Abstract
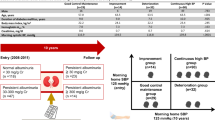
Our previous study showed that the morning systolic blood pressure target should be <120 mmHg to prevent the onset or progression of diabetic nephropathy in patients with type 2 diabetes. In this study, we examined the prognostic values of home and clinical blood pressure for first cardiovascular events in the same cohort. Morning and evening home blood pressure measurements were obtained in triplicate for 14 consecutive days from the beginning of the study in a retrospective cohort of 1081 type 2 diabetes patients (44.5% women; median age 66.0 years) with no history of macrovascular complications. The first major cardiovascular event was the primary endpoint; the risk was examined by the Cox proportional hazards model. After a mean follow-up of 6.63 years, first-time cardiovascular events occurred in 119 patients (incidence, 16.6/1000 patient-years). Baseline morning systolic blood pressure (hazard ratio: 1.14, 95% CI 1.01–1.28) significantly predicted cardiovascular events, whereas clinical blood pressure did not. The adjusted hazard ratio (95% CI) for the incidence of cardiovascular events in patients with morning systolic blood pressure ≥135 mmHg tended to be higher than that in those with morning systolic blood pressure <125 mmHg [1.67 (0.94–2.97)]. Elevated home blood pressure measurement is a predictor of future cardiovascular events in type 2 diabetes patients and may be superior to clinical blood pressure measurement in this regard.
Similar content being viewed by others
Introduction
Type 2 diabetes is a complex and heterogeneous metabolic disease requiring continuous medical care and comprehensive and multifactorial strategies to reduce cardiovascular risk. Patients with type 2 diabetes have a higher cardiovascular morbidity and mortality than individuals in the general population [1]. It has been reported that patients with type 2 diabetes and no previous history of cardiovascular disease have a similar risk for cardiovascular events as nondiabetic patients with previous myocardial infarction [2]. However, subsequent studies have revealed that patients with type 2 diabetes with risk-related variables, including blood pressure (BP), within target ranges appeared to have little or no excess cardiovascular risk compared with that among the general population [3]. This highlights the necessity for a multivariate approach for the prevention of cardiovascular disease in patients with type 2 diabetes.
The importance of adequate BP control in the context of cardiovascular disease has been well documented [4]. Moreover, compared to clinical BP measurement, home BP (HBP) measurement has been shown to have better predictive power for target organ damage. In addition, HBP-based BP treatment was associated with a significantly larger reduction in the average systolic BP than clinical BP-based BP treatment [5]. We have previously reported results from the KAMOGAWA-HBP cohort demonstrating that the preferable morning systolic BP target should be <120 mmHg for preventing the onset or progression of diabetic nephropathy in patients with type 2 diabetes [6]. The objective of the present study was to examine the prognostic values of home and clinical BP measurements for the first onset of cardiovascular events using the same cohort as that mentioned above.
Methods
Study design
This retrospective cohort study was based on data from a cohort of patients with type 2 diabetes who had been participating in an HBP measurement program since 2008 and regularly attended the diabetes outpatient clinical at the Kyoto Prefectural University of Medicine Hospital or one of four other general hospitals, all of which are located in the Kansai region of Japan (KAMOGAWA-HBP study). The details of the study project have been previously described [7, 8]. All procedures were approved by the local Research Ethics Committee and were conducted in accordance with the Declaration of Helsinki. Informed consent was obtained from all patients.
Patients
We sequentially recruited 1473 patients with type 2 diabetes with a minimum age of 20 years who visited the facilities beginning in March 2008. We collected follow-up data until December 2018. There was no BP-level criterion for study inclusion. We excluded patients who failed to measure their HBP. To examine the risk of first-onset cardiovascular events, 388 patients who had a previous history of cardiovascular events, defined as the presence of previous cardiovascular disease, cerebrovascular disease, or arteriosclerosis obliterans based on clinical history or physical examination, were further excluded from the present analysis. Therefore, the study population comprised 1081 patients (577 male, 504 female). The diagnosis of type 2 diabetes was based on the American Diabetes Association criteria [9].
