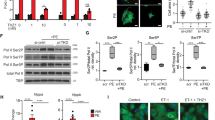
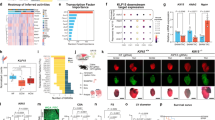
Abstract
Hypertensive heart failure highlights an urgent need for effective therapeutic strategies. Protein kinases regulate multiple pathways in cardiac pathophysiology and may provide promising therapeutic targets. Here, we identified a Cyclin-dependent kinase, CDK9, promoting inflammation and cardiac remodeling in terminally differentiated cardiomyocytes. Firstly, kinase enrichment analysis and experimental evidence revealed CDK9 phosphorylation at Thr-186 in both human and mouse hypertrophic heart tissues. CDK9 loss of function via T186A mutation in cardiomyocytes attenuated Ang II-induced heart remodeling and NF-κB-mediated inflammation, whereas CDK9 overactivation by T186E mutation induces. This regulatory function of CDK9 in cardiac remodeling is cell cycle-independent. Further studies demonstrate that the kinase domain of CDK9 directly binds to NF-κB P65 protein, which leads to the CDK9/P65 complex nuclear translocation, P65 phosphorylation, and transcription of inflammatory and hypertrophic genes in cardiomyocytes. This process requires CDK9 Thr-186 phosphorylation and Cyclin T1 presence, but is independent on IKKβ and CDK9-RNAPII pathways. Pharmacological inhibition of CDK9 phosphorylation significantly attenuated Ang II-induced cardiac inflammation, remodeling, and dysfunction in mice. Collectively, Ang II-activated CDK9 directly binds to and phosphorylates P65 to drive cardiac inflammation and remodeling. This study identifies CDK9 as a potential target in heart failure therapeutics.
Similar content being viewed by others
Data availability
The raw bulk RNA-sequencing data have been deposited in the Sequence Read Archive (SRA) under BioProject accession number PRJNA1012513. The raw data of mass spectrometry Data are available via the PRIDE database under accession code PXD074483, and the raw data of mass spectrometry Data of Quantitative Phosphoproteomics are available via the PRIDE database under accession code PXD074496. Source data are provided with this paper.
References
GBD 2016 Risk Factors Collaborators Global, regional, and national comparative risk assessment of 84 behavioural, environmental and occupational, and metabolic risks or clusters of risks, 1990-2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet 390, 1345–1422 (2017).
Cleland, J. G. F. et al. The effect of spironolactone on cardiovascular function and markers of fibrosis in people at increased risk of developing heart failure: the heart ‘OMics’ in AGEing (HOMAGE) randomized clinical trial. Eur. Heart J. 42, 684–696 (2021).
Higashikuni, Y. et al. NLRP3 Inflammasome activation through heart-brain interaction initiates cardiac inflammation and hypertrophy during pressure overload. Circulation https://doi.org/10.1161/circulationaha.122.060860 (2022).
Wang, J., Gareri, C. & Rockman, H. A. G-Protein-coupled receptors in heart disease. Circ. Res. 123, 716–735 (2018).
Patel, V. B., Lezutekong, J. N., Chen, X. & Oudit, G. Y. Recombinant human ACE2 and the angiotensin 1-7 axis as potential new therapies for heart failure. Can. J. Cardiol. 33, 943–946 (2017).
Manning, G., Whyte, D. B., Martinez, R., Hunter, T. & Sudarsanam, S. The protein kinase complement of the human genome. Science 298, 1912–1934 (2002).
Johnson, J. L. et al. An atlas of substrate specificities for the human serine/threonine kinome. Nature 613, 759–766 (2023).
Graña, X. et al. PITALRE, a nuclear CDC2-related protein kinase that phosphorylates the retinoblastoma protein in vitro. Proc. Natl. Acad. Sci. USA 91, 3834–3838 (1994).
Anshabo, A. T., Milne, R., Wang, S. & Albrecht, H. CDK9: A comprehensive review of its biology, and its role as a potential target for anti-cancer agents. Front. Oncol. 11, 678559 (2021).
O’Brien, T., Hardin, S., Greenleaf, A. & Lis, J. T. Phosphorylation of RNA polymerase II C-terminal domain and transcriptional elongation. Nature 370, 75–77 (1994).
Fujinaga, K., Huang, F. & Peterlin, B. M. P-TEFb: The master regulator of transcription elongation. Mol. Cell 83, 393–403 (2023).
Chen, R. et al. Cyclin-dependent kinase inhibitor fadraciclib (CYC065) depletes anti-apoptotic protein and synergizes with venetoclax in primary chronic lymphocytic leukemia cells. Leukemia 36, 1596–1608 (2022).
Thieme, E. et al. CDK9 inhibition induces epigenetic reprogramming revealing strategies to circumvent resistance in lymphoma. Mol. cancer 22, 64 (2023).
Li, X. et al. The methyltransferase METTL3 negatively regulates nonalcoholic steatohepatitis (NASH) progression. Nat. Commun. 12, 7213 (2021).
