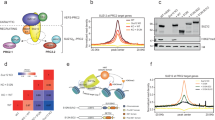
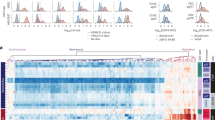
Abstract
Discriminating regulatory functions of chromatin composition from those of chromatin-modifying complexes is a central problem in gene regulation. This question remains unexplored in the context of histone variants and their dedicated chromatin remodelers. Here we dissect the distinct and cell cycle-dependent functions of Snf2 Related CREBBP Activator Protein (SRCAP) and H2A.Z in gene regulation of pluripotent stem cells. Using acute degradation of endogenous SRCAP, we uncover dynamic changes of H2A.Z occupancy and continuous requirement of SRCAP over the cell cycle. We also engineered an SRCAP mutant, defective for H2A.Z deposition, allowing us to distinguish H2A.Z-dependent and independent functions of SRCAP. We discover that SRCAP exhibits essential H2A.Z-independent functions in inhibiting DNA binding of dozens of pioneer transcription factors at enhancers by steric hindrance. In contrast, H2A.Z acts mainly as a transcriptional repressor gatekeeping the expression of lineage-specific genes. Our study establishes the catalytic-independent role of a chromatin remodeler in broadly regulating transcription factor binding, and demonstrates how a chromatin remodeler-histone variant pair orchestrates transcription to maintain self-renewal and plasticity of pluripotent stem cells.
Similar content being viewed by others
Data availability
All sequencing data were deposited at NCBI Gene Expression Omnibus (GEO), accession number GSE269310 [https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE269305]. Proteomics data were deposited at ProteomeXchange, accession number PXD052934. Source Data have been deposited at Zenodo, under https://doi.org/10.5281/zenodo.18161555.
References
Ibarra-Morales, D. et al. Histone variant H2A.Z regulates zygotic genome activation. Nat. Commun. 12, 7002 (2021).
Hickey, G. J. et al. Establishment of developmental gene silencing by ordered polycomb complex recruitment in early zebrafish embryos. eLife 11, e67738 (2022).
Liu, X. et al. Hierarchical accumulation of histone variant H2A.Z regulates transcriptional states and histone modifications in early mammalian embryos. Adv. Sci. 9, 2200057 (2022).
Matsuda, R. et al. Identification and characterization of the two isoforms of the vertebrate H2A.Z histone variant. Nucleic Acids Res. 38, 4263–4273 (2010).
Hu, G. et al. H2A.Z facilitates access of active and repressive complexes to chromatin in embryonic stem cell self-renewal and differentiation. Cell Stem Cell 12, 180–192 (2013).
Yu, H. et al. INO80 promotes H2A.Z occupancy to regulate cell fate transition in pluripotent stem cells. Nucleic Acids Res. 49, 6739–6755 (2021).
Zhao, B. et al. Znhit1 controls intestinal stem cell maintenance by regulating H2A.Z incorporation. Nat. Commun. 10, 1071 (2019).
Wong, M. M., Cox, L. K. & Chrivia, J. C. The chromatin remodeling protein, SRCAP, is critical for deposition of the histone variant H2A.Z at promoters. J. Biol. Chem. 282, 26132–26139 (2007).
Gévry, N., Chan, H. M., Laflamme, L., Livingston, D. M. & Gaudreau, L. p21 transcription is regulated by differential localization of histone H2A.Z. Genes Dev. 21, 1869–1881 (2007).
Dijkwel, Y. & Tremethick, D. J. The role of the histone variant H2A.Z in metazoan development. J. Dev. Biol. 10, 28 (2022).
Johnston, H., Kneer, J., Chackalaparampil, I., Yaciuk, P. & Chrivia, J. Identification of a novel SNF2/SWI2 protein family member, SRCAP, which interacts with CREB-binding protein. J. Biol. Chem. 274, 16370–16376 (1999).
Xu, X. et al. Adenovirus DNA binding protein interacts with the SNF2-related CBP activator protein (SrCap) and inhibits SrCap-mediated transcription. J. Virol. 75, 10033–10040 (2001).