BP measurements
BP was measured using HEM-70801C automatic BP measuring devices (Omron Healthcare Co., Ltd, Kyoto, Japan) using the cuff-oscillometric principle, which generates a digital display of systolic and diastolic BP and heart rate values. The HEM-70801C employs identical components and an identical BP-determining algorithm as the HEM-705IT, which was previously used to validate the accuracy and satisfied the criteria of the British Hypertension Society protocol [10]. Study patients were seated at rest for ≥2 min, and then clinical BP was measured three times consecutively by physicians or well-trained nurses. The clinical BP of each patient was the average of three clinical BP readings. Patients were taught how to measure their HBP. After their ability to measure HBP was verified, patients were asked to perform triplicate morning and evening BP measurements with at least 1 min between recordings for 14 consecutive days while they were in a sitting position after ≥2 min of rest. They were instructed to perform the morning measurements of BP within 1 h of waking and before eating breakfast or taking any drugs, and they were instructed to perform the evening measurements of BP in a similar fashion just before going to bed (eating was prohibited for over 1 h before measurements) [11]. We calculated the mean of three measurements per morning and three measurements per evening each day, and then the level of HBP was computed from those 14 means per patient. Patients did not need to keep a diary of the record of the values measured because the BP device was equipped with a memory to store the date, time, and readings. We used the electronic readout from the device for our analysis. These procedures were described in detail in our previous report [7, 8].
Data collection
Blood samples were taken in the morning for biochemical measurements at the time of study entry. Each patient’s characteristics, including age, sex, body mass index, duration of diabetes, smoking status, alcohol consumption status, antihypertensive medication, antidiabetic medication, and past history of cardiovascular events, were obtained from the initial questionnaire survey and regular check-up results concurrently with HBP measurements. The details of the methodology of the study have been previously published [7, 8]. Nephropathy was defined as urinary albumin excretion greater than 30 mg/g Cr. Retinopathy was assessed from chart reviews. Neuropathy was defined by the diagnostic criteria for diabetic neuropathy proposed by the Diagnostic Neuropathy Study Group [12].
Definitions
We coded endpoints according to the tenth revision of the International Classification of Diseases (ICD-10). In the present study, a composite cardiovascular endpoint was defined based on the patient’s clinical history or physical examination and included cardiovascular death (ICD-10 codes I00 to I99), angina pectoris (I20), nonfatal myocardial infarction (I21), nonfatal stroke (I60, I61, and I63), occlusion and stenosis of unspecified carotid artery (I65), transient ischemic attack (G45), arteriosclerosis obliterans (I73) and heart failure (I50). Moreover, hard major adverse cardiac events (MACEs) included a composite of cardiovascular death, nonfatal myocardial infarction, and nonfatal stroke. We considered only the first event in the individual when assessing outcome results.
Statistical analysis
Baseline characteristics are summarized by medians with interquartile ranges or numbers with proportions. The risk of the first event was examined by the Cox proportional hazards model. The dependent variable was the number of months from study entry to the event or censoring for survivors until December 2018. In addition to the unadjusted model (Model 1), the multivariate model (Model 2), adjusted for sex, duration of diabetes mellitus, hemoglobin A1c, low-density lipoprotein cholesterol, smoking status, and antihypertensive medication [13], was established. To express the change in risk associated with a 10 mmHg increase in HBP measurements or clinical BP measurements, we derived standardized hazard ratios (HRs) from Cox regression. Moreover, we conducted categorical analysis with patients classified into three groups according to the following criteria: systolic BP (SBP) <125 mmHg, 125–135 mmHg, and ≥135 mmHg.
We performed subgroup analyses according to BP (SBP <135 mmHg and ≥ 135 mmHg).
The SPSS statistical package, version 11.0 J (SPSS, Inc., Chicago, IL) and JMP version 11.2.0 software (SAS Institute Inc., Cary, North Carolina) were used for statistical analyses, and P values < 0.05 (two-sided) were considered statistically significant.
