Abstract
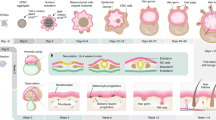
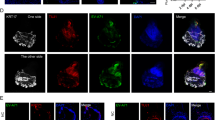
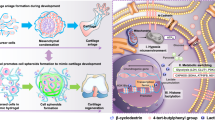
Self-organization in organoid morphogenesis involves the coordinated arrangement of interacting cells into higher-order structures, yet the underlying principles remain elusive. Here, we investigate how epidermal and dermal cells respond distinctively to elevated levels of hypoxia during skin organoid morphogenesis that largely resembles the skin development during embryogenesis. We unveil that autonomously generated hypoxic environment-induced metabolic adaptation drives the transition from coalesced spheroids to a planarized structure in skin organoids through the following three levels. Hif1a-mediated anaerobic metabolism positions epidermal cells in the liquid phase of the cultures under lower oxygen levels, facilitating tissue phase separation of the epidermal layer from the dermal layer. Hypoxia-driven activation of lysosomal hydrolases eliminates suprabasal keratin debris during planar epidermis formation. Fibroblasts adjacent to the basal epidermis have differential metabolic adaptation to hypoxia, which exhibit enhanced retinoid metabolism and become putative papillary dermis. Together, these hypoxia-induced metabolic adaptations contribute to reconstructing skin architecture similar to physiological development. Our findings highlight the ability of hypoxia-induced metabolic alteration to trigger varied cellular responses, leading to self-organizing coalesced spheroids-to-planar topological transformations and the restoration of tissue homeostasis.
Similar content being viewed by others
Data availability
ScRNA-seq data of skin organoids that support the findings of this study have been deposited in the Gene Expression Omnibus (GEO) under the accession code GSE215980. Bulk RNA-seq data of skin organoids have been deposited under the accession code GSE86955. The ATAC-seq and ChIP-seq raw data have been deposited in the NCBI Sequence Read Archive (SRA) under accession numbers PRJNA1392000 and PRJNA1393772. All statistically relevant source data are provided in the Source Data file.
Code availability
All single-cell RNA-seq data were analyzed following the standard workflow of the Seurat package (https://satijalab.org/seurat/). Custom codes used to generate the results reported in this paper are available from the corresponding authors upon reasonable request.
References
Xavier da Silveira Dos Santos, A. & Liberali, P. From single cells to tissue self-organization. FEBS J. 286, 1495–1513 (2019).
Ramos, R. et al. Parsing patterns: emerging roles of tissue self-organization in health and disease. Cell 187, 3165–3186 (2024).
Yang, S. et al. Morphogens enable interacting supracellular phases that generate organ architecture. Science 382, eadg5579 (2023).
Shahbazi, M. N., Siggia, E. D. & Zernicka-Goetz, M. Self-organization of stem cells into embryos: A window on early mammalian development. Science 364, 948–951 (2019).
Lei, M. et al. Self-organization process in newborn skin organoid formation inspires strategy to restore hair regeneration of adult cells. Proc. Natl. Acad. Sci. USA. 114, E7101–e7110 (2017).
Wang, M. et al. Mechanical force drives the initial mesenchymal-epithelial interaction during skin organoid development. Theranostics 13, 2930–2945 (2023).
Lei, M. et al. Epidermal-dermal coupled spheroids are important for tissue pattern regeneration in reconstituted skin explant cultures. NPJ Regen. Med. 8, 65 (2023).
Lei, M. et al. The mechano-chemical circuit drives skin organoid self-organization. Proc. Natl. Acad. Sci. USA. 120, e2221982120 (2023).
Toyoshima, K. E. et al. Fully functional hair follicle regeneration through the rearrangement of stem cells and their niches. Nat. Commun. 3, 784 (2012).
Hendrix, S., Handjiski, B., Peters, E. M. & Paus, R. A guide to assessing damage response pathways of the hair follicle: lessons from cyclophosphamide-induced alopecia in mice. J. Invest. Dermatol. 125, 42–51 (2005).
Lee, J. et al. Hair-bearing human skin generated entirely from pluripotent stem cells. Nature 582, 399–404 (2020).
Jonz, M. G., Buck, L. T., Perry, S. F., Schwerte, T. & Zaccone, G. Sensing and surviving hypoxia in vertebrates. Ann. N. Y Acad. Sci. 1365, 43–58 (2016).
Wan, C. et al. Activation of the hypoxia-inducible factor-1alpha pathway accelerates bone regeneration. Proc. Natl. Acad. Sci. USA. 105, 686–691 (2008).
Qian, X. et al. Sliced human cortical organoids for modeling distinct cortical layer formation. Cell Stem Cell 26, 766–781.e769 (2020).
Hosseini, M., Koehler, K. R. & Shafiee, A. Biofabrication of human skin with its appendages. Adv. Health. Mater. 11, e2201626 (2022).
Colson, A., Sonveaux, P., Debiève, F. & Sferruzzi-Perri, A. N. Adaptations of the human placenta to hypoxia: opportunities for interventions in fetal growth restriction. Hum. Reprod. Update 27, 531–569 (2021).
Lee, P., Chandel, N. S. & Simon, M. C. Cellular adaptation to hypoxia through hypoxia inducible factors and beyond. Nat. Rev. Mol. Cell Biol. 21, 268–283 (2020).
Ebner-Peking, P. et al. Self-assembly of differentiated progenitor cells facilitates spheroid human skin organoid formation and planar skin regeneration. Theranostics 11, 8430–8447 (2021).
Wickersham, M. et al. Metabolic stress drives keratinocyte defenses against staphylococcus aureus infection. Cell Rep. 18, 2742–2751 (2017).
