Abstract
Skin Neglected Tropical Diseases (skin NTDs) are a critical, underestimated source of chronic pathology, severely impacting vulnerable, rural populations with poor healthcare access. These diseases heighten the risk of systemic infection, amputation, and premature mortality. Their visibility and co-endemic nature present a pivotal opportunity for integrated diagnostics and management at the primary care level. Aligned with the WHO roadmap, this review advocates for a robust One Health framework, examining the complex interplay of human, animal, and environmental transmission factors. We contrast diseases with understood transmission against those requiring research. Leveraging the skin as a common pathway, we argue for strategic, integrated public health action to substantially improve patient outcomes globally.
Similar content being viewed by others
Introduction
Acute and chronic cutaneous wounds represent a significant global health burden, affecting millions annually and substantially contributing to disability1,2,3. Neglected tropical skin diseases (skin NTDs) comprise an underestimated but significant percentage of chronic cutaneous wounds and often affect people living in rural areas in low-income countries4. These pathologies, frequently compounded by inadequate healthcare access and infrastructure, significantly elevate the risk of consequential adverse outcomes, including secondary systemic infections, non-traumatic limb amputation, and premature mortality. However, the skin’s visibility offers a critical opportunity for integrated case detection and management, especially for skin NTDs, which often present with prominent lesions and are co-endemic in the same communities.
The stigma and mental distress attached to visible skin conditions often discourage patients from seeking care. Crucially, this health burden is exacerbated by severe financial barriers to care, including high out-of-pocket costs for diagnosis, treatment, and travel, often resulting in catastrophic expenditure and loss of income, which can pose a barrier to care sometimes greater than stigma itself 5,6 (Fig. 1). Furthermore, a lack of robust health surveillance systems and insufficient awareness among healthcare workers hinders early detection and effective intervention.
These diseases are characterized by visible skin changes, frequent co-existence in the same geographical areas, significant psychosocial impact, common diagnostic challenges, and a high burden in low- and middle-income countries, necessitating integrated approaches for management.
The WHO call to action: A need for integration
The World Health Organization (WHO) has underscored the need for an integrated approach to tackling skin NTDs, promoting coordinated efforts across different healthcare sectors. The 2021-2030 roadmap promotes innovative strategies, with a focus on early case detection and treatment, and this commitment culminated in a May 2025 resolution recognizing skin diseases as a global public health priority, urging member states to prioritize them through integrated efforts7,8.
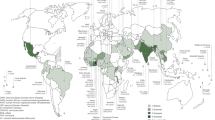
Despite this mandate, skin NTDs remain underreported, making it difficult to fully understand their true burden and transmission dynamics (Figs. 1, 2)9. Skin NTDs often occur alongside other burdensome diseases such as malaria, HIV, and tuberculosis, requiring a comprehensive strategy to effectively manage diagnosis and treatment in areas where healthcare resources are limited10.
This map shows the global distribution of countries that reported cases of at least one skin neglected tropical disease between 2017 and 2022, with the shading indicating the number of different diseases reported by each country. This figure was reproduced from reproduced from: Skin health for all: update on skin neglected tropical diseases with a focus on Buruli ulcer and yaws Weekly Epidemiological Record, 13 June 2025, vol. 100, 24/25 (pp. 239–250).
The World Health Organization (WHO) has recognized several NTDs with dermatological manifestations, such as onchocerciasis, cutaneous leishmaniasis, and leprosy4,11. Other skin NTDs include Buruli ulcer, lymphatic filariasis, mycetoma, tungiasis, yaws, chromoblastomycosis, sporotrichosis, scabies, and other ectoparasitic infections (Table 1)6. These conditions all present with visible clinical features, may be co-endemic, and many can be addressed using integrated strategies for case detection and disease management (Fig. 1).
This review examines the rationale for an integrated approach to skin NTDs, specifically through the lens of transmission complexity and the One Health framework. We highlight the urgent need to address critical knowledge gaps in transmission dynamics by fostering trans-disciplinary research. Finally, we propose a set of concrete recommendations necessary to translate One Health insights into actionable, effective public health strategies.
A One Health Approach
Transmission risk of many skin NTDs is complex and occurs at the nexus of landscape and aquatic ecosystem disturbances; pathogen ecological dynamics; food web interactions; human activity, movement, and behavior; and individual genetic and host factors. Many skin NTDs are zoonotic or influenced by environmental factors such as water quality and sanitation, soil composition, or spread through vectors that thrive in specific environmental conditions and interact with human activities12. For instance, leprosy, yaws, and cutaneous leishmaniasis, exhibit zoonotic or environmentally mediated transmission cycles that link humans, animals, and shared habitats, and non-human primates have been identified as potential reservoirs for Mycobacterium leprae and Treponema pallidum pertenue, the causative agents of leprosy and yaws, respectively, highlighting the role of wildlife in maintaining these pathogens outside the human population13,14,15.
However, there is often a broad lack of fundamental knowledge on specific transmission mechanisms or exposure rates that inhibit the development of effective prevention strategies. Measuring drivers of ecological and evolutionary interactions with humans, animals and the environment will identify biological processes generating disease occurrence patterns in space and time. These patterns can and do influence environmental persistence, colonization, virulence, and transmission heterogeneity. With this in mind, an integrated One Health approach through a trans-disciplinary perspective will undoubtedly provide a holistic understanding of the complex interdependencies of human, animal, microbial and ecosystem health--influencing transmission of skin NTD diseases: a key strategy for disease management breakthroughs.
The One Health framework recognizes the interconnectedness of human, animal, and environmental health, emphasizing that diseases and their associated conditions and symptoms arise from complex interactions16,17. A One Health approach also supports global health security by improving coordination, collaboration and communication at the human-animal-environment interface in addressing shared health threats such as zoonotic diseases, antimicrobial resistance, and food and agriculture safety. Therefore, adopting a One Health framework allows public health efforts to address the broader ecological and social factors affecting skin NTDs, promoting more sustainable disease control strategies. Incorporating these complex processes in transmission dynamic models can help inform policy and disease control interventions.
For several skin NTDs, such as onchocerciasis, lymphatic filariasis (LF), cutaneous leishmaniasis (CL), yaws, and scabies, the primary transmission mechanism is well-understood (i.e., vector-borne or person-to-person contact). However, even for these diseases, a deeper understanding is needed regarding strain heterogeneity, environmental co-factors influencing transmission rates (e.g., climate, land use), and the full extent of non-human reservoirs. This complexity necessitates increased investment in targeted transmission research within a One Health framework. In contrast, the transmission mechanisms for other skin NTDs, such as Buruli ulcer (BU), leprosy, mycetoma, chromoblastomycosis, and sporotrichosis, are not yet fully elucidated, complicating the development of effective, non-pharmaceutical control strategies.
Challenges with integration
Co-endemicity and co-morbidity
The simultaneous presence of multiple NTDs in the same geographic region complicates healthcare delivery in resource-limited settings, making it difficult to address specific conditions without addressing broader systemic health challenges18. The shared presence of skin NTDs however, provides an opportunity for integrating case detection and morbidity management, reducing the need for disease-specific interventions and streamlining healthcare delivery in these regions19.
