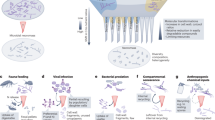
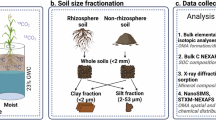
Abstract
Microbial necromass is increasingly recognized as a major source of stable soil organic matter (SOM), and its persistence is often attributed to interactions with clay-sized minerals. However, the mechanisms underlying this mineral-mediated stabilization remain poorly understood. Here, we conducted an in situ dual-labeled (13C and 15N) microbial necromass experiment across a clay gradient to quantify how clay content and necromass origin (bacterial vs. fungal) regulate necromass persistence. We find that higher clay content markedly enhances necromass retention by strengthening mineral protection, suppressing microbial activity and diversity, and limiting leaching losses. NanoSIMS imaging shows that new necromass preferentially associates with organic matter coatings on the rough mineral surfaces, highlighting organo-organic interfaces as important stabilization pathways. Necromass origin exerts little effect on retention despite marked differences in C:N ratios and bulk chemical composition, indicating that finer-scale molecular features, rather than broad compositional differences, govern necromass stabilization in soils.
Similar content being viewed by others
Data availability
Microbial amplicon sequencing data have been deposited in the National Center for Biotechnology Information (NCBI) Sequence Read Archive (SRA) data repository under accession no. PRJNA1191639. All aggregated data supporting the findings of this study are available in the Figshare repository: https://doi.org/10.6084/m9.figshare.27896424.
Code availability
The main R code used in this study is available in the Figshare repository: https://doi.org/10.6084/m9.figshare.27896424.
References
Ciais, P et al. Carbon and other biogeochemical cycles. In Climate Change 2013: The Physical Science Basis. Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change (eds Stocker, T. F.) (Cambridge University Press, 2014).
Melillo, J. M. et al. Long-term pattern and magnitude of soil carbon feedback to the climate system in a warming world. Science 358, 101–105 (2017).
Paustian, K. et al. Climate-smart soils. Nature 532, 49–57 (2016).
Simpson, A. J., Simpson, M. J., Smith, E. & Kelleher, B. P. Microbially derived inputs to soil organic matter: are current estimates too low? Environ. Sci. Technol. 41, 8070–8076 (2007).
Liang, C. & Balser, T. C. Microbial production of recalcitrant organic matter in global soils: implications for productivity and climate policy. Nat. Rev. Microbiol. 9, 75 (2011).
Wang, B., An, S., Liang, C., Liu, Y. & Kuzyakov, Y. Microbial necromass as the source of soil organic carbon in global ecosystems. Soil Biol. Biochem. 162, 108422 (2021).
Lehmann, J. & Kleber, M. The contentious nature of soil organic matter. Nature 528, 60 (2015).
Schmidt, M. W. et al. Persistence of soil organic matter as an ecosystem property. Nature 478, 49–56 (2011).
Camenzind, T., Mason-Jones, K., Mansour, I., Rillig, M. & Lehmann, J. Formation of necromass-derived soil organic carbon determined by microbial death pathways. Nat. Geosci. 16, 1–8 (2023).
Sokol, N. W., Sanderman, J. & Bradford, M. A. Pathways of mineral-associated soil organic matter formation: integrating the role of plant carbon source, chemistry, and point of entry. Glob. Change Biol. 25, 12–24 (2018).
Kleber, M. et al. Mineral–organic associations: formation, properties, and relevance in soil environments. Adv. Agron. 130, 1–140 (2015).
Xiao, K.-Q. et al. Introducing the soil mineral carbon pump. Nat. Rev. Earth Environ. 4, 135–136 (2023).
Liang, C., Schimel, J. & Jastrow, J. The importance of anabolism in microbial control over soil carbon storage. Nat. Microbiol. 2, 17105 (2017).
Ni, X. et al. The vertical distribution and control of microbial necromass carbon in forest soils. Glob. Ecol. Biogeogr. 29, 1829–1839 (2020).