Choudhry, H. & Harris, A. L. Advances in hypoxia-inducible factor biology. Cell Metab. 27, 281–298 (2018).
Cibrian, D., de la Fuente, H. & Sánchez-Madrid, F. Metabolic pathways that control skin homeostasis and inflammation. Trends Mol. Med. 26, 975–986 (2020).
Kierans, S. J. & Taylor, C. T. Regulation of glycolysis by the hypoxia-inducible factor (HIF): implications for cellular physiology. J. Physiol. 599, 23–37 (2021).
Zhao, X. et al. Metabolic regulation of dermal fibroblasts contributes to skin extracellular matrix homeostasis and fibrosis. Nat. Metab. 1, 147–157 (2019).
Monks, J. et al. Epithelial cells as phagocytes: apoptotic epithelial cells are engulfed by mammary alveolar epithelial cells and repress inflammatory mediator release. Cell Death Differ. 12, 107–114 (2005).
Zaarour, R. F. et al. Role of hypoxia-mediated autophagy in tumor cell death and survival. Cancers 13, 533 (2021).
Reinheckel, T. et al. The lysosomal cysteine protease cathepsin L regulates keratinocyte proliferation by control of growth factor recycling. J. Cell Sci. 118, 3387–3395 (2005).
Driskell, R. R. et al. Distinct fibroblast lineages determine dermal architecture in skin development and repair. Nature 504, 277–281 (2013).
Capolupo, L. et al. Sphingolipids control dermal fibroblast heterogeneity. Science 376, eabh1623 (2022).
Smedberg, J. L. et al. Ras/MAPK pathway confers basement membrane dependence upon endoderm differentiation of embryonic carcinoma cells. J. Biol. Chem. 277, 40911–40918 (2002).
Semenza, G. L. Hypoxia-inducible factors in physiology and medicine. Cell 148, 399–408 (2012).
Kaelin, W. G. Jr. & Ratcliffe, P. J. Oxygen sensing by metazoans: the central role of the HIF hydroxylase pathway. Mol. Cell 30, 393–402 (2008).
Evans, S. M., Schrlau, A. E., Chalian, A. A., Zhang, P. & Koch, C. J. Oxygen levels in normal and previously irradiated human skin as assessed by EF5 binding. J. Invest. Dermatol. 126, 2596–2606 (2006).
Nelson, J. K. et al. USP25 promotes pathological HIF-1-driven metabolic reprogramming and is a potential therapeutic target in pancreatic cancer. Nat. Commun. 13, 2070 (2022).
Kozlov, A. M., Lone, A., Betts, D. H. & Cumming, R. C. Lactate preconditioning promotes a HIF-1α-mediated metabolic shift from OXPHOS to glycolysis in normal human diploid fibroblasts. Sci. Rep. 10, 8388 (2020).
Midha, A. D. et al. Organ-specific fuel rewiring in acute and chronic hypoxia redistributes glucose and fatty acid metabolism. Cell Metab. 35, 504–516.e505 (2023).
Cheng, S. C. et al. mTOR- and HIF-1α-mediated aerobic glycolysis as metabolic basis for trained immunity. Science 345, 1250684 (2014).
Gao, Y. et al. LRG1 promotes keratinocyte migration and wound repair through regulation of HIF-1α stability. J. Invest. Dermatol. 140, 455–464.e458 (2020).
Zhang, J. et al. BNIP3 promotes the motility and migration of keratinocyte under hypoxia. Exp. Dermatol. 26, 416–422 (2017).
Kim, C. S. et al. Glutamine metabolism controls stem cell fate reversibility and long-term maintenance in the hair follicle. Cell Metab. 32, 629–642.e628 (2020).
Tierney, M. T. et al. Vitamin A resolves lineage plasticity to orchestrate stem cell lineage choices. Science 383, eadi7342 (2024).
Cho, S. et al. Long-term treatment of photoaged human skin with topical retinoic acid improves epidermal cell atypia and thickens the collagen band in papillary dermis. J. Am. Acad. Dermatol. 53, 769–774 (2005).
Aumailley, M. Laminins and interaction partners in the architecture of the basement membrane at the dermal-epidermal junction. Exp. Dermatol. 30, 17–24 (2021).
Lei, M., Yang, L. & Chuong, C. M. Getting to the core of the dermal papilla. J. Invest. Dermatol. 137, 2250–2253 (2017).
Acknowledgements
This work was supported by the National Key Research and Development Program of China (2023YFC2508200), the National Natural Science Foundation of China (82373509, 82574005, and 82404181), the Natural Science Foundation of Chongqing (cstc2021jcyj-cxttX0002, 2024NSCQ-MSX1985), and the Inheritance and Innovation Team of TCM Treatment of Immune Diseases, China. CMC was supported by a research contract (GR1035751) between USC and China Medical University in Taiwan and a US NIH grant R37 AR060306. We thank Professor Qiaoli Xie from Chongqing University for her assistance with the schematic illustrations in this paper.
Author information
Authors and Affiliations
Contributions
M.L. and C.-M.C. supervised this work and approved the final version of the manuscript. M.L., C.-M.C. and J.J. initiated the study, designed the experiments, analyzed data, and wrote the manuscript. W.L., M.W., D.W., W.W., M.Z., S.Z., X.L., X.Y, X.X. and W.A. performed experiments and generated figures. H.Z., G. L., K.C. and R. Z. provided clinical insight and edited the manuscript. L.Y. provided suggestions.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Pawel Swietach and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Jiang, J., Liu, W., Wang, M. et al. Metabolic adaptation drives self-organization during skin organoid morphogenesis. Nat Commun (2026). https://doi.org/10.1038/s41467-026-71709-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-71709-0