This overlap necessitates a holistic approach, where healthcare providers consider all the diseases a patient may have, ensuring that diagnosis and treatment address the full spectrum of conditions. Failing to recognize these connections can lead to misdiagnosis, ineffective treatment, and the continued spread of diseases, further exacerbating the health and socioeconomic challenges faced by affected communities.
Training needs
Insufficient awareness among healthcare workers also delays early detection and treatment, underscoring the shortage of training and other resources in affected regions. Fundamental challenges for skin NTD programs include a lack of epidemiologic data to determine burden at national, let alone local levels and limited guidance on how to sustainably and equitably implement resource-intensive case detection and management interventions within primary healthcare services20. Strengthening surveillance systems is crucial for gathering reliable data, which can inform more effective public health strategies and improve early intervention efforts21. Given the complex issues surrounding skin NTDs—such as co-endemicity with other diseases, co-morbidity, underreporting, and environmental influences—an integrated, multi-sectoral approach is essential. Public health strategies should not only focus on treating individual diseases but also prioritize improving healthcare infrastructure, enhancing surveillance systems, and fostering cross-sector collaboration. By doing so, public health authorities can more effectively combat these widespread yet neglected public health challenges and improve the overall well-being of affected communities.
Transmission standard operating procedures
When considering the integration of skin NTDs, particularly from the perspective of designing protocols for detection, quantification, and prevention across co-endemic skin NTDs, several challenges need to be overcome. Particular challenges arise from heterogeneities in host-environment contact patterns, susceptibility and infectiousness of causative pathogens, environmental factors and climate change, and possibly pathogen phenotypes or genotypes. For instance, when assessing pathogen or parasite emergence, it will be necessary to consider those capable of sustaining human-to-human transmission, versus those that are transmitted due to direct environmental (reservoir or vector) contact, as abiotic and biotic factors driving emergence and persistence will likely be different between the two. An additional challenge includes the number of strains, and individual or shared hosts, intermediate hosts or other environmental reservoirs across skin NTDs. This may be particularly challenging when asking evolutionary questions. While it is not logistically or economically feasible to sample everything in all environments, integrating pathogen/parasite detection across environmental studies could be a way to overcome limitations and aid in the detection of host and environmental ranges.
New approaches for integrating skin NTD transmission models that take into account multiple infectious disease dynamics, epidemiology, and ecological parameters will most certainly have its challenges, but may also present a number of solutions. For instance, if infection occurs, it may be possible to incorporate ecological complexities of multiple reservoirs into models measuring the overall impact of environmental burden and ultimately to predict and understand risk.
Other considerations, particularly when considering skin NTDs, is the level of stigma associated with some of these diseases, where case reporting and case finding may be a limitation. It is in this area where integrated education and outreach will be incredibly important. And while integration should facilitate the development of more resilient health systems, there will need to be a high level of support at national and local levels of health systems for integrated approaches, rather than remaining limited by disease-specific program goals.
Skin NTDs with Known Transmission Routes
Onchocerciasis
Onchocerciasis, also known as river blindness, presents with a diverse array of dermatological and ocular manifestations. Clinical signs commonly present as intense pruritus, papular and/or lichenified dermatitis, and subcutaneous nodules (onchocercomata). Ocular involvement frequently presents as visual impairment, ranging from subtle optic nerve changes to irreversible blindness, contingent upon the chronicity and intensity of the parasitic load. The spectrum and severity of these clinical presentations are directly correlated with the stage of infection and the host’s immunological response to the parasite22. Onchocerciasis is caused when the filarial worm Onchocerca volvulus is transmitted by bite by infective Simulium (blackflies), that breed in fast-flowing rivers. The role of black flies in transmitting onchocerciasis was identified in the early 20th century has driven the implementation of several control and elimination programs with success in many regions and countries of the globe23,24.
Tungiasis
Tungiasis has a different mode of transmission since the insect itself is the parasite. The parasite is the female sand flea Tunga penetrans, and to a lesser extent T. trimamillata, transmitted through direct contact with the flea in infested soil25,26. The female flea penetrates host skin, embedding itself in the subcutaneous layer where it feeds on the host’s blood and grows rapidly as eggs develop, and are shed into the environment. Larvae, pupae and adult emergence take place in soil27. Multiple risk factor studies have found tungiasis to be associated with living in a house with an unpaved floor28,29,30, and the majority of soil samples containing off-host stages have been collected from the floors inside sleeping rooms27,31. A wide range of domestic and wild animal species can act as reservoirs maintaining the parasite in the environment32,33,34,35, but the cycle can also persist in homes without any animal involvement. Preventing flea penetration into host skin through protective footwear has been suggested as a preventative measure, but was ineffective in one trial36. Other prevention attempts have included application of coconut oil based insect repellent36,37, sealing of house floors38 and a combination of flooring, penning pigs, footwear use and environmental hygiene39. Integrating the control of Tungiasis into other skin NTD programs has yet to be attempted.
Scabies
Scabies is also a parasitic infection caused by an ectoparasite, Sarcoptes scabiei var. hominis, that burrows into the skin40. Itching is often severe and usually worse at night. The rash can appear in different colors depending on the patient skin tone, and can resemble hives, insect bites, or pimples. Scabies is highly transmissible and spreads person-to-person through direct skin contact with an infested individual or less commonly, by sharing bedding, clothes, or towels with someone who has the disease41. The risk of transmission is highest with individuals with crusted scabies, who may harbor thousands to millions of mites. Scabies is most common in low-income tropical areas, but occurs worldwide with children and older people in resource-poor areas are at higher risk.
Yaws
The etiological agent for Yaws is Treponema pallidum subspecies pertenue42. This bacterium causes chronic relapsing nonvenereal treponematosis characterized by highly contagious primary and secondary cutaneous lesions and noncontagious tertiary destructive lesions of the bones. While yaws is predominantly anthroponotic, non-human primates in certain regions (e.g., parts of West Africa) are known to be naturally infected with a genomically identical pathogen, acting as a potential zoonotic reservoir13. This primate reservoir represents a critical human-animal interface that must be factored into elimination strategies and highlights a key application of the One Health approach13. Yaws affects mostly children between 2 and 15 years old, who are considered reservoirs, and latent infection can occur after remaining asymptomatic for several years41,42.
Epidemiological evidence links the highest incidence of yaws within the tropics to seasonality with increased cases during rainy seasons43. This could be due to humid conditions creating an ideal environment for disease spread, leading to a higher prevalence of open, infectious lesions on the skin compared to drier seasons; or due to an increase in human contact during the rainy season (i.e., closed door environments), facilitating increased transmission through direct skin-to-skin contact with infected individuals.
