newman NJ, Wallace DC. Ocular pathology in mitochondrial superoxide dismutase (Sod2)–deficient mice. Investig Ophthalmol Vis Sci. 2001;42:2173–8.
Kortuem K, Geiger LK, Levin LA. Differential susceptibility of retinal ganglion cells to reactive oxygen species. Investig Ophthalmol Vis Sci. 2000;41:3176–82.
Jackson GR, Barber AJ. Visual dysfunction associated with diabetic retinopathy. Curr Diab Rep. 2010;10:380–4.
Trento M, Durando O, Lavecchia S, Charrier L, Cavallo F, Costa MA, et al. Vision related quality of life in patients with type 2 diabetes in the EUROCONDOR trial. Endocrine. 2017;57:83–88.
Zhao H, Zhou L, Lai K, Yu M, Huang C, Xu F, et al. Comparison of functional changes of retina after subthreshold and threshold pan-retinal photocoagulation in severe non-proliferative diabetic retinopathy. Lasers Med Sci. 2022;37:3561–9.
Funding
This work was supported by Chungnam National University.
Author information
Authors and Affiliations
Contributions
Contributions to Authors in each of these areas Design and conduct of the study (MWL); Collection of data (MWL and JTK); Analysis and interpretation of data (MWL, JTK, and JYS); Writing the article (MWL, JTK, and JYS); Critical revision of the article (MWL, JTK, and JYS); Final approval of the article (MWL, JTK, and JYS).
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self−archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sung, JY., Kim, JT. & Lee, MW. Longitudinal changes in each retinal layer thickness in diabetic retinopathy patients treated with pan-retinal photocoagulation. Eye 40, 376–382 (2026). https://doi.org/10.1038/s41433-025-04173-7
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41433-025-04173-7