Abstract
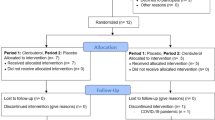
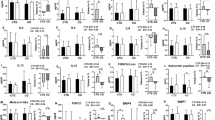
Impaired post-prandial skeletal muscle glucose uptake plays a pivotal role in the development of type 2 diabetes mellitus (T2DM), yet pharmacological strategies to enhance muscle glucose uptake are limited. Previous (pre)clinical research revealed that β2-adrenergic receptor (β2-AR) stimulation enhances glucose uptake, but its clinical relevance in individuals susceptible to developing T2DM is unknown. Here we determined in a double-blinded, placebo-controlled, crossover study (ClinicalTrials.gov-identifier: NCT04921306), the effects of a 4-week treatment with the β2-adrenergic agonist clenbuterol (40 μg/day) on insulin-stimulated glucose uptake in the quadriceps muscle (primary outcome) and brown adipose tissue (BAT) (secondary outcome) using 18F-FDG PET-MRI during a hyperinsulinemic-euglycemic clamp in individuals with overweight or obesity (age: 40-70 years, BMI: 25-35 kg/m2). A total of 14 participants were recruited and randomized. Insulin-stimulated glucose uptake tended to improve in vastus lateralis (15%, p = 0.072) and increased significantly in the hamstring (13%, p = 0.039) muscle, while BAT uptake (p = 0.720) remained unaffected. These findings suggest potential therapeutic benefits of β2-AR stimulation for improving muscle-specific glucose uptake in individuals with or at risk for developing diabetes.
Similar content being viewed by others
Data availability
The study protocol and statistical analysis plan are available in the Supplementary Material. Source data are provided in this paper. Upon scientific request, de-identified and processed participant data can be requested from the corresponding author (j.hoeks@maastrichtuniversity.nl), with no end date, following the completion of a signed data transfer agreement. De-identified data will be shared due to participant privacy.
References
Hossain, M. J., Al-Mamun, M. & Islam, M. R. Diabetes mellitus, the fastest growing global public health concern: Early detection should be focused. Health Sci. Rep. 7, e2004 (2024).
DeFronzo, R. A. & Tripathy, D. Skeletal muscle insulin resistance is the primary defect in type 2 diabetes. Diabetes Care 32, S157–S163 (2009).
Ferrannini, E. et al. The disposal of an oral glucose load in patients with non-insulin-dependent diabetes. Metabolism 37, 79–85 (1988).
Thiebaud, D. et al. The effect of graded doses of insulin on total glucose uptake, glucose oxidation, and glucose storage in man. Diabetes 31, 957–963 (1982).
Merz, K. E. & Thurmond, D. C. Role of skeletal muscle in insulin resistance and glucose uptake. Compr. Physiol. 10, 785–809 (2020).
Meister, J. et al. Clenbuterol exerts antidiabetic activity through metabolic reprogramming of skeletal muscle cells. Nat. Commun. 13, 22 (2022).
Sato, M. et al. Improving type 2 diabetes through a distinct adrenergic signaling pathway involving mTORC2 that mediates glucose uptake in skeletal muscle. Diabetes 63, 4115–4129 (2014).
Nevzorova, J., Bengtsson, T., Evans, B. A. & Summers, R. J. Characterization of the beta-adrenoceptor subtype involved in mediation of glucose transport in L6 cells. Br. J. Pharmacol. 137, 9–18 (2002).
Nevzorova, J., Evans, B. A., Bengtsson, T. & Summers, R. J. Multiple signalling pathways involved in beta2-adrenoceptor-mediated glucose uptake in rat skeletal muscle cells. Br. J. Pharmacol. 147, 446–454 (2006).
Kalinovich, A. et al. Treatment with a beta-2-adrenoceptor agonist stimulates glucose uptake in skeletal muscle and improves glucose homeostasis, insulin resistance and hepatic steatosis in mice with diet-induced obesity. Diabetologia 63, 1603–1615 (2020).
