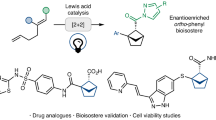
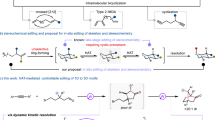
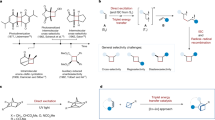
Abstract
Due to the ubiquitous presence of ortho-disubstituted benzenes in pharmaceuticals and agrochemicals, the exploration of their bioisosteres incorporating three-dimensional bridged cyclic scaffolds has attracted significant interest among chemists in recent years. As a result, 4,5-disubstituted 2-oxabicyclo[2.1.1]hexanes have recently emerged as promising candidates owing to their enhanced pharmacological potency, improved metabolic stability, and, importantly, superior aqueous solubility. Herein, we report our recent progress on enantioselective synthesis of such compounds via an intramolecular [2 + 2] photocycloaddition enabled by cooperative photoinduced energy transfer (EnT) and chiral Brønsted acid catalysis. A broad range of valuable pyridine-functionalized 2-oxabicyclo[2.1.1]hexane derivatives have been obtained in high yields with good to excellent enantioselectivity and diastereoselectivity. In addition, this transition metal-free approach offers an efficient and modular route to pyridine-based bicyclo[2.1.1]hexanes, which contain a significant yet challenging structural motif—pyridine rings directly attached to the bridgehead carbon atoms. Mechanistic studies demonstrate that the photosensitizer and low reaction temperature are crucial for rendering the chiral catalyst indispensable in facilitating substrate activation to the triplet state, thereby enabling enantiocontrol in this highly reactive transformation.
Similar content being viewed by others
Data availability
All data are available from the corresponding author. The authors declare that all data supporting the findings of this study are available in the paper and its Supplementary Information files. Crystallographic data for the structures reported in this Article have been deposited at the Cambridge Crystallographic Data Centre, under deposition numbers (CCDC 2496083) (2 g). Copies of the data can be obtained free of charge via https://www.ccdc.cam.ac.uk/structures/.
References
Meanwell, N. A. Fluorine and fluorinated motifs in the design and application of bioisosteres for drug design. J. Med. Chem. 61, 5822–5880 (2018).
Kumari, S., Carmona, A. V., Tiwari, A. K. & Trippier, P. C. Amide bond bioisosteres: strategies, synthesis, and successes. J. Med. Chem. 63, 12290–12358 (2020).
Mykhailiuk, P. K. Saturated bioisoteres of benzene: where to go next? Org. Biomol. Chem. 17, 2839–2849 (2019).
Subbaiah, M. A. M. & Meanwell, N. A. Biosiosteres of the phenyl ring: recent strategic applications in lead optimization and drug design. J. Med. Chem. 64, 14046–14128 (2021).
Tsien, J., Hu, C., Merchant, R. R. & Qin, T. Three-dimensional saturated C(sp3)-rich bioisosteres for benzene. Nat. Rev. Chem. 8, 605–627 (2024).
Zhao, J.-X. et al. 1,2-Difuntionalized bicyclo[1.1.1]pentanes: long-sought-after mimetic for ortho/meta-substituted arenes. Proc. Natl. Acad. Sci. USA. 118, e2108881118 (2021).
Dargazanli, G., Estenne-Bouhtou, G. & Mafroud, A.-K. N-[(2-Azabicyclo[2.1.1]hex-1-y)-aryl-methyl]-benzamide derivatives, preparation thereof, and therapeutic use thereof. WO2010092286A (2010).
Yang, B., Yang, G., Wang, Q. & Zhu, J. Pd-catalyzed strain-releasing dyotropic rearrangement: ring-expanding amidofluorination of methylenecyclobutanes. J. Am. Chem. Soc. 147, 8969–8977 (2025).
Mingzhe, J. T., Martin, A. T., Desai, M. C., Jin, H. & Pyun, H.-J. Polycyclic-carbamoylpyridone compounds and their use for the treatment of HIV infections. WO2015006733A1 (2015).
Qiao, J. X. et al. Achieving structural diversity using the perpendicular conformation of alpha-substituted phenylcyclopropanes to mimic the bioactive conformation of ortho-substituted biphenyl P4 moieties: Discovery of novel, highly potent inhibitors of Factor Xa. Bioorg. Med. Chem. Lett. 18, 4118–4123 (2008).
Shinozuka, T. et al. Discovery of DS-1971a, a potent, selective NaV1.7 inhibitor. J. Med. Chem. 63, 10204–10220 (2020).