Results
The patients were followed up for a median of 7.0 (interquartile 4.3–9.0) years, with a maximum follow-up of 10.6 years. The clinical characteristics of the patients at baseline are shown in Table 1. The median age and hemoglobin A1c were 65.0 (IQR: 58.0–72.0) years and 7.1 (IQR: 6.6–7.8) % [54.1 (IQR: 48.6–61.7) mmol/mol], respectively. The median number of days of morning and evening BP measurements per patient during the 14 consecutive days were 13.0 (IQR: 12.0–14.0) and 12.0 (IQR: 12.0–14.0), respectively. The total median measurements of morning and evening BP per patient during the 14 consecutive days were 39.0 (IQR: 36.0–42.0) and 36.0 (IQR: 36.0–42.0), respectively.
We observed 119 incident cardiovascular events among the 1081 patients: 5 (4%) cardiovascular deaths, 42 (35%) cases of angina pectoris, 17 (14%) nonfatal myocardial infarctions, 37 (31%) nonfatal strokes, 3 (2.5%) cases of occlusion and stenosis of precerebral arteries, 2 (1.7%) transient ischemic attacks, 11 (9.2%) cases of arteriosclerosis obliterans and 2 (1.7%) cases of heart failure.
Among patients with morning SBP ≥135 mmHg, 72 (14.3%) out of 503 patients had cardiovascular events, whereas 47 (8.1%) out of 578 patients with morning SBP <135 mmHg had cardiovascular events.
The unadjusted HRs (95% CI), which expresses the risk associated with a 10 mmHg increase in BP, of morning SBP, evening SBP and clinic SBP for cardiovascular events were 1.21 (1.11–1.33), 1.11 (1.00–1.22), and 1.12 (1.02–1.22), respectively. The adjusted HR (95% CI) of morning SBP for cardiovascular events was 1.14 (1.01–1.28) (Table 2). The unadjusted HR for cardiovascular events in patients with morning SBP ≥135 mmHg was significantly higher than that in those with morning SBP <125 mmHg [2.31 (95% CI: 1.42–3.77)]. The adjusted HR for cardiovascular events in patients with morning SBP ≥135 mmHg was higher than that in those with morning SBP <125 mmHg, although this difference was not significant [1.67 (95% CI: 0.94–2.97)] (Table 3). On the other hand, the unadjusted HRs (95% CIs) of morning SBP, evening SBP and clinical SBP for hard MACEs were 1.24 (1.09–1.40), 1.15 (1.00–1.31), and 1.07 (0.93–1.21), respectively. In the adjusted model, there was no significant relationship between BP variables and hard MACEs.
The adjusted HR of morning SBP for cardiovascular events was 1.40 (95% CI: 0.94–2.08) in patients with morning SBP <135 mmHg and 1.09 (95% CI: 0.87–1.37) in patients with morning SBP ≥135 mmHg.
Discussion
Principal findings
This prospective cohort study of 1081 patients with type 2 diabetes addressed for the first time the prognostic value of HBP for cardiovascular events. The primary finding of our study was that every 10 mmHg increase in morning HBP increased the risk of first-onset cardiovascular events by 14%, and the HR for first-onset cardiovascular events in patients with morning SBP ≥135 mmHg tended to be higher than that in those with morning SBP <125 mmHg.