Ye, B. et al. The chromatin remodeler SRCAP promotes self-renewal of intestinal stem cells. EMBO J. 39, e103786 (2020).
Creyghton, M. P. et al. H2AZ Is enriched at polycomb complex target genes in ES cells and is necessary for lineage commitment. Cell 135, 649–661 (2008).
Ku, M. et al. H2A.Z landscapes and dual modifications in pluripotent and multipotent stem cells underlie complex genome regulatory functions. Genome Biol. 13, R85 (2012).
Murphy, K. E., Meng, F. W., Makowski, C. E. & Murphy, P. J. Genome-wide chromatin accessibility is restricted by ANP32E. Nat. Commun. 11, 5063 (2020).
Berta, D. G. et al. Deficient H2A.Z deposition is associated with genesis of uterine leiomyoma. Nature 596, 398–403 (2021).
Rudnizky, S., Khamis, H., Malik, O., Melamed, P. & Kaplan, A. The base pair-scale diffusion of nucleosomes modulates binding of transcription factors. Proc. Natl. Acad. Sci. USA 116, 12161–12166 (2019).
Pünzeler, S. et al. Multivalent binding of PWWP2A to H2A.Z regulates mitosis and neural crest differentiation. EMBO J. 36, 2263–2279 (2017).
Mylonas, C., Lee, C., Auld, A. L., Cisse, I. I. & Boyer, L. A. A dual role for H2A.Z.1 in modulating the dynamics of RNA polymerase II initiation and elongation. Nat. Struct. Mol. Biol. 28, 435–442 (2021).
Wen, Z., Zhang, L., Ruan, H. & Li, G. Histone variant H2A.Z regulates nucleosome unwrapping and CTCF binding in mouse ES cells. Nucleic Acids Res. 48, 5939–5952 (2020).
Cole, L. et al. Multiple roles of H2A.Z in regulating promoter chromatin architecture in human cells. Nat. Commun. 12, 2524 (2021).
Lukauskas, S. et al. Decoding chromatin states by proteomic profiling of nucleosome readers. Nature 627, 671–679 (2024).
Shen, T. et al. Brain-specific deletion of histone variant H2A.z results in cortical neurogenesis defects and neurodevelopmental disorder. Nucleic Acids Res. 46, 2290–2307 (2018).
Greenberg, R. S., Long, H. K., Swigut, T. & Wysocka, J. Single amino acid change underlies distinct roles of H2A.Z subtypes in human syndrome. Cell 178, 1421–1436.e24 (2019).
Zilberman, D., Coleman-Derr, D., Ballinger, T. & Henikoff, S. Histone H2A.Z and DNA methylation are mutually antagonistic chromatin marks. Nature 456, 125–129 (2008).
Conerly, M. L. et al. Changes in H2A.Z occupancy and DNA methylation during B-cell lymphomagenesis. Genome Res. 20, 1383–1390 (2010).
Bagchi, D. N., Battenhouse, A. M., Park, D. & Iyer, V. R. The histone variant H2A.Z in yeast is almost exclusively incorporated into the +1 nucleosome in the direction of transcription. Nucleic Acids Res. 48, 157–170 (2020).
Bowman, T. A., Wong, M. M., Cox, L. K., Baldassare, J. J. & Chrivia, J. C. Loss of H2A.Z is not sufficient to determine transcriptional activity of Snf2-related CBP activator protein or p400 complexes. Int. J. Cell Biol. 2011, e715642 (2011).
Nishimura, K., Fukagawa, T., Takisawa, H., Kakimoto, T. & Kanemaki, M. An auxin-based degron system for the rapid depletion of proteins in nonplant cells. Nat. Methods 6, 917–922 (2009).
Raccaud, M. et al. Mitotic chromosome binding predicts transcription factor properties in interphase. Nat. Commun. 10, 487 (2019).
Patty, B. J., Dhungana, R. & Hainer, S. J. Widespread impact of nucleosome remodelers on transcription at cis-regulatory elements. Cell Rep. 44, 115767 (2025).
