Fig. 1: Study design and oral rotavirus vaccine response.
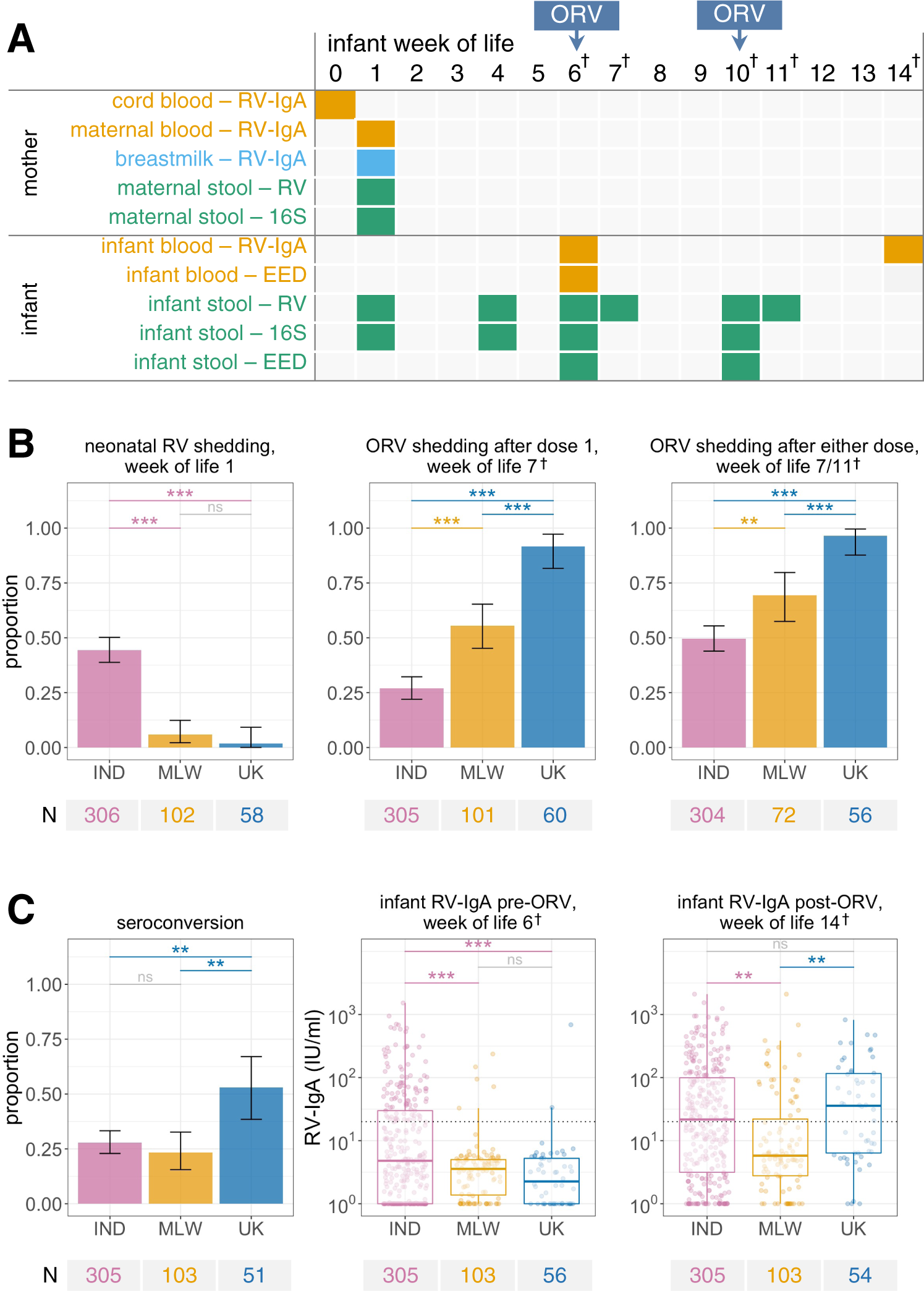
A Study design. The final study population comprised 307 infants in India, 119 in Malawi, and 60 in the UK. B, C Geographic differences in B rotavirus shedding and C ORV immunogenicity. Rotavirus shedding was detected via quantitative PCR using a pan-rotavirus assay targeting the VP6 gene of group A rotaviruses (week of life 1) and an assay for vaccine virus shedding targeting the Rotarix NSP2 gene (1 week after each dose). Seroconversion was defined as detection of RV-IgA at ≥20 IU/ml post-vaccination among infants who were seronegative at baseline or a 4-fold increase in RV-IgA concentration among infants who were seropositive at baseline. Error bars represent Clopper–Pearson 95% confidence intervals. Groups were compared by two-sided Fisher’s exact test with FDR correction (binary outcomes) or ANOVA with post-hoc Tukey tests (continuous outcomes). The dotted lines at 20 IU/ml indicate the standard cut-off for RV-IgA seropositivity. Box plots display median (centre line), upper and lower quartiles (box limits), the minimum value greater than or equal to the lower quartile − 1.5 × interquartile range (lower whisker), and the largest value less than or equal to the upper quartile + 1.5 × interquartile range (upper whisker). EED, environmental enteric dysfunction markers; IND, India; MLW, Malawi; ns, not significant; ORV, oral rotavirus vaccine; RV, rotavirus; †, +2 weeks in UK due to later vaccination schedule; *p < 0.05; **p < 0.005; ***p < 0.0005.