Lymphatic filariasis
Lymphatic filariasis (LF), commonly known as elephantiasis, is a neglected tropical disease caused by parasitic filarial nematodes, primarily Wuchereria bancrofti, Brugia malayi, and B. timori. These parasites are transmitted to humans through the bites of infected mosquitoes, with key vector genera including Anopheles, Aedes, Culex, and Mansonia, varying by geographical region44,45. The transmission cycle is intricately linked to the nocturnal or diurnal periodicity of the parasite in the human host’s peripheral blood, maximizing the likelihood of vector uptake and subsequent spread46. While the understanding of LF transmission is robust, specific risk factors driving the chronic conditions like lymphedema and the formation of hydroceles remain less clear.
Despite widespread infection, only a subset of individuals develop overt clinical manifestations, which often emerge years after initial exposure, by which point many individuals test negative for circulating filarial antigens, complicating diagnosis. Emerging evidence suggests potential zoonotic reservoirs, particularly for Brugian species in primates and companion animals; however, their epidemiological significance in human transmission requires further investigation47,48. The integration of LF into the skin NTD framework is primarily driven by the dermatological implications of chronic disease, with management focusing on preventing lymphedema progression through rigorous hygiene, skin care, exercise, and elevation, and surgical intervention for hydrocele49,50.
Cutaneous and Post-kala-azar dermal Leishmaniasis
Leishmaniasis is caused by protozoan parasites from over 20 Leishmania species, transmitted through bites of infected female phlebotomine sandflies inhabiting forested or rural areas where humans may interact closely with natural reservoirs such as rodents or domestic animals51. The primary vectors are Phlebotomus and Lutzomyia sandflies, though there are over 90 sandfly species thought to transmit Leishmania parasites52. Sandfly vectors acquire Leishmania by feeding on infected animals or humans. Rodents, dogs, and other mammals can serve as reservoirs, allowing the parasites to be transmitted from animal to sandfly to human. In some cases, humans are the primary or only reservoir, and anthroponotic transmission of Leishmania can occur (e.g., L. tropica, and L. donovani)53. Infected sandflies inject promastigotes (the motile form of the parasite) into the skin as they bite. These promastigotes are then engulfed by macrophages (or other mononuclear phagocytic cells) and transform into amastigotes (the non-motile form), which multiply inside these cells, causing disease. Symptoms typically present weeks to months after the bite, though may be delayed for years. Intensive exposure to sandflies is a risk factor for being infected, and those with immunocompromise are at a higher risk of developing disease54. Though rare, transmission can also occur via blood transfusion or shared needles carrying infected blood55. Cutaneous leishmaniasis (CL) and post-kala-azar dermal leishmaniasis are both dermal forms of leishmaniasis but differ in origin and clinical presentation. Cutaneous leishmaniasis presents primarily as skin ulcers, while post-kala-azar dermal leishmaniasis is a manifestation of visceral leishmaniasis (also known as kala-azar), that may develop during or after treatment. Post-kala-azar dermal leishmaniasis may present as macules, papules, or nodules on the face, upper arms and trunk.
Skin NTDs whose transmission are not fully understood
Leprosy
Leprosy, caused by M. leprae and M. lepromatosis, is thought to be primarily transmitted through continuous close contact with an infected individual, particularly via respiratory droplets56. Evidence suggests that bacteria can enter the body via the respiratory tract or broken skin, resulting in disease. Although human-to-human transmission is considered the primary mechanism, environmental reservoirs may also contribute to disease persistence57,58. Mycobacterium leprae can persist in damp soil, water, or other environmental niches for extended periods of time, creating potential secondary sources of transmission59,60,61. Furthermore, several animal species, most notably armadillos in the Americas, red squirrels in Europe, and widely-distributed non-human primates, are known zoonotic reservoirs that may, in the case of the armadillo, be involved occasionally in the transfer of the disease to people15,56,62. The role, if any, of environmental and animal reservoirs in facilitating bacterial persistence and spread is still not understood, but a continuing environmental presence may hinder efforts to eradicate leprosy by allowing alternative ways for bacterial persistence and spread. A better understanding of animal and environmental reservoirs is needed, because transmission from such reservoirs might partly explain the persistence of leprosy despite effective and widespread surveillance and multidrug therapy.
Buruli ulcer
Buruli ulcer is a necrotizing skin disease caused by infection with M. ulcerans41. The bacterium produces a lipid toxin, mycolactone, responsible for disease pathology63. The exact transmission mechanisms of M. ulcerans are elusive and might differ between endemic areas. However, a large-scale case-control study reported by Combe et al.64 identified significant associations between Buruli ulcer and exposure to slow-flowing or stagnant water bodies, consistent with the hypothesis of waterborne transmission. Epidemiological studies have strengthened the evidence for environmental reservoirs and the bacterium persisting in aquatic environments, particularly in the water column, biofilms, algae and aquatic plants65,66. There is evidence that fish and amphibians also act as reservoirs carrying the bacteria in their skin or tissues67,68. Emerging evidence also suggests that climate change and land use alterations influence Buruli ulcer transmission in West Africa69. Direct human-to-human transmission is still considered rare or nonexistent. In African settings, insect bites, as well as deep cuts and scratches near or in water bodies, have been linked with an increased risk of infection and lab studies have shown inoculation through skin puncture or puncture of contaminated skin is sufficient for initiating disease pathology70,71,72. In southeastern Australia, possums play a crucial role as an animal reservoir that can sustain clinical disease and shed viable M. ulcerans through feces73 and there are multiple lines of evidence suggesting a role of mosquitoes in M. ulcerans transmission in Australia as either biological or mechanical vectors41,66,74. However, other publications suggest that alternative hypotheses are warranted to truly determine transmission of M. ulcerans to humans75,76. The WHO’s 2021 report on Buruli ulcer noted that children under 15 years old are disproportionately affected in many endemic regions, however, age distribution can vary by geographic location77. A systematic review documented by Tchatchouang, 2024 found that lower socioeconomic status, including poor housing conditions and limited access to clean water, was associated with increased Buruli ulcer risk69.
Mycetoma, chromoblastomycosis, and sporotrichosis
Mycetoma, chromoblastomycosis, and sporotrichosis are known to be acquired through implantation of the causative organism after trauma to the skin78. Although the route of acquisition is known, the geographical niches and distribution for most of the causative organisms are less well known, along with drivers of transmission and pathophysiology of the infections.
Mycetoma, a subcutaneous infection, is caused by the traumatic implantation of environmental bacteria (Actinomycetoma) and fungi (Eumycetoma) after a break in skin (e.g., following thorn pricks, minor trauma). Eumycetoma is more prevalent in arid regions with characteristic vegetation (e.g., Acacia bushes), such as in Sudan and Northern India79,80. Actinomycetoma seems to be mainly found in humid, tropical areas, such as in Mexico. However, these organisms are likely to have species-specific environmental and climatic niches which are not well-understood81. The frequency and distribution of the organism in the soil may increase the risk of transmission by providing higher chances of inoculation after minor injury although other factors such as the presence of cattle may contribute82. Mycetoma principally occurs in adults engaged in rural occupations such as agriculture, although anyone, including children, can be affected including children.
