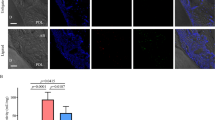
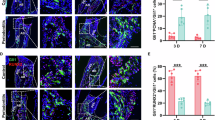
Abstract
Excessive neutrophil extracellular traps (NETs) induce an intense inflammatory response in periodontitis. Recently, Tregs were shown to be essential for attenuating inflammation-driven bone resorption. However, the regulation of Tregs differentiation by NETs in periodontitis is still unclear and needs further investigation. In this study, a murine experimental periodontitis model was established either without or with NETs depletion via DNase I. Firstly, we revealed that NETs accumulated significantly in both periodontal tissues and sera of mice models with periodontitis, while the depletion of NETs alleviated alveolar bone resorption. Moreover, RNA sequencing and bioinformatics analysis revealed that NETs depletion regulated the immune response of gingival tissue, especially affecting T-cell differentiation, and identified potential regulatory pathways. Subsequently, we verified that inhibition of NETs promoted the infiltration of Tregs and increased expression levels of IL-10 and TGF-β in periodontal tissue. Furthermore, in vitro studies demonstrated that NETs produced by P. g-LPS-stimulated neutrophils impeded the differentiation of co-cultured naive CD4+ T cells into Tregs, which could be restored by DNase I-mediated digestion of NETs. In conclusion, excessive NETs could exacerbate alveolar bone resorption in periodontitis by interfering with the differentiation of Tregs, and DNase I provides a novel targeted strategy for the immunotherapy of periodontitis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 6 digital issues and online access to articles
$119.00 per year
only $19.83 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
All data analyzed during this study are included in the published article as well as the supplemental files online. Further enquiries are available by contacting the corresponding author upon reasonable request.
References
Hajishengallis G, Chavakis T. Local and systemic mechanisms linking periodontal disease and inflammatory comorbidities. Nat Rev Immunol. 2021;21:426–40.
Silva LM, Brenchley L, Moutsopoulos NM. Primary immunodeficiencies reveal the essential role of tissue neutrophils in periodontitis. Immunol Rev. 2019;287:226–35.
Khoury W, Glogauer J, Tenenbaum HC, Glogauer M. Oral inflammatory load: Neutrophils as oral health biomarkers. J Periodontal Res. 2020;55:594–601.
Hajishengallis G. New developments in neutrophil biology and periodontitis. Periodontology. 2020;82:78–92.
Thiam HR, Wong SL, Wagner DD, Waterman CM. Cellular mechanisms of NETosis. Annu Rev Cell Dev Biol. 2020;36:191–218.
Brinkmann V, Reichard U, Goosmann C, Fauler B, Uhlemann Y, Weiss DS, et al. Neutrophil extracellular traps kill bacteria. Science. 2004;303:1532–5.
Souza FW, Miao EA. Neutrophils only die twice. Sci Adv. 2023;9:eadm8715.
Ebersole JL, Dawson D 3rd, Emecen-Huja P, Nagarajan R, Howard K, Grady ME, et al. The periodontal war: microbes and immunity. Periodontology. 2017;75:52–115.
Chapple ILC, Hirschfeld J, Kantarci A, Wilensky A, Shapira L. The role of the host-neutrophil biology. Periodontology. 2023;00:1–47.
Silva LM, Doyle AD, Greenwell-Wild T, Dutzan N, Tran CL, Abusleme L, et al. Fibrin is a critical regulator of neutrophil effector function at the oral mucosal barrier. Science. 2021;374:eabl5450.
Bonilha CS, Veras FP, de Queiroz Cunha F. NET-targeted therapy: effects, limitations, and potential strategies to enhance treatment efficacy. Trends Pharm Sci. 2023;44:622–34.
O’Neil LJ, Barrera-Vargas A, Sandoval-Heglund D, Merayo-Chalico J, Aguirre-Aguilar E, Aponte AM, et al. Neutrophil-mediated carbamylation promotes articular damage in rheumatoid arthritis. Sci Adv. 2020;6:eabd2688.