Buckeridge, K. M., Creamer, C. & Whitaker, J. Deconstructing the microbial necromass continuum to inform soil carbon sequestration. Funct. Ecol. 36, 1396–1410 (2022).
Cai, Y. et al. Assessing the accumulation efficiency of various microbial carbon components in soils of different minerals. Geoderma 407, 115562 (2022).
Mao, H.-R. et al. Dual role of silt and clay in the formation and accrual of stabilized soil organic carbon. Soil Biol. Biochem. 192, 109390 (2024).
Kögel-Knabner, I. et al. Organo-mineral associations in temperate soils: Integrating biology, mineralogy, and organic matter chemistry. J. Plant Nutr. Soil Sci. 171, 61–82 (2008).
Schweizer, S. A., Bucka, F. B., Graf-Rosenfellner, M. & Kögel-Knabner, I. Soil microaggregate size composition and organic matter distribution as affected by clay content. Geoderma 355, 113901 (2019).
Kleber, M. et al. Dynamic interactions at the mineral–organic matter interface. Nat. Rev. Earth Environ. 2, 402–421 (2021).
Oades, J. The retention of organic matter in soils. Biogeochemistry 5, 35–70 (1988).
von Lüetzow, M. et al. Stabilization of organic matter in temperate soils: mechanisms and their relevance under different soil conditions – a review. Eur. J. Soil Sci. 57, 426–445 (2006).
Rakhsh, F., Golchin, A., Beheshti Al Agha, A. & Nelson, P. N. Mineralization of organic carbon and formation of microbial biomass in soil: effects of clay content and composition and the mechanisms involved. Soil Biol. Biochem. 151, 108036 (2020).
Kindler, R. et al. Dissolved carbon leaching from soil is a crucial component of the net ecosystem carbon balance. Glob. Change Biol. 17, 1167–1185 (2011).
Lieberman, H., Rothman, M., Sperber, C. & Kallenbach, C. Experimental flooding shifts carbon, nitrogen, and phosphorus pool distribution and microbial activity. Biogeochemistry 165, 1–16 (2023).
Keiluweit, M., Nico, P. S., Kleber, M. & Fendorf, S. Are oxygen limitations under recognized regulators of organic carbon turnover in upland soils? Biogeochemistry 127, 157–171 (2016).
Krull, E., Baldock, J. & Skjemstad, J. Importance of mechanisms and processes of the stabilisation of soil organic matter for modelling carbon turnover. Funct. Plant Biol. 30, 207–222 (2003).
Cuadros, J. Clay minerals interaction with microorganisms: a review. Clay Min. 52, 235–261 (2017).
Schimel, J. Life in dry soils: effects of drought on soil microbial communities and processes. Annu. Rev. Ecol. Evol. Syst. 49, 409–432 (2018).
Shabtai, I. et al. Soil organic carbon accrual due to more efficient microbial utilization of plant inputs at greater long-term soil moisture. Geochim. Cosmochim. Acta 327, 170–185 (2022).
Wei, H. et al. High clay content accelerates the decomposition of fresh organic matter in artificial soils. Soil Biol. Biochem. 77, 100–108 (2014).
Wu, S., Konhauser, K. O., Chen, B. & Huang, L. “Reactive Mineral Sink” drives soil organic matter dynamics and stabilization. Npj Mater. Sustain 1, 3 (2023).
Fernandez, C. W. & Koide, R. T. Initial melanin and nitrogen concentrations control the decomposition of ectomycorrhizal fungal litter. Soil Biol. Biochem. 77, 150–157 (2014).
Fernandez, C. W. & Kennedy, P. G. Melanization of mycorrhizal fungal necromass structures microbial decomposer communities. J. Ecol. 106, 468–479 (2018).
Buckeridge, K. M. et al. Sticky dead microbes: rapid abiotic retention of microbial necromass in soil. Soil Biol. Biochem. 149, 107929 (2020).
Schweigert, M., Herrmann, S., Miltner, A. & Fester, T. Kaestner M. Fate of ectomycorrhizal fungal biomass in a soil bioreactor system and its contribution to soil organic matter formation. Soil Biol. Biochem. 88, 120–127 (2015).