Interpretations
Our study showed that lower SBP was associated with lower risks of cardiovascular outcomes, which is in accordance with previous studies [14, 15]. In the Tanno–Sobetsu Cho Study, the rate of cardiovascular death was significantly higher in the group with an SBP of ≥130 mmHg and a diastolic BP of ≥80 mmHg than in the group with a BP of <120/80 mmHg in patients with borderline diabetes and diabetes, which also showed the predictive value of BP [16]. Increased BP due to arterial stiffness contributes to cardiovascular events. The Systolic Blood Pressure Intervention Trial, which was an interventional study and compared the benefit of treatment of SBP to a target of <120 mmHg with treatment to a target of <140 mmHg among patients at high risk for cardiovascular events but without diabetes, showed that targeting an SBP of <120 mmHg resulted in lower rates of cardiovascular events and death [17]. On the other hand, in the Action to Control Cardiovascular Risk in Diabetes (ACCORD) BP trial, which was also an interventional study and examined whether intensive control of BP might reduce the risk of cardiovascular events and death compared with the risk associated with standard treatment in patients with type 2 diabetes, an intensive BP control strategy to achieve SBP <120 mmHg did not significantly reduce the composite of cardiovascular events and death compared with the outcome associated with a standard SBP control goal of <140 mmHg [18]. An alternative explanation for the apparent discordance between our study and the ACCORD BP findings may be their study design. The ACCORD BP trial was a double two-by-two factorial trial in which the mean SBP and DBP of the participants at baseline were 139.2 mm Hg and 76.0 mm Hg, respectively, and participants were randomly assigned to either intensive or standard BP control. Our study, on the other hand, was an observational study conducted to examine the optimal HBP, assessed by a baseline measurement, to prevent first-onset cardiovascular events. This might suggest the importance of early intervention. Moreover, BP was assessed as clinical BP in the ACCORD BP trial, whereas it was assessed as HBP in our study. Last, the difference in disease structure might have influenced the study results. The incidence of stroke is higher in Japan than in the United States. In ACCORD-BP, the incidence of cerebral infarction was approximately one-quarter that of myocardial infarction, whereas in the present study, there were three cerebral infarction cases for every five cases of angina pectoris or myocardial infarction combined. Meta-analyses of randomized trials showed that no single optimal SBP level can be identified in patients with type 2 diabetes or impaired fasting glucose/impaired glucose tolerance [19]. An intensive BP control goal of <135 mmHg was associated with a significant reduction in all-cause mortality and stroke compared with a standard BP control goal of <140 mmHg. The continued stroke risk reduction of an SBP <120 mm Hg was shown, but there was no benefit regarding the risk of cardiac, renal or retinal outcomes. In the Japan Diabetes Outcome Intervention Trial (J-DOIT)3 [20], which investigated whether stricter control than the current targets in the Japanese guidelines for blood glucose, BP, and lipids could efficiently and safely reduce cardiovascular disease risk in patients with type 2 diabetes, the achieved SBP values were 123 mmHg in the intensive therapy group and 129 mmHg in the conventional therapy group. In the intensive therapy group, strokes decreased by 58% and coronary artery disease decreased by 14% compared to the conventional therapy group during the intervention, and the incidence of stroke decreased significantly with decreasing SBP.
Regarding BP control based on HBP, strict antihypertensive therapy with an HBP target of <125/75 mmHg was shown to be effective from the viewpoint of the prevention of target organ damage in patients with impaired glucose tolerance or diabetes [21]. The Hypertension Objective Treatment Based on Measurement by Electrical Devices of Blood Pressure trial also showed that the risk of cardiovascular disease tended to increase in patients with an HBP ≥125/75 mmHg in hypertensive patients with impaired glucose tolerance or diabetes [22]. Our study also showed that patients with home SBP <125 mmHg had a significantly lower risk of first-onset cardiovascular events, supporting the 2019 JSH Guidelines for the Management of Hypertension [23], in which the target of HBP control in patients with diabetes was set to <125/75 mmHg. In the present study, clinical BP, which is often affected by psychological stress, did not predict cardiovascular events.
We previously reported that the risk for the onset or progression of diabetic nephropathy in 851 patients with morning SBP ≥120 mmHg was significantly higher than that in those with morning SBP <120 mmHg in a retrospective 2-year study of the KAMOGAWA-HBP cohort [6]. The BP value for preventing the progression of nephropathy is lower than that shown in the present study for the prevention of cardiovascular events. Appel et al. reported that intensive BP control significantly retarded disease progression in patients with baseline proteinuria, according to the primary outcome (a doubling of the serum creatinine level, end-stage renal disease, or death); on the other hand, intensive BP control had no significant effect among patients without proteinuria [24]. It might be that strict BP control is desirable for hyperfiltration conditions such as diabetic nephropathy.