Xia Y, Cheng T, Zhang C, Zhou M, Hu Z, Kang F, et al. Human bone marrow mesenchymal stem cell-derived extracellular vesicles restore Th17/Treg homeostasis in periodontitis via miR-1246. FASEB J. 2023;37:e23226.
Deng J, Lu C, Zhao Q, Chen K, Ma S, Li Z. The Th17/Treg cell balance: crosstalk among the immune system, bone and microbes in periodontitis. J Periodontal Res. 2022;57:246–55.
Miron RJ, Bohner M, Zhang Y, Bosshardt DD. Osteoinduction and osteoimmunology: emerging concepts. Periodontology. 2024;94:9–26.
Kim TS, Silva LM, Theofilou VI, Greenwell-Wild T, Li L, Williams DW, et al. Neutrophil extracellular traps and extracellular histones potentiate IL-17 inflammation in periodontitis. J Exp Med. 2023;220:1–15.
Wang H, Zhang H, Wang Y, Brown ZJ, Xia Y, Huang Z, et al. Regulatory T-cell and neutrophil extracellular trap interaction contributes to carcinogenesis in non-alcoholic steatohepatitis. J Hepatol. 2021;75:1271–83.
Lambert S, Hambro CA, Johnston A, Stuart PE, Tsoi LC, Nair RP, et al. Neutrophil extracellular traps induce human Th17 cells: effect of psoriasis-associated TRAF3IP2 genotype. J Investig Dermatol Symp Proc. 2019;139:1245–53.
Mikami N, Kawakami R, Chen KY, Sugimoto A, Ohkura N, Sakaguchi S. Epigenetic conversion of conventional T cells into regulatory T cells by CD28 signal deprivation. Proc Natl Acad Sci USA. 2020;117:12258–68.
Marchesan J, Girnary MS, Jing L, Miao MZ, Zhang S, Sun L, et al. An experimental murine model to study periodontitis. Nat Protoc. 2018;13:2247–67.
Zhou B, Zhang M, Ma H, Wang Y, Qiu J, Liu Y, et al. Distinct palmitoylation of Foxp3 regulates the function of regulatory T cells via palmitoyltransferases. Cell Mol Immunol. 2024;21:787–9.
Li MO, Rudensky AY. T cell receptor signalling in the control of regulatory T cell differentiation and function. Nat Rev Immunol. 2016;16:220–33.
Wan MC, Jiao K, Zhu YN, Wan QQ, Zhang YP, Niu LZ. et al. Bacteria-mediated resistance of neutrophil extracellular traps to enzymatic degradation drives the formation of dental calculi. Nat Biomed Eng. 2024;8:1503
Liu B, Deng Y, Duan Z, Chu C, Wang X, Yang C, et al. Neutrophil extracellular traps promote intestinal barrier dysfunction by regulating macrophage polarization during trauma/hemorrhagic shock via the TGF-β signaling pathway. Cell Signal. 2024;113:110941.
Chen W, Chen H, Yang ZT, Mao EQ, Chen Y, Chen EZ. Free fatty acids-induced neutrophil extracellular traps lead to dendritic cells activation and T cell differentiation in acute lung injury. Aging. 2021;13:26148–60.
Hajishengallis G. Illuminating the oral microbiome and its host interactions: animal models of disease. FEMS Microbiol Rev. 2023;47:fuad018
Xiao E, Mattos M, Vieira GHA, Chen S, Corrêa JD, Wu Y, et al. Diabetes enhances IL-17 expression and alters the oral microbiome to increase its pathogenicity. Cell Host Microbe. 2017;22:120–8.e4.
Kimura S, Nagai A, Onitsuka T, Koga T, Fujiwara T, Kaya H, et al. Induction of experimental periodontitis in mice with Porphyromonas gingivalis-adhered ligatures. J Periodontol. 2000;71:1167–73.