Chromoblastomycosis is an implantation mycosis that causes infection, usually in warm or tropical climates, after dematiaceous (black) fungi enter a break in the skin. The most common causes of chromoblastomycosis are Fonsecaea pedrosoi (most commonly reported), F. monophora, F. nubica, Cladophialophora carrionii, Rhinocladiella aquaspersa, and Phialophora verrucosa83; However, species distribution varies by region and climate, which are incompletely understood. Fonsecaea pedrosoi and F. nubica infections are commonly acquired in tropical humid regions, while C. carrionii infections are more common in semiarid regions suggesting these fungi are climate-sensitive84. Chromoblastomycosis may also be associated with natural disasters such as hurricanes or flooding85,86.
Chromoblastomycosis and its causative fungi have been associated with certain plants, including thorns of Mimosa pudica, several species of the Palmacea family, including Madagascar palm house plant, Jurubeba, Murta tree, Tucum tree, Vassourinha tree, Bacuri tree, and babassu coconut, along with hydrocarbon-polluted environments such as wood treated with phenolic preservatives, toxic mine waste, oil-polluted soils83. Some infections are associated with animals like birds, insect’s stings, and snakes. However, most inoculation sources or events are unknown, and the patient seldom recalls any injury let alone its source.
Sporotrichosis is another implantation mycosis usually acquired after skin lacerations such as scrapes from thorns or plant matter or from bites or scratches of infected animals, particularly cats. It is caused by environmental dimorphic fungi of the Sporothrix schenckii species complex: S. schenckii sensu stricto, S. brasiliensis, S. globosa, and S. luriei87. Sporothrix schenckii and S. globosa are commonly acquired after contact with plant matter and have been linked to outbreaks with plant matter such as sphagnum moss and corn stalks, although handling fish and working in gold mines have been implicated in outbreaks87. Climatic events such as flooding have also been linked to outbreaks88. Outside of outbreaks, little is known if Sporothrix species have preferential environmental conditions (e.g., soil characteristics, altitude, temperature) making it more suitable in this area, or if infection is more related to occupational and recreational activities.
The emergence of S. brasiliensis in Brazil since the late 1990s has shown that some forms of sporotrichosis are zoonotic. Animals, mainly felines, are serving as the vector for transmission from cats to humans and from cats to cats and causing an epidemic of disease. This species can be transmitted via bites, scratches, exudates, and even sneezes from infected cats89,90 Interestingly, whole genome sequencing has revealed that S. brasiliensis has emerged independently several times in Brazil, suggesting that felines may lead to the spread of this fungus, but may not be responsible for introducing it to the area91. The role of other animals (e.g., mice, armadillos) or the environment can be explored further to better prevent introduction in areas currently without disease92,93,94. In Argentina, where S. brasiliensis is spreading amongst cats, the species was first discovered in soil, although it is unknown what role soil played in the introduction of the fungus95.
Opportunities for modeling multiple diseases
Integrating models for policy and impact assessment
Mathematical models incorporating morbidity outcomes are crucial in guiding public health policy for skin NTDs. They assess the impact of interventions, screen protocols, and quantify cost-effectiveness using metrics like disability-adjusted life years (DALYs). Furthermore, models facilitate examination of how diseases interact with broader social determinants of health, including poverty and malnutrition, which may exacerbate the prevalence of skin NTDs96. A notable success is the predictive modeling for LF, which has informed effective mass drug administration strategies aimed at control97,98. However, for other skin NTDs, particularly those with complex or unknown transmission cycles like Buruli ulcer or mycetoma, modeling remains severely underutilized99. We strongly argue that a One Health framework must be leveraged to drive next-generation modeling by integrating socio-economic and, crucially, One Health data (animal reservoir distribution, ecological drivers, environmental persistence rates) to define the key transmission parameters, are necessary for building new, reliable transmission models. There is also a need to quantify the full spectrum of disease burden, including social stigma and the financial barriers (cost of illness, loss of productivity), to enable robust cost-effectiveness modeling. And finally, establishing standardized protocols for data collection will be imperative to enhance model validity and applicability, as predictions are contingent upon high-quality, standardized input data.
Enhancing model validity using standardized data protocols
As previously stated, the standardization of sample collection, preservation, and data reporting in skin NTD research is essential for enhancing the comparability of results and ensuring data quality100. As an example of success, laboratory networks such as BU LabNet and SkinNTD LabNet have played a pivotal role in developing and implementing standardized protocols for the diagnosis and monitoring of diseases, including Buruli ulcer, leprosy, and mycetoma101. These initiatives provide a common framework that guarantees consistency in procedures across laboratories and field settings.
BU LabNet is a global network dedicated to Buruli ulcer research, supported by the WHO. Its primary objective is to improve Buruli ulcer diagnosis through standardized polymerase chain reaction (PCR) testing protocols and external quality assessment programs. BU LabNet aims to broaden its molecular platform to incorporate other skin NTDs and laboratories, enhancing cost-effectiveness and expanding access to quality-assured diagnostic services101. By adhering to these protocols, researchers can reliably compare data across different regions, facilitating collaborative studies that inform global public health policy.
In 2023, BU LabNet evolved into SkinNTD LabNet. While the network currently plays a key role in advancing the diagnosis and research of skin NTDs in Africa, its ultimate goal is to harmonize diagnostic procedures globally for multiple skin NTDs, including yaws, cutaneous leishmaniasis, mycetoma, and leprosy. To support its expansion, the network provides laboratories with standardized PCR-based protocols, external quality assessments, and training for capacity-building. Crucially, future efforts must address the logistical barriers to global implementation. This includes developing sustainable methods for sample transport and cold-chain maintenance in challenging field conditions, which currently limit access to advanced testing outside of Africa and away from central laboratories. Expanding protocols to be practical and scalable across diverse global environments, including Asia, Latin America, and Oceania, will be necessary to foster the cross-regional collaboration required to address the global burden of skin NTDs effectively.
In addition to standardizing sample collection, these laboratory networks also establish uniform data reporting practices, which are important for building reliable epidemiological models and facilitate more comprehensive analyses and model validation, leading to better-informed public health interventions. Future efforts should focus on adapting these protocols to evolving field conditions to ensure their practicality and scalability across diverse environments. Standardized approaches not only enhance scientific rigor but also foster the cross-regional collaboration necessary to address the global burden of skin NTDs effectively.
While formalized laboratory networks have driven some success in standardized protocols of some diseases discussed above, modeling disease distribution and intervention impact of several other skin NTDs, including tungiasis, have been attempted but are undermined by the severe lack of data on prevalence and distribution29,102. There are no standardized protocols for surveys, clinical examinations, nor reporting. Facility-based records and information systems do not capture tungiasis, even if there is an indicator in the system, since cases rarely seek care at facilities due to the lack of suitable treatment options, poor access to facilities and stigma. Recommendations from a WHO informal meeting on tungiasis included identifying affected areas through key informants in community health networks, followed by school-based or community surveys103. Since tungiasis prevalence is highest among school-age children28,104, surveys could be integrated into school-based surveys of other diseases, such as scabies, schistosomiasis and soil-transmitted helminths, for which funding is available4,11. These types of interventions could equally serve other skin NTDs with similar challenges.
