Abstract
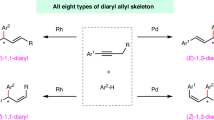
2,5-Dihydropyrroles are prevalent structural motifs in various natural products and biologically active molecules. Conventional methods for constructing these heterocycles often rely on elaborate multi-step procedures or complex starting materials. Herein, we describe a palladium-catalyzed chemodivergent protocol for synthesizing functionalized 2,5-dihydropyrrole scaffolds from readily accessible 1,3-enynes and anilines. By modulating the relative rates of the selectivity-determining steps, either two-component annulation or three-component telomerization can be selectively achieved, affording two distinct types of functionalized 2,5-dihydropyrroles in excellent yields and with high selectivities. Mechanistic studies reveal that the reaction initially proceeds through 1,4-hydroamination of 1,3-enynes to generate an aminomethyl allene intermediate, followed by an intramolecular annulation affording 2-substituted 2,5-dihydropyrroles. This process is significantly accelerated in the presence of Pd(II) catalysts. However, employing Pd(0) precursors, strong acids, and excess ligand effectively decelerates this pathway, diverting the selectivity towards reaction with a second equivalent of 1,3-enyne to yield the telomeric products. This study not only provides an atom-economical method for constructing the 2,5-dihydropyrrole core but also offers a valuable strategy for the development of telomerization chemistry.
Similar content being viewed by others
Data availability
The X-ray crystallographic data for compounds have been deposited in the Cambridge Crystallographic Data Centre (CCDC) under deposition numbers CCDC 2468082 (3aa), 2468080 (4ja), 2472318 (21), 2468081 (22 E) and 2519020 (S-3aa). Data relating to the characterization data of materials and products, general methods, optimization studies, experimental procedures, mechanistic studies, mass spectrometry and NMR spectra are available in the Supplementary Information. All data are also available from the corresponding author upon request.
References
Beletskaya, I. P., Nájera, C. & Yus, M. Stereodivergent catalysis. Chem. Rev. 118, 5080–5200 (2018).
Li, L., Chen, Z., Zhang, X.-W. & Jia, Y.-X. Divergent strategy in natural product total synthesis. Chem. Rev. 118, 3752–3832 (2018).
Nájera, C., Beletskaya, I. P. & Yus, M. Metal-catalyzed regiodivergent organic reactions. Chem. Soc. Rev. 48, 4515–4618 (2019).
Beletskaya, I. P., Nájera, C. & Yus, M. Chemodivergent reactions. Chem. Soc. Rev. 49, 7101–7166 (2020).
Chintawar, C. C., Yadav, A. K., Kumar, A., Sancheti, S. P. & Patil, N. T. Divergent gold catalysis: Unlocking molecular diversity through catalyst control. Chem. Rev. 121, 8478–8558 (2021).
Viji, M. et al. Regiodivergent organocatalytic reactions. Catalysts 11, 1013–1058 (2021).
Sakakibara, Y. & Murakami, K. Switchable divergent synthesis using photocatalysis. ACS Catal. 12, 1857–1878 (2022).
Chaudhary, H. R. & Patel, D. M. Recent trends for chemoselectivity modulation in one-pot organic transformations. RSC Adv. 14, 31072–31116 (2024).
Chakraborty, G. & Maity, S. Divergent hydroelementation of unsaturated C−C bonds using 3d transition metal based catalysts. Adv. Synth. Catal. 367, e202401239 (2025).
Bhutani, P. et al. U.S. FDA approved drugs from 2015–june 2020: A perspective. J. Med. Chem. 64, 2339–2381 (2021).
Marshall, C. M., Federice, J. G., Bell, C. N., Cox, P. B. & Njardarson, J. T. An update on the nitrogen heterocycle compositions and properties of U.S. FDA-approved pharmaceuticals (2013–2023). J. Med. Chem. 67, 11622–11655 (2024).
Vitaku, E., Smith, D. T. & Njardarson, J. T. Analysis of the structural diversity, substitution patterns, and frequency of nitrogen heterocycles among U.S. FDA approved pharmaceuticals. J. Med. Chem. 57, 10257–10274 (2014).
Pennington, L. D. & Moustakas, D. T. The necessary nitrogen atom: A versatile high-impact design element for multiparameter optimization. J. Med. Chem. 60, 3552–3579 (2017).
