Abstract
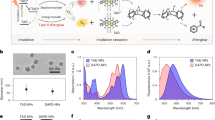
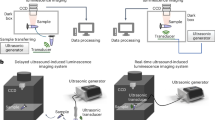
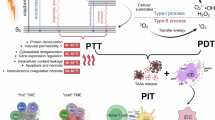
Persistent luminescence is a promising approach for photodynamic therapy (PDT) in deep-seated tumors, as it provides sustained light within tissues, eliminating the need for continuous external illumination. However, the uncontrollability of light within the body complicates precise spatiotemporal regulation. In this study, we report X-ray preactivated elimusertib-loaded tumor-targeted photodynamic nanoparticles (ETPNs), featuring reversible “on-off” afterglow properties. The excellent afterglow properties of X-ray-activated porous NaYF4:Er@NaGdF4 persistent luminescence nanoparticles enable the continuous activation of chlorin e6 (Ce6) to generate reactive oxygen species (ROS), leading to DNA damage. The integration of elimusertib potentiates ROS-induced DNA damage and activates the cGAS-STING pathway, thereby enhancing immuno-photodynamic therapeutic efficacy. All in vivo experiments were conducted using female mice. Our findings highlight the potential of ETPNs to advance the therapeutic landscape for deep-seated tumors, offering a robust and controllable platform for combined immuno-photodynamic therapy.
Similar content being viewed by others
Data availability
All data underlying this study are available from the corresponding author upon request (and its Supplementary Information files). Source data are provided with this paper.
References
Li, Z., Liu, H. & Zhang, X.-B. Reactive oxygen species-mediated organic long-persistent luminophores light up biomedicine: from two-component separated nano-systems to integrated uni-luminophores. Chem. Soc. Rev. https://doi.org/10.1039/D4CS00443D (2024).
Wang, Y. et al. Cyclic amplification of the afterglow luminescent nanoreporter enables the prediction of anti-cancer efficiency. Angew. Chem. Int. Ed. 60, 19779–19789 (2021).
Fan, W. et al. Enhanced afterglow performance of persistent luminescence implants for efficient repeatable photodynamic therapy. ACS Nano 11, 5864–5872 (2017).
Lucky, S. S., Soo, K. C. & Zhang, Y. Nanoparticles in photodynamic therapy. Chem. Rev. 115, 1990–2042 (2015).
Nestoros, E., Sharma, A., Kim, E., Kim, J. S. & Vendrell, M. Smart molecular designs and applications of activatable organic photosensitizers. Nat. Rev. Chem. https://doi.org/10.1038/s41570-024-00662-7 (2024).
Liang, L. et al. Controlling persistent luminescence in nanocrystalline phosphors. Nat. Mater. 22, 289–304 (2023).
Song, L. et al. Low-dose X-ray activation of W(VI)-doped persistent luminescence nanoparticles for deep-tissue photodynamic therapy. Adv. Funct. Mater. 28, 1707496 (2018).
Juengpanich, S. et al. Pre-activated nanoparticles with persistent luminescence for deep tumor photodynamic therapy in gallbladder cancer. Nat. Commun. 14, 5699 (2023).
Hu, J.-J., Lei, Q. & Zhang, X.-Z. Recent advances in photonanomedicines for enhanced cancer photodynamic therapy. Prog. Mater. Sci. 114, 100685 (2020).
Zuo, L. et al. Smart tumor-cell-derived microparticles provide on-demand photosensitizer synthesis and hypoxia relief for photodynamic therapy. Angew. Chem. Int. Ed. 60, 25365–25371 (2021).
Zhao, X., Liu, J., Fan, J., Chao, H. & Peng, X. Recent progress in photosensitizers for overcoming the challenges of photodynamic therapy: from molecular design to application. Chem. Soc. Rev. 50, 4185–4219 (2021).
Grossberg, A. J. et al. Multidisciplinary standards of care and recent progress in pancreatic ductal adenocarcinoma. Cancer J. Clin. 70, 375–403 (2020).
Klein, A. P. Pancreatic cancer epidemiology: understanding the role of lifestyle and inherited risk factors. Nat. Rev. Gastroenterol. Hepatol. 18, 493–502 (2021).
Hu, Z. I. & O’Reilly, E. M. Therapeutic developments in pancreatic cancer. Nat. Rev. Gastroenterol. Hepatol. 21, 7–24 (2024).
Mizrahi, J. D., Surana, R., Valle, J. W. & Shroff, R. T. Pancreatic cancer. Lancet 395, 2008–2020 (2020).