King, H. W. & Klose, R. J. The pioneer factor OCT4 requires the chromatin remodeller BRG1 to support gene regulatory element function in mouse embryonic stem cells. eLife 6, e22631 (2017).
Friman, E. T. et al. Dynamic regulation of chromatin accessibility by pluripotency transcription factors across the cell cycle. eLife 8, e50087 (2019).
Niwa, H., Miyazaki, J. & Smith, A. G. Quantitative expression of Oct-3/4 defines differentiation, dedifferentiation or self-renewal of ES cells. Nat. Genet. 24, 372–376 (2000).
Pundhir, S. et al. The impact of SWI/SNF and NuRD inactivation on gene expression is tightly coupled with levels of RNA polymerase II occupancy at promoters. Genome Res. 33, 332–345 (2023).
Ranjan, A. et al. Live-cell single particle imaging reveals the role of RNA polymerase II in histone H2A.Z eviction. eLife 9, e55667 (2020).
Placzek, S., Vanzan, L., Deluz, C. & Suter, D. M. Orchestration of pluripotent stem cell genome reactivation during mitotic exit. Cell Rep. 44, 115486 (2025).
Kelly, T. K. et al. H2A.Z maintenance during mitosis reveals nucleosome shifting on mitotically silenced genes. Mol. Cell 39, 901–911 (2010).
Nekrasov, M. et al. Histone H2A.Z inheritance during the cell cycle and its impact on promoter organization and dynamics. Nat. Struct. Mol. Biol. 19, 1076–1083 (2012).
Giaimo, B. D., Ferrante, F., Herchenröther, A., Hake, S. B. & Borggrefe, T. The histone variant H2A.Z in gene regulation. Epigenetics Chromatin 12, 37 (2019).
Palozola, K. C. et al. Mitotic transcription and waves of gene reactivation during mitotic exit. Science 358, 119–122 (2017).
Gaidatzis, D., Burger, L., Florescu, M. & Stadler, M. B. Analysis of intronic and exonic reads in RNA-seq data characterizes transcriptional and post-transcriptional regulation. Nat. Biotechnol. 33, 722–729 (2015).
Yang, Y. et al. HIRA complex presets transcriptional potential through coordinating depositions of the histone variants H3.3 and H2A.Z on the poised genes in mESCs. Nucleic Acids Res. 50, 191–206 (2022).
Schoenfelder, S. & Fraser, P. Long-range enhancer–promoter contacts in gene expression control. Nat. Rev. Genet. 20, 437–455 (2019).
Schneider, J. et al. Molecular regulation of histone H3 trimethylation by COMPASS and the regulation of gene expression. Mol. Cell 19, 849–856 (2005).
Policarpi, C., Munafò, M., Tsagkris, S., Carlini, V. & Hackett, J. A. Systematic epigenome editing captures the context-dependent instructive function of chromatin modifications. Nat. Genet. 56, 1168–1180 (2024).
Wang, H. et al. H3K4me3 regulates RNA polymerase II promoter-proximal pause-release. Nature 615, 339–348 (2023).
Willhoft, O. et al. Structure and dynamics of the yeast SWR1-nucleosome complex. Science 362, eaat7716 (2018).
Poyton, M. F. et al. Coordinated DNA and histone dynamics drive accurate histone H2A.Z exchange. Sci. Adv. 8, eabj5509 (2022).
Kubik, S. et al. Nucleosome stability distinguishes two different promoter types at all protein-coding genes in yeast. Mol. Cell 60, 422–434 (2015).
Brahma, S. & Henikoff, S. RSC-associated subnucleosomes define MNase-sensitive promoters in yeast. Mol. Cell 73, 238–249.e3 (2019).
Jin, C. & Felsenfeld, G. Nucleosome stability mediated by histone variants H3.3 and H2A.Z. Genes Dev. 21, 1519–1529 (2007).
Jin, C. et al. H3.3/H2A.Z double variant–containing nucleosomes mark ‘nucleosome-free regions’ of active promoters and other regulatory regions. Nat. Genet. 41, 941–945 (2009).