Predictive modeling using one health and environmental data
Beyond epidemiological case-control approaches to understanding the transmission of many skin NTDs, there has been increasing interest in employing strategies within the One Health concept105, using a multidisciplinary approach to understanding the disease agent biology, ecology and evolution to better predict how it is transmitted and causes pathogenesis, with the goal of identifying multiple areas of intervention to better manage disease. As an example, this approach was used for Chagas disease transmission in Guatemala with notable success in breaking transmission for at least 20 years106. Similarly, new environmental protocols could be developed within this concept to better inform future management of skin NTDs. There has been some effort to take a One Health/EcoHealth approach to understanding and managing skin NTDs and other NTDs107, but the current understanding of many of these conditions is limited by a deficit in inclusive, multidisciplinary research108. This research imperative is consistent with the WHO’s evolving policy, which champions an integrated approach to the control and management of skin NTDs109.
One major area of transmission research within the One Health/EcoHealth concept is integrating case mapping with advances in landscape cover technologies that allow for both simple and complex statistical and mathematical model development that can describe patterns of cases in space and time to better identify environmental associations of transmission107. Such landscape models can also incorporate host, pathogen and reservoir densities, how they are distributed and what factors induce changes that lead to increased transmission to humans. For instance, the damming of waterbodies has been shown to cause changes in vector populations like those associated with lymphatic filariasis. Using established methods of case and pathogen surveillance and detection, these multivariate models integrate formerly independent studies (e.g., human Buruli ulcer cases mapped by household and aquatic habitat surveys for its pathogen, M. ulcerans) into more comprehensive and quantitative methods of predicting where and how transmission may occur. By understanding these multiple dimensions of the disease system, scientists can identify areas to break the transmission chain or correct factors that may be leading to increased rates of pathogen/vector contact with hosts (e.g., deforestation107).
Integrating landscape and ecological mapping with data derived from case-control studies, allows for the placement of the pathogen within the broader scope of how humans use the space around them, what they come into contact with, how they interact with each other, what is changing around them to estimate what some have called exposomes, risk surfaces, or the living environment, and activities that expose people to agents of disease. The exposome has been investigated as important to many other skin ailments110,111,112 and having the ability to predict them for each of the skin NTDs would provide substantial advancements for achieving efficient and affordable management of them. Thus, there is a need for established environmental and epidemiological approaches for studying skin NTDs to be integrated with growing multidisciplinary collaborative efforts and for the resulting comprehensive datasets to be incorporated into predictive models to better understand and predict transmission.
Recommendations for integrated skin NTD control
Co-endemicity and similarities in skin NTD disability, stigma, and exacerbation of poverty highlight the essential need to integrate insights from many disciplines, including epidemiology, ecology, social sciences, public health and laboratory science at multiple scales to manage and control skin NTDs. This underscores the need for more focused and standardized field and laboratory studies to link disparate data, explore case studies, and define priorities in data collection, handling and reporting. There will likely be serious challenges arising from missing information – and from not adequately addressing these data gaps. At the same time, expanded sampling efforts and new technologies that leverage funding across multiple skin NTDs will bring a wealth of high-value data that must be analyzed with a focus on mechanistic principles and possible imbalances in sampling design. The long-term goal is to draw robust conclusions about risk across skin NTDs that will allow integrated management and control in resource limited environments.
Based on the necessity of integrated action and the demonstrated transmission knowledge gaps, we propose three core recommendations to champion the One Health approach and advance control strategies. First, we need to prioritize funding for integrated studies that link human cases with animal reservoirs (e.g., yaws in primates, M. ulcerans in possums) and environmental determinants (e.g., fungal niches for mycetoma, climate drivers for Buruli ulcer). The goal is to identify key parameters that allow for the development of targeted, non-pharmaceutical interventions. There is also a need to expand the model established by SkinNTD LabNet beyond Africa, focusing on global implementation of standardized diagnostic protocols. Specifically, attention must be paid to addressing the logistical challenges of sample transport and decentralized testing in resource-limited settings worldwide. Data reporting should also include standardized epidemiological, environmental, and socio-economic variables to ensure model validity. Finally, systematic data collection should be conducted on the financial barriers to care, loss of productivity, and social disability associated with all skin NTDs. This evidence is essential for robust cost-effectiveness modeling and policy advocacy, ensuring that morbidity management is adequately resourced and sustained.
Disclaimer
The findings and conclusions of this report are those of the authors and do not necessarily represent the official position of the Centers for Disease Control (CDC).
References
Hay, R. J. et al. The global burden of skin disease in 2010: an analysis of the prevalence and impact of skin conditions. J. Invest. Dermatol. 134, 1527–1534 (2014).
Giona, G., Cooper, L. & Butt, A. Changing Demographics and Disease Burden in Low Resource Countries. (Springer, Cham., 2024).
Martinengo, L. et al. Prevalence of chronic wounds in the general population: systematic review and meta-analysis of observational studies. Ann. Epidemiol. 29, 8–15 (2019).
Organization, W. H. (World Health Organization, 2020).
Hay, R. J. & Asiedu, K. Skin-related neglected tropical diseases (Skin NTDs)-A new challenge. Trop. Med. Infect. Dis. 4, https://doi.org/10.3390/tropicalmed4010004 (2018).
Chandler, D. J. & Fuller, L. C. The skin-A common pathway for integrating diagnosis and nanagement of NTDs. Trop. Med. Infect. Dis. 3, (2018).
Vieri, M. K., Logora, M. Y., Rafiq, K. & Colebunders, R. The World Health Organization road map for neglected tropical diseases 2021-2030: implications for onchocerciasis elimination programs. Infect. Dis. Poverty 10, 70 (2021).
Organization, W. H. Skin Diseases as a Global Public Health Priority. (2025).
Antonio Ruiz Postigo, J., Pathak, P. & Asiedu, K. Skin health for all: update on skin neglected tropical diseases with a focus on Buruli ulcer and yaws. 239–250 (World Health Organization, 2025).
Branda, F. et al. Assessing the burden of neglected tropical diseases in low-income communities: challenges and solutions. Viruses 17, 29 (2025).
Organization, W. H. (Geneva, Switzerland, 2020).
Threats, I. O. N. U. F. O. M. The Causes and Impacts of Neglected Tropical and Zoonotic Diseases. (National Academies Press, 2011).
Goldberg, T. L. et al. Yaws circulating in nonhuman primates, Uganda and Rwanda. Emerg. Infect. Dis. 31, 799–803 (2025).
Romero-Alvarez, D. et al. Mycobacterium leprae in Nine-Banded Armadillos (Dasypus novemcinctus), Ecuador. Emerg. Infect. Dis. 30, 2629–2632 (2024).
Honap, T. P. et al. Mycobacterium leprae genomes from naturally infected nonhuman primates. PLoS Negl. Trop. Dis. 12, e0006190 (2018).
Zinsstag, J. et al. Climate change and one health. FEMS Microbiol. Lett. 365, https://doi.org/10.1093/femsle/fny085 (2018).