Ma, X., Han, Y. & Bennett, D. J. Selective synthesis of 1-dialkylamino-2-alkylbicyclo-[1.1.1]pentanes. Org. Lett. 22, 9133–9138 (2020).
Denisenko, A., Garbuz, P., Shishkina, S. V., Voloshchuk, N. M. & Mykhailiuk, P. K. Saturated bioisosteres of ortho-substituted benzenes. Angew. Chem. Int. Ed. 59, 20515–20521 (2020).
Yang, Y. et al. An intramolecular coupling approach to alkyl bioisosteres for the synthesis of multisubstituted bicycloalkyl boronates. Nat. Chem. 13, 950–955 (2021).
Liang, Y., Kleinmans, R., Daniliuc, C. G. & Glorius, F. Synthesis of polysubstituted 2-oxabicyclo[2.1.1]hexanes via visible-light-induced energy transfer. J. Am. Chem. Soc. 144, 20207–20213 (2022).
Harmata, A. S., Spiller, T. E., Sowden, M. J. & Stephenson, C. R. J. Photochemical formal [4 + 2]-cycloaddition of imine-substituted bicycle[1.1.1]pentanes and alkenes. J. Am. Chem. Soc. 143, 21223–21228 (2021).
Guo, R. et al. Strain-release [2π + 2σ] cycloadditions for the synthesis of bicycle[2.1.1]hexanes initiated by energy transfer. J. Am. Chem. Soc. 144, 7988–7994 (2022).
Kleinmans, R. et al. Intermolecular [2π + 2σ]-photocycloaddition enabled by triplet energy transfer. Nature 605, 477–482 (2022).
Agasti, S. et al. Catalytic alkene insertion approach to bicycle[2.2.1]hexane bioisosteres. Nat. Chem. 15, 535–541 (2023).
Sailer, J. K., Ly, D., Musaev, D. G. & Davies, H. M. L. Direct synthesis of bicyclo[1.1.1]pentanes by sequential C=C, C‒C functionalization reactions. J. Am. Chem. Soc. 147, 31034–31041 (2025).
Yang, J. et al. Asymmetric dearomative [2 + 2] photocycloaddition of quinoline and indole derivatives with bicyclo[1.1.0]butanes. J. Am. Chem. Soc. 147, 35755–35766 (2025).
Yi, L. et al. Modular access to sp3-rich bicyclo[1.1.1]pentane bioisosteres via energy-transfer-mediated carbene insertion. Angew. Chem. Int. Ed. 65, e18508 (2026).
Denisenko, A. et al. 2-Oxabicyclo[2.1.1]hexanes as saturated bioisosteres of the ortho-substituted phenyl ring. Nat. Chem. 15, 1155–1163 (2023).
Zhou, Q.-Q., Zou, Y.-Q., Lu, L.-Q. & Xiao, W.-J. Visible-light-induced organic photochemical reactions through energy-transfer pathways. Angew. Chem. Int. Ed. 58, 1586–1604 (2019).
Yao, W., Bazan-Bergamino, E. A. & Ngai, M.-Y. Asymmetric photocatalysis enabled by chiral organocatalysts. ChemCatChem 14, e202101292 (2022).
Mondal, S. et al. Enantioselective radical reactions using chiral catalysts. Chem. Rev. 122, 5842–5976 (2022).
Lv, X., Xu, H., Yin, Y., Zhao, X. & Jiang, Z. Visible light-driven cooperative DPZ and chiral hydrogen-bonding catalysis. Chin. J. Chem. 38, 1480–1488 (2020).
Yin, Y., You, M., Li, X. & Jiang, Z. Catalytic asymmetric photocycloaddition reactions mediated by enantioselective radical approaches. Chem. Soc. Rev. 54, 2246–2274 (2025).
Rigotti, T., Schwinger, D. P., Graßl, R., Jandl, C. & Bach, T. Enantioselective crossed intramolecular [2 + 2] photocycloaddition reactions mediated by a chiral chelating Lewis acid. Chem. Sci. 13, 2378–2384 (2022).
Qin, T., He, M. & Zi, W. Palladium-catalysed [2σ + 2π] cycloaddition reactions of bicycle[1.1.0]butanes with aldehydes. Nat. Synth. 4, 124–133 (2025).
Garrido-García, P. et al. Enantioselective photocatalytic synthesis of bicycle[2.2.1]hexanes as ortho-disubstituted benzene bioisosteres with improved biological activity. Nat. Chem. 17, 734–745 (2025).