Xi, Y., Yao, J., Chen, R., Li, W. & He, X. Nucleosome fragility reveals novel functional states of chromatin and poises genes for activation. Genome Res. 21, 718–724 (2011).
Bentsen, M. et al. ATAC-seq footprinting unravels kinetics of transcription factor binding during zygotic genome activation. Nat. Commun. 11, 4267 (2020).
Lemma, R. B. et al. Pioneer transcription factors are associated with the modulation of DNA methylation patterns across cancers. Epigenetics Chromatin 15, 13 (2022).
Peng, Y. et al. Detection of new pioneer transcription factors as cell-type-specific nucleosome binders. eLife 12, RP88936 (2024).
Ugur, E. et al. Comprehensive chromatin proteomics resolves functional phases of pluripotency and identifies changes in regulatory components. Nucleic Acids Res. 51, 2671–2690 (2023).
Becker, J. S. et al. Genomic and proteomic resolution of heterochromatin and its restriction of alternate fate genes. Mol. Cell 68, 1023–1037.e15 (2017).
Cauchy, P., Koch, F. & Andrau, J.-C. Two possible modes of pioneering associated with combinations of H2A.Z and p300/CBP at nucleosome-occupied enhancers. Transcription 8, 179–184 (2017).
Michael, A. K. et al. Mechanisms of OCT4-SOX2 motif readout on nucleosomes. Science 368, 1460–1465 (2020).
Dodonova, S. O., Zhu, F., Dienemann, C., Taipale, J. & Cramer, P. Nucleosome-bound SOX2 and SOX11 structures elucidate pioneer factor function. Nature 580, 669–672 (2020).
Masui, S. et al. Pluripotency governed by Sox2 via regulation of Oct3/4 expression in mouse embryonic stem cells. Nat. Cell Biol. 9, 625–635 (2007).
Murphy, P. J., Wu, S. F., James, C. R., Wike, C. L. & Cairns, B. R. Placeholder nucleosomes underlie germline-to-embryo DNA methylation reprogramming. Cell 172, 993–1006.e13 (2018).
Spruijt, C. G. et al. Dynamic readers for 5-(Hydroxy)methylcytosine and its oxidized derivatives. Cell 152, 1146–1159 (2013).
Domcke, S. et al. Competition between DNA methylation and transcription factors determines binding of NRF1. Nature 528, 575–579 (2015).
Azevedo Portilho, N. et al. The DNMT1 inhibitor GSK-3484862 mediates global demethylation in murine embryonic stem cells. Epigenetics Chromatin 14, 56 (2021).
Dror, I., Zhou, T., Mandel-Gutfreund, Y. & Rohs, R. Covariation between homeodomain transcription factors and the shape of their DNA binding sites. Nucleic Acids Res. 42, 430–441 (2014).
Kim, S. et al. Probing allostery through DNA. Science 339, 816–819 (2013).
Yu, J. et al. Structural insights into histone exchange by human SRCAP complex. Cell Discov. 10, 1–18 (2024).
Park, G., Patel, A. B., Wu, C. & Louder, R. K. Structures of H2A.Z-associated human chromatin remodelers SRCAP and TIP60 reveal divergent mechanisms of chromatin engagement. Preprint at bioRxiv https://doi.org/10.1101/2024.07.30.605802 (2024).
Jalal, A. S. B. et al. Stabilization of the hexasome intermediate during histone exchange by yeast SWR1 complex. Mol. Cell 84, 3871–3884.e9 (2024).
Louder, R. K. et al. Molecular basis of global promoter sensing and nucleosome capture by the SWR1 chromatin remodeler. Cell 187, 6849–6864.e18 (2024).
Hong, L. & Lei, J. Scaling law for the radius of gyration of proteins and its dependence on hydrophobicity. J. Polym. Sci. B Polym. Phys. 47, 207–214 (2009).