Sinclair, J. R. Importance of a One Health approach in advancing global health security and the Sustainable Development Goals. Rev. Sci. Tech. 38, 145–154 (2019).
Hudu, S. A. et al. An insight into the Success, Challenges, and Future perspectives of eliminating Neglected tropical disease. Sci. African 24, e02165 (2024).
Barnowska, E. J. et al. Diagnosing skin neglected tropical diseases with the aid of digital health tools: A scoping review. PLOS Digit Health 3, e0000629 (2024).
Timothy, J. W. S. et al. Quantifying population burden and effectiveness of secentralized surveillance strategies for skin-presenting neglected tropical diseases, liberia. Emerg. Infect. Dis. 28, 1755–1764 (2022).
Yotsu, R. R. Integrated management of Skin NTDs-lessons learned from existing practice and field research. Trop. Med. Infect. Dis. 3, https://doi.org/10.3390/tropicalmed3040120 (2018).
Lakwo, T., Oguttu, D., Ukety, T., Post, R. & Bakajika, D. Onchocerciasis elimination: progress and challenges. Res. Rep. Trop. Med. 11, 81–95 (2020).
Crump, A., Morel, C. M. & Omura, S. The onchocerciasis chronicle: from the beginning to the end? Trends Parasitol. 28, 280–288 (2012).
Mackenzie, C. D., Homeida, M. M., Hopkins, A. D. & Lawrence, J. C. Elimination of onchocerciasis from Africa: possible? Trends Parasitol. 28, 16–22 (2012).
Eisele, M. et al. Investigations on the biology, epidemiology, pathology and control of Tunga penetrans in Brazil: I. Natural history of tungiasis in man. Parasitol. Res. 90, 87–99 (2003).
Pampiglione, S., Trentini, M., Fioravanti, M. L., Onore, G. & Rivasi, F. Additional description of a new species of Tunga (Siphonaptera) from Ecuador. Parasite 10, 9–15 (2003).
Nagy, N. et al. Investigations on the life cycle and morphology of Tunga penetrans in Brazil. Parasitol Res 101, S233–S242 (2007).
Muehlen, M. et al. Investigations on the biology, epidemiology, pathology and control of Tunga penetrans in Brazil. II. Prevalence, parasite load and topographic distribution of lesions in the population of a traditional fishing village. Parasitol Res. 90, 449–455 (2003).
Nyangacha, R. M. et al. Spatial distribution, prevalence and potential risk factors of Tungiasis in Vihiga County, Kenya. PLoS Negl. Trop. Dis. 13, e0007244 (2019).
Ugbomoiko, U. S., Ariza, L., Ofoezie, I. E. & Heukelbach, J. Risk factors for tungiasis in Nigeria: identification of targets for effective intervention. PLoS Negl. Trop. Dis. 1, e87 (2007).
Matharu, A. K. et al. Identification of tungiasis infection hotspots with a low-cost, high-throughput method for extracting Tunga penetrans (Siphonaptera) off-host stages from soil samples-An observational study. PLoS Negl. Trop. Dis. 18, e0011601 (2024).
Mutebi, F. et al. Animal Reservoirs of Zoonotic Tungiasis in Endemic Rural Villages of Uganda. PLoS Negl. Trop. Dis. 9, e0004126 (2015).
Frank, R. et al. Tunga penetrans and further parasites in the giant anteater (Myrmecophaga tridactyla) from Minas Gerais, Brazil. Parasitol. Res. 111, 1907–1912 (2012).
Heukelbach, J., Costa, A. M., Wilcke, T., Mencke, N. & Feldmeier, H. The animal reservoir of Tunga penetrans in severely affected communities of north-east Brazil. Med. Vet. Entomol. 18, 329–335 (2004).
Schott, D. et al. Clinical and pathological aspects of first report of Tunga penetrans infestation on southern brown howler monkey (Alouatta guariba clamitans) in Rio Grande do Sul, Brazil. J. Med. Primatol. 49, 315–321 (2020).
Thielecke, M. et al. Prevention of tungiasis and tungiasis-associated morbidity using the plant-based repellent Zanzarin: a randomized, controlled field study in rural Madagascar. PLoS Negl. Trop. Dis. 7, e2426 (2013).
Buckendahl, J. et al. Control of tungiasis through intermittent application of a plant-based repellent: an intervention study in a resource-poor community in Brazil. PLoS Negl. Trop. Dis. 4, e879 (2010).
Elson, L., Nyawa, S. M., Matharu, A. & Fillinger, U. Developing low-cost house floors to control tungiasis in Kenya - a feasibility study. BMC Public Health 23, 2483 (2023).
Heukelbach, J., Ariza, L., Adegbola, R. Q. & Ugbomoiko, U. S. Sustainable control of tungiasis in rural Nigeria: a case for One Health. One Health & Implement. Res. 1, 4–13 (2021).
Arlian, L. G. & Morgan, M. S. A review of Sarcoptes scabiei: past, present and future. Parasites Vectors 10, 297 (2017).
Röltgen, K. & Pluschke, G. in Buruli Ulcer: Mycobacterium Ulcerans Disease (eds Gerd Pluschke & Katharina Röltgen) 1–41 (Springer International Publishing, 2019).
Maxfield, L. & Corley, J., Crane, J. S. in StatPearls [Internet] (StatPearls Publishing, 2023).
Vicar, E. K., Simpson, S. V., Mensah, G. I., Addo, K. K. & Donkor, E. S. Yaws in Africa: Past, Present and Future. Diseases 13, 14 (2025).
Bhuvaneswari, A., Shriram, A. N., Raju, K.H.K. & Kumar, A. Mosquitoes, lymphatic filariasis, and public health: A systematic review of anopheles and aedes surveillance strategies. Pathogens 12, 1406 (2023).
de Souza, D. K. et al. Diversity and transmission competence in lymphatic filariasis vectors in West Africa, and the implications for accelerated elimination of Anopheles-transmitted filariasis. Parasit Vectors 5, 259 (2012).
Moulia-Pelat, J. P. et al. Periodicity of Wuchereria bancrofti var. pacifica filariasis in French Polynesia. Trop. Med. Parasitol. 44, 83–85 (1993).
Mallawarachchi, C. H. et al. A preliminary survey of filarial parasites in dogs and cats in Sri Lanka. PLoS ONE 13, e0206633 (2018).
Evans, C. C., Pilotte, N. & Moorhead, A. R. Current status of the diagnosis of Brugia spp. Infections. Pathogens 13, 714 (2024).
Stocks, M. E., Freeman, M. C. & Addiss, D. G. The effect of hygiene-based lymphedema management in lymphatic filariasis-endemic areas: a systematic review and meta-analysis. PLoS Negl. Trop. Dis. 9, e0004171 (2015).
Beard, J. H., Ohene-Yeboah, M., devries, C. R. & Schecter, W. P. in Essential Surgery: Disease Control Priorities, Third Edition (Volume 1) (eds H. T. Debas) et al. (The International Bank for Reconstruction and Development / The World Bank© 2015 International Bank for Reconstruction and Development / The World Bank., 2015).