Ma, Y., Zhang, F. & Yang, Y. Recent advances in the treatment of pancreatic cancer. Holist. Integr. Oncol. 3, 34 (2024).
Yap, T. A. et al. First-in-human trial of the oral ataxia telangiectasia and RAD3-related (ATR) inhibitor BAY 1895344 in patients with advanced solid tumors. Cancer Discov. 11, 80–91 (2021).
Yap, T. A. et al. Phase I trial of first-in-class ATR inhibitor M6620 (VX-970) as monotherapy or in combination with carboplatin in patients with advanced solid tumors. J. Clin. Oncol. 38, 3195–3204 (2020).
Topatana, W. et al. Advances in synthetic lethality for cancer therapy: cellular mechanism and clinical translation. J. Hematol. Oncol. 13, 118 (2020).
Taniguchi, H. et al. ATR inhibition activates cancer cell cGAS/STING-interferon signaling and promotes antitumor immunity in small-cell lung cancer. Sci. Adv. 10, eado4618 (2024).
Liu, H., Shi, Y. & Qian, F. Opportunities and delusions regarding drug delivery targeting pancreatic cancer-associated fibroblasts. Adv. Drug Deliv. Rev. 172, 37–51 (2021).
O’Leary, P. C. et al. Resistance to ATR inhibitors is mediated by loss of the nonsense-mediated decay factor UPF2. Cancer Res. 82, 3950–3961 (2022).
Li, Y., Gecevicius, M. & Qiu, J. Long persistent phosphors-from fundamentals to applications. Chem. Soc. Rev. 45, 2090–2136 (2016).
Huang, K. et al. Designing next generation of persistent luminescence: recent advances in uniform persistent luminescence nanoparticles. Adv. Mater. 34, e2107962 (2022).
Wu, W., Shao, X., Zhao, J. & Wu, M. Controllable photodynamic therapy implemented by regulating singlet oxygen efficiency. Adv. Sci. 4, 1700113 (2017).
Ou, X. et al. High-resolution X-ray luminescence extension imaging. Nature 590, 410–415 (2021).
Pei, P. et al. X-ray-activated persistent luminescence nanomaterials for NIR-II imaging. Nat. Nanotechnol. 16, 1011–1018 (2021).
Li, L. et al. Persistent- and mechanoluminescence of Er(3+)-doped NaYF(4) for multipurpose use. Small Methods, e2401615, https://doi.org/10.1002/smtd.202401615 (2025).
Han, S., Deng, R., Xie, X. & Liu, X. Enhancing luminescence in lanthanide-doped upconversion nanoparticles. Angew. Chem. 53, 11702–11715 (2014).
Park, Y. I. et al. Theranostic probe based on lanthanide-doped nanoparticles for simultaneous in vivo dual-modal imaging and photodynamic therapy. Adv. Mater. 24, 5755–5761 (2012).
Wang, F. et al. Tuning upconversion through energy migration in core-shell nanoparticles. Nat. Mater. 10, 968–973 (2011).
Jiang, Z. Y. et al. Synthesis of single-crystalline ZnO polyhedral submicrometer-sized hollow beads using laser-assisted growth with ethanol droplets as soft templates. Adv. Mater. 16, 904–907 (2004).
Peng, Q., Dong, Y. & Li, Y. ZnSe semiconductor hollow microspheres. Angew. Chem. Int. Ed. 42, 3027–3030 (2003).
Zhou, L. et al. Mesoporous NaYbF4@NaGdF4 core-shell up-conversion nanoparticles for targeted drug delivery and multimodal imaging. Biomaterials 35, 7666–7678 (2014).
Liu, Y., Wang, D., Li, L., Peng, Q. & Li, Y. Energy upconversion in lanthanide-doped core/porous-shell nanoparticles. Inorg. Chem. 53, 3257–3259 (2014).
Lv, R. et al. Mesoporous NaYF4:Yb,Er@Au–Pt(iv)-FA nanospheres for dual-modal imaging and synergistic photothermal/chemo-anti-cancer therapy. RSC Adv. 5, 43391–43401 (2015).
Li, F. et al. Responsive assembly of upconversion nanoparticles for pH-activated and near-infrared-triggered photodynamic therapy of deep tumors. Adv. Mater. 30, e1802808 (2018).
Kim, B. et al. Tuning payload delivery in tumour cylindroids using gold nanoparticles. Nat. Nanotechnol. 5, 465–472 (2010).
