Abstract
The reverse water-gas shift reaction (RWGSR) is essential for converting CO2 into fuels using renewable hydrogen, but it remains challenged by the difficulty of simultaneously maximizing catalyst activity, selectivity, and stability. These limitations stem from thermodynamic constraints – specifically, the Gibbs-Curie-Wulff theorem - which restricts the synthetic accessibility of high-energy micro-faceted nanocrystals via conventional methods. To address this, we introduce a near-surface “quasi-hyperbaric” ammonia strategy that integrates atmospheric-pressure processing with in-situ ammonia decomposition. This approach enables the controlled synthesis of molybdenum nitride nanocrystals with preferentially exposed high-energy (112) microfacets. These facets promote CO2 activation through a hydrogen-assisted redox mechanism, driven by geometrically confined and stabilized Mo-N/M-O hybrid active sites. The resulting catalyst outperforms the benchmark Pt/CeO₂, which typically suffers from CO selectivity below 92%. Our catalyst achieves near-equilibrium conversion (56%) at a space velocity (24000 ml/gcat/h), with 100% CO selectivity and outstanding stability (≤ 1% deactivation over 250 hours).
Similar content being viewed by others
Introduction
Catalytic conversion of CO₂ to CO via the reverse water-gas shift reaction (RWGSR) is a key pathway for sustainable fuel production and advancing carbon neutrality1,2,3. Despite its thermodynamic favorability at elevated temperatures, practical implementation of RWGSR is limited by the lack of catalysts that concurrently offer near-equilibrium conversion, perfect selectivity and industrial-grade stability4,5,6. This performance paradox arises from intrinsic trade-offs between the reactivity of catalytically active sites and their thermodynamic stability during synthesis and reaction. Although high-energy active sites, such as the high-energy microfacets of nanocrystals, are expected to enhance CO production7, the creation of these metastable states poses significant challenges. Unfortunately, they also accelerate catalyst sintering and side reactions, which compromise selectivity and stability.
Conventional catalyst systems exemplify this dilemma. While Pt-group catalysts are active and robust, they suffer from CO selectivity loss above 400 oC due to unfavorable electronic state transitions under reducing conditions8,9,10. Conversely, Cu-based catalysts offer high selectivity but lack sufficient activity and thermal instability, limited by their low Tamman temperature and a tendency for excessive hydrogenation3,11,12. Emerging transition metal nitrides and carbides, particularly molybdenum nitride (Mo₂N) nanocrystals, show promise in bridging this gap by mimicking noble metal behavior while offering superior thermal stability1,5,13,14,15,16,17,18. However, conventional synthesis routes for Mo₂N nanocrystals constrained by Gibbs-Curie-Wulff thermodynamic principles, inevitably expose low-energy microfacets as global surface energy minima through the predictive Wulff reconstruction, compromising the activity towards the RWGSR reaction19,20. To address the thermodynamic constraint, it is crucial to implement a kinetic control strategy that stabilizes highly active metastable microfacets by overriding the surface termination configurations at global energy minima. A viable approach involves introducing an intermediate stage characterized by a non-equilibrium steady-state chemical environment, guiding unconventional crystal growth during chemical synthesis. This strategy facilitates the formation of high-energy metastable microfacets in nanocrystals under synthetic conditions, allowing them to be effectively maintained and stabilized upon quenching.
As a proof-of-concept study, we introduce a near-surface “quasi-hyperbaric” ammonia atmosphere as a non-equilibrium steady-state chemical environment that completely alters the growth kinetics of Mo₂N nanocrystals restricted by the Gibbs-Curie-Wulff theorem. This is achieved by imposing strong surface-adsorbate interaction onto the intermediate structures during crystal growth, which completely alters the micro-faceting behaviors of nanocrystals and leads to the formation of high-energy (112) microfacets instead of thermodynamically stable (001)/(111) microfacets as predicted by Wulff construction. The high-energy-micro-faceted molybdenum nitride nanocrystal (HEMN) performs as a catalyst and demonstrates exceptional CO₂ hydrogenation performance, achieving 56% near-equilibrium CO₂ conversion with 100% CO selectivity at 550 °C and a space velocity of 24,000 mL·g⁻¹·h⁻¹, while maintaining excellent stability (< 1% activity loss over 250 + hours). Notably, HEMN outperforms noble-metal benchmarks, sustaining 100% CO selectivity compared to < 92% for Pt-based catalysts under equivalent conditions. The in situ spectroscopy, reaction kinetic analysis, and DFT calculations reveal that Mo-N/M-O hybrid structures on (112) microfacets act as active sites for CO₂ activation via an H₂-assisted redox mechanism.
Results
The creation of HEMN catalysts
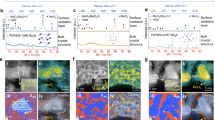
The growth of Mo₂N nanocrystals is influenced by the point symmetry of the tetragonal crystal lattice and thermodynamic constraints defined by the Gibbs-Curie-Wulff theorem. Consequently, conventional chemical synthesis typically results in Mo₂N nanocrystals featuring low-energy microfacets, as predicted by Wulff construction. This characteristic can be assessed through the surface free energy plot, or γ-plot, illustrated in Fig. 1a for the Mo₂N crystal structure projected along the (110) direction under vacuum conditions (red contour). Notably, the γ-plot under vacuum displays multiple cusped minima at 0.1003, 0.1121, 0.0977, and 0.1009 eV/Ų along the normals of the (001), (112), (111), and (110) microfacets. These cusped minima emerge from the lateral growth of Mo₂N nanocrystals, following the classical terrace-ledge-kink model. The surface energies of these microfacets are ranked as (111) < (001) < (110) < (112) (Fig. 1 and Supplementary Figs. S1, S2). The Wulff construction under vacuum would therefore lead to an enclosure of the low-energy (001) and (111) microfacets, resulting in Mo₂N nanocrystals shaped like truncated elongated octahedra. To overcome the thermodynamic restrictions associated with these stable microfacets, a near-surface “quasi-hyperbaric” ammonia atmosphere was introduced as a non-equilibrium steady-state, integrating both atmospheric and on-site decomposed ammonia (Supplementary Fig. S3a). This “quasi-hyperbaric” environment modulates the growth kinetics of Mo₂N nanocrystals by significantly enhancing the surface-adsorbate interaction. Investigating the γ-plot of Mo₂N surfaces bonded with ammonia adsorbates (purple contour) reveals that the interaction dramatically alters the surface energies of the nanocrystals. The chemical bonding of ammonia molecules decreases the surface energies for all microfacets at the cusped minima of the γ-plot, with a particularly pronounced effect on the (112) microfacets (approximately a 45% reduction in surface energy, from 0.1121 eV/Ų to 0.0648 eV/Ų). Consequently, the surface energies are re-ranked to (112) < (111) < (110) < (001), leading to the tentative stabilization of the high-energy (112) microfacets under synthetic conditions. Metastable nanocrystals in HEMN catalysts with exposed (112) microfacets and an octahedral shape can be synthesized by creating a near-surface “quasi-hyperbaric” ammonia atmosphere based on Wulff construction.
a γ-plot under vacuum (red contour) and ammonia environmental conditions (purple contour), corresponding surface energy mapping. b 3D schematic of HEMN catalyst, with the terminating crystal facet as (112); c Electron tomography image of HEMN catalyst; Fine structure of HEMN catalyst along the [021] zone axis (d) HAADF-STEM and (e) iDPC-STEM imaging, as well as along the [001] zone axis (h) HAADF-STEM and (i) iDPC-STEM imaging, with orange lines indicating the particle shape in the projection direction; Magnified (f) HAADF-STEM image and (j) iDPC-STEM image along the [021] zone axis; (g) Structural model of HEMN and (k) corresponding simulated projected potential image (Mo atomic columns and N atomic columns are marked in yellow and blue solid circles, respectively).