Yotsu, R. R. et al. A global call for action to tackle skin-related neglected tropical diseases (skin NTDs) through integration: An ambitious step change. PLoS Negl. Trop. Dis. 17, e0011357 (2023).
de Vries, H. J. C. & Schallig, H. D. Cutaneous leishmaniasis: A 2022 updated narrative review into diagnosis and management developments. Am. J. Clin. Dermatol. 23, 823–840 (2022).
Akhoundi, M. et al. A historical overview of the classification, evolution, and dispersion of Leishmania parasites and sandflies. PLoS Negl. Trop. Dis. 10, e0004349 (2016).
Scott, P. & Novais, F. O. Cutaneous leishmaniasis: immune responses in protection and pathogenesis. Nat. Rev. Immunol. 16, 581–592 (2016).
Perales-Gonzalez, A., Perez-Garza, D. M., Garza-Davila, V. F. & Ocampo-Candiani, J. Cutaneous leishmaniasis by a needlestick injury, an occupational infection? PLoS Negl. Trop. Dis. 17, e0011150 (2023).
Alrehaili, J. Leprosy classification, clinical features, epidemiology, and host immunological responses: failure of eradication in 2023. Cureus 15, e44767 (2023).
Ploemacher, T., Faber, W. R., Menke, H., Rutten, V. & Pieters, T. Reservoirs and transmission routes of leprosy; A systematic review. PLoS Negl. Trop. Dis. 14, e0008276 (2020).
Bratschi, M. W., Steinmann, P., Wickenden, A. & Gillis, T. P. Current knowledge on Mycobacterium leprae transmission: a systematic literature review. Lepr. Rev. 86, 142–155 (2015).
Arraes, M. et al. Natural environmental water sources in endemic regions of northeastern Brazil are potential reservoirs of viable Mycobacterium leprae. Mem. Inst. Oswaldo Cruz 112, 805–811 (2017).
Lavania, M. et al. Detection of viable Mycobacterium leprae in soil samples: insights into possible sources of transmission of leprosy. Infect. Genet. Evol. 8, 627–631 (2008).
Turankar, R. P., Lavania, M., Singh, M., Siva Sai, K. S. & Jadhav, R. S. Dynamics of Mycobacterium leprae transmission in environmental context: deciphering the role of environment as a potential reservoir. Infect. Genet. Evol. 12, 121–126 (2012).
Rojas-Espinosa, O. & Løvik, M. Mycobacterium leprae and Mycobacterium lepraemurium infections in domestic and wild animals. Rev. Sci. Tech. 20, 219–251 (2001).
George, K. M. et al. Mycolactone: a polyketide toxin from Mycobacterium ulcerans required for virulence. Science 283, 854–857 (1999).
Combe, M. et al. Global and local environmental changes as drivers of Buruli ulcer emergence. Emerg. Microbes Infect. 6, e21 (2017).
Narh, C. A. et al. Source tracking Mycobacterium ulcerans infections in the Ashanti region, Ghana. PLoS Negl. Trop. Dis. 9, e0003437 (2015).
Dhungel, L., Benbow, M. E. & Jordan, H. R. Linking the Mycobacterium ulcerans environment to Buruli ulcer disease: Progress and challenges. One Health 13, 100311 (2021).
Willson, S. J. et al. Fish and amphibians as potential reservoirs of Mycobacterium ulcerans, the causative agent of Buruli ulcer disease. Infect. Ecol. Epidemiol. 3, https://doi.org/10.3402/iee.v3i0.19946 (2013).
Mosi, L. et al. Mycobacterium ulcerans causes minimal pathogenesis and colonization in medaka (Oryzias latipes): an experimental fish model of disease transmission. Microbes Infect. 14, 719–729 (2012).
Tchatchouang, S. et al. Systematic review: Global host range, case fatality and detection rates of Mycobacterium ulcerans in humans and potential environmental sources. J. Clin. Tuberc. Other Mycobact. Dis. 36, 100457 (2024).
Maman, I. et al. Risk factors for Mycobacterium ulcerans infection (Buruli Ulcer) in Togo ─ a case-control study in Zio and Yoto districts of the maritime region. BMC Infect. Dis. 18, 48 (2018).
Williamson, H. R. et al. Mycobacterium ulcerans fails to infect through skin abrasions in a guinea pig infection model: implications for transmission. PLoS Negl. Trop. Dis. 8, e2770 (2014).
Wallace, J. R. et al. Mycobacterium ulcerans low infectious dose and mechanical transmission support insect bites and puncturing injuries in the spread of Buruli ulcer. PLoS Negl. Trop. Dis. 11, e0005553 (2017).
McNamara, B. J. et al. Comprehensive case-control study of protective and risk factors for Buruli Ulcer, Southeastern Australia. Emerg. Infect. Dis. 29, 2032–2043 (2023).
Mee, P. T. et al. Mosquitoes provide a transmission route between possums and humans for Buruli ulcer in southeastern Australia. Nat. Microbiol. 9, 377–389 (2024).
Receveur, J. P. et al. A need for null models in understanding disease transmission: the example of Mycobacterium ulcerans (Buruli ulcer disease). FEMS Microbiol. Rev. 46, https://doi.org/10.1093/femsre/fuab045 (2022).
Guégan, J.-F. et al. Canonical fact versus hypothesis testing to decipher transmission of non-tuberculous and tuberculous mycobacteria: a comparative review. Clin. Microbiol. Rev. 0, e00228–00224 (2025).
Amofah, G. et al. Buruli ulcer in Ghana: results of a national case search. Emerg Infect. Dis. 8, 167–170 (2002).
Smith, D. J. et al. South-East Asia regional neglected tropical disease framework: improving control of mycetoma, chromoblastomycosis, and sporotrichosis. Lancet Reg. Health Southeast Asia 35, 100561 (2025).
Hassan, R. et al. Modelling the spatial distribution of mycetoma in Sudan. Trans. R. Soc. Trop. Med. Hyg. 115, 1144–1152 (2021).
Bakshi, R. & Mathur, D. R. Incidence and changing pattern of mycetoma in western Rajasthan. Indian J. Pathol. Microbiol. 51, 154–155 (2008).
Hao, X., Cognetti, M., Burch-Smith, R., Mejia, E. O. S. & Mirkin, G. Mycetoma: Development of Diagnosis and Treatment. J. Fungi 8, 743 (2022).
Hashizume, H. et al. Environmental detection of eumycetoma pathogens using multiplex real-time PCR for soil DNA in Sennar State, Sudan. Trop. Med. Health 51, 71 (2023).
Smith, D. J. et al. A global chromoblastomycosis strategy and development of the global chromoblastomycosis working group. PLoS Negl. Trop. Dis. 18, e0012562 (2024).
Gadre, A., Enbiale, W., Andersen, L. K. & Coates, S. J. The effects of climate change on fungal diseases with cutaneous manifestations: A report from the International Society of Dermatology Climate Change Committee. J. Clim. Change Health 6, 100156 (2022).