Yuan, Y.-Y. et al. Surface charge switchable nanoparticles based on zwitterionic polymer for enhanced drug delivery to tumor. Adv. Mater. 24, 5476–5480 (2012).
Park, W. et al. Intermolecular structural change for thermoswitchable polymeric photosensitizer. J. Am. Chem. Soc. 138, 10734–10737 (2016).
Chen, M. et al. Bortezomib-encapsulated dual responsive copolymeric nanoparticles for gallbladder cancer targeted therapy. Adv. Sci. 9, e2103895 (2022).
Qian, C. et al. Light-activated hypoxia-responsive nanocarriers for enhanced anticancer therapy. Adv. Mater. 28, 3313–3320 (2016).
Tavakkoli Yaraki, M., Liu, B. & Tan, Y. N. Emerging strategies in enhancing singlet oxygen generation of nano-photosensitizers toward advanced phototherapy. Nano-Micro Lett. 14, 123 (2022).
Yang, M. et al. Multipath ROS storm and immune activation via sulfur vacancy-optimized ZnIn(2)S(4) nanosheets for piezocatalytic tumor therapy. Angew. Chem. e202507502, https://doi.org/10.1002/anie.202507502 (2025).
Yao, Y. et al. Photo-triggered fluorescence polyelectrolyte nanoassemblies: manipulate and boost singlet oxygen in photodynamic therapy. Angew. Chem. Int, Ed, n/a, e202416963, https://doi.org/10.1002/anie.202416963 (2024).
Ling, D. et al. Multifunctional tumor pH-sensitive self-assembled nanoparticles for bimodal imaging and treatment of resistant heterogeneous tumors. J. Am. Chem. Soc. 136, 5647–5655 (2014).
Seeliger, H. et al. Expression of estrogen receptor beta correlates with adverse prognosis in resected pancreatic adenocarcinoma. BMC Cancer 18, 1049 (2018).
Xie, X. et al. Tamoxifen enhances the anticancer effect of cantharidin and norcantharidin in pancreatic cancer cell lines through inhibition of the protein kinase C signaling pathway. Oncol. Lett. 9, 837–844 (2015).
Srinivas, U. S., Tan, B. W. Q., Vellayappan, B. A. & Jeyasekharan, A. D. ROS and the DNA damage response in cancer. Redox Biol. 25, 101084 (2019).
Liu, C. et al. Combining radiation and the ATR inhibitor berzosertib activates STING signaling and enhances immunotherapy via inhibiting SHP1 function in colorectal cancer. Cancer Commun. 43, 435–454 (2023).
Tang, Z. et al. ATR inhibition induces CDK1-SPOP signaling and enhances Anti-PD-L1 cytotoxicity in prostate cancer. Clin. Cancer Res. 27, 4898–4909 (2021).
Dvorkin, S., Cambier, S., Volkman, H. E. & Stetson, D. B. New frontiers in the cGAS-STING intracellular DNA-sensing pathway. Immunity 57, 718–730 (2024).
Kwon, J. & Bakhoum, S. F. The cytosolic DNA-sensing cGAS-STING pathway in cancer. Cancer Discov. 10, 26–39 (2020).
Wang, M. et al. Inhibition of tumor intrinsic BANF1 activates antitumor immune responses via cGAS-STING and enhances the efficacy of PD-1 blockade. J. Immunother. Cancer 11, https://doi.org/10.1136/jitc-2023-007035 (2023).
Hopp, M.-T. et al. Heme determination and quantification methods and their suitability for practical applications and everyday use. Anal. Chem. 92, 9429–9440 (2020).
Keum, H., Yoo, D. & Jon, S. Photomedicine based on heme-derived compounds. Adv. drug Deliv. Rev. 182, 114134 (2022).
Bourquin, J. et al. Biodistribution, clearance, and long-term fate of clinically relevant nanomaterials. Adv. Mater. 30, e1704307 (2018).
Hu, X. et al. Tailor-made nanomaterials for diagnosis and therapy of pancreatic ductal adenocarcinoma. Adv. Sci. 8, 2002545 (2021).
Agostinis, P. et al. Photodynamic therapy of cancer: an update. Cancer J. Clin. 61, 250–281 (2011).
Brown, S. B., Brown, E. A. & Walker, I. The present and future role of photodynamic therapy in cancer treatment. Lancet Oncol. 5, 497–508 (2004).