Tsukasaki M, Takayanagi H. Osteoimmunology: evolving concepts in bone–immune interactions in health and disease. Nat Rev Immunol. 2019;19:626–42.
Jia L, Jiang Y, Wu L, Fu J, Du J, Luo Z, et al. Porphyromonas gingivalis aggravates colitis via a gut microbiota-linoleic acid metabolism-Th17/Treg cell balance axis. Nat Commun. 2024;15:1617.
Oliveira SR, de Arruda JAA, Schneider AH, Bemquerer LM, de Souza RMS, Barbim P, et al. Neutrophil extracellular traps in rheumatoid arthritis and periodontitis: Contribution of PADI4 gene polymorphisms. J Clin Periodontol. 2023;51:e13921.
Onuora S. NETs implicated in periodontitis-associated bone loss. Nat Rev Rheumatol. 2023;19:463.
Schneider AH, Taira TM, Públio GA, da Silva Prado D, Donate Yabuta PB, Dos Santos JC, et al. Neutrophil extracellular traps mediate bone erosion in rheumatoid arthritis by enhancing RANKL-induced osteoclastogenesis. Br J Pharm. 2024;181:429–46.
Guilherme Neto JL, Rodrigues Venturini LG, Schneider AH, Taira TM, Duffles Rodrigues LF, Veras FP, et al. Neutrophil extracellular traps aggravate apical periodontitis by stimulating osteoclast formation. J Endod. 2023;49:1514–21.
Zou H, Zhou N, Huang Y, Luo A, Sun J. Phenotypes, roles, and modulation of regulatory lymphocytes in periodontitis and its associated systemic diseases. J Leukoc Biol. 2022;111:451–67.
Hascoët E, Blanchard F, Blin-Wakkach C, Guicheux J, Lesclous P, Cloitre A. New insights into inflammatory osteoclast precursors as therapeutic targets for rheumatoid arthritis and periodontitis. Bone Res. 2023;11:26.
Liu Z, Chen X, Zhang Z, Zhang X, Saunders L, Zhou Y, et al. Nanofibrous spongy microspheres to distinctly release miRNA and growth factors to enrich regulatory T cells and rescue periodontal bone loss. ACS Nano. 2018;12:9785–99.
Ha DY, Jung JS, Choi GH, Ji S. Polarization of human gingival fibroblasts by Th1-, Th2-, Th17-, and Treg-derived cytokines. J Periodontal Res. 2022;57:487–501.
Tsukasaki M, Komatsu N, Nagashima K, Nitta T, Pluemsakunthai W, Shukunami C, et al. Host defense against oral microbiota by bone-damaging T cells. Nat Commun. 2018;9:701.
Liu, Hayashi M, Ohsugi Y, Katagiri S, Akira S, Iwata T, et al. The IL-33/ST2 axis is protective against acute inflammation during the course of periodontitis. Nat Commun. 2024;15:2707.
Qin X, Liu JY, Wang T, Pashley DH, Al-Hashim AH, Abdelsayed R, et al. Role of indoleamine 2,3-dioxygenase in an inflammatory model of murine gingiva. J Periodontal Res. 2017;52:107–13.
Breedveld A, Groot Kormelink T, van Egmond M, de Jong EC. Granulocytes as modulators of dendritic cell function. J Leukoc Biol. 2017;102:1003–16.
Wilson AS, Randall KL, Pettitt JA, Ellyard JI, Blumenthal A, Enders A, et al. Neutrophil extracellular traps and their histones promote Th17 cell differentiation directly via TLR2. Nat Commun. 2022;13:1–12.
Esberg A, Kindstedt E, Isehed C, Lindquist S, Holmlund A, Lundberg P. LIGHT protein: A novel gingival crevicular fluid biomarker associated with increased probing depth after periodontal surgery. J Clin Periodontol. 2024;51:852–62.