Ao, M. T. et al. The CDK9-targeting degrader PO-8 alleviates LPS-induced inflammation by inhibiting JAK-STAT signaling in vitro and in vivo. Biochem. Pharm. 242, 117341 (2025).
Espinoza-Derout, J. et al. Pivotal role of cardiac lineage protein-1 (CLP-1) in transcriptional elongation factor P-TEFb complex formation in cardiac hypertrophy. Cardiovasc. Res. 75, 129–138 (2007).
Lin, J. R. et al. Brown adipocyte ADRB3 mediates cardioprotection via suppressing exosomal iNOS. Circul. Res. 131, 133–147 (2022).
Egloff, S. CDK9 keeps RNA polymerase II on track. Cell Mol. Life Sci. 78, 5543–5567 (2021).
Simpson, P., McGrath, A. & Savion, S. Myocyte hypertrophy in neonatal rat heart cultures and its regulation by serum and by catecholamines. Circul. Res. 51, 787–801 (1982).
Olson, C. M. et al. Pharmacological perturbation of CDK9 using selective CDK9 inhibition or degradation. Nat. Chem. Biol. 14, 163–170 (2018).
Breuer, D. et al. CDK2 regulates HIV-1 transcription by phosphorylation of CDK9 on serine 90. Retrovirology 9, 94 (2012).
Mbonye, U. et al. Cyclin-dependent kinase 7 (CDK7)-mediated phosphorylation of the CDK9 activation loop promotes P-TEFb assembly with Tat and proviral HIV reactivation. J. Biol. Chem. 293, 10009–10025 (2018).
Baumli, S. et al. The structure of P-TEFb (CDK9/cyclin T1), its complex with flavopiridol and regulation by phosphorylation. EMBO J. 27, 1907–1918 (2008).
Liu, Y. et al. Macrophage CARD9 mediates cardiac injury following myocardial infarction through regulation of lipocalin 2 expression. Signal Transduct. Target. Ther. 8, 394 (2023).
Enzan, N. et al. ZBP1 Protects Against mtDNA-Induced Myocardial Inflammation in Failing Hearts. Circul. Res. 132, 1110–1126 (2023).
Tahirov, T. H. et al. Crystal structure of HIV-1 Tat complexed with human P-TEFb. Nature 465, 747–751 (2010).
Walker, R. L., Hornicek, F. J. & Duan, Z. Transcriptional regulation and therapeutic potential of cyclin-dependent kinase 9 (CDK9) in sarcoma. Biochem. Pharm. 226, 116342 (2024).
Guhan, S. M. et al. The molecular context of vulnerability for CDK9 suppression in triple wild-type melanoma. J. Invest. Dermatol. 141, 2018–2027 (2021).
Milletti, G., Colicchia, V. & Cecconi F. Cyclers’ kinases in cell division: from molecules to cancer therapy. Cell Death Differ. 30, 2035–2052 (2023).
Malumbres, M. Cyclin-dependent kinases. Genome Biol. 15, 122 (2014).
Hsu, A. et al. Targeting transcription in heart failure via CDK7/12/13 inhibition. Nat. Commun. 13, 4345 (2022).
Sano, M. & Schneider, M. D. Cyclin-dependent kinase-9: an RNAPII kinase at the nexus of cardiac growth and death cascades. Circul. Res. 95, 867–876 (2004).
Core, L. & Adelman, K. Promoter-proximal pausing of RNA polymerase II: a nexus of gene regulation. Genes Dev. 33, 960–982 (2019).
Zhou, Q., Li, T. & Price, D. H. RNA polymerase II elongation control. Annu. Rev. Biochem. 81, 119–143 (2012).
Parua, P. K. & Fisher, R. P. Dissecting the Pol II transcription cycle and derailing cancer with CDK inhibitors. Nat. Chem. Biol. 16, 716–724 (2020).
Funamoto, M. et al. Pyrazole-curcumin suppresses cardiomyocyte hypertrophy by disrupting the CDK9/cyclinT1 complex. Pharmaceutics. 14, 1269 (2022).
Sano, M. et al. Activation and function of cyclin T-Cdk9 (positive transcription elongation factor-b) in cardiac muscle-cell hypertrophy. Nat. Med. 8, 1310–1317 (2002).
Zhu, C., Yuan, T. & Krishnan, J. Targeting cardiomyocyte cell cycle regulation in heart failure. Basic Res. Cardiol. 119, 349–369 (2024).
Larochelle, S. et al. Cyclin-dependent kinase control of the initiation-to-elongation switch of RNA polymerase II. Nat. Struct. Mol. Biol. 19, 1108–1115 (2012).
Bacon, C. W. & D’Orso, I. CDK9: a signaling hub for transcriptional control. Transcription 10, 57–75 (2019).
Zhou, M. et al. The Tat/TAR-dependent phosphorylation of RNA polymerase II C-terminal domain stimulates cotranscriptional capping of HIV-1 mRNA. Proc. Natl. Acad. Sci. USA 100, 12666–12671 (2003).
Elagib, K. E. et al. Cross-talk of GATA-1 and P-TEFb in megakaryocyte differentiation. Blood 112, 4884–4894 (2008).
