Fig. 1: Engineered SynTag-Tau undergoes phase separation coupled to fibril formation.
From: Decoupling phase separation and fibrillization preserves activity of biomolecular condensates
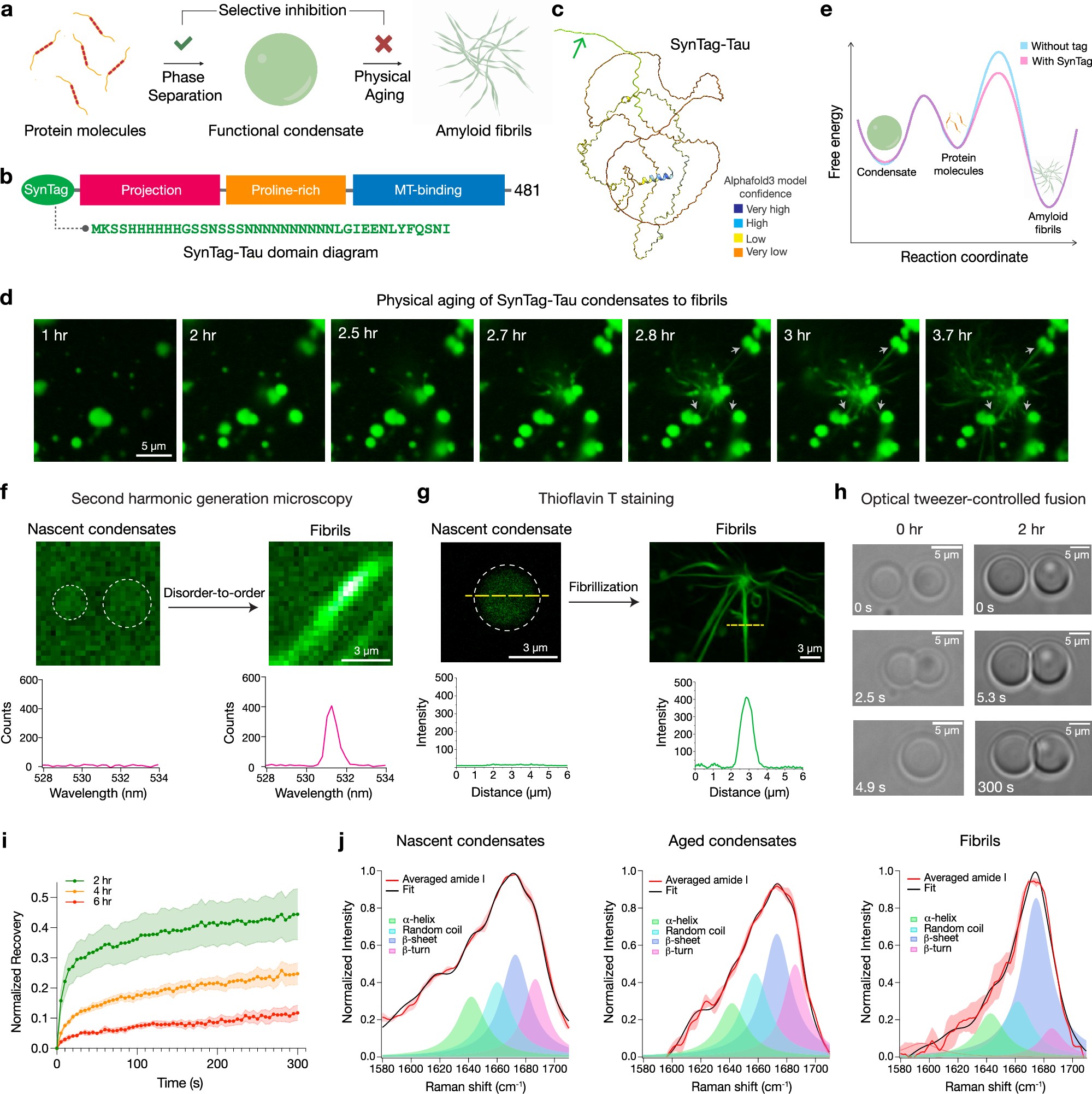
a Schematic depicting the formation of phase-separated protein condensates and their time-dependent transition to form amyloid fibrils. b Domain diagram of SynTag-Tau consisting of full-length Tau (2N4R isoform) with an N-terminal synthetic prionogenic tag (SynTag; sequence provided in green). c Alphafold368 predicted structure of SynTag-Tau. The green arrow marks the location of the tag. d Time-lapse imaging of Atto488-labeled SynTag-Tau condensates. Grey arrows indicate the attachment of fibrils emerging from one condensate to neighboring condensates. Also, see Supplementary Video 1. e Schematic of free energy landscape illustrating the relative stability of monomeric Tau, liquid-like Tau condensates, and amyloid fibrils, either with or without SynTag. f Second harmonic generation (SHG) microscopy shows the absence of molecular ordering in nascent SynTag-Tau condensates (indicated by dashed circles; sample age = 2 h) and the presence of spatial ordering in fibrils (sample age = 10 h). The corresponding quantification is also shown. g Representative Thioflavin T (ThT) fluorescence line profiles from a nascent SynTag-Tau condensate (indicated by a dashed circle; sample age = 2 h) and fibrils (sample age = 10 h). The contrast was adjusted independently in order to better visualize the nascent condensate that is very dim. h Optical tweezer-controlled fusion of nascent SynTag-Tau condensates (sample age = 30 mins, indicated as the 0-h time point) and condensates at 2 h of age. i FRAP measurements of SynTag-Tau condensates at various time points as indicated. The individual dots represent the mean value at each time point, based on data from at least three independent replicates (sample size: 2 h, 4 droplets; 4 h and 6 h, 5 droplets each). Intensity error bars were plotted based on the standard error of the mean (S.E.M.) at each time point and are represented as the shaded regions. j Peak fitting of averaged amide I spectra obtained from broadband coherent anti-Stokes Raman (BCARS) hyperspectral imaging shows the structural profile of SynTag-Tau in condensates at 2 h (nascent condensates) and at 6 h (aged condensates), as well as fibrils formed at 10 h of age. Spectra from multiple samples within each independent experiment (n ≥ 6 per experiment) were averaged prior to analysis. Data are presented as mean values with S.E.M. Each experiment was independently repeated at least three times. Source data are provided as a Source Data file.