Maeshima, K. et al. The physical size of transcription factors is key to transcriptional regulation in chromatin domains. J. Phys. Condens. Matter 27, 064116 (2015).
Girvan, P. et al. Nucleosome flipping drives kinetic proofreading and processivity by SWR1. Nature 636, 251–257 (2024).
Kuhn, T. et al. Single-molecule tracking of Nodal and Lefty in live zebrafish embryos supports hindered diffusion model. Nat. Commun. 13, 6101 (2022).
Kuhn, T., Hettich, J., Davtyan, R. & Gebhardt, J. C. M. Single molecule tracking and analysis framework including theory-predicted parameter settings. Sci. Rep. 11, 9465 (2021).
Reisser, M. et al. Inferring quantity and qualities of superimposed reaction rates from single molecule survival time distributions. Sci. Rep. 10, 1758 (2020).
Hettich, J. & Gebhardt, J. C. M. Transcription factor target site search and gene regulation in a background of unspecific binding sites. J. Theor. Biol. 454, 91–101 (2018).
Huynh, D. et al. Effective in vivo binding energy landscape illustrates kinetic stability of RBPJ-DNA binding. Nat. Commun. 16, 1259 (2025).
McNally, J. G., Müller, W. G., Walker, D., Wolford, R. & Hager, G. L. The glucocorticoid receptor: rapid exchange with regulatory sites in living cells. Science 287, 1262–1265 (2000).
Gebhardt, J. C. M. et al. Single-molecule imaging of transcription factor binding to DNA in live mammalian cells. Nat. Methods 10, 421–426 (2013).
Morisaki, T., Müller, W. G., Golob, N., Mazza, D. & McNally, J. G. Single-molecule analysis of transcription factor binding at transcription sites in live cells. Nat. Commun. 5, 4456 (2014).
Schick, S. et al. Acute BAF perturbation causes immediate changes in chromatin accessibility. Nat. Genet. 53, 269–278 (2021).
Iurlaro, M. et al. Mammalian SWI/SNF continuously restores local accessibility to chromatin. Nat. Genet. 53, 279–287 (2021).
Lashgari, A., Millau, J.-F., Jacques, P. -É & Gaudreau, L. Global inhibition of transcription causes an increase in histone H2A.Z incorporation within gene bodies. Nucleic Acids Res. 45, 12715–12722 (2017).
Brahma, S. & Henikoff, S. The BAF chromatin remodeler synergizes with RNA polymerase II and transcription factors to evict nucleosomes. Nat. Genet. 56, 100–111 (2024).
Kaur, U., Muñoz, E. N. & Narlikar, G. J. Hexasomal particles: consequence or also consequential?. Curr. Opin. Genet. Dev. 85, 102163 (2024).
Hoffman, J. A., Trotter, K. W. & Archer, T. K. RNA polymerase II coordinates histone deacetylation at active promoters. Sci. Adv. 11, eadt3037 (2025).
Chen, Z. et al. High-resolution and high-accuracy topographic and transcriptional maps of the nucleosome barrier. eLife 8, e48281 (2019).
Dull, T. et al. A third-generation lentivirus vector with a conditional packaging system. J. Virol. 72, 8463–8471 (1998).
Suter, D. M. et al. Rapid generation of stable transgenic embryonic stem cell lines using modular lentivectors. Stem Cells 24, 615–623 (2006).
Concordet, J.-P. & Haeussler, M. CRISPOR: intuitive guide selection for CRISPR/Cas9 genome editing experiments and screens. Nucleic Acids Res. 46, W242–W245 (2018).
Kadauke, S. et al. Tissue-specific mitotic bookmarking by hematopoietic transcription factor GATA1. Cell 150, 725–737 (2012).
Schindelin, J. et al. Fiji: an open-source platform for biological-image analysis. Nat. Methods 9, 676–682 (2012).
Egan, B. et al. An alternative approach to ChIP-seq normalization enables detection of genome-wide changes in histone H3 lysine 27 trimethylation upon EZH2 inhibition. PLoS ONE 11, e0166438 (2016).