The experimental validation of HEMN catalysts can be achieved through combined high-resolution scanning transmission electron microscopy (HR-STEM) and electron tomography (ET). Multimodal STEM imaging, which incorporates high-angle annular dark-field (HAADF) and integrated differential phase contrast (iDPC) modes, enables the simultaneous visualization of both heavy and light elements within the Mo₂N nanocrystals. HAADF-STEM images captured along the [021] and [001] zone axes (Fig. 1d, h) clearly depict projected polyhedral nanocrystals measuring 5–10 nm (Supplementary Figs. S4, S5) with regular shapes. The angular gaps measured between the projected microfacets of these nanocrystals are 75° and 91°, respectively, aligning with the crystallographic interplanar angle for (112) microfacets. This observation supports the octahedral model predicted by Wulff construction (Fig. 1a). In addition, the explicit three-dimensional (3D) morphology of the nanocrystals can be reconstructed using the ET method based on tilt-series of STEM images, as illustrated in Fig. 1c and Supplementary Fig. S6. The Gaussian surface curvature of the 3D tomogram reveals that a randomly selected Mo₂N nanocrystal exhibits an octahedral shape, enclosed by microfacets shown in blue and connected by edges depicted in yellow. These microfacets can be indexed to the (112) group of crystal faces by comparing the projections of the reconstructed tomogram with the HAADF-STEM image along the [021] axis (Fig. 1b, c and Supplementary Fig. S5). Such observations confirm the (112) micro-faceted structure of HEMN catalysts stabilized under a “quasi-hyperbaric” ammonia atmosphere as a non-equilibrium steady-state. Furthermore, atomic-resolution structural elucidation of Mo₂N nanocrystals is facilitated through combined HAADF- and iDPC-STEM imaging, as demonstrated in Fig. 1d, h and Fig. 1e, i. An enlarged view of the HAADF-STEM image along the [021] axis reveals hexagonally arranged bright spots indicative of Mo atomic columns (Fig. 1f). The nitrogen atoms residing in the octahedral voids of the Mo sublattice can be visualized as atomic columns in the iDPC-STEM image (Fig. 1j). The phase contrast images align well with both the structural projections and the simulated projected potential of the Mo₂N crystal structure (Fig. 1g, k). In summary, these observations robustly corroborate the proposed concept, strategy, and structural model of HEMN catalysts.
Evaluation of the RWGSR Performance
Systematic optimization of calcination conditions, support materials, and loading levels identified 30 wt% Mo₂N on silica, formed via ammonia calcination, as the optimal catalyst, exhibiting outstanding RWGSR activity (Supplementary Figs. S7, S8). We further conducted a comprehensive comparative analysis of the synthesized HEMN catalyst against representative RWGSR catalysts from the literature, evaluating key metrics including reaction rate (mol/g−1*h−1), CO selectivity, CO2/H2 ratio, deactivation rate, and catalyst synthesis complexity (Supplementary Table S1). The HEMN catalyst demonstrated superior performance across all dimensions (Fig. 2a and Supplementary Fig. S10)9,18,21,22. Notably, within the high-temperature range (450–600 °C), the catalyst maintained nearly 100% CO selectivity across the full CO₂ conversion range (41.9%–62.6%). In contrast, Pt/CeO₂ exhibited a notable decrease in CO selectivity from 97.2% to 91.6% under the same reaction conditions (Fig. 2b and Supplementary Fig. S9). When evaluated under the same conditions, its overall performance exceeds that of other commonly reported catalysts (Supplementary Fig. S10). Additionally, the stoichiometric CO2/H2 ratio for the RWGSR is 1:1. However, in practical applications, a CO2/H2 ratio below 1:1 is often used to promote CO2 reaction and generate syngas compatible with downstream conversion processes. Increasing the H2 partial pressure thermodynamically favors the formation of methane formation23. To evaluate the effect of reactant composition, we investigated the influence of the CO2/H2 ratio on the catalytic performance. Increasing the H2 partial pressure significantly enhanced CO2 turnover rates while maintaining nearly 100% CO selectivity across both low (400 oC, in Supplementary Fig. S11) and high (550 oC, in Fig. 2c) temperature ranges. This behavior is a distinct feature of the Mo2N-based catalysts. Overall, the synthesized HEMN catalyst demonstrates robust performance over a broad temperature window (300–600 oC), wide space velocity range (12000 to 48000 ml/gcat/h, shown in Supplementary Fig. S12), and varied CO2/H2 ratios (1:1 to 1:4).
a Radar chart shows comparing the synthesized HEMN catalyst with representative RWGSR catalysts reported in the references, where the number of synthesis steps is determined by the number of high-temperature calcination and post-treatment process (Supplementary Table S1 corresponds to the radar chart presented in Fig. 2a). b CO selectivity of the noble metal-based Pt/CeO2 catalyst and the HEMN catalyst as a function of reaction temperature, under similar CO2 conversion conditions, with a space velocity of 12600 ml/gcat/h. c The effect of CO2/H2 ratio on CO2 conversion and CO selectivity of this catalyst at 550 oC, with a space velocity of 18000 ml/gcat/h. d Stability of this catalyst at 550 oC with a space velocity of 24000 mL/gcat/h. e Cyclic stability test of this catalyst (repeated temperature cycling from 300 oC to 550 oC), conducted at a space velocity of 24000 mL/gcat/h.