In the present study, there was no relationship between BP variables and hard MACEs. We observed 59 incident cases of hard MACEs among the 1081 patients, which may limit the statistical power. To make valid assessments concerning the prognostic significance of BP variables for hard MACEs, a long-term follow-up study or a validation study is desirable.
In this study, the adjusted HR of morning SBP for cardiovascular events was higher in patients with morning SBP <135 mmHg than that in patients with morning SBP ≥135 mmHg. In this cohort, the adjusted HR for cardiovascular events in patients with morning SBP ≥130 mmHg was significantly higher than that in those with morning SBP <130 mmHg [1.69 (95% CI: 1.03–2.78)]; therefore, patients with morning SBP ≥135 mmHg composed a high-risk group for cardiovascular events, and the HR associated with a 10 mmHg increase in morning SBP was low in patients with morning SBP ≥135 mmHg.
The strength of this study was that it was retrospective by design and that the device used was equipped to store measurements, so poor adherence to patient logbooks was avoided [25]. In addition, the HBP measurements were performed for 14 consecutive days, which is a relatively long consecutive period.
Our study has several limitations that need to be mentioned. First, our study only assessed the association of a single baseline measurement of BP with cardiovascular events, as the objective of the study was to examine cardiovascular risk defined according to initial baseline BP and treatment status. Moreover, it has been reported that the association of cardiovascular risk was confirmed by BP at baseline or during follow-up, which might suggest that single baseline BP assessment was sufficient when the addition of subsequent values did not change the result remarkably [26]. Second, the generalizability of our results to other ethnic groups is uncertain because of the difference in disease structure explained by differential environmental and genetic risk factors. Last, morning measurements of BP were performed before taking antihypertensive drugs, and clinical BP measurements were performed after taking antihypertensive drugs. Therefore, antihypertensive drugs may have influenced the results in patients taking antihypertensive medication.
Perspectives
In conclusion, to the best of our knowledge, this retrospective cohort study of patients with type 2 diabetes revealed, for the first time, the prognostic values of morning and evening BP for cardiovascular events. The results may contribute to the development of guidelines and be valuable to clinicians involved in the management of patients with diabetes. HBP would be a better tool to assess cardiovascular risk associated with BP levels.
References
Rawshani A, Rawshani A, Franzén S, Eliasson B, Svensson AM, Miftaraj M, et al. Mortality and cardiovascular disease in type 1 and type 2 diabetes. N Engl J Med. 2017;376:1407–18.
Haffner SM, Lehto S, Rönnemaa T, Pyörälä K, Laakso M. Mortality from coronary heart disease in subjects with type 2 diabetes and in nondiabetic subjects with and without prior myocardial infarction. N Engl J Med. 1998;339:229–34.
Patel AV, Bernstein L, Deka A, Feigelson HS, Campbell PT, Gapstur SM, et al. Leisure time spent sitting in relation to total mortality in a prospective cohort of US adults. Am J Epidemiol. 2010;172:419–29.
Takashima N, Ohkubo T, Miura K, Okamura T, Murakami Y, Fujiyoshi A, et al. NIPPON DATA80 Research Group. Long-term risk of BP values above normal for cardiovascular mortality: a 24-year observation of Japanese aged 30 to 92 years. J Hypertens. 2012;30:2299–306.
Satoh M, Maeda T, Hoshide S, Ohkubo T. Is antihypertensive treatment based on home blood pressure recommended rather than that based on office blood pressure in adults with essential hypertension? (meta-analysis). Hypertens Res. 2019;42:807–16.
Ushigome E, Hamaguchi M, Matsumoto S, Oyabu C, Omoto A, Tanaka T, et al. Optimal home SBP targets for preventing the progression of diabetic nephropathy in patients with type 2 diabetes mellitus. J Hypertens. 2015;33:1853–9.