Kerru, N., Gummidi, L., Maddila, S., Gangu, K. K. & Jonnalagadda, S. B. A review on recent advances in nitrogen-containing molecules and their biological applications. Molecules 25, 1909–1951 (2020).
Anderson, W. K. & Milowsky, A. S. 3-Pyrroline N-oxide bis(carbamate) tumor inhibitors as analogues of indicine N-oxide. J. Med. Chem. 30, 2144–2147 (1987).
Fraley, M. E. et al. Kinesin spindle protein (KSP) inhibitors. Part 2: The design, synthesis, and characterization of 2,4-diaryl-2,5-dihydropyrrole inhibitors of the mitotic kinesin KSP. Bioorg. Med. Chem. Lett. 16, 1775–1779 (2006).
Shi, W. et al. Design, synthesis, and antibacterial activity of 2,5-dihydropyrrole formyl hydroxyamino derivatives as novel peptide deformylase inhibitors. Bioorg. Med. Chem. Lett. 20, 3592–3595 (2010).
Medran, N. S., La-Venia, A. & Testero, S. A. Metal-mediated synthesis of pyrrolines. RSC Adv. 9, 6804–6844 (2019).
Fu, G. C., Nguyen, S. T. & Grubbs, R. H. Catalytic ring-closing metathesis of functionalized dienes by a ruthenium carbene complex. J. Am. Chem. Soc. 115, 9856–9857 (1993).
Moonen, K., Dieltiens, N. & Stevens, C. V. Synthesis of 2-phosphonopyrroles via a one-pot RCM/oxidation sequence. J. Org. Chem. 71, 4006–4009 (2006).
Bunrit, A., Sawadjoon, S., Tšupova, S., Sjöberg, P. J. R. & Samec, J. S. M. A general route to β-substituted pyrroles by transition-metal catalysis. J. Org. Chem. 81, 1450–1460 (2016).
Groso, E. J. et al. 3-Aryl-2,5-Dihydropyrroles via catalytic carbonyl-olefin metathesis. ACS Catal. 8, 2006–2011 (2018).
Zhang, D. H., Yao, L. F., Wei, Y. & Shi, M. Gold(I)-catalyzed cycloisomerization of 1,6-diynes: synthesis of 2,3-disubstituted 3-pyrroline derivatives. Angew. Chem. Int. Ed. 50, 2583–2587 (2011).
Zhang, D.-H., Zhang, Z. & Shi, M. Transition metal-catalyzed carbocyclization of nitrogen and oxygen-tethered 1,n-enynes and diynes: Synthesis of five or six-membered heterocyclic compounds. Chem. Commun. 48, 10271–10279 (2012).
Yang, J.-M. et al. Rhodium(III)-catalyzed intramolecular cyclization and sequential aromatization of ynamides with propargyl esters: Access to 2,5-dihydropyrroles and pyrroles. Org. Lett. 26, 6191–6196 (2024).
Morit, N. & Krause, N. Gold catalysis in organic synthesis: Efficient cycloisomerization of α-aminoallenes to 3-pyrrolines. Org. Lett. 6, 4121–4123 (2004).
Sai, M., Yorimitsu, H. & Oshima, K. Allyl-, allenyl-, and propargyl-transfer reactions through cleavage of c-c bonds catalyzed by an n-heterocyclic carbene/copper complex: Synthesis of multisubstituted pyrroles. Angew. Chem. Int. Ed. 50, 3294–3298 (2011).
Brioche, J., Meyer, C. & Cossy, J. Synthesis of functionalized allenamides from ynamides by enolate claisen rearrangement. Org. Lett. 15, 1626–1629 (2013).
Rajasekaran, T., Karthik, G., Sridhar, B. & Reddy, B. V. S. Dual behavior of isatin-based cyclic ketimines with dicarbomethoxy carbene: Expedient synthesis of highly functionalized spirooxindolyl oxazolidines and pyrrolines. Org. Lett. 15, 1512–1515 (2013).
Xie, H.-D. et al. A copper-catalyzed annulation reaction of diazo esters with propargyl amines for the synthesis of 2,5-dihydropyrroles. Org. Chem. Front. 11, 4546–4555 (2024).
Zhang, Z. et al. Enantioselective propargylic amination and related tandem sequences to α-tertiary ethynylamines and azacycles. Nat. Chem. 16, 521–532 (2024).
Radhakrishnan, U., AI-Masum, M. & Yamamoto, Y. Palladium catalyzed hydroamination of conjugated enynes. Tetrahedron letters 39, 1037–1040 (1998).