Catalyst stability is a critical factor for its industrial application. To evaluate the stability of this HEMN catalyst in the RWGSR, we conducted long-term tests at both low (400 oC, in Supplementary Fig. S13) and high (550 °C, Fig. 2d) temperatures. For example, at 550 oC with a space velocity of 24000 ml/gcat/h and a CO2/H2 ratio of 1/3, the catalyst exhibited only a slight decrease in conversion (from 52.6% to 52.1%) over 250 h, corresponding to just ~ 1% deactivation, which is significantly lower than values reported for similar catalytic systems18. Moreover, the catalyst demonstrated excellent cyclic stability under repeated temperature cycling between 300 oC and 550 oC (Fig. 2e). XRD analysis of the catalyst after thermal cycling revealed no significant changes in the catalyst’s microstructure (Supplementary Fig. S14), further confirming its high-temperature stability. Moreover, the HEMN catalyst demonstrated exceptional high-temperature stability during a 12 h test at 600 °C, exhibiting no deactivation while maintaining consistent performance (Supplementary Fig. S15). Comprehensive characterization via in-situ and ex situ XRD (Supplementary Figs. S16, 17) and HRTEM (Supplementary Figs. S18, 19) confirmed that both the phase structure and the specific (112) crystal facet remained stable without detectable thermodynamic reconstruction. These results collectively affirm the catalyst’s robust performance and structural integrity under demanding operational conditions.
Active sites of the HEMN catalyst in the RWGSR
To elucidate the reasons for the high RWGSR activity of this HEMN catalyst, we performed surface structure analysis on the synthesized catalyst using XPS. As shown in Supplementary Fig. S20, the O 1 s results revealed a peak at a binding energy of 530.6 eV, in addition to the characteristic peak of the support Si-O at 532.9 eV, which can be attributed to Mo-O species on the catalyst surface24. The Mo 3 d results showed peaks at 228.9 eV and 231.8 eV, which can be attributed to Mo-N species, and peaks at 229.9 eV, 232.7 eV, and 232.5 eV, 235.2 eV, which correspond to two oxidation states of Mo-O species, Mo(IV) and Mo(VI)25. This further confirms the presence of a certain amounts of Mo-O species on the surface of this catalyst. To further investigate the redox properties of M-O species, we performed H2-TPR-MS tests on the HEMN catalyst (Supplementary Figs. S21, S22). The results showed that compared to pure-phase MoO3 (with a reduction onset temperature of ~ 600 °C), the Mo-O species on the HEMN catalyst surface are more easily reduced (with a reduction onset temperature of ~ 350 oC). This indicates that the Mo-O species on this HEMN catalyst are highly dispersed, consistent with the previously discussed HAADF-STEM structure (Supplementary Figs. S4, S5). These species have more active oxygen species, which can easily form a rich oxygen vacancy structure with high electron-donating capacity, acting as a scissor to directly cleave the C = O double bond of CO2 to form CO26.
To further explore the impact of Mo-O content on the catalytic performance of the RWGSR, we reduced the HEMN catalyst at different temperatures to construct a series of catalysts with varying Mo-O contents without destroying the Mo2N crystalline phase structure (Supplementary Figs. S23 and S24). Supplementary Fig. S25 indicates that as the reduction temperature increases, the catalytic performance of the RWGSR gradually increases, reaching an optimal level when reduced at approximately 500 oC. When the reduction temperature is further increased, the CO2 conversion decreases significantly (from 21.5% to 14.5%). Supplementary Fig. S26 shows a volcano-like relationship between Mo-O content and CO yield, indicating that the catalytic activity is optimal only when the Mo-O content (~ 4%, as determined by quantitative XPS analysis) on this catalyst surface is moderate. The activation of the C − O bond is crucial in the CO₂ hydrogenation reaction27, and oxygen vacancies play a critical role28. At low reduction temperatures, the surface Mo-O concentration is high, resulting in weak CO2 adsorption and hindered C-O bond cleavage, which collectively lead to reduced catalytic activity. At higher reduction temperatures, the surface Mo-O concentration decreases, exposing more oxygen vacancies and enhancing CO2 adsorption. However, this also causes stronger adsorption of intermediate species, which hinders subsequent reactions29 and reduces the overall reaction rate (Supplementary Fig. S27). Therefore, the Mo-O/Mo-N species on the surface of the HEMN catalyst are the key determinants of its catalytic activity, a conclusion supported by studies of other similar systems30,31
Reaction mechanism of the catalytic reaction
Depending on the interaction between CO2 and H2, the RWGSR can proceed via different mechanisms, including: the redox mechanism (where H* does not assist in C-O bond cleavage), the HCOO* pathway (H* directly hydrogenates C*), and the COOH* mechanism (H* directly hydrogenates O*)5. To investigate the reaction mechanism of the RWGSR on the HEMN catalyst, we tracked the intermediate species on the catalyst surface during the reaction using in situ IR experiments, as shown in Fig. 3a. The results showed that when the reaction gas (CO2/H2/N2 = 24/72/4) was introduced, signals at 2175 cm−1 and 2120 cm−1 gradually appeared as the reaction temperature increased. These characteristic peaks can be attributed to the asymmetric and symmetric stretching vibrations of CO, respectively, as shown in Fig. 3a. Throughout the temperature ramping process (300 oC–600 oC), we did not observe the formation of any species related to COOH* or HCOO* (Supplementary Fig. S28), indicating that the hydrogenation of CO2 on the HEMN catalyst does not follow the formate or carboxylate pathways. We hypothesize that the HEMN catalyst operates via the redox mechanism, in which CO2 adsorbs on active sites, such as oxygen vacancies, followed by C-O bond cleavage to form CO and O species. To test this hypothesis, we introduced a CO2/N2 (30/70) mixture over the HEMN catalyst. If the redox mechanism were operative, CO* intermediates should be observed. However, unexpectedly, no CO* signals were detected under any of the following conditions: i) gradual temperature increase under CO2/N2 (Supplementary Fig. S29a), ii) The experiments involved either temperature ramping in 5% H₂/Ar after CO₂ adsorption at room temperature, or H₂ pre-adsorption followed by CO₂/N₂ exposure at varying temperatures (Supplementary Fig. S29b and c), or iii) after high temperatures reduction at 600 oC followed by similar in-situ experiments (Supplementary Figs. S30, S31). In addition, we complemented these studies with temperature-programmed surface reaction (TPSR) experiments and in-situ Raman measurements under CO₂/Ar atmosphere on the HEMN catalyst (Supplementary Fig. S32). Collectively, these results from both gas-phase and surface-sensitive characterization techniques consistently demonstrate that the reaction does not proceed via a conventional redox pathway, indicating a distinct mechanism deviating from the simple redox route.
In situ DRIFTS at different temperatures: (a) 2250 cm⁻¹ to 2000 cm⁻¹, (b) 4000 cm⁻¹ to 2500 cm⁻¹; (c) H₂-TPR-MS profile of HEMN catalyst. d In situ DRIFTS of the HEMN catalyst in H₂/Ar atmosphere at different temperatures. Reaction conditions for catalysis: Catalyst mass: 12 mg, Gas composition: CO₂/H₂/N₂ = 24/72/4.