Minns D, Smith KJ, Alessandrini V, Hardisty G, Melrose L, Jackson-Jones L, et al. The neutrophil antimicrobial peptide cathelicidin promotes Th17 differentiation. Nat Commun. 2021;12:1285.
Apolinário Vieira GH, Aparecida Rivas AC, Figueiredo Costa K, Ferreira Oliveira LF, Tanaka Suzuki K, Reis Messora M, et al. Specific inhibition of IL-6 receptor attenuates inflammatory bone loss in experimental periodontitis. J Periodontol. 2021;92:1460–9.
Piao W, Lee ZL, Zapas G, Wu L, Jewell CM, Abdi R. et al. Regulatory T cell and endothelial cell crosstalk. Nat Rev Immunol. 2025;25:588–607.
Ohl L, Mohaupt M, Czeloth N, Hintzen G, Kiafard Z, Zwirner J, et al. CCR7 governs skin dendritic cell migration under inflammatory and steady-state conditions. Immunity. 2004;21:279–88.
Davalos-Misslitz AC, Rieckenberg J, Willenzon S, Worbs T, Kremmer E, Bernhardt G, et al. Generalized multi-organ autoimmunity in CCR7-deficient mice. Eur J Immunol. 2007;37:613–22.
Chen Y, Liang R, Shi X, Shen R, Liu L, Liu Y, et al. Targeting kinase ITK treats autoimmune arthritis via orchestrating T cell differentiation and function. Biomed Pharmacother. 2023;169:115886.
Stakos D, Skendros P, Konstantinides S, Ritis K. Traps N’ Clots: NET-mediated thrombosis and related diseases. Thromb Haemost. 2020;120:373–83.
Hu Y, Wang H, Liu Y. NETosis: sculpting tumor metastasis and immunotherapy. Immunol Rev. 2024;321:263–79.
Jain S, Darveau RP. Contribution of Porphyromonas gingivalis lipopolysaccharide to periodontitis. Periodontology. 2010;54:53–70.
Herath TD, Darveau RP, Seneviratne CJ, Wang CY, Wang Y, Jin L. Tetra- and penta-acylated lipid A structures of Porphyromonas gingivalis LPS differentially activate TLR4-mediated NF-κB signal transduction cascade and immuno-inflammatory response in human gingival fibroblasts. PloS One. 2013;8:e58496.
de Bont CM, Koopman WJH, Boelens WC, Pruijn GJM. Stimulus-dependent chromatin dynamics, citrullination, calcium signalling and ROS production during NET formation. Biochim Biophys Acta Mol Cell Res. 2018;1865:1621–9.
Tamura K, Miyato H, Kanamaru R, Sadatomo A, Takahashi K, Ohzawa H, et al. Activated neutrophils inhibit chemotactic migration of activated T lymphocytes to CXCL11 by multiple mechanisms. Cell Immunol. 2023;384:104663.
Funding
This study was supported by National Natural Science Foundation of China (82470956, 82001008); Shanghai Municipal Health Commission (JKKPZX-2023-A07); Chinese Stomatological Association (Young Talent Project of Orthodontics, COS-B202105); China Oral Health Foundation (A2023-029); Fundamental Research Funds for the Central Universities (22120240282) to CL.
Author information
Authors and Affiliations
Contributions
BBC and CSL conceived the study. BBC, DNS, CSL and YML designed the project and experiment. BBC and DNS collected samples and performed the experiment. BBC wrote the manuscript and prepared the figures. BBC, CSL, YML, TFC and LJJ revised the manuscript. All authors agreed to publish the final version.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
The studies involving all animal experiments were reviewed and approved from the Ethics Committee of Stomatology Hospital, Tongji University (Approval No. 2022-DW-013). All methods were performed in accordance with the relevant guidelines and regulations.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chen, B., Song, D., Cheng, T. et al. Neutrophil extracellular traps aggravate periodontitis by disturbing regulatory T-cell differentiation. Genes Immun 26, 475–485 (2025). https://doi.org/10.1038/s41435-025-00350-6
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41435-025-00350-6