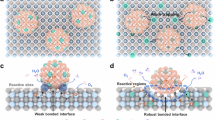
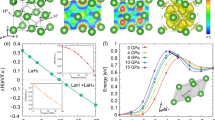
Abstract
Proton ceramic electrochemical cells PCECs hold promise for efficient, sustainable production and use of hydrogen. The positive electrodes are mixed proton conducting perovskites that facilitate water splitting and oxygen reduction, but the factors that determine the protonation are poorly understood. Here, we establish the governing principles of protonation through a study of hydration of 45 double perovskites with the general formula \({A}^{{{{\rm{I}}}}}{A}^{{{{\rm{II}}}}}{{{{\rm{Co}}}}}_{2}{{{{\rm{O}}}}}_{6-\delta }\), having Ba or Ba+Sr on AI and a mix of rare earths (Y and lanthanides Ln = La, Pr, Nd, Sm, Gd, Dy, Tb, Lu) on AII. We show how hydration is coupled to the A-site basicity and disorder as well as population of electron holes in the Co-O bond (Co oxidation state), promoted by a closed or semi-closed Ln 4 f shell, i.e., Ln = La, Gd, Lu.
Similar content being viewed by others
Data availability
The experimental data generated in this study are provided in the Supplementary Information/Source Data file. Source data are provided with this paper.
References
Choi, S. et al. Exceptional power density and stability at intermediate temperatures in protonic ceramic fuel cells. Nat. Energy 3, 202–210 (2018).
Duan, C. et al. Readily processed protonic ceramic fuel cells with high performance at low temperatures. Science 349, 1321–1326 (2015).
Vøllestad, E. et al. Mixed proton and electron conducting double perovskite anodes for stable and efficient tubular proton ceramic electrolysers. Nat. Mater. 18, 752–759 (2019).
Strandbakke, R. et al. Gd- and Pr-based double perovskite cobaltites as oxygen electrodes for proton ceramic fuel cells and electrolyser cells. Solid State Ion 278, 120–132 (2015).
Ding, H. et al. Self-sustainable protonic ceramic electrochemical cells using a triple conducting electrode for hydrogen and power production. Nat. Commun. 11, 1907 (2020).
Seong, A. et al. Electrokinetic proton transport in triple (H+/O2−/e−) conducting oxides as a key descriptor for highly efficient protonic ceramic fuel cells. Adv. Sci. 8, 2004099 (2021).
Geng, C. et al. A new in situ synthetic triple-conducting core–shell electrode for protonic ceramic fuel cells. ACS Sustain. Chem. Eng. 9, 11070–11079 (2021).
Zhang, Z. et al. In situ formation of a 3D core-shell and triple-conducting oxygen reduction reaction electrode for proton-conducting SOFCs. J. Power Sources 385, 76–83 (2018).
Kim, J. et al. Triple-conducting layered perovskites as cathode materials for proton-conducting solid oxide fuel cells. ChemSusChem 7, 2811–2815 (2014).
Papac, M., Stevanović, V., Zakutayev, A. & O’Hayre, R. Triple ionic–electronic conducting oxides for next-generation electrochemical devices. Nat. Mater. 20, 301–313 (2021).
Bjørheim, T. S., Hoedl, M. F., Merkle, R., Kotomin, E. A. & Maier, J. Proton, hydroxide ion, and oxide ion affinities of closed-shell oxides: importance for the hydration reaction and correlation to electronic structure. J. Phys. Chem. C 124, 1277–1284 (2020).
Poetzsch, D., Merkle, R. & Maier, J. Proton uptake in the H + -SOFC cathode material Ba0.5Sr0.5Fe0.8Zn0.2O3−δ: transition from hydration to hydrogenation with increasing oxygen partial pressure. Faraday Discuss. 182, 129–143 (2015).
Raimondi, G. et al. X-ray spectroscopy of (Ba,Sr,La)(Fe,Zn,Y)O3-δ identifies structural and electronic features favoring proton uptake. Chem. Mater. https://doi.org/10.1021/acs.chemmater.0c02655 (2020).
Hoedl, M. F., Gryaznov, D., Merkle, R., Kotomin, E. A. & Maier, J. Interdependence of oxygenation and hydration in mixed-conducting (Ba,Sr)FeO3−δ perovskites studied by density functional theory. J. Phys. Chem. C 124, 11780–11789 (2020).
Gryaznov, D., Merkle, R., Kotomin, E. A. & Maier, J. Ab initio modelling of oxygen vacancies and protonic defects in La1−xSrxFeO3−δ perovskite solid solutions. J. Mater. Chem. A 4, 13093–13104 (2016).
Zohourian, R., Merkle, R., Raimondi, G. & Maier, J. Mixed-conducting perovskites as cathode materials for protonic ceramic fuel cells: understanding the trends in proton uptake. Adv. Funct. Mater. 28, 1801241 (2018).
Raimondi, G., Merkle, R. & Maier, J. Impact of first-row transition metals in (Ba,La)(Fe,TM)O3-δ on proton uptake and electronic conductivity. Solid State Ion 391, 116143 (2023).
Grimaud, A. et al. Hydration properties and rate determining steps of the oxygen reduction reaction of perovskite-related oxides as H + -SOFC cathodes. J. Electrochem. Soc. 159, B683–B694 (2012).
Strandbakke, R. et al. Structural properties of mixed conductor Ba1 − xGd1−yLax+yCo2O6−δ. Dalton Trans. 51, 18667–18677 (2022).
Wachowski, S. L. et al. Structure and water uptake in BaLnCo2O6−δ (Ln =La, Pr, Nd, Sm, Gd, Tb and Dy). Acta Mater. 199, 297–310 (2020).