Buenrostro, J. D., Giresi, P. G., Zaba, L. C., Chang, H. Y. & Greenleaf, W. J. Transposition of native chromatin for fast and sensitive epigenomic profiling of open chromatin, DNA-binding proteins and nucleosome position. Nat. Methods 10, 1213–1218 (2013).
Picelli, S. et al. Tn5 transposase and tagmentation procedures for massively scaled sequencing projects. Genome Res. 24, 2033–2040 (2014).
Esnault, C., Magat, T., García-Oliver, E. & Andrau, J.-C. in Enhancers and Promoters: Methods and Protocols (eds. Borggrefe, T. & Giaimo, B. D.) Chapter 5 (Springer, 2021).
Tokunaga, M., Imamoto, N. & Sakata-Sogawa, K. Highly inclined thin illumination enables clear single-molecule imaging in cells. Nat. Methods 5, 159–161 (2008).
Mazza, D., Abernathy, A., Golob, N., Morisaki, T. & McNally, J. G. A benchmark for chromatin binding measurements in live cells. Nucleic Acids Res. 40, e119 (2012).
Shaban, H. A., Barth, R., Recoules, L. & Bystricky, K. Hi-D: nanoscale mapping of nuclear dynamics in single living cells. Genome Biol. 21, 95 (2020).
Chen, J. et al. Single-molecule dynamics of enhanceosome assembly in embryonic stem cells. Cell 156, 1274–1285 (2014).
Loffreda, A. et al. Live-cell p53 single-molecule binding is modulated by C-terminal acetylation and correlates with transcriptional activity. Nat. Commun. 8, 313 (2017).
Tang, X. et al. Kinetic principles underlying pioneer function of GAGA transcription factor in live cells. Nat. Struct. Mol. Biol. 29, 665–676 (2022).
Dobin, A. et al. STAR: ultrafast universal RNA-seq aligner. Bioinformatics 29, 15–21 (2013).
Li, H. et al. The sequence alignment/Map format and SAMtools. Bioinformatics 25, 2078–2079 (2009).
Zhang, Y. et al. Model-based analysis of ChIP-Seq (MACS). Genome Biol. 9, R137 (2008).
Ramírez, F., Dündar, F., Diehl, S., Grüning, B. A. & Manke, T. deepTools: a flexible platform for exploring deep-sequencing data. Nucleic Acids Res. 42, W187–W191 (2014).
Gao, T. & Qian, J. EnhancerAtlas 2.0: an updated resource with enhancer annotation in 586 tissue/cell types across nine species. Nucleic Acids Res. 48, D58–D64 (2020).
Hsieh, T.-H. S. et al. Resolving the 3D landscape of transcription-linked mammalian chromatin folding. Mol. Cell 78, 539–553.e8 (2020).
Chen, K. et al. DANPOS: Dynamic analysis of nucleosome position and occupancy by sequencing. Genome Res. 23, 341–351 (2013).
Lee, S. et al. Covering all your bases: incorporating intron signal from RNA-seq data. NAR Genom. Bioinform. 2, lqaa073 (2020).
Liao, Y., Smyth, G. K. & Shi, W. featureCounts: an efficient general purpose program for assigning sequence reads to genomic features. Bioinformatics 30, 923–930 (2014).
Love, M. I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 15, 550 (2014).
Eisenberg, E. & Levanon, E. Y. Human housekeeping genes, revisited. Trends Genet. 29, 569–574 (2013).
Akalin, A. et al. methylKit: a comprehensive R package for the analysis of genome-wide DNA methylation profiles. Genome Biol. 13, R87 (2012).
Castro-Mondragon, J. A. et al. JASPAR 2022: the 9th release of the open-access database of transcription factor binding profiles. Nucleic Acids Res. 50, D165–D173 (2022).
Bailey, T. L. & Grant, C. E. SEA: simple enrichment analysis of motifs. Preprint at https://doi.org/10.1101/2021.08.23.457422 (2021).