Tian, D., Pan, Y., Zhao, X., Yin, Y. & Jiang, Z. Chiral Lewis acid-catalyzed intramolecular [2 + 2] photocycloaddition: enantioselective synthesis of azaarene-functionalized azabicyclo[2.1.1]hexanes and bicyclo[1.1.1]pentanes. J. Am. Chem. Soc. 147, 12410–12417 (2025).
Fu, Q. et al. Enantioselective [2π + 2σ] cycloadditions of bicycle[1.1.0]butanes with vinylazaarenes through asymmetric photoredox catalysis. J. Am. Chem. Soc. 146, 8372–8380 (2024).
Yin, Y. et al. Conjugate addition–enantioselective protonation of N-aryl glycines to α-branched 2-vinylazaarenes via cooperative photoredox and asymmetric catalysis. J. Am. Chem. Soc. 140, 6083–6087 (2018).
Cao, K. et al. Catalytic enantioselective addition of prochiral radcials to vinylpyridines. J. Am. Chem. Soc. 141, 5437–5443 (2019).
Chai, X. et al. Asymmetric hydroaminoalkylation of alkenylazaarenes via cooperative photoredox and chiral hydrogen-bonding catalysis. Angew. Chem. Int. Ed. 61, e202115110 (2022).
Sun, X. et al. Asymmetric photoredox catalytic formal de Mayo reaction enabled by sensitization-initiated electron transfer. Nat. Chem. 16, 1169–1176 (2024).
Tian, D. et al. Catalytic asymmetric [4 +2] dearomative photocycloadditions of anthracene and its derivatives with alkenylazaarenes. Nat. Commun. 15, 4563 (2024).
Bai, X. et al. Kinetic resolution for 1,4-spin-center shift-based reduction of azaarene-functionalized secondary and tertiary allylic alcohols. J. Am. Chem. Soc. 147, 38942–38950 (2025).
Le, D. P. Insecticidal 6-arylpyridinecarboxaldehyde thiosemicarbazones. US 4696938 (1987).
Smith, N. D. et al.Preparation of diaryltetrazoles as modulators of metabotropic glutamate receptor-5. WO 2003077918 (2003).
Oi, S., Maezaki, H. & Suzuki, N. Preparation of pyridines as inhibitors of dipeptidyl peptidase IV useful for the prophylaxis or treatment of diabetes. WO 2005042488 (2005).
Zhou, Q., Zhang, B., Chen, R., Jiang, H. & Yu, X. Aryl pyridine derivative and its preparation. CN 10335133 (2013).
Yoshimoto, Y., Arimori, S. & Hou, Z. Preparation of tetrazolinone compounds as pesticides. WO 2014175465 (2014).
Li, D., Wang, Y. & Zhang, Q. Application of diarylketone compound in preparation of anti-tumor drug. CN 106632000 (2017).
Shannon, J., Thom, S., Carrillo Arregui, J. & Alanine, T. Novel sulfone urea compounds as NLRP3 inhibitors and their preparation. WO 2020035465 (2020).
Gallagher, R., Qudah, T., Balle, T., Chebib, M. & Mcleod, M. D. Methyllycaconitine analogues selective for the alpha4beta2 over alpha7 nicotinic acetylcholine receptors. Bioorg. Med. Chem. 51, 116516 (2021).
Sakakibara, Y. & Murakami, K. Switchable divergent synthesis using photocatalysis. ACS Catal 12, 1857–1878 (2022).
Zeng, G. et al. Triplet energy transfer-based deracemization of axially chiral alkenes enabled by a dual catalyst system. J. Am. Chem. Soc. 147, 26079–26088 (2025).
Acknowledgements
This work was financially supported by the National Science Foundation of China (Nos. 22471064, 22401086, 22301061, 22171072, and 22201068), Central Government Guided Local Science and Technology Development Fund Projects (Z20231811081), and the Key Project of the Henan Provincial Natural Science Foundation (Nos. 252300421286 and 254000510005).
Author information
Authors and Affiliations
Contributions
Z.J. conceived and designed the study. D.T. and C.Q. performed the experiments. D.T., C.Q., S.C., B.Y., X.Z. and J.Z. analyzed and interpreted the data. S.C. carried out the DFT calculations. D.T., J.Z., and Z.J. prepared the Supplementary Information. Z.J. wrote the manuscript. All authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Shunxi Dong, liang yi and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Tian, D., Qu, C., Cao, S. et al. Enantioselective Synthesis of 2-Oxabicyclo[2.1.1]hexanes and Bicyclo[2.1.1]hexanes via Catalytic Asymmetric Intramolecular Photocycloadditions. Nat Commun (2026). https://doi.org/10.1038/s41467-026-71590-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-71590-x