Bandino, J. P., Hang, A. & Norton, S. A. The infectious and noninfectious dermatological consequences of flooding: A field manual for the responding provider. Am. J. Clin. Dermatol. 16, 399–424 (2015).
Riddel, C. E. et al. Fungal foes: presentations of chromoblastomycosis post-hurricane Ike. Cutis 87, 269–272 (2011).
Marimon, R. et al. Sporothrix brasiliensis, S. globosa, and S. mexicana, Three New Sporothrix Species of Clinical Interest. J. Clin. Microbiol. 45, 3198–3206 (2007).
Song, Y. et al. Report of 457 sporotrichosis cases from Jilin province, northeast China, a serious endemic region. J. Eur. Acad. Dermatol. Venereol. 27, 313–318 (2013).
Almeida-Paes, R. et al. The present and future research agenda of sporotrichosis on the silver anniversary of zoonotic sporotrichosis in Rio de Janeiro, Brazil. Mem. Inst. Oswaldo Cruz 119, e230208 (2024).
de Andrade Galliano Daros Bastos, F. et al. Spread of Sporothrix spp. through respiratory droplets from infected cats: A potential route of transmission. Med. Mycol. 60, https://doi.org/10.1093/mmy/myac079 (2022).
Ribeiro dos Santos, A. et al. Emergence of zoonotic sporotrichosis in Brazil: a genomic epidemiology study. Lancet Microbe 5, e282–e290 (2024).
Baes Pereira, S. et al. Sporotrichosis in dogs: epidemiological and clinical-therapeutic profile and the emergence of itraconazole-resistant isolates. Med. Mycol. 60, https://doi.org/10.1093/mmy/myac089 (2022).
Cabeza, E. et al. Clinical and epidemiological characteristics of Sporotrichosis in a Reference Center of Uruguay. J. Fungi 8, 322 (2022).
Moore, J. J. & Davis, D. J. Sporotrichosis following Mouse Bite with Certain Immunologic Data. J. Infect. Dis. 23, 252–265 (1918).
Córdoba, S. et al. Molecular identification and susceptibility profile of Sporothrix schenckii sensu lato isolated in Argentina. Mycoses 61, 441–448 (2018).
George, N. S. et al. Addressing neglected tropical diseases in Africa: a health equity perspective. Glob. Health Res. Policy 8, 30 (2023).
Borlase, A., Prada, J. M. & Crellen, T. Modelling morbidity for neglected tropical diseases: the long and winding road from cumulative exposure to long-term pathology. Philos. Trans. R Soc. Lond. B Biol. Sci. 378, 20220279 (2023).
Shaw, C., McLure, A., Graves, P. M., Lau, C. L. & Glass, K. Lymphatic filariasis endgame strategies: Using GEOFIL to model mass drug administration and targeted surveillance and treatment strategies in American Samoa. PLoS Negl. Trop. Dis. 17, e0011347 (2023).
Sharma, Y. et al. Models and data used to predict the abundance and distribution of Ixodes scapularis (blacklegged tick) in North America: a scoping review. Lancet Reg. Health Am. 32, 100706 (2024).
Toor, J. et al. Strengthening data collection for neglected tropical diseases: What data are needed for models to better inform tailored intervention programmes? PLoS Negl. Trop. Dis. 15, e0009351 (2021).
Marion, E. et al. A combined effort of 11 laboratories in the WHO African region to improve quality of Buruli ulcer PCR diagnosis: The BU-LABNET. PLoS Negl. Trop. Dis. 16, e0010908 (2022).
Girma, A. & Abdu, I. Prevalence and Risk Factors of Tungiasis Among Selected Regions of Ethiopia: A Meta-Analysis. Health Sci. Rep. 8, e71233 (2025).
Organization, W. H. Report of a WHO informal meeting on the development of a conceptual framework for tungiasis control: virtual meeting, 11–13 January 2021. (World Health Organization, 2021).
Wiese, S., Elson, L., Reichert, F., Mambo, B. & Feldmeier, H. Prevalence, intensity and risk factors of tungiasis in Kilifi County, Kenya: I. Results from a community-based study. PLoS Negl. Trop. Dis. 11, e0005925 (2017).
Organization, W. H. (2025).
Pereira, F. M., Penados, D., Dorn, P. L., Alcántara, B. & Monroy, M. C. The long-term impact of an Ecohealth intervention: Entomological data suggest the interruption of Chagas disease transmission in southeastern Guatemala. Acta Tropica 235, 106655 (2022).
Guégan, J. F. et al. Emerging infectious diseases and new pandemics: dancing with a ghost! Lessons in inter- and transdisciplinary research in French Guiana, South America. Int. J. Infect. Dis. 133, 9–13 (2023).
Chevillon, C. et al. Ecological and evolutionary perspectives advance understanding of mycobacterial diseases. Lancet Microbe 5, 100906 (2024).
Mitjà, O. et al. Integrated Control and Management of Neglected Tropical Skin Diseases. PLoS Negl. Trop. Dis. 11, e0005136 (2017).
Cecchi, L., D’Amato, G. & Annesi-Maesano, I. External exposome and allergic respiratory and skin diseases. J Allergy Clin. Immunol. 141, 846–857 (2018).
Molina-García, M., Granger, C., Trullàs, C. & Puig, S. Exposome and Skin: Part 1. Bibliometric Analysis and Review of the Impact of Exposome Approaches on Dermatology. Dermatol. Ther. 12, 345–359 (2022).
Molina-García, M. et al. Exposome and Skin. Part 2. The Influential Role of the Exposome, Beyond UVR, in Actinic Keratosis, Bowen’s Disease and Squamous Cell Carcinoma: A Proposal. Dermatol. Ther. 12, 361–380 (2022).
Acknowledgements
We thank the WHO NTD department for map production. Additionally, we appreciate the assistance of Mr. Yorm Edem Doe and Miss Susanna Apraku for their help in organizing literature resources. HJ is supported, in part, by NSF Industry University Cooperative Research Center grant 2052788 and USDA NIFA grant 2023-67019-39248, and H.J. and M.E.B. were supported, in part, by the NSF-NIH-NIFA Ecology and Evolution of Infectious Disease program [DEB #1911457]; any opinions, findings, and conclusions or recommendations expressed in this material are those of the author(s) and do not necessarily reflect the views of the National Science Foundation.
Author information
Authors and Affiliations
Contributions
H.J. and L.M. conceived and coordinated the conceptual framework for the manuscript. H.J., L.M., B.A., K.A., A.B., M.E.B., D.D., L.E., A.F., R.H., R.N., C.Q., A.S., and D.J.S. all contributed sections of the paper and/or critically reviewed, edited, and refined the various drafts of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Mosi, L., Acharya, B., Asiedu, K. et al. Integrated action for skin NTDs: Deconstructing transmission, addressing knowledge gaps, and championing one health strategies. Nat Commun 17, 2271 (2026). https://doi.org/10.1038/s41467-026-69065-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-026-69065-0