Fig. 1: Design of light-induced PANAC conjugation enables modular functionalization of small molecules and native biomolecules in temporal control.
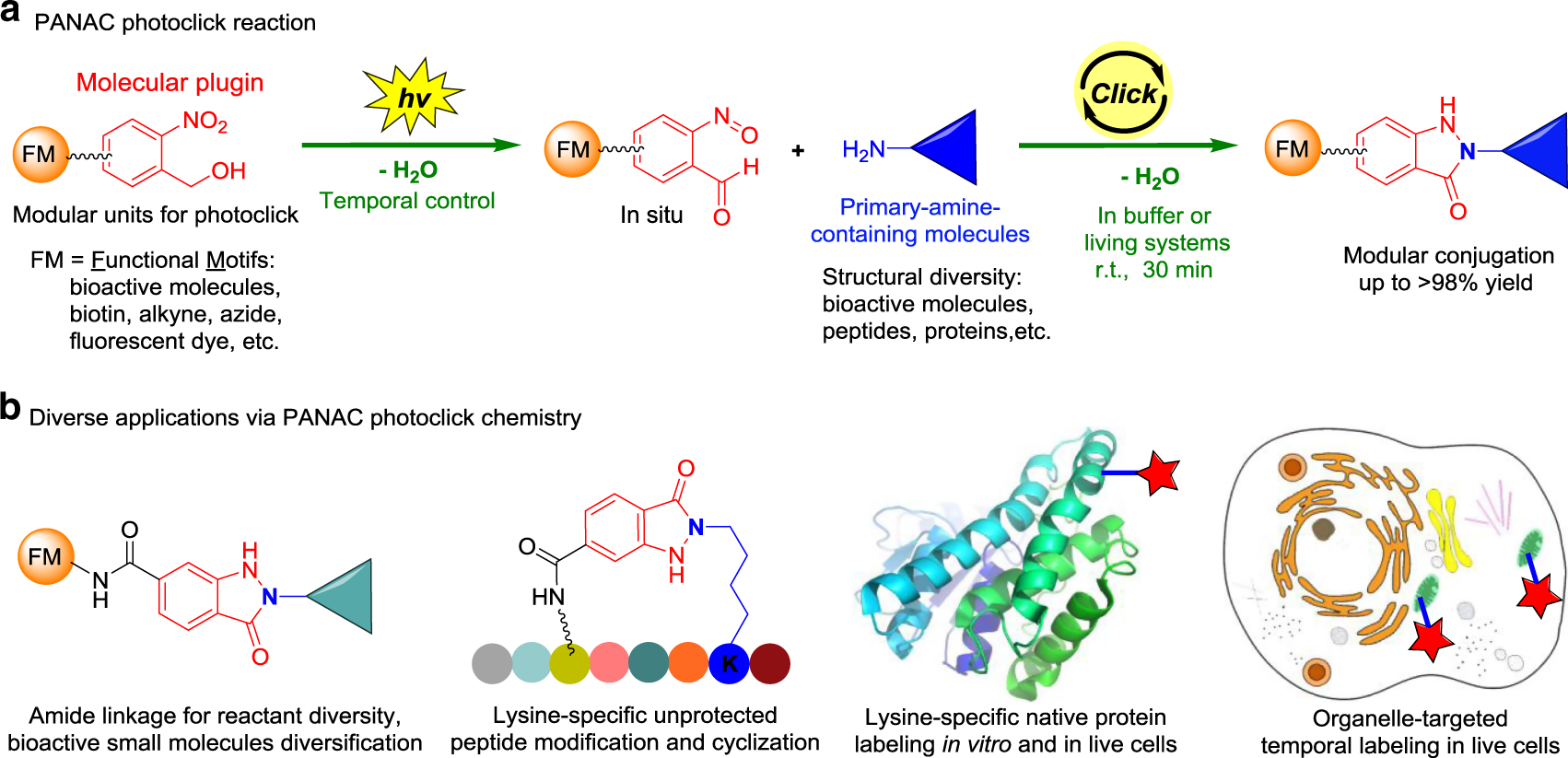
a o-Nitrobenzyl alcohol (o-NBA) was designed as molecular plugin and amide formations, as general linkage to rapidly access diverse reactants as modular units, and primary amines as straightforward click handle for PANAC photoclick reaction in vitro and in living systems. b Diverse applications of this PANAC photoclick chemistry.