Based on in situ experiments under reactive conditions (Fig. 3a), we conclude that CO* formation requires the presence of H2, indicating a likely H2-assisted redox mechanism, which is consistent with previous reports on the Mo2C system5. Furthermore, throughout the reaction process (Fig. 3b and Supplementary Fig. S33), we observed no peaks forming or consuming in the range of 3500 cm−1 to 3200 cm−1, which is attributed to the stretching vibrations of hydroxyl groups32. This indicates that the consumption or generation of OH/H2O is a fast step in the elementary reactions and occurs after the rate-determining step33.
During the H2-TPR-MS test of the HEMN catalysts, we observed a significant formation of the H2O peak before 300 oC (Fig. 3c). Since the samples were pre-treated in an inert atmosphere at 300 oC before the experiment, the adsorbed water on the samples was eliminated, ruling out the possibility that the H2O peak was from desorption of adsorbed water. Simultaneous online mass spectrometry detection revealed no signal peaks at m/z = 2 (H2 species), indicating that the H2O peak did not originate from the reduction of surface metal oxide species. Infrared transmission spectroscopy of the HEMN catalyst revealed distinct surface Mo-OH species (Supplementary Fig. S34), suggesting that the H2O peak detected by mass spectrometry likely originates from hydrogen-assisted dehydration of surface OH groups on the support.
To further verify this hypothesis, we conducted in situ IR experiments on the catalyst with H2/Ar (Fig. 3d). The results showed a significant consumption of OH in a temperature range similar to that observed in the H2-TPR-MS experiment, which is consistent with our hypothesis. When the HEMN samples were pretreated with hydrogen reduction at temperatures above 300 oC, there was no consumption of hydrogen observed in the in situ IR spectra. This finding confirms that surface Mo-OH species undergo hydrogen-assisted dehydration, forming Mo-O/Mo-N sites that constitute the active site structure, further supporting our earlier conclusions (Figs. S24–S27). Theoretical calculations further support this finding (Supplementary Figs.S35, S36), showing that the presence of H2 lowers the dehydration energy of Mo-OH from 0.76/0.74 eV to 0.45/0.46 eV on Mo2N (111) or (112) facets.
To further investigate the reaction mechanism of the RWGSR on the HEMN catalyst, we studied the kinetic dependences of CO2 and H2 at 300 °C and atmosphere pressure with 50.7–76.0 kPa H2, and 10.1-35.5 kPa CO2, when maintaining the CO2 conversion below 10% and approach-to-equilibrium less than 10%. As shown in Fig. 4a, b, the turnover rate on HEMN catalyst gradually increased with the increasing partial pressure of CO2, demonstrating a reaction order of 0.37 ( ± 0.003). The effect of H2 partial pressure on the reaction rate showed a similar trend, with a reaction order of 0.28 ( ± 0.01). Combining the results from H2-TPR-MS, CO-TPD, and in-situ IR spectroscopy experiments, we proposed the elementary reaction steps of the RWGSR on the HEMN catalyst, as shown in Fig. 4c. The reaction begins with the adsorption of CO₂ onto the Mo-O/N-Mo active site pair (denoted as *), forming adsorbed CO2* (Step 1). Concurrently, H₂ undergoes dissociative adsorption at an adjacent site, producing an H* adatom and a Mo-OH* (Step 2). Subsequently, the H* species performs a nucleophilic attack on the oxygen atom of a nearby CO₂* molecule, cleaving the C = O bond to generate OH* and CO* intermediates (Step 3). The CO* intermediate then desorbs into the gas phase as CO (Step 4). Finally, two OH* species combine to form an H₂O* intermediate, which subsequently desorbs as a water molecule (Steps 5 and 6). Based on the above findings, together with the assumption of Step 3 as the rate-determining step and the application of the pseudo steady-state approximation, we can obtain the following turnover rate equation, with the detailed derivation process given in the Supporting Information:
where k and Kj (subscript j = CO2 or H2;) denote the rate constant of the kinetically relevant step (Step 3) and the equilibrium constant of the relevant elementary step (Step 1 or 2), respectively. The non-linear fitting of the experimental data with the turnover rate Eq. (1) determined \({{kK}}_{{{CO}}_{2}}{({K}_{{H}_{2}})}^{0.5}\), \({K}_{{{CO}}_{2}}\), and \({K}_{{H}_{2}}\) to be 3.32 ( ± 0.30) × 10-4 mol h−1 gcat−1 kPa−1.5, 0.041 ( ± 0.002) kPa−1, and 5.01 ( ± 1.26) × 10−3 kPa−1, respectively, as present in Fig. 4d. The predicted turnover rates closely match the experimental results, as confirmed by the parity plot (Fig. 4e). Under the operating conditions (50.7–76.0 kPa H2, and 10.1-35.5 kPa CO2), the coverage of unoccupied active site pairs on the catalyst surface varies from 33.3% to 52.1%, that of CO2* from 20.4% to 49.2%, and that of H* from 17.0% to 30.4%. We also investigated catalytic systems with lower M-O content on HEMN reduced at 600oC, as shown in Supplementary Fig. S37, S38. The results indicate that the different M-O contents do not affect the reaction kinetics, such as the reaction orders and the rate-determining step.
a Effect of CO2. b Effect of H2. c Elementary steps on the HEMN catalyst. d Kinetic parameters derived from rate data regression. e Parity plot for the predicted and measured intrinsic rates on the HEMN catalyst. All turnover rates were corrected to 0.4–1.8 h time-on-stream. Kinetic testing conditions of the RWGSR over the HEMN catalyst reacted at 300 oC, ambient pressure with 50.7 kPa (■) or 60.8 kPa H2 (●), and 10.1 kPa (▲) or 20.3 kPa CO2 (▼), and a space velocity of 48,000 ml/gcat/h.
To gain deeper mechanistic insights into the RWGSR catalyzed by the HEMN system, we conducted comprehensive theoretical investigations of the hydrogen-assisted CO2 activation pathway, as illustrated in Fig. 5. Advanced electron microscopy coupled with 3D structural reconstruction unambiguously identified the preferential exposure of the (112) crystallographic facet in the HEMN catalyst, overriding the conventional thermodynamic stability of the (111) surface. Building on these experimental insights, we constructed DFT-optimized models of the dominant (112) and reference (111) surfaces of Mo₂N (Supplementary Fig. S39). To this end, we systematically probed the influence of Mo–O/N vacancy concentration on the H₂ binding energy, thereby identifying the Mo–O···H–Mo configuration as the initial active site for CO₂ activation (Supplementary Fig. S40). Comparative analysis of surface reactivity (Fig. 5 and Supplementary Fig. S41) revealed a striking reduction in the CO2 dissociation energy barrier on the (112) surface (0.66 eV vs 1.14 eV on (111) facet), providing fundamental rationale for the exceptional catalytic performance of our engineered material compared to conventional Mo2N systems.
a Reaction energy profiles for different pathways on the (111) and (112) facets. b–e Corresponding initial state, transition state, and final state structures.