Ushigome E, Fukui M, Hamaguchi M, Senmaru T, Sakabe K, Tanaka M, et al. The coefficient variation of home blood pressure is a novel factor associated with macroalbuminuria in type 2 diabetes mellitus. Hypertens Res. 2011;34:1271–5.
Ushigome E, Matsumoto S, Oyabu C, Kitagawa N, Tanaka T, Hasegawa G, et al. Prognostic significance of day-by-day variability of home blood pressure on progression to macroalbuminuria in patients with diabetes. J Hypertens. 2018;36:1068–75.
Report of the expert committee on the diagnosis and classification of diabetes mellitus. Diabetes Care 2003;26(Suppl 1):S5–20.
Coleman A, Freeman P, Steel S, Shennan A. Validation of the Omron 705IT (HEM-759-E) oscillometric blood pressure monitoring device according to the British Hypertension Society protocol. Blood Press Monit. 2006;11:27–32.
Imai Y, Kario K, Shimada K, Kawano Y, Hasebe N, Matsuura H, et al. Japanese Society of Hypertension Committee for Guidelines for Self-monitoring of Blood Pressure at Home. The Japanese society of hypertension guidelines for self-monitoring of blood Pressure at home (second edition). Hypertens Res. 2012;35:777–95.
Yasuda H, Sanada M, Kitada K, Terashima T, Kim H, Sakaue Y, et al. Rationale and usefulness of newly devised abbreviated diagnostic criteria and staging for diabetic polyneuropathy. Diabetes Res Clin Pract. 2007;77(Suppl 1):S178–83.
Maruhashi T, Kinoshita Y, Kajikawa M, Kishimoto S, Matsui S, Hashimoto H, et al. Relationship between home blood pressure and vascular function in patients receiving antihypertensive drug treatment. Hypertens Res. 2019;42:1175–85.
Stevens RJ, Kothari V, Adler AI, Stratton IM. United Kingdom Prospective Diabetes Study (UKPDS) Group. The UKPDS risk engine: a model for the risk of coronary heart disease in type II diabetes (UKPDS 56). Clin Sci. 2001;101:671–9.
Rawshani A, Rawshani A, Franzén S, Sattar N, Eliasson B, Svensson A-M, et al. Risk factors, mortality, and cardiovascular outcomes in patients with type 2 diabetes. N Engl J Med. 2018;379:633–44.
Obara F, Saitoh S, Takagi S, Shimamoto K. Influence of Hypertension on the Incidence of Cardiovascular Disease in Two Rural Communities in Japan: The Tanno-Sobetsu [Corrected] Study. Hypertens Res. 2007;30:677–82.
SPRINT Research Group, Wright JT Jr, Williamson JD, Whelton PK, et al. A randomized trial of intensive versus standard blood-pressure control. N Engl J Med. 2015;373:2103–16.
ACCORD Study Group, Cushman WC, Evans GW, Byington RP, Goff DC Jr, Grimm RH Jr, et al. Effects of intensive blood-pressure control in type 2 diabetes mellitus. N Engl J Med. 2015;373:2103–16.
Bangalore S, Kumar S, Lobach I, Messerli FH. Blood pressure targets in subjects with type 2 diabetes mellitus/impaired fasting glucose: observations from traditional and bayesian random-effects meta-analyses of randomized trials. Circulation. 2011;123:2799–810.
Ueki K, Sasako T, Okazaki Y, Kato M, Okahata S, Katsuyama H, et al. J-DOIT3 Study Group. Effect of an intensified multifactorial intervention on cardiovascular outcomes and mortality in type 2 diabetes (J-DOIT3): an open-label, randomised controlled trial. Lancet Diabetes Endocrinol. 2017;5:951–64.