Kindler, R., Miltner, A., Richnow, H.-H. & Kästner, M. Fate of gram-negative bacterial biomass in soil—mineralization and contribution to SOM. Soil Biol. Biochem. 38, 2860–2870 (2006).
Kopittke, P. M. et al. Nitrogen-rich microbial products provide new organo-mineral associations for the stabilization of soil organic matter. Glob. Change Biol. 24, 1762–1770 (2018).
Miltner, A., Bombach, P., Schmidt-Brücken, B. & Kästner, M. SOM genesis: microbial biomass as a significant source. Biogeochemistry 111, 41–55 (2012).
Tong, Y., Xiang, H., Jiang, J. & Chen, W. Interfacial interactions between minerals and organic matter: Mechanisms and characterizations. Chemosphere 359, 142383 (2024).
Gow, N. A. R., Latge, J.-P. & Munro, C. A. The fungal cell wall: structure, biosynthesis, and function. Microbiol. Spectr. 5, FUNK-0035-2016 (2017).
Kleber, M., Sollins, P. & Sutton, R. A conceptual model of organo-mineral interactions in soils: self-assembly of organic molecular fragments into zonal structures on mineral surfaces. Biogeochemistry 85, 9–24 (2007).
Possinger, A. R. et al. Organo–organic and organo–mineral interfaces in soil at the nanometer scale. Nat. Commun. 11, 6103 (2020).
Throckmorton, H. M., Bird, J. A., Dane, L., Firestone, M. K. & Horwath, W. R. The source of microbial C has little impact on soil organic matter stabilisation in forest ecosystems. Ecol. Lett. 15, 1257–1265 (2012).
Rakhsh, F., Golchin, A., Ali, B. A. A. & Alamdri, P. Effects of exchangeable cations, mineralogy and clay content on the mineralization of plant residue carbon. Geoderma 307, 150–158 (2017).
Witzgall, K. et al. Particulate organic matter as a functional soil component for persistent soil organic carbon. Nat. Commun. 12, 4115 (2021).
Torn, M., Trumbore, S., Chadwick, O., Vitousek, P. & Hendricks, D. Mineral control of soil organic carbon storage and turnover. Nature 389, 170–173 (1997).
Vogel, C. et al. Submicron structures provide preferential spots for carbon and nitrogen sequestration in soils. Nat. Commun. 5, 2947 (2014).
Manzoni, S., Taylor, P., Richter, A., Porporato, A. & Agren, G. Environmental and stoichiometric controls on microbial carbon-use efficiency in soils. N. Phytologist 196, 79–91 (2012).
Matzner, B. okenW. E. Reappraisal of drying and wetting effects on C and N mineralization and fluxes in soils. Glob. Change Biol. 15, 808–824 (2009).
Picek, T., Šimek, M. & Šantrůčková, H. Microbial responses to fluctuation of soil aeration status and redox conditions. Biol. Fert. Soils 31, 315–322 (2000).
Herbst, M., Tappe, W., Kummer, S. & Vereecken, H. The impact of sieving on heterotrophic respiration response to water content in loamy and sandy topsoils. Geoderma 272, 73–82 (2016).
Spence, A. et al. The degradation characteristics of microbial biomass in soil. Geochim Cosmochim. Acta 75, 2571–2581 (2011).
Yang, N. et al. Leaf litter species identity influences biochemical composition of ectomycorrhizal fungi. Mycorrhiza 29, 85–96 (2019).
Sokol, N. W. et al. Life and death in the soil microbiome: how ecological processes influence biogeochemistry. Nat. Rev. Microbiol. 20, 415–430 (2022).
Cotrufo, M. F., Wallenstein, M. D., Boot, C. M., Denef, K. & Paul, E. The Microbial Efficiency-Matrix Stabilization (MEMS) framework integrates plant litter decomposition with soil organic matter stabilization: do labile plant inputs form stable soil organic matter? Glob. Change Biol. 19, 988–995 (2013).
Buckeridge, K. M. et al. Microbial necromass carbon and nitrogen persistence are decoupled in agricultural grassland soils. Commun. Earth Environ. 3, 114 (2022).