To corroborate the hydrogen-mediated activation mechanism suggested by kinetic investigation and operando spectroscopic characterization, we systematically probed the hydrogen participation in CO2 activation thermodynamics. DFT calculations demonstrate that on the (112) facet, the spontaneous conversion of adsorbed CO2* to CO* and O* proceeds with an energy change of − 1.20 eV under hydrogen-free conditions. Remarkably, the co-adsorption of H* species amplifies the thermodynamic favorability, lowering the energy change to − 1.45 eV. Crucially, kinetic analyses revealed that hydrogen participation reduces the activation barrier from 0.66 eV to 0.45 eV, decisively confirming the viability of the H*-assisted activation pathway. In summary, our calculations demonstrate that the (112) facet is more active than the (111) facet for C–O bond cleavage, and that the H₂-assisted pathway enables CO₂ dissociation with a substantially lower energy barrier compared to direct dissociation. Furthermore, the catalytic cycle maintains sustainability through dynamic surface regeneration: The transient Mo-OH species formed during reaction undergoes rapid dehydrogenation in the H2-rich environment (Fig. 3 and Supplementary Fig. S35), reconstructing the active Mo-O/Mo-N configuration that perpetuates the catalytic cycle. The superior catalytic performance and stability of the HEMN system arise from a synergistic combination of favorable thermodynamics, lowered kinetic barriers, and self-repairing surface architecture. Based on the structural model of the (112) facet, we further evaluated alternative reaction pathways, namely, the HCOO* and *COOH routes, to theoretically exclude competing mechanisms. The results show that the H₂-assisted pathway features a significantly lower activation barrier (2.22 eV for the *COOH route and 1.45 eV for the *HCOO route, as shown in Supplementary Fig. S42), in good agreement with our in-situ DRIFTS measurements (Supplementary Fig. S28).
Discussion
In summary, we report a novel synthesis strategy that integrates atmospheric modulation with self-driven ammonia decomposition to create a near-surface “quasi-hyperbaric” ammonia microenvironment, enabling in-situ formation of HEMN with dominant (112)-oriented facets. The resulting catalyst combines cost-effectiveness and synthetic simplicity with outstanding performance, achieving near-equilibrium CO2 conversion (56%) and 100% CO selectivity at 550 °C with a space velocity at 24,000 ml/gcat/h. It also demonstrates excellent stability, with ≤ 1% activity loss over 250 + hours of continuous operation and cycling. Remarkably, HEMN exceeds noble-metal behavior, maintaining 100% CO selectivity versus < 92% for a benchmark Pt-based catalyst at 550 °C. Mechanistic studies, including in-situ spectroscopy and DFT calculations, reveal that hybrid Mo-N/M-O motifs on (112) microfacets act as dual-functional active sites, enabling CO₂ activation via a hydrogen-assisted redox mechanism. This work not only deciphers crucial structure-activity relationships in transition metal nitrides but also establishes a robust design framework for developing non-precious, thermally robust catalysts with industrial-grade activity-selectivity – advancing the prospects of sustainable CO₂ valorization.
Methods
The synthesis of catalysts
The high-energy-micro-faceted molybdenum nitride (HEMN) catalysts
A precise amount of ammonium molybdate (H₈MoN₂O₄, Aladdin, AR grade) was dissolved in deionized water and uniformly dispersed onto silica or other supports using the incipient wetness impregnation method. The resulting slurry was dried at 100 °C for 12 h to obtain the precursor. Subsequent calcination was performed in a tubular furnace under flowing NH₃ at 600 °C (with a heating rate: 5 °C/min) for 4 h to yield the corresponding supported HEMN catalysts.
Synthesis of reference catalysts
(i) The pure Mo₂N catalyst was synthesized via a co-precipitation method. First, 3.5 g of ammonium molybdate (H₈MoN₂O₄, Aladdin, AR grade) and 3.6 g of melamine (C₃H₆N₆, Sigma, 99%) were separately dissolved in 50 mL deionized water. The solutions were combined under continuous magnetic stirring to induce complete precipitation. The resulting precipitate was isolated by centrifugation, dried at 120 °C for 12 h, and subjected to temperature-programmed calcination in a tubular furnace under N₂ flow. The thermal profile included a ramp from ambient temperature to 800 °C at 10 °C/min for 7 h; (ii) The CeO₂ support was prepared by calcining 6 g of cerium nitrate (Ce(NO₃)₃, Macklin, AR grade) in a muffle furnace at 350 °C at 5 °C/min for 2 h. Separately, 14.88 mg of platinum tetra-ammine nitrate (H₁₂N₆O₆Pt, Aladdin, 99.9%) was dissolved in 525 μL deionized water and impregnated onto 1.5 g of the pre-synthesized CeO₂ support using the incipient wetness impregnation method. The impregnated material was dried at 80 °C for 12 h and activated in a muffle furnace under static air with a thermal ramp of 5 °C/min to 800 °C, where it was held for 1 h; (iii) The Ni/Al₂O₃ catalyst was prepared by dissolving 148.5 mg of nickel nitrate hexahydrate (Ni(NO₃)₂·6H₂O, Macklin, AR grade) in 1150 μL deionized water. The solution was impregnated onto 1.5 g of γ-Al₂O₃ support via the incipient wetness method. The composite was dried at 100 °C for 12 h and calcined in a muffle furnace with a 5 °C/min ramp to 600 °C for 4 h. (iv) The CuZnAl catalyst was prepared by co-precipitation. An aqueous solution of Cu, Zn, and Al nitrates and a separate K₂CO₃ solution were simultaneously added dropwise into a beaker at 80 °C with vigorous stirring. The pH was maintained at approximately 9 during this process. The resulting suspension was aged at 80 °C for 12 h, then the precipitate was collected, washed with deionized water, dried at 100 °C for 12 h, and finally calcined at 550 °C for 4 h in air. (v) The Ni/CeO₂ catalyst was prepared by incipient wetness impregnation of the CeO₂ support with an aqueous solution of nickel nitrate. The material was subsequently dried at 100 °C for 12 h and calcined at 300 °C for 2 h (ramp rate: 5 °C/min). Prior to catalytic testing, the catalyst was reduced in a 5% H₂/Ar flow at 550 °C for 2 h. (vi) The Fe/ZrO₂ catalyst with an iron loading of 1.43 wt% was prepared by the incipient wetness impregnation of a ZrO₂ support with an aqueous solution of ferric nitrate. The precursor was dried at 100 °C for 12 h and subsequently calcined at 450 °C for 4 h in static air with a heating rate of 5 °C/min. Prior to catalytic testing, the catalyst was reduced at 550 °C for 2 h in a 5% H₂/Ar (30 mL/min). (vii) The Pt/SiO₂ catalyst was prepared by the incipient wetness impregnation of a SiO₂ support with an aqueous solution of tetra-ammine-platinum(II) nitrate. Following impregnation, the material was dried at 100 °C for 12 h and subsequently calcined at 450 °C for 2 h in static air with a heating rate of 10 °C/min. (viii) The Ru/γ-Al₂O₃ catalyst was prepared by the incipient wetness impregnation of a γ-Al₂O₃ support with an aqueous solution of ruthenium(III) chloride. The resulting material was dried at 100 °C for 12 h and subsequently calcined at 500 °C for 2 h in static air with a heating rate of 10 °C/min. Prior to catalytic testing, the catalyst was reduced in a 5% H₂/Ar flow (30 mL/min) at 550 °C for 1 h.