Pomaznoy, M., Ha, B. & Peters, B. GOnet: a tool for interactive Gene Ontology analysis. BMC Bioinformatics 19, 470 (2018).
de Dieuleveult, M. et al. Genome-wide nucleosome specificity and function of chromatin remodellers in ES cells. Nature 530, 113–116 (2016).
Hon, G. C. et al. 5mC oxidation by Tet2 modulates enhancer activity and timing of transcriptome reprogramming during differentiation. Mol. Cell 56, 286–297 (2014).
Deluz, C. et al. A role for mitotic bookmarking of SOX2 in pluripotency and differentiation. Genes Dev. 30, 2538–2550 (2016).
Neph, S. et al. BEDOPS: high-performance genomic feature operations. Bioinformatics 28, 1919–1920 (2012).
Quinlan, A. R. & Hall, I. M. BEDTools: a flexible suite of utilities for comparing genomic features. Bioinformatics 26, 841–842 (2010).
Pedersen, B. S., Eyring, K., De, S., Yang, I. V. & Schwartz, D. A. Fast and accurate alignment of long bisulfite-seq reads. Preprint at http://arxiv.org/abs/1401.1129 (2014).
Robinson, M. D., McCarthy, D. J. & Smyth, G. K. edgeR: a Bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics 26, 139–140 (2010).
Ritchie, M. E. et al. limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res. 43, e47 (2015).
Yu, G., Wang, L.-G., Han, Y. & He, Q.-Y. clusterProfiler: an R package for comparing biological themes among gene clusters. OMICS 16, 284–287 (2012).
Wickham, H. Ggplot2: Elegant Graphics for Data Analysis (Springer, 2009).
Durinck, S. et al. BioMart and Bioconductor: a powerful link between biological databases and microarray data analysis. Bioinformatics 21, 3439–3440 (2005).
Mitternacht, S. FreeSASA: an open source C library for solvent accessible surface area calculations. F1000Res. https://doi.org/10.12688/f1000research.7931.1 (2016).
Sehnal, D. et al. Mol* Viewer: modern web app for 3D visualization and analysis of large biomolecular structures. Nucleic Acids Res. 49, W431–W437 (2021).
Acknowledgements
We thank the Gene Expression, Flow Cytometry, BioImaging and Biomolecular Screening core facilities at EPFL. We thank Dr Matthias Mann for kindly providing access to MS instrumentation, and to Dr Igor Paron and Dr Tim Heymann for outstanding MS technical assistance. We thank Dr Martin Pacesa for help in designing cdSRCAP. We thank Dr Ludovica Vanzan, Dr Nadine Vastenhouw and Dr Nicolas Thomä for their input. We thank Dr Marcel Tisch and Dr Pauline Franz for respectively sharing the drawing of stem cells colonies and nucleosomes. We are thankful for funding by the Swiss National Science Foundation grant#310030_184782 and# 310030_212197 to DMS. HL and EU are funded by the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation, 213249687 - SFB1064 to H.L. (A17)). E.U. gratefully acknowledges the International Max Planck Research School for Molecular Life Sciences (IMPRS-LS) and the Research Training Group 1721 (RTG 1721) for training and support. The work from S.D.L., D.A., and J.C.M.G. was funded by the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation no. 468578170 and CRC 1506 C05 no. 450627322 to J.C.M.G.).
Author information
Authors and Affiliations
Contributions
Conceptualization, A.T. and D.M.S.; Methodology, A.T., E.U., S.D.L., and D.M.S.; Formal analysis, A.T., E.U., S.D.L., and D.M.S.; Investigation, A.T., E.U., S.D.L., and C.D.; Instrumentation (single-molecule imaging microscope), D.A.; Resources, D.M.S., H.L., and J.C.M.G.; Writing original manuscript, A.T. and D.M.S.; Funding acquisition, D.M.S., H.L., and J.C.M.G.; Supervision, D.M.S., H.L., and J.C.M.G.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Xiaotao Dong and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Tollenaere, A., Ugur, E., Dalla Longa, S. et al. Mechanisms of gene regulation by SRCAP and H2A.Z. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70087-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70087-x