Zhang, W., Werness, J. B. & Tang, W. Intramolecular hydroamination of conjugated enynes. Tetrahedron 65, 3090–3095 (2009).
Zhang, Y., Yu, B., Gao, B., Zhang, T. & Huang, H. Triple-bond insertion triggers highly regioselective 1,4-aminomethylamination of 1,3-enynes with aminals enabled by Pd-catalyzed C–N bond activation. Org. Lett. 21, 535–539 (2019).
Li, L. et al. Direct access to spirocycles by Pd/wingphos-catalyzed enantioselective cycloaddition of 1,3-enynes. Nat. Commun. 12, 5667–5677 (2021).
Cera, G. & Maestri, G. Palladium/brønsted acid catalysis for hydrofunctionalizations of alkynes: From tsuji-trost allylations to stereoselective methodologies. ChemCatChem 14, e202200295 (2022).
Li, Q.-Y., Fang, X.-X., Pan, R., Yao, H.-Q. & Lin, A.-J. Palladium-catalyzed asymmetric sequential hydroamination of 1,3-enynes: Enantioselective syntheses of chiral imidazolidinones. J. Am. Chem. Soc. 144, 11364–11376 (2022).
Lu, W.-X., Zhao, Y.-M. & Meng, F.-K. Cobalt-catalyzed sequential site- and stereoselective hydrosilylation of 1,3- and 1,4-enynes. J. Am. Chem. Soc. 144, 5233–5240 (2022).
Li, L., Wang, S., Jakhar, A. & Shao, Z.-H. Pd-catalyzed Functionalization of 1,3-enynes via alkylene-π-allylpalladium intermediates. Green Synth. Catal. 4, 124–134 (2023).
Li, R., Zhang, H., Yu, B. & Huang, H. Chemo- and regioselective cyclization of diene-tethered enynesviapalladium-catalyzed aminomethylamination. Org. Chem. Front. 10, 2988–2993 (2023).
Wang, H., Jie, X.-F., Chong, Q.-L. & Meng, F.-K. Pathway-divergent coupling of 1,3-enynes with acrylates through cascade cobalt catalysis. Nat. Commun. 15, 3427–3436 (2024).
Wang, S. et al. Stereodivergent synthesis of chiral spiropyrazolones through Pd-catalyzed asymmetric sequential hydroalkylation of 1,3-enynes: unusual solvent effects on the enantioselectivity. Org. Chem. Front. 11, 3033–3040 (2024).
Xie, B.-Y. & He, Z.-T. Chemodivergent tandem hydroalkylation and hydroalkenoxylation of conjugated enynes. ACS catal. 14, 9742–9751 (2024).
Hu, Y. et al. Nickel-catalyzed asymmetric three-component reaction of unactivated alkyl halides, 1,3-enynes and aldehydes. Angew. Chem. Int. Ed. 65, e20212 (2026).
Jin, P.-S., He, X.-Q., Liu, S.-S., Qi, X.-T. & Shen, X. Synthesis of trifluoromethylated 2H-pyrans enabled by pd-catalyzed cascade cyclization of trifluoroacetylsilanes and 1,3-enynes. CCS Chem. 8, 1267-1275 (2026).
Pagar, V. V. & RajanBabu, T. V. Tandem catalysis for asymmetric coupling of ethylene and enynes to functionalized cyclobutanes. Science 361, 68–72 (2018).
Zatolochnaya, O. V. & Gevorgyan, V. Synthesis of fluoro- and perfluoroalkyl arenes via palladium-catalyzed [4 + 2] benzannulation reaction. Org. Lett. 15, 2562–2565 (2013).
Zhang, Z. & Gevorgyan, V. Escape from hydrofunctionalization: Palladium hydride-enabled difunctionalization of conjugated dienes and enynes. Angew. Chem. Int. Ed. 62, e202311848 (2023).
Yang, Z.-R. et al. Onestone three birds: Ni-catalyzed asymmetric allenylic substitution of allenic ethers, hydroalkylation of 1,3-enynes and double alkylation of enynyl ethers. Chin. Chem. Lett. 35, 109518–109523 (2024).
Hu, Y. C., Ji, D. W., Zhao, C. Y., Zheng, H. & Chen, Q. A. Catalytic prenylation and reverse prenylation of indoles with isoprene: Regioselectivity manipulation through choice of metal hydride. Angew. Chem. Int. Ed. 58, 5438–5442 (2019).