The performance of catalysts
After synthesizing the HEMN catalyst under an ammonia atmosphere, we evaluated its performance of RWGSR. The RWGSR tests were conducted in a home-made fixed-bed reactor system, which consists of a quartz tube with an inner diameter of 8 mm. The catalyst was prepared by pelletizing, crushing, and sieving to 60–80 mesh, followed by homogeneous mixing with quartz sand. The reaction was carried out at 1 atm with a feed gas mixture of CO₂, H₂, and N₂ in a volumetric ratio of 24:72:4. During the temperature-programmed process, the reaction gas mixture was continuously fed into the reactor. After reaching the target temperature, the system was stabilized for 30 minutes before data collection. Product analysis was performed using an online gas chromatograph (GC2060, Shanghai Ruimin Instrument Co., Ltd.) equipped with a flame ionization detector (FID; Rt-Q-BOND column) and a thermal conductivity detector (TCD; TDX-01 column). A high-temperature conversion furnace was integrated into the system, and all product lines were maintained at 150 oC to ensure accurate online detection. CO₂ conversion and product selectivity were calculated via the carbon normalization method, with specific formulas provided as (2) and (3).
Here, i denotes any gaseous species in the reaction system; N represents the number of carbon atoms in the component; Ai and fi correspond to the chromatographic peak area and calibration factor of species i, respectively; and p denotes the products.
Detailed kinetic testing parameters are provided in Supplementary Tables S3, S4, with corresponding conversion and product selectivity data available in Supplementary Tables S7–S14.
The characterization of catalysts
X-ray diffraction (XRD) measurements were conducted on a Rigaku SmartLab 9 kW Advanced powder X-ray diffractometer using Cu Kα radiation (λ = 0.154 nm). The 2θ scanning range was 10° to 80° at a rate of 5°/min. X-ray photoelectron spectroscopy (XPS) analysis was performed using a Thermo Scientific K-Alpha instrument. Binding energy results were calibrated by referencing the C1s peak to 284.6 eV. The specific surface area was determined using the Brunauer–Emmett–Teller (BET) equation by analyzing the adsorption branch of the isotherm in the pressure range of P/P₀ = 0.04–0.32. Pore size distribution was derived from the adsorption branch using the Barrett–Joyner–Halenda (BJH) method, and the total pore volume was measured at P/P₀ = 0.99. Temperature-programmed reduction and desorption analyses (H₂-TPR, H₂-TPD, CO₂-TPD) were conducted on a home-made system equipped with an online mass spectrometer (Tilon SRD200M). For all experiments, 100 mg of catalyst was pretreated in Ar at 300 °C for 2 h to eliminate surface impurities. In H₂-TPR, the sample was reduced with 5% H₂/Ar while heating from room temperature to 900 °C at 10 °C/min, monitoring H₂ (m/z = 2) and H₂O (m/z = 18). For H₂-TPD, after H₂ adsorption (5% H₂/Ar, 1 h) and Ar purging, H2 desorption was tracked (m/z = 2) during heating to 800 °C at 10 °C/min. Similarly, CO₂-TPD involved CO₂ adsorption (30% CO₂/N₂, 1 h), Ar purging, and monitoring CO₂ (m/z = 44) during heating to 800 °C at 10 °C/min under Ar flow. High-angle annular dark-field scanning transmission electron microscopy (HAADF-STEM) and integrated differential phase contrast imaging (iDPC-STEM) were performed using a Thermo Scientific Spectra 300 double spherical aberration-corrected transmission electron microscope operated at 300 kV with a convergence semi-angle of 25 mrad. The sample was dispersed in anhydrous ethanol via ultrasonication, deposited onto a grid using a micropipette, dried, and loaded into the microscope. In-situ diffuse reflectance infrared Fourier transform spectroscopy (DRIFTS) experiments were conducted using a Thermo Scientific Nicolet iS50 spectrometer equipped with an MCT detector. For RWGSR studies, the HEMN catalyst was pretreated in N₂ at 300 °C for 1 h, cooled to room temperature, and background spectra were collected at 300–600 °C under N₂. Reaction gas (CO₂/H₂/N₂ = 24/72/4) was then introduced into the in-situ cell to monitor intermediates and products. To investigate surface hydroxyl species interacted with H₂, the pretreated catalyst was exposed to 5% H₂/Ar from 25 to 300 °C, with spectra recorded in the range of 4000–2500 cm⁻¹. In-situ XRD was performed using a PANalytical Empyrean diffractometer equipped with a Cu Kα radiation source (λ = 1.5418 Å) at 40 kV and 40 mA. In-situ experiments were operated with a diffractometer equipped with a high-temperature reaction cell (XRK 900, Anton Paar GmbH). All XRD patterns were scanned from 10° to 80° (2θ) at a rate of 5° min⁻¹. Temperature-programmed in-situ XRD measurements of the HEMN catalyst were performed under a mixed gas atmosphere of CO₂:H₂:Ar = 72:24:4 (vol%) with a total flow rate of 40 mL min⁻¹. The catalyst was heated from 25 °C to 50 °C, 400 °C, 500 °C, 550 °C, and 600 °C sequentially at a ramp rate of 10 °C min⁻¹, with one diffraction pattern collected at each target temperature. Subsequently, the catalyst was held at 600 °C for 60 min, during which diffraction patterns were collected every 20 minutes (3 patterns in total). For the in-situ HRTEM characterization, the data were acquired using an FEI ETEM equipped with a GATAN K3 direct electron detection camera, combined with a DENS MEMS-based gas sample holder. First, a small amount of an aqueous solution of the HEMN catalyst was precisely drop-cast onto the silicon nitride membrane window of the gas chip and subsequently air-dried at room temperature. Finally, the gas chip loaded with the sample was carefully installed into the in-situ gas holder. At the beginning of the in-situ experiment, a mixed gas of CO2 and H2 was introduced, while the temperature was simultaneously and gradually increased to 600 °C and held stable for 1 h. During this process, dynamic HRTEM images of the sample were acquired along the [021] zone axis. The TPSR experiment was conducted using a custom-built system equipped with an online mass spectrometer (Tilon SRD200M). Prior to the reaction, 100 mg of the catalyst was pretreated in argon at 300 °C for 2 h to remove surface impurities. The sample was then exposed to a 30% CO₂/N₂ gas mixture (flow rate: 20 mL/min) while heating from room temperature to 600 °C at a rate of 10 °C/min. The desorption signals of CO₂ (m/z = 44) and CO (m/z = 28) were continuously monitored. The In situ Raman spectra with CO2/Ar were recorded on a Renishaw in Via spectrometer with 532 nm laser excitation, coupled with a Linkam TS 1500 micro in-situ cell. The HEMN was exposed to a 10% CO₂/Ar flow (30 mL/min) during the heating process. A reference spectrum was first collected at room temperature. The sample was then heated at 10 °C/min to 300 °C to 600 °C and then kept for 60 min. At each target temperature, the sample was stabilized, repositioned under the microscope, and two consecutive Raman scans were collected (60 s integration, spectral range: 100–1200 cm⁻¹).