Mooshammer, M., Wanek, W., Zechmeister-Boltenstern, S. & Richter, A. Stoichiometric imbalances between terrestrial decomposer communities and their resources: mechanisms and implications of microbial adaptations to their resources. Front. Microbiol. 5, 22 (2014).
Mooshammer, M. et al. Adjustment of microbial nitrogen use efficiency to carbon:nitrogen imbalances regulates soil nitrogen cycling. Nat. Commun. 5, 3694 (2014).
Liu, J. et al. Different fates of deposited NH4+ and NO3− in a temperate forest in northeast China: a 15N tracer study. Glob. Change Biol. 23, 2441–2449 (2017).
Pronk, G. J. et al. Interaction of minerals, organic matter, and microorganisms during biogeochemical interface formation as shown by a series of artificial soil experiments. Biol. Fert. Soils 53, 9–22 (2017).
Kallenbach, C. M., Frey, S. D. & Grandy, A. S. Direct evidence for microbial-derived soil organic matter formation and its ecophysiological controls. Nat. Commun. 7, 13630 (2016).
Cotrufo, M. F. et al. Formation of soil organic matter via biochemical and physical pathways of litter mass loss. Nat. Geosci. 8, 776–781 (2015).
North, P. Towards an absolute measurement of soil structural stability using ultrasound. Soil Sci. 27, 451–459 (1976).
Vance, E. D., Brookes, P. C. & Jenkinson, D. S. An extraction method for measuring soil microbial biomass C. Soil Biol. Biochem. 19, 703–707 (1987).
Parikh, S. J., Goyne, K. W., Margenot, A.J., Mukome, F. N. & Calderón, F. J. Soil chemical insights provided through vibrational spectroscopy. In Advances in Agronomy (eds Sparks, D. L.) (Academic Press, 2014).
Gao, D. et al. Small and transient response of winter soil respiration and microbial communities to altered snow depth in a mid-temperate forest. Appl. Soil Ecol. 130, 40–49 (2018).
Wiesenberg, G. L. B., Schwarzbauer, J., Schmidt, M. W. I. & Schwark, L. Source and turnover of organic matter in agricultural soils derived from n-alkane/n-carboxylic acid compositions and C-isotope signatures. Org. Geochem. 35, 1371–1393 (2004).
Oksanen, J. Vegan: community ecology package. https://cran.r-project.org/web/packages/vegan/index.html (2013).
Lai, J., Zou, Y., Zhang, J. & Peres-Neto, P. R. Generalizing hierarchical and variation partitioning in multiple regression and canonical analyses using the rdacca.hp R package. Methods Ecol. Evol. 13, 782–788 (2022).
Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (42322306, 32201412), the National Key Research and Development Program of China (2022YFF1300501), the International Partnership Program of Chinese Academy of Sciences (064GJHZ2022054FN), Liaoning Revitalization Talents Program (XLYC2403021) and the Natural Science Foundation Program of Liaoning Province (2025JH6/101100019, 2023-MSBA-142), the Youth Innovation Promotion Association CAS to Chao Wang (Y2022064), and the CAS (Chinese Academy of Sciences) Project for Young Scientists in Basic Research (YSBR-108). We thank Guanghui Yu and Bohao Yin from Tianjin University for technical assistance and Jie Kang and Jiaxin Zhao from Huazhong Agricultural University) for NanoSIMS analysis. Additionally, we thank Weixing Zhu from Binghamton University for helpful discussions.
Author information
Authors and Affiliations
Contributions
X.W., C.W., and E.B. designed the study. X.W., C.W., and P.J. performed the experiment. X.W., C.P.S., and L.F.S. performed the laboratory analyses. X.W., Y.L., and C.W. analyzed the data. X.W., C.M.K., M.A., K.G., E.B., and C.W. wrote the manuscript. All authors contributed to the article and approved the submitted version.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Javier Cuadros and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wang, X., Kallenbach, C.M., Almaraz, M. et al. Clay-organic matter interactions drive microbial necromass preservation in soils. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70156-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70156-1