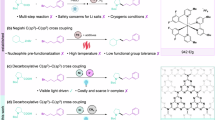
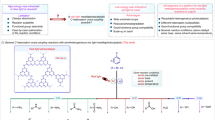
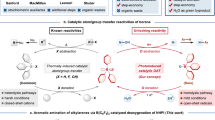
Abstract
Photoinduced transition metal catalysis offers innovative strategies for fostering novel chemical reactions and improving established ones. In this work, we present a highly efficient, photoinduced Mn(II)-bipyridine catalyzed C–N, C-O and C-S coupling reaction between aryl halides—particularly less reactive aryl chlorides—and nucleophiles containing nitrogen, oxygen, and sulfur. This protocol does not need an external photocatalyst, as the single Mn(II)–bipyridine complex simultaneously serves as both the light-harvester and the metal catalyst. This method exhibits excellent substrate scope, covering eight different nitrogen sources for C-N coupling, as well as C-O coupling with alcohols, C-S coupling with thiophenols, encompassing more than 150 examples, with yields reaching up to 94%. Mechanistic studies suggest that this reaction may be initiated and sustained by the Mn(I) species through the photoinduced homolysis of the catalyst precursor bipyridine-Mn(II)(OAc)2, likely proceeding via a Mn(I)/Mn(III) catalytic cycle.
Similar content being viewed by others
Data availability
Data available in this study are provided in the supplementary information. Crystallographic data coordinates for structures reported in this article has been deposited at the Cambridge Crystallographic Data Center (CCDC), under deposition numbers CCDC 2224253 (Mn complex 174). The sedata can be obtained free of charge from the Cambridge Crystallographic Data Center via https://www.ccdc.cam.ac.uk/structures/.
References
Crabtree, R. H. et al. The Organometallic chemistry of the Transition Metals (2009).
Molnár, Á. et al. Palladium-Catalyzed Coupling Reactions: Practical Aspects and Future Developments (2013).
Meijere, A., Bräse, S. & Oestreich, M. Metal-Catalyzed Cross-Coupling Reactions and More (2014).
Hartwig, J. F. Carbon–heteroatom bond formation catalyzed by organometallic complexes. Nature 455, 314 (2008).
Bariwal, J. & Van der Eycken, E. V. C–N bond forming cross-coupling reactions: an overview. Chem. Soc. Rev. 42, 9283 (2013).
Ruiz-Castillo, P. & Buchwald, S. L. Applications of palladium-catalyzed C–N cross-coupling reactions. Chem. Rev. 116, 12564 (2016).
Forero-Cortés, P. A. & Haydl, A. M. The 25th anniversary of the Buchwald-Hartwig amination: Development, applications, and outlook. Org. Process Res. Dev. 23, 1478 (2019).
Dorel, R., Grugel, C. P. & Haydl, A. M. The Buchwald-Hartwig amination after 25 years. Angew. Chem. Int. Ed. 58, 17118 (2019).
Sortais, J.-B. Manganese Catalysis in Organic Synthesis. (2021).
Wenger, O. S. Photoactive nickel complexes in cross-coupling catalysis. Chem. Eur. J. 27, 2270 (2021).
Wu, H. et al. Recent advances in non-precious metal catalysis. Org. Process Res. Dev. 26, 2281 (2022).
Philip, R. M., Saranya, P. V. & Anilkumar, G. Nickel-catalyzed amination of arenes and heteroarenes. Eur. J. Org. Chem. 2022, e202200184 (2022).
Zhu, S., Li, H., Li, Y., Huang, Z. & Chu, L. Exploring visible light for carbon–nitrogen and carbon–oxygen bond formation via nickel catalysis. Org. Chem. Front. 10, 548 (2023).
Monnier, F. & Taillefer, M. Catalytic C-C, C-N, and C-O Ullmann-type coupling reactions. Angew. Chem. Int. Ed. 48, 6954 (2009).
Sambiagio, C., Marsden, S. P., Blacker, A. J. & McGowan, P. C. Copper catalyzed Ullmann type chemistry: from mechanistic aspects to modern development. Chem. Soc. Rev. 43, 3525 (2014).
Bhunia, S., Pawar, G. G., Kumar, S. V., Jiang, Y. & Ma, D. Selected copper-based reactions for C−N, C−O, C−S, and C−C bond formation. Angew. Chem. Int. Ed. 56, 16136 (2017).
Teo, Y.-C. & Chua, G.-L. Cobalt-catalyzed N-arylation of nitrogen nucleophiles in water. Chem. Eur. J. 15, 3072 (2017).