Derivation of rate expression
The rate expression is derived based on the sequence of elementary steps proposed in Fig. 4c in the manuscript, considering the interaction between CO2* and H* as the kinetically relevant step (Step 3, main manuscript) and the pseudo steady-state approximation of all intermediates.
where r and k represent the turnover rate and rate constant of Step 3, respectively. θj denotes the fractional coverage of surface species j (j = *, CO2*, H*). According to the Langmuirian adsorption model, the overall site balance is
The quasi-equilibrium assumption on CO2 adsorption (Step 1) and H2 dissociative adsorption (Step 2) gives
where Ki (i = CO2 or H2) is the equilibrium constant of CO2 adsorption or H2 dissociative adsorption.
Substituting Eqs. 6–7 into Eq. 5 gives
Thus,
Substituting (6), (7), and (9) into (4) gives (1) in the main manuscript.
Density functional theory (DFT)
Density functional theory (DFT) calculations were conducted using the Vienna ab initio simulation package (VASP 5.4.4)34. The exchange-correlation energy was modeled with the Perdew-Burke-Ernzerhof (PBE) functional under the generalized gradient approximation (GGA)35. Core electrons were described using projected-augmented wave (PAW) pseudopotentials, and a plane-wave cutoff energy of 450 eV was employed36. After conducting k-point convergence tests (as shown in Table S5), we ultimately determined that a Γ-centered 2 × 1 × 1 k-point grid was employed for the (111) facet, and a 2 × 2 × 1 grid was used for the (112) facet in the k-point sampling process. Convergence criteria were set to 10⁻⁵ eV for electronic relaxation and -0.05 eV/Å for ionic relaxation. Based on electron microscopy analysis, the synthesized Mo₂N material predominantly exposes the high-surface-energy (112) facet (Fig. 1 in the main manuscript). To explore its surface properties, we constructed a computational model of the Mo₂N (112) facet with lattice parameters of a = 12.0366 Å, b = 9.9712 Å, c = 21.0606 Å and angles of α = 90.0 o, β = 90.0 o, γ = 90 o. For comparative purposes, a thermodynamically stable Mo₂N (111) supercell model was also established (Supplementary Figs. S39, 40), with lattice parameters of a = 12.0366 Å, b = 18.0350 Å, c = 19.9984 Å, and angles of α = 90.0 o, β = 90.0 o, γ = 109.5 o. In the computational setup, the bottom atomic layers were fixed to simulate the bulk catalyst’s structural rigidity, while the top surface layers were fully relaxed to capture local atomic rearrangements during adsorption and surface reactions. This approach strikes a balance between computational efficiency and accuracy, ensuring the identification of the most stable configurations during adsorption-reaction processes. The climbing image nudged-elastic-band method (CI-NEB), including six replicas, was used to search the transition states (TS) for all of the elementary reactions. The TS was denoted by the highest image along the minimum energy path. The energy barrier (Ea) of each elementary reaction was calculated by the energy difference between the TS and the initial state (IS) without inclusion of the zero-point energy. Vibrational frequencies were computed at the optimized transition state structure using the finite-displacement method to determine the Hessian (force constant) matrix (Supplementary Table S6). The calculation was performed with IBRION = 5, a displacement step of POTIM = 0.015 Å, and the same exchange-correlation functional and pseudopotentials as used in the transition state optimization. NSW was set to 1 to prevent any further geometry relaxation. The vibrational mode corresponding to the imaginary frequency was visualized using “Jmol” (an open-source Java viewer), confirming that the atomic displacements align with the expected reaction coordinate.
The adsorption energy (Eads) of adsorbates on all slab models was calculated as below:
where Etotal, Eslab, and Eg represent the total energy of the adsorbed system, the pristine slab energy, and the gas-phase adsorbate energy, respectively. Correspondingly, the desorption energy (Edes) is defined as below:
The vacancy formation energy (ΔEf) of HEMN catalysts was calculated as below:
where, Ev denotes the energy of the vacancy-containing surface, and EH2O(g) represents the energy of an isolated water molecule in the gas phase.
The surface energy calculation involves three key steps: First, both the bulk material and the surface models (as illustrated in Supplementary Fig. S1) undergo geometric optimization to ensure they correspond to their lowest-energy stable configurations. Second, the bulk energy Ebulk is calculated, and the average energy per atom Ebulk, per atom is determined using the formula (\({E}_{{bulk},{peratom}}={E}_{{bulk}}/{N}_{{bulk}}\), where \({N}_{{bulk}}\) is the number of atoms in the bulk material). Third, the total energy of the surface model Eslab is computed. The surface energy γsurface is then calculated using the following formula:
where: A is the surface area, \({N}_{{slab}}\) is the number of atoms in the surface model. For the surface model with adsorbed ammonia, which simulates the surface energy under an ammonia atmosphere, the calculation formula is as follows:
where \({E}_{{slab},{{NH}}_{3}}\) represents the total energy of the surface model with adsorbed ammonia, \({N}_{{{NH}}_{3}}\) is the number of ammonia molecules adsorbed on the surface, and \({E}_{{{NH}}_{3}}\) is the energy of a single ammonia molecule in the gas phase.
Data availability
All data generated in this study are provided in the Supplementary Information/Source Data. All data are available from the corresponding author upon request.
References
Xin, H. et al. Reverse water gas-shift reaction product driven dynamic activation of molybdenum nitride catalyst surface. Nat. Commun. 15, 3100 (2024).
Wang, H. et al. Synergistic interactions of neighboring platinum and iron atoms enhance reverse water–gas shift reaction performance. J. Am. Chem. Soc. 145, 2264–2270 (2023).
Liu, H. et al. Partially sintered copper-ceria as excellent catalyst for the high-temperature reverse water gas shift reaction. Nat. Commun. 13, 867 (2022).
Bahmanpour, A. M. et al. Recent progress in syngas production via catalytic CO2 hydrogenation reaction. Appl. Catal. B Environ. 295, 120319 (2021).
Ahmadi, K. M. et al. An active, stable cubic molybdenum carbide catalyst for the high-temperature reverse water-gas shift reaction. Science 384, 540–546 (2024).