Barboric, M., Nissen, R. M., Kanazawa, S., Jabrane-Ferrat, N. & Peterlin, B. M. NF-kappaB binds P-TEFb to stimulate transcriptional elongation by RNA polymerase II. Mol. cell 8, 327–337 (2001).
Abdullah, M., Berthiaume, J. M. & Willis, M. S. Tumor necrosis factor receptor-associated factor 6 as a nuclear factor kappa B-modulating therapeutic target in cardiovascular diseases: at the heart of it all. Transl. Res. J. Lab. Clin. Med. 195, 48–61 (2018).
Bettelli, E., Dastrange, M. & Oukka, M. Foxp3 interacts with nuclear factor of activated T cells and NF-kappa B to repress cytokine gene expression and effector functions of T helper cells. Proc. Natl. Acad. Sci. USA 102, 5138–5143 (2005).
Fukui, T. et al. Bromodomain-containing-protein-4 and cyclin-dependent-kinase-9 inhibitors interact synergistically in vitro and combined treatment reduces post-traumatic osteoarthritis severity in mice. Osteoarthr. Cartil. 29, 68–77 (2021).
Diamond, J. R. et al. First-in-human dose-escalation study of cyclin-dependent kinase 9 inhibitor VIP152 in patients with advanced malignancies shows early signs of clinical efficacy. Clin. Cancer Res. 28, 1285–1293 (2022).
McDonagh, T. A. et al. 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur. Heart J. 42, 3599–3726 (2021).
Nakamura, K. et al. Inhibitory effects of antioxidants on neonatal rat cardiac myocyte hypertrophy induced by tumor necrosis factor-alpha and angiotensin II. Circulation 98, 794–799 (1998).
Roffel, M. P. et al. Identification of asthma-associated microRNAs in bronchial biopsies. Eur. Respir. J. 59, 2101294 (2022).
Weng, G. et al. HawkDock: a web server to predict and analyze the protein-protein complex based on computational docking and MM/GBSA. Nucleic Acids Res. 47, W322–w330 (2019).
Salomon-Ferrer, R., Case, R. A. & Walker, R. C. An overview of the Amber biomolecular simulation package. WIREs Comput. Mol. Sci. 3, 198–210 (2012).
Chen, J. et al. Trimetazidine prevents macrophage-mediated septic myocardial dysfunction via activation of the histone deacetylase sirtuin 1. Br. J. Pharmacol. 173, 545–561 (2016).
Percie du Sert, N. et al. Reporting animal research: Explanation and elaboration for the ARRIVE guidelines 2.0. PLoS Biol. 18, e3000411 (2020).
Wang, L. et al. CXCL1-CXCR2 axis mediates angiotensin II-induced cardiac hypertrophy and remodelling through regulation of monocyte infiltration. Eur. Heart J. 39, 1818–1831 (2018).
Kandalam, V. et al. TIMP2 deficiency accelerates adverse post-myocardial infarction remodeling because of enhanced MT1-MMP activity despite lack of MMP2 activation. Circul. Res. 106, 796–808 (2010).
Fernandez, N. F. et al. Clustergrammer, a web-based heatmap visualization and analysis tool for high-dimensional biological data. Sci. Data 4, 170151 (2017).
Kuleshov, M. V. et al. Enrichr: a comprehensive gene set enrichment analysis web server 2016 update. Nucleic Acids Res. 44, W90–W97 (2016).
Sahar, S. et al. Cooperation of SRC-1 and p300 with NF-kappaB and CREB in angiotensin II-induced IL-6 expression in vascular smooth muscle cells. Arterioscler. Thromb. Vasc. Biol. 27, 1528–1534 (2007).
Wang, H. et al. The transcriptional coactivator RUVBL2 regulates Pol II clustering with diverse transcription factors. Nat. Commun. 13, 5703 (2022).
Acknowledgements
This study was supported by the National Natural Science Foundation of China (U24A20814 to G.L., 82570395 to S.Y., 82370334 to D.L.), the China National Postdoctoral Program for Innovative Talents (BX20240329 to S.Y.), the Postdoctoral Science Foundation (2025M772361 to S.Y.), and the Zhejiang Provincial Natural Science Foundation of China (LMS26H020006 to S.Y.).
Author information
Authors and Affiliations
Contributions
Guang Liang, Guosheng Fu and Yanbo Zhao contributed to the literature search and study design. Guang Liang and Shiju Ye participated in the drafting of the article. Shiju Ye, Hanxiao Tu, Xue Han, Tong Liu, Yingchao Gong, Jiangting Lu, Tingting Jin and Wu Luo carried out the experiments. Yingchao Gong and Dongwu Lai collected the Clinical samples. Yanbo Zhao, Xuefeng Qu, and Guang Liang revised the manuscript. Shiju Ye, Yanbo Zhao and Hanxiao Tu contributed to data collection and analysis.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Sergei Nekhai and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Ye, S., Zhao, Y., Tu, H. et al. Cardiomyocyte Cyclin-dependent kinase 9 directly binds to and phosphorylates NF-κB p65 subunit to drive cardiac inflammation and remodeling. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70410-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70410-6