Toma, G. & Yamaguchi, R. Cobalt-catalyzed C–N bond-forming reaction between chloronitrobenzenes and secondary amines. Eur. J. Org. Chem. 2010, 6404–6408 (2010).
Tan, B. Y.-H. & Teo, Y.-C. Efficient cobalt-catalyzed C–N cross-coupling reaction between benzamide and aryl iodide in water. Org. Biomol. Chem. 12, 7478 (2014).
Lobie, E., Subbegowda, R. K. & Basappa Development of a new arylamination reaction catalyzed by polymer bound 1,3-(bisbenzimidazolyl) benzene Co(II) complex and generation of bioactive adamanate amines. Catalysts 10, 1315 (2017).
Ahmad, K., Chang, C.-R. & Li, J. Mechanistic investigations of Co(II)-Catalyzed C-N coupling reactions. Organomet. Chem. 868, 144 (2018).
Song, G. et al. Adaptive photochemical amination via Co(II) catalysis. J. Am. Chem. Soc. 146, 26936 (2024).
Institute of Medicine. Food and Nutrition Board. Dietary Reference Intakes for Vitamin A, Vitamin K, Arsenic, Boron, Chromium, Copper, Iodine, Iron, Manganese, Molybdenum, Nickel, Silicon, Vanadium, and Zinc (National Academy Press, 2001).
Lin, S. et al. Mapping the dark space of chemical reactions with extended nanomole synthesis and MALDI-TOF MS. Science 361, eaar6236 (2018).
Pohl, P., Bielawska-Pohl, A., Dzimitrowicz, A., Jamroz, P. & Welna, M. Impact and practicability of recently introduced requirements on elemental impurities. Trends Anal. Chem. 101, 43 (2018).
Egorova, K. S. & Ananikov, V. P. Toxicity of metal compounds: Knowledge and myths. Organometallics 36, 4071 (2017).
Teo, Y. -C., Yong, F.-F., Poh, C.-Y., Yan, Y.-K. & Chua, G.-L. Manganese-catalyzed cross-coupling reactions of nitrogen nucleophiles with aryl halides in water. Chem. Commun. 41, 6258–6260 (2009).
Yong, F.-F. & Teo, Y.-C. Manganese-catalyzed cross-coupling reactions of aliphatic amines with aryl halides. Tetrahedron Lett. 52, 3910 (2010).
Teo, Y.-C., Yong, F.-F. & Lim, G. S. A manganese/copper bimetallic catalyst for C–N coupling reactions under mild conditions in water. Tetrahedron Lett. 53, 7171 (2011).
Yong, F.–F. & Teo, Y.-C. Manganese-mediated N-heteroarylation of indoles and indazoles in water. Synlett 23, 2106 (2012).
Teo, Y.-C., Yong, F.-F., Ithnin, I. K., Yio, S.-H. T. & Lin, Z. Efficient manganese/copper bimetallic catalyst for N-arylation of amides and sulfonamides under mild conditions in water. Eur. J. Org. Chem. 2013, 515–524 (2013).
Santilli, C. et al. The manganese-catalyzed cross-coupling reaction and the influence of trace metals. Eur. J. Org. Chem. 2017, 5269–5274 (2017).
Carney, J. R., Dillon, B. R. & Thomas, S. P. Recent advances of manganese catalysis for organic synthesis. Eur. J. Org. Chem. 2016, 3912–3929 (2016).
Garbe, M., Junge, K. & Beller, M. Homogeneous catalysis by manganese-based pincer complexes. Eur. J. Org. Chem. 2017, 4344–4362 (2017).
Liu, X., Jaroschik, F. & Taillefer, M. Manganese-Catalyzed Cross-Coupling Processes (2021).
Rohit, K. R., Saranya, S., Harry, N. A. & Anilkumar, G. A Novel ligand-free manganese-catalyzed C-O coupling protocol for the synthesis of biaryl ethers. ChemistrySelect 4, 5150 (2019).
Pichette Drapeau, M., Ollevier, T. & Taillefer, M. On the frontier between nucleophilic aromatic substitution and catalysis. Chem. Eur. J. 20, 5231 (2014).
Bandaru, M., Sabbavarpu, N. M., Katla, R. & Yadavalli, V. D. N. MnCl2·4H2O-catalyzed potential protocol for the synthesis of aryl/vinyl sulfides. Chem. Lett. 39, 1149 (2010).
Liu, T.–J., Yi, C.–L., Chan, C.–C. & Lee, C.–F. Manganese-catalyzed cross-coupling of thiols with aryl iodides. Chem. Asian J. 8, 1029 (2013).