Guo, J. et al. In–Ni intermetallic compounds derived from layered double hydroxides as efficient catalysts toward the reverse water gas shift reaction. ACS Catal. 12, 4026–4036 (2022).
Wang, R. et al. High-energy facet engineering for electrocatalytic applications. Small 20, 2401546 (2024).
Zhang, R. et al. Synergism of ultrasmall Pt clusters and basic La2O2CO3 supports boosts the reverse water gas reaction efficiency. Adv. Energy Mater. 13, 2203806 (2023).
Zhao, Z. et al. Atomically dispersed Pt/CeO2 catalyst with superior CO selectivity in reverse water gas shift reaction. Appl. Catal. B Environ. 291, 120101 (2021).
Yang, X. et al. Promotion effects of potassium on the activity and selectivity of Pt/zeolite catalysts for reverse water gas shift reaction. Appl. Catal. B Environ. 216, 95–105 (2017).
Zakharova, A. et al. Reverse microemulsion-synthesized high-surface-area Cu/γ-Al2O3 catalyst for CO2 conversion via reverse water gas shift. ACS Appl. Mater. Interfaces 14, 22082–22094 (2022).
Gu, M. et al. Structure-activity relationships of copper-and potassium-modified iron oxide catalysts during reverse water-gas shift reaction. ACS Catal. 11, 12609–12619 (2021).
Lin, Z. et al. Transition metal carbides and nitrides as catalysts for thermochemical reactions. J. Catal. 404, 929–942 (2021).
Tian, D. et al. Density functional theory studies of transition metal carbides and nitrides as electrocatalysts. Chem. Soc. Rev. 50, 12338–12376 (2021).
Wang, H. et al. Transition metal nitrides for electrochemical energy applications. Chem. Soc. Rev. 50, 1354–1390 (2021).
Zhou, H. et al. Two-dimensional molybdenum carbide 2D-Mo2C as a superior catalyst for CO2 hydrogenation. Nat. Commun. 12, 5510 (2021).
Jurado, A. et al. Molecular mechanism and microkinetic analysis of the reverse water gas shift reaction heterogeneously catalyzed by the Mo2C MXene. ACS Catal. 12, 15658–15667 (2022).
Wu, Y. et al. The highly selective catalytic hydrogenation of CO2 to CO over transition metal nitrides. Chin. J. Chem. Eng. 43, 248–254 (2022).
Gao, Z. et al. Shielding Pt/γ-Mo2N by inert nano-overlays enables stable H2 production. Nature 638, 690–696 (2025).
Nagae, M. et al. Microstructure of a molybdenum nitride layer formed by nitriding molybdenum metal. J. Am. Ceram. Soc. 84, 1175–1177 (2001).
Zhang, X. et al. Highly dispersed copper over β-Mo2C as an efficient and stable catalyst for the reverse water gas shift (RWGS) reaction. ACS Catal. 7, 912–918 (2016).
Hameed, G. et al. Experimental optimization of Ni/P atomic ratio for nickel phosphide catalysts in reverse water-gas shift. J. CO2 Util. 77, 102606 (2023).
Gao, P. et al. Novel heterogeneous catalysts for CO2 hydrogenation to liquid fuels. ACS Cent. Sci. 6, 1657–1670 (2020).
Wang, Q. et al. One-dimensional MoO3 coated by carbon for supercapacitor with enhanced electrochemical performance. J. Mater. Sci. Mater El. 30, 6643–6649 (2019).
Yu, Z. et al. Formic acid as a bio-CO carrier: selective dehydration with γ-Mo2N catalysts at low temperatures. ACS Sustain. Chem. Eng. 8, 13956–13963 (2020).
Liu, H. et al. Direct cleavage of C=O double bond in CO2 by the subnano MoO(x) surface on Mo2N. Nat. Commun. 15, 9126 (2024).
Ye, R. et al. CO2 hydrogenation to high-value products via heterogeneous catalysis. Nat. Commun. 10, 5698 (2019).
Jin, F. et al. Unraveling the influence of oxygen vacancies in MoOx catalysts on CO2 hydrogenation. Chem. Eng. J. 495, 153333 (2024).
Wang, Y. et al. Breaking the linear scaling relationship of the reverse water–gas–shift reaction via construction of dual-atom Pt–Ni pairs. ACS Catal. 13, 3735–3742 (2023).
Sun, X. et al. Reaction-induced unsaturated Mo oxycarbides afford highly active CO2 conversion catalysts. Nat. Chem. 16, 2044–2053 (2024).
Gong, Y. et al. In Situ Surface Restructuring of γ-Mo2N for Enhanced Reverse Water-GasShift Reaction. ACS Catal. 15, 8337–8344 (2025).
Santiago, C. M. et al. FT-IR examination of the development of secondary cell wall in cotton fibers. Fibers 3, 30–40 (2015).
Langer, M. et al. Kinetic modeling of dynamically operated heterogeneously catalyzed reactions: Microkinetic model reduction and semi-mechanistic approach on the example of the CO2 methanation. Chem. Eng. J. 467, 143217 (2023).
Kresse, G. et al. Efficient iterative schemes for Ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B. 54, 11169 (1996).
Perdew, J. P. et al. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Kresse, G. et al. From ultrasoft pseudopotentials to the projector augmented-wave method. Phys. Rev. B. 59, 1758–1775 (1999).
Acknowledgements
This work was supported in part by the National Key Research and Development Program of China (2022YFE0113800), the National Natural Science Foundation of China (No. 22279115, 22122505, 22075250, 22208299), the Natural Science Foundation of Zhejiang Province (No. LBMHZ25B030005) and the Zhejiang Province Leading Earth Goose Program (2025c02224). The authors acknowledge Prof. Jianguo Wang’ Group for providing computational resources and technical support.
Author information
Authors and Affiliations
Contributions
J.T., H.C., X.L., Y.W., and Y.Z. conceived and designed the project. L.F., L.X., and Z.W. carried out the catalytic reactions and performed initial characterizations. L.F. conducted the X-ray related analyses (XRD, and XPS), DRIFTS studies, and temperature-programmed experiments. H.C. performed and analyzed the kinetic experiments. Q.F., L.X., and X.J. carried out electron microscopy measurements and analysis. N.O. and J.T. performed the DFT calculations. J.T. wrote the original manuscript. J.T., L.F., N.O., L.X., Z.W., H.C., Q.F., X.J., J.Z., M.T., L.Z., Y.W., X.L., and Y.Z. participated in discussing the results and revising the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Tian, J., Fang, L., Ouyang, N. et al. Breaking activity-selectivity-stability trade-offs in reverse water-gas shift reaction via high-energy micro-faceted Mo2N nanocrystals. Nat Commun 17, 2240 (2026). https://doi.org/10.1038/s41467-026-68756-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-026-68756-y