Ishizawa, N. et al. Structural evolution of GdBaCo2O5+δ (δ = 7/18) at elevated temperatures. Chem. Mater. 26, 6503–6517 (2014).
Jia, Y. Q. Crystal radii and effective ionic radii of the rare earth ions. J. Solid State Chem. 95, 184–187 (1991).
Frati, F., Hunault, M. O. J. Y. & de Groot, F. M. F. Oxygen K-edge X-ray absorption spectra. Chem. Rev. 120, 4056–4110 (2020).
Bernuy-Lopez, C., Høydalsvik, K., Einarsrud, M.-A. & Grande, T. Effect of A-site cation ordering on chemical stability, oxygen stoichiometry and electrical conductivity in layered LaBaCo2O5+δ double perovskite. Materials 9, 154 (2016).
Maignan, A., Martin, C., Pelloquin, D., Nguyen, N. & Raveau, B. Structural and magnetic studies of ordered oxygen-deficient perovskitesLnBaCo2O5+δ, closely related to the “112” structure. J. Solid State Chem. 142, 247–260 (1999).
Grimaud, A. et al. Double perovskites as a family of highly active catalysts for oxygen evolution in alkaline solution. Nat. Commun. 4, 2439 (2013).
Suntivich, J. et al. Estimating hybridization of transition metal and oxygen states in perovskites from O K-edge X-ray absorption spectroscopy. J. Phys. Chem. C 118, 1856–1863 (2014).
Toyoda, K., Hinogami, R., Miyata, N. & Aizawa, M. Calculated descriptors of catalytic activity for water electrolysis anode: application to delafossite oxides. J. Phys. Chem. C 119, 6495–6501 (2015).
Wilson, R. G., Stevie, F. A. & Magee, C. W. Secondary Ion Mass Spectrometry: A Practical Handbook for Depth Profiling and Bulk Impurity Analysis (eds Wilson, R. G. et al.) (Wiley, New York, 1989).
Merkle, R., Hoedl, M. F., Raimondi, G., Zohourian, R. & Maier, J. Oxides with mixed protonic and electronic conductivity. Annu. Rev. Mater. Res. 51, 461–493 (2021).
Andersen, H. Functional Properties and Electrochemical Performance of New Positrode Materials for Proton Ceramic Electrochemical Cells. MsC thesis, Department of Chemistry, University of Oslo, 2020).
Liu, C.-Y. et al. Orbital-overlap-driven hybridization in 3d-transition metal perovskite oxides LaMO3 (M = Ti-Ni) and La2CuO4. Commun. Phys. 7, 156 (2024).
Norby, T. EMF method determination of conductivity contributions from protons and other foreign ions in oxides. Solid State Ion 28–30, 1586–1591 (1988).
Szpunar, I. et al. High-temperature structural and electrical properties of BaLnCo2O6 positrodes. Materials 13, 4044 (2020).
Acknowledgements
This work is financed by The Research Council of Norway (Grant nᵒ 272797 “GoPHy MiCO” and nᵒ 299736 “FunKeyCat”) through the M-ERA.NET Joint Call 2016 and 2018 (R.S., M.H.S., T.N.). Project “FunKeyCat” is supported by the National Science Centre, Poland, under the M-ERA.NET 2, which has received funding from the European Union’s Horizon 2020 research and innovation programme under grant agreement no 685451 (2016/22/Z/ST5/00691) (S.L.W., A.M.G., M.G.), and the Spanish Government through the National Research Agency AEI (grant PCIN-2017-125) (M.B., J.A.S.). This publication was partially developed under the provision of the Polish Ministry and Higher Education project “Support for research and development with the use of research infrastructure of the National Synchrotron Radiation Centre SOLARIS” under contract nr 1/SOL/2021/2 (S.L.W., A.M.G., M.G.). We acknowledge the SOLARIS Centre for access to the Beamline PIRX, where the measurements were performed. We thank Dr. Marcin Zając for assistance (proposal nos. 191011 and 201036). We also thank Dr. Vegar Øygarden for Rietveld refinements of NPD data.
Author information
Authors and Affiliations
Contributions
R.S.: Conceptualisation (lead), data curation (equal), formal analysis (equal), funding acquisition (lead), investigation (equal), methodology (equal), project administration (lead), supervision, validation (lead), visualisation (lead), writing original draft (lead). T.N.S.: Investigation – SIMS measurements on BGLC82. L.V.: Investigation – SIMS measurements on BGLC37. P.A.C.: Investigation – STEM-EDS. M.H.S.: SR-PXD and NPD, data curation (equal), formal analysis (equal), funding acquisition (equal), investigation (equal), methodology (equal). S.L.W.: Formal analysis (equal), funding acquisition (equal), investigation (equal), methodology (equal), project administration (equal), supervision, validation (equal), writing (equal). I.S.: Formal analysis (equal), investigation (equal), methodology (equal), validation (equal). A.M.G.: Formal analysis (equal), funding acquisition (equal), investigation (equal), methodology (equal), project administration (equal), supervision, validation (equal). M.B.: Formal analysis (equal), funding acquisition (equal), investigation (equal), methodology (equal), project administration (equal), supervision, validation (equal). T.N., M.G. and J.M.S.: funding acquisition (equal), investigation (equal), methodology (equal), project administration (equal), supervision, Writing – Review & Editing.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Guntae Kim and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Strandbakke, R., Wachowski, S.L., Balaguer, M. et al. Governing principles of hydration of mixed proton conducting Co-based double perovskites. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70212-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70212-w