Li, L.-J. et al. Recent advances in Mn, Fe, Co, and Ni-catalyzed organic reactions. CCS Chem. 6, 537 (2024).
Zhou, H. & Fu, C. Manganese-oxidizing microbes and biogenic manganese oxides: characterization, Mn(II) oxidation mechanism and environmental relevance. Rev. Environ. Sci. Biotechnol. 19, 489 (2020).
Everson, D. A., Shrestha, R. D. & Weix, J. Nickel-Catalyzed Reductive Cross-coupling of aryl halides with alkyl halides. J. Am. Chem. Soc. 132, 920 (2010).
Cherney, A. H. & Reisman, S. E. Nickel-catalyzed asymmetric reductive cross-coupling between vinyl and benzyl electrophiles. J. Am. Chem. Soc. 136, 14365 (2014).
Wu, B.-B., Xu, J., Bian, K.-J., Gao, Q. & Wang, X.-S. Enantioselective synthesis of secondary β-trifluoromethyl alcohols via catalytic asymmetric reductive trifluoroalkylation and diastereoselective reduction. J. Am. Chem. Soc. 144, 6543 (2022).
Nuhant, P. et al. Visible-light-initiated manganese catalysis for C-H alkylation of heteroarenes: applications and mechanistic studies. Angew. Chem. Int. Ed. 56, 15309 (2017).
Liang, Y.-F., Steinbock, R., Yang, L. & Ackermann, L. Continuous visible-light photoflow approach for a manganese-catalyzed (het)arene C-H arylation. Angew. Chem., Int. Ed. 57, 10625 (2018).
Yang, Y., Wang, C. Mn-Catalyzed C–C Coupling Reactions. (2023).
Narayanam, J. M. R. & Stephenson, C. R. J. Visible light photoredox catalysis: Applications in organic synthesis. Chem. Soc. Rev. 40, 102 (2011).
Prier, C. K., Rankic, D. A. & MacMillan, D. W. C. Visible light photoredox catalysis with transition metal complexes: Applications in organic synthesis. Chem. Rev. 113, 5322 (2013).
Twilton, J. et al. The merger of transition metal and photocatalysis. Nat. Rev. Chem. 1, 0052 (2017).
Candish, L. et al. Keesset. S. Photocatalysis in the life science industry. Chem. Rev. 122, 2907 (2022).
Li, G. et al. Light-promoted C–N coupling of aryl halides with nitroarenes. Angew. Chem. Int. Ed. 60, 5230 (2021).
Song, G. et al. Photochemical synthesis of anilines via Ni-catalyzed coupling of aryl halides with ammonium salts. ACS Catal. 12, 15590 (2022).
Song, G. et al. Werner salt as nickel and ammonia source for photochemical synthesis of primary aryl amines. Angew. Chem. Int. Ed. 63, e202314355 (2024).
Li, J. et al. Synthesis, characterization, and catalytic activity of Ni(0)(DQ)dtbbpy, an air-Stable, bifunctional red-light-sensitive precatalyst. J. Am. Chem. Soc. 147, 5851 (2025).
Song, G. et al. General C-heteroatom Cross-Coupling Reactions by Semi-heterogeneous Red-Light Metallaphotocatalysis. Nat. Commun. 16, 7045 (2025).
Wang, L., Lear, J. M., Rafferty, S. M., Fosu, S. C. & Nagib, D. A. Ketyl radical reactivity via atom transfer catalysis. Science 362, 225 (2018).
Britton, L. et al. sp2)-H borylation of furan and thiophene derivatives. ACS Catal. 11, 6857 (2021).
Zhao, C.-G. et al. Manganese(I)-catalyzed enantioselective C(sp2)-C(sp3) bond-forming for the synthesis of skipped dienes with synergistic aminocatalysis. Angew. Chem. Int. Ed. 63, e202400177 (2024).
Herr, P., Kerzig, C., Larsen, C. B., Häussinger, D. & Wenger, O. S. Manganese(I) complexes with metal-to-ligand charge transfer luminescence and photoreactivity. Nat. Chem. 13, 956 (2021).
Patra, K., Bhattacherya, A., Li, C., Bera, J. K. & Soo, H. S. Understanding the visible-light-initiated manganese-catalyzed synthesis of quinolines and naphthyridines under ambient and aerobic conditions. ACS Catal. 12, 15168 (2022).
Vivien, A., Veyre, L., Mirgalet, R., Camp, C. & Thieuleux, C. Mn2(CO)10 and UV light: a promising combination for regioselective alkene hydrosilylation at low temperature. Chem. Commun. 58, 4091 (2022).