Gotoh, K. et al. A novel image-guided surgery of hepatocellular carcinoma by indocyanine green fluorescence imaging navigation. J. Surg. Oncol. 100, 75–79 (2009).
Qiu, R. et al. Deploying indocyanine green fluorescence-guided navigation system in precise laparoscopic resection of pediatric hepatoblastoma. Cancers 14, https://doi.org/10.3390/cancers14246057 (2022).
Liu, Y. et al. Modulating cancer-stroma crosstalk by a nanoparticle-based photodynamic method to pave the way for subsequent therapies. Biomaterials 289, 121813 (2022).
Zhang, T. et al. Synergistic effects of photodynamic therapy and chemotherapy: activating the intrinsic/extrinsic apoptotic pathway of anoikis for triple-negative breast cancer treatment. Biomater. Adv. 160, 213859 (2024).
Wengner, A. M. et al. The novel ATR inhibitor BAY 1895344 is efficacious as monotherapy and combined with DNA damage-inducing or repair-compromising therapies in preclinical cancer models. Mol. Cancer Ther. 19, 26–38 (2020).
Tao, H. et al. cGAS-STING pathway activation and systemic anti-tumor immunity induction via photodynamic nanoparticles with potent toxic platinum DNA intercalator against uveal melanoma. Adv. Sci. 10, e2302895 (2023).
Reisländer, T., Groelly, F. J. & Tarsounas, M. DNA damage and cancer immunotherapy: a STING in the tale. Mol. Cell 80, 21–28 (2020).
Alicea-Torres, K. et al. Immune suppressive activity of myeloid-derived suppressor cells in cancer requires inactivation of the type I interferon pathway. Nat. Commun. 12, 1717 (2021).
Li, K. et al. Myeloid-derived suppressor cells as immunosuppressive regulators and therapeutic targets in cancer. Signal Transduct. Target. Ther. 6, 362 (2021).
Li, S. et al. Adavosertib-encapsulated metal-organic frameworks for p53-mutated gallbladder cancer treatment via synthetic lethality. Sci. Bull. 69, 1286–1301 (2024).
Xie, T. et al. Targeted intracellular copper reservoir enhances liver cancer immunotherapy. Small e02783, https://doi.org/10.1002/smll.202502783 (2025).
Acknowledgements
This work was supported by the National Natural Science Foundation of China (Nos. 82473007, 82202873, 52173290, T2122003, and W2433188); Zhejiang Provincial Natural Science Foundation of China (No. LR25H160001); The Fundamental Research Funds for the Central Universities (No. 226-2025-00172); and State Key Laboratory of Advanced Drug Delivery and Release Systems (No. DSQZ-QN-202303). The authors would like to thank Huiwen Wang and Chaogang Xing from the Analysis Center of Agrobiology and Environmental Sciences, Zhejiang University; Yayu Qiu from the Department of Chemistry, Zhejiang University; Yu Liu and Xinhang Jiang from the College of Life Sciences, Zhejiang University; Lucheng Cai from the School of Materials Science and Engineering, Zhejiang University, and Guoqing Zhu from the Center for Electron Microscopy of Zhejiang University for their technical assistance in nanoparticle characterization; Yuchen Zhang from the Center of Cryo-Electron Microscopy (CCEM), Zhejiang University for technical assistance on Cryo-TEM; as well as Qike Jiang from the Instrumentation and Service Center for Physical Sciences, Westlake University for technical assistance on HAADF-STEM. The figures in this article were created using Adobe Illustrator, Adobe Photoshop, BioRender, Blender v4.0, and Microsoft PowerPoint.
Author information
Authors and Affiliations
Contributions
W. Topatana, Y. Sun, T. Xie, Y. Zhu, S. Juengpanich, and M. Chen designed the experiments, analyzed the data, and wrote the manuscript. X. Cai, R. Deng, S. Juengpanich, and M. Chen supervised the project and revised the manuscript. W. Topatana, T. Xie, Y. Zhu, T. Yang, P. Ran, C. Li, J. Chen, and Z. Lu synthesized and characterized the nanoparticles. W. Topatana, Y. Sun, R. Shen, X. Shen, Y. Han, Y. Shan, S. Li, and T. Chen performed in vitro and in vivo experiments. All authors discussed the results throughout the project and approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Yongsheng Li, Guang Peng, and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Topatana, W., Sun, Y., Xie, T. et al. X-ray preactivated reversible persistent luminescence enables photodynamic immunotherapy of deep tumors. Nat Commun (2026). https://doi.org/10.1038/s41467-026-71028-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-71028-4