Eguchi K, Hoshide S, Ishikawa S, Shimada K, Kario K. Aggressive blood pressure-lowering therapy guided by home blood pressure monitoring improves target organ damage in hypertensive patients with type 2 diabetes/prediabetes. J Clin Hypertens. 2012;14:422–8.
Noguchi Y, Asayama K, Staessen JA, Inaba M, Ohkubo T, Hosaka M.HOMED-BP study group et al. Predictive power of home blood pressure and clinic blood pressure in hypertensive patients with impaired glucose metabolism and diabetes. J Hypertens. 2013;31:1593–602.
Umemura S, Arima H, Arima S, Asayama K, Dohi Y, Hirooka Y, et al. The Japanese society of hypertension guidelines for the management of hypertension (JSH 2019). Hypertens Res. 2019;42:1235–481.
Appel LJ, Wright JT, Jr., Greene T, Agodoa LY, Astor BC, Bakris GL. AASK Collaborative Research Group. et al. Intensive blood-pressure control in hypertensive chronic kidney disease. N Engl J Med. 2010;363:918–29 .
Matsumoto S, Fukui M, Hamaguchi M, Ushigome E, Matsushita K, Fukuda T, et al. Is home blood pressure reporting in patients with type 2 diabetes reliable? Hypertens Res. 2014;37:741–5.
Zhao W, Katzmarzyk PT, Horswell R, Wang Y, Li W, Johnson J, et al. Aggressive blood pressure control increases coronary heart disease risk among diabetic patients. Diabetes Care. 2013;36:3287–96.
Acknowledgements
We acknowledge Dr. Atsushi Omoto and Dr. Wataru Fukuda of Kyoto First Red Cross Hospital, Dr. Shin-ichi Mogami and Dr. Yoshihiro Kitagawa of Osaka General Hospital of West Japan Railway Company for their assistance in collecting data. We would like to thank Naoko Higo, Machiko Hasegawa and Terumi Kaneko at the Kyoto Prefectural University of Medicine for teaching the patients how to measure their BP and Sayoko Tanaka, also at the Kyoto Prefectural University of Medicine, for her secretarial assistance. We would like to thank Editage (www.editage.com) for English language editing.
Funding
EU received grant support from the Japanese Study Group for Physiology and Management of Blood Pressure and the Astellas Foundation for Research on Metabolic Disorders (Grant number: 4024).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
EU received grants from the Japan Society for the Promotion of Science and ONO PHARMACEUTICAL CO., LTD. and received personal fees from AstraZeneca plc; Astellas Pharma Inc.; DAIICHI SANKYO COMPANY, LIMITED; Kyowa Hakko Kirin Company Ltd.; KOWA PHARMACEUTICAL COMPANY LTD.; MSD K.K.; Mitsubishi Tanabe Pharma Corp.; Novo Nordisk Pharma Ltd.; Taisho Toyama Pharmaceutical Co., Ltd.; Takeda Pharmaceutical Co., Ltd.; Nippon Boehringer Ingelheim Co., Ltd.; Sumitomo Dainippon Pharma Co., Ltd.; and Johnson & Johnson K.K. outside the submitted work. Masahide Hamaguchi received grants from Asahi Kasei Pharma; Nippon Boehringer Ingelheim Co., Ltd.; Mitsubishi Tanabe Pharma Corporation; DAIICHI SANKYO COMPANY, LIMITED; Sanofi K.K.; Takeda Pharmaceutical Company Limited; Astellas Pharma Inc.; Kyowa Kirin Co., Ltd.; Sumitomo Dainippon Pharma Co., Ltd.; Novo Nordisk Pharma Ltd.; and Eli Lilly Japan K.K. Masahide Hamaguchi also received personal fees from ONO PHARMACEUTICAL CO., LTD.; AstraZeneca plc; KOWA PHARMACEUTICAL COMPANY LTD.; MSD K.K.; Mitsubishi Tanabe Pharma Corp.; and Sumitomo Dainippon Pharma Co., Ltd. outside the submitted work. Masahiro Yamazaki received personal fees from MSD K.K.; Sumitomo Dainippon Pharma Co., Ltd.; Kowa