Huang, T., Du, P., Cheng, X. & Lin, Y.-M. Manganese complexes with consecutive Mn(IV)-Mn(III) excitation for versatile photoredox. J. Am. Chem. Soc. 146, 24515 (2024).
For details, see SI.
Choury, M., Basilio Lopes, A., Blond, G. & Gulea, M. Synthesis of medium-sized heterocycles by transition-metal-catalyzed intramolecular cyclization. Molecules 25, 3147 (2020).
Song, G. et al. Chiral arylated amines via C−N coupling of chiral amines with aryl bromides promoted by light. Angew. Chem. Int. Ed. 60, 21536 (2021).
Jenkins, C. A., Murphy, D. M., Rowlands, C. C. & Egerton, T. A. EPR study of spin-trapped free radical intermediates formed in the heterogeneously-assisted photodecomposition of acetaldehyde. J. Chem. Soc. Perkin Trans. 2, 2479 (1997).
Valyaev, D. A., Lavigne, G. & Lugan, N. Manganese organometallic compounds in homogeneous catalysis: Past, present, and prospects. Coord. Chem. Rev. 308, 191 (2016).
Cahiez, G., Duplais, C. & Buendia, J. Chemistry of organomanganese(II) compounds. Chem. Rev. 109, 1434 (2009).
He, Y.-T. et al. C–C bond elimination from high-valent Mn aryl complexes. Organometallics 40, 2320 (2021).
Liu, W. et al. Oxidative aliphatic C-H fluorination with fluoride ion catalyzed by a manganese porphyrin. Science 337, 1322 (2012).
Paradine, S. M. et al. A manganese catalyst for highly reactive yet chemoselective intramolecular C(sp3)–H amination. Nat. Chem. 7, 987 (2015).
Kumar, A., Janes, T., Espinosa-Jalapa, A. N. & Milstein, D. Manganese catalyzed hydrogenation of organic carbonates to methanol and alcohols. Angew. Chem. Int. Ed. 57, 12076 (2018).
Zhang, L., Wang, Z., Han, Z. & Ding, K. Manganese-catalyzed anti-selective asymmetric hydrogenation of α-substituted β-ketoamides. Angew. Chem. Int. Ed. 59, 15565 (2020).
Liu, C. et al. Manganese-catalyzed asymmetric hydrogenation of quinolines enabled by π-π interaction. Angew. Chem. Int. Ed. 60, 5108 (2021).
Liu, C., Wang, M., Xu, Y., Li, Y. & Liu, Q. Manganese-catalyzed asymmetric hydrogenation of 3H-indoles. Angew. Chem. Int. Ed. 61, e202202814 (2022).
Perez, J. M. et al. Manganese(I)-catalyzed H–P bond activation via metal–ligand cooperation. J. Am. Chem. Soc. 143, 20071 (2021).
Acknowledgements
This research is supported by the National Natural Science Foundation of China (Grant No. 22171174, 22471150 to D.X.; Grant No. 22402113 to G.S.), the Fundamental Research Funds for the Central Universities (Grant No. GK202406026 to T.K.; Grant No. GK202505023 to G.S.; Grant No. GK202505026 to D.X.), the Innovation Capability Support Program of Shaanxi (Grant No. 2023-CX-TD-28 to D.X.), the Fundamental Science Research Project of Shaanxi for Chemistry, Biology (Grant No. 22JHZ0027 to D.X.), the Natural Science Foundation of Shaanxi Province (Grant No. 2024JC-YBQN-0075 to G.S.; Grant No. 2025JC-YBQN-144 to T.K.), the State Key Laboratory of Natural and Biomimetic Drugs (Grant No. K202437 to G.S.), the Key Research and Development Program of Shaanxi (Grant No. 2024CY-JJQ-26 to D.X.), and the S&T Program of Energy Shaanxi Laboratory (Grant No. ESLB202420 00 to D.X.).
Author information
Authors and Affiliations
Contributions
D.X. conceived and directed the project. G.S. designed and conducted experiments, analyzed data. J.S. participated in the substrate scope expansion. Q.L., X.S., X.L., D.P., J.F., and H.S. helped analyzed data. T.K., J.D., G.L., and C.W. contributed to the project discussion. G.S. prepared the manuscript. D.X. wrote the manuscript. All authors discussed the experimental results and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Haoyue Xiang and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Song, G., Song, J., Li, Q. et al. Photoinduced Mn catalysis for efficient platform for C-heteroatom bond coupling of aryl halides. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70925-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70925-y