Jiang, W. S. et al. Orthogonal regulation of nucleophilic and electrophilic sites in Pd-catalyzed regiodivergent couplings between indazoles and isoprene. Angew. Chem. Int. Ed. 60, 8321–8328 (2021).
Zhao, C. Y. et al. Bioinspired and ligand-regulated unnatural prenylation and geranylation of oxindoles with isoprene under Pd. Catalysis. Angew. Chem. Int. Ed. 61, e202207202 (2022).
Yang, S.-N. et al. Ni-catalyzed regiodivergent hydrophosphorylation of enynes. Chin. Chem. Lett. 34, 107914 (2023).
Zhang, W.-N. et al. Ligand-controlled regiodivergence in Pd-catalyzed coupling of azlactones with isoprene. Cell Rep. Phys. Sci. 5, 101908 (2024).
Adamson, N. J., Jeddi, H. & Malcolmson, S. J. Preparation of chiral allenes through Pd-catalyzed intermolecular hydroamination of conjugated enynes: enantioselective synthesis enabled by catalyst design. J. Am. Chem. Soc. 141, 8574–8583 (2019).
Sakai, N., Ridder, A. & Hartwig, J. F. Tropene derivatives by sequential intermolecular and transannular, intramolecular palladium-catalyzed hydroamination of cycloheptatriene. J. Am. Chem. Soc. 128, 8134–8135 (2006).
Yang, X.-H., Lu, A. & Dong, V. M. Intermolecular hydroamination of 1,3-dienes to generate homoallylic amines. J. Am. Chem. Soc. 139, 14049–14052 (2017).
Tang, M.-Q., Yang, Z.-J. & He, Z.-T. Asymmetric formal sp2-hydrocarbonations of dienes and alkynes via palladium hydride catalysis. Nat. Commun. 14, 6303 (2023).
Kuai, C.-S., Wang, Y.-R., Yang, T. & Wu, X.-F. Multimodal precise control over multiselective carbonylation of 1,3-enynes. J. Am. Chem. Soc. 147, 7950–7964 (2025).
Asao, N., Nogami, T., Takahashi, K. & Yamamoto, Y. Pd(II) Acts simultaneously as a lewis acid and as a transition-metal catalyst: Synthesis of cyclic alkenyl ethers from acetylenic aldehydes. J. Am. Chem. Soc. 124, 764–765 (2002).
Xiao, Y.-J. & Zhang, J.-L. Tetrasubstituted furans by a PdII-catalyzed three-component michael addition/cyclization/cross-coupling reaction. Angew. Chem. Int. Ed. 47, 1903–1906 (2008).
Zhang, J.-B. & Han, X.-L. An unexpected addition of acetic acid to ortho-electron-deficient alkynyl-substituted aryl aldehydes catalyzed by palladium(ii) acetate. Adv. Synth. Catal. 356, 2465–2470 (2014).
Li, L., Luo, P.-F., Deng, Y.-H. & Shao, Z.-H. Regioselectivity switch in palladium-catalyzed allenylic cycloadditions of allenic esters: [4+1] or [4+3] Cycloaddition/cross-coupling. Angew. Chem. Int. Ed. 58, 4710–4713 (2019).
Luo, P.-F. et al. Switchable chemo-, regio- and pseudo-stereodivergence in palladium-catalyzed cycloaddition of allenes. Angew. Chem. Int. Ed. 63, e202412179 (2024).
Zhou, X.-Y., Liu, L., Lyu, H. & wang, x.-c. Modular alkyl growth in amines via the selective insertion of alkynes into C–C bonds. Nat. Chem. 17, 1323–1330 (2025).
Acknowledgements
Financial support from the National Natural Science Foundation of China (22201281) and the Natural Science Foundation of Liaoning Province (2025-MS-055) is acknowledged.
Author information
Authors and Affiliations
Contributions
Q.-A. C. conceived and supervised the project. Q.-A. C., S.-Y. X. and D.-W. J. designed the experiments. S.-Y. X., D.-W. J., X.-T. L. and Z.-H. W. performed the experiments and analyzed the data. All authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Zhihui Shao, and the other, anonymous, reviewer for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Xu, SY., Li, XT., Wang, ZH. et al. Chemodivergent Coupling of 1,3-Enynes with Anilines to Access Dihydropyrrole Skeleton under Palladium Catalysis. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70201-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70201-z