Company, Ltd.; AstraZeneca PLC; Takeda Pharmaceutical Company Ltd.; Kyowa Hakko Kirin Co., Ltd.; DAIICHI SANKYO COMPANY, LIMITED; KOWA PHARMACEUTICAL COMPANY LTD.; and ONO PHARMACEUTICAL CO., LTD. outside the submitted work. Michiaki Fukui received grants from the Japan Society for the Promotion of Science; Nippon Boehringer Ingelheim Co., Ltd.; KISSEI PHARMACEUTICAL CO., LTD.; Mitsubishi Tanabe Pharma Corporation; DAIICHI SANKYO COMPANY, LIMITED; Sanofi K.K.; Takeda Pharmaceutical Company Limited; Astellas Pharma Inc.; MSD K.K.; Kyowa Hakko Kirin Co., Ltd.; Sumitomo Dainippon Pharma Co., Ltd.; KOWA PHARMACEUTICAL COMPANY LTD.; Novo Nordisk Pharma Ltd.; ONO PHARMACEUTICAL CO., LTD.; SANWA KAGAKU KENKYUSHO CO., LTD.; Eli Lilly Japan K.K.; Taisho Pharmaceutical Co., Ltd.; TERUMO CORPORATION; TEIJIN PHARMA LIMITED; Nippon Chemiphar Co., Ltd.; and Johnson & Johnson k.k. Medical Company. Michiaki Fukui also received honoraria from Nippon Boehringer Ingelheim Co., Ltd.; KISSEI PHARMACEUTICAL CO., LTD.; Mitsubishi Tanabe Pharma Corporation; DAIICHI SANKYO COMPANY, LIMITED; Sanofi K.K.; Takeda Pharmaceutical Company Limited; Astellas Pharma Inc.; MSD K.K.; Kyowa Kirin Co., Ltd.; Sumitomo Dainippon Pharma Co., Ltd.; KOWA PHARMACEUTICAL COMPANY LTD.; Novo Nordisk Pharma Ltd.; ONO PHARMACEUTICAL CO., LTD.; SANWA KAGAKU KENKYUSHO CO., LTD.; Eli Lilly Japan K.K.; Taisho Pharmaceutical Co., Ltd.; Bayer Yakuhin, Ltd.; AstraZeneca K.K.; MOCHIDA PHARMACEUTICAL CO., LTD.; and Combi Corporation outside the submitted work. IY received speaking fees from Chugai Pharmaceutical Co., Ltd. and Japan Tobacco Inc. Pharmaceutical Division outside the submitted work. The sponsors were not involved in the study design; in the collection, analysis, or interpretation of the data; in the writing of this manuscript; or in the decision to submit the article for publication. The authors, their immediate families, and any research foundations with which they are affiliated have not received any financial payments or other benefits from any commercial entity related to the subject of this article. The authors declare that although they are affiliated with a department that is supported financially by a pharmaceutical company, the authors received no current funding for this study, and their department affiliation does not alter their adherence to all the full journal policies on sharing data and materials.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ushigome, E., Kitagawa, N., Kitagawa, N. et al. Predictive power of home blood pressure measurement for cardiovascular outcomes in patients with type 2 diabetes: KAMOGAWA-HBP study. Hypertens Res 44, 348–354 (2021). https://doi.org/10.1038/s41440-020-00584-z
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41440-020-00584-z
Keywords
This article is cited by
-
Impact of diabetic status and contribution of office and home blood pressure across diabetic status for cardiovascular disease: the J-HOP study
Hypertension Research (2023)
-
Home blood pressure measurement: the original and the best for predicting the risk
Hypertension Research (2023)
-
Lifetime home BP-centered approach is the core from onset to aggravation of hypertension
Hypertension Research (2023)
-
The clinical significance of home and office blood pressure in diabetic nephropathy
Hypertension Research (2023)
-
Annual reports on hypertension research 2020
Hypertension Research (2022)