Castle, A., Yaspelkis, B. B. 3rd, Kuo, C. H. & Ivy, J. L. Attenuation of insulin resistance by chronic beta2-adrenergic agonist treatment possible muscle specific contributions. Life Sci. 69, 599–611 (2001).
Pan, S. J. et al. Effects of clenbuterol on insulin resistance in conscious obese Zucker rats. Am. J. Physiol. Endocrinol. Metab. 280, E554–E561 (2001).
Jacob, S., Fogt, D. L., Dietze, G. J. & Henriksen, E. J. The beta2-adrenergic modulator celiprolol reduces insulin resistance in obese Zucker rats. Life Sci. 64, 2071–2079 (1999).
Smith, S. A., Levy, A. L., Sennitt, M. V., Simson, D. L. & Cawthorne, M. A. Effects of BRL 26830, a novel beta-adrenoceptor agonist, on glucose tolerance, insulin sensitivity and glucose turnover in Zucker (fa/fa) rats. Biochem. Pharm. 34, 2425–2429 (1985).
van Beek, S. M. M. et al. Effect of beta2-agonist treatment on insulin-stimulated peripheral glucose disposal in healthy men in a randomised placebo-controlled trial. Nat. Commun. 14, 173 (2023).
Jessen, S. et al. Muscle hypertrophic effect of inhaled beta(2) -agonist is associated with augmented insulin-stimulated whole-body glucose disposal in young men. J. Physiol. 600, 2345–2357 (2022).
Scheidegger, K., Robbins, D. C. & Danforth, E. Jr Effects of chronic beta receptor stimulation on glucose metabolism. Diabetes 33, 1144–1149 (1984).
Chondronikola, M. et al. Brown adipose tissue improves whole-body glucose homeostasis and insulin sensitivity in humans. Diabetes 63, 4089–4099 (2014).
Blondin, D. P. et al. Human brown adipocyte thermogenesis is driven by beta2-AR stimulation. Cell Metab. 32, 287–300 e287 (2020).
Straat, M. E. et al. Stimulation of the beta-2-adrenergic receptor with salbutamol activates human brown adipose tissue. Cell Rep. Med. 4, 100942 (2023).
Cipolletta, E., Del Giudice, C., Santulli, G., Trimarco, B. & Iaccarino, G. Opposite effects of beta(2)-adrenoceptor gene deletion on insulin signaling in liver and skeletal muscle. Nutr. Metab. Cardiovasc Dis. 27, 615–623 (2017).
Hagstrom-Toft, E., Enoksson, S., Moberg, E., Bolinder, J. & Arner, P. beta-Adrenergic regulation of lipolysis and blood flow in human skeletal muscle in vivo. Am. J. Physiol. 275, E909–E916 (1998).
Vincent, M. A. et al. Microvascular recruitment is an early insulin effect that regulates skeletal muscle glucose uptake in vivo. Diabetes 53, 1418–1423 (2004).
Ugwoke, C. K., Cvetko, E. & Umek, N. Skeletal muscle microvascular dysfunction in obesity-related insulin resistance: pathophysiological mechanisms and therapeutic perspectives. Int J. Mol. Sci. 23, 847 (2022).
Jessen, S. et al. Beta(2) -adrenergic agonist clenbuterol increases energy expenditure and fat oxidation, and induces mTOR phosphorylation in skeletal muscle of young healthy men. Drug Test. Anal. 12, 610–618 (2020).
Onslev, J. et al. Beta(2)-adrenergic stimulation increases energy expenditure at rest, but not during submaximal exercise in active overweight men. Eur. J. Appl Physiol. 117, 1907–1915 (2017).
Hoeks, J. et al. Effect of beta1- and beta2-adrenergic stimulation on energy expenditure, substrate oxidation, and UCP3 expression in humans. Am. J. Physiol. Endocrinol. Metab. 285, E775–E782 (2003).
Lee, P., Day, R. O., Greenfield, J. R. & Ho, K. K. Formoterol, a highly beta2-selective agonist, increases energy expenditure and fat utilisation in men. Int. J. Obes. 37, 593–597 (2013).
Blaak, E. E., van Baak, M. A. & Saris, W. H. Beta-adrenergically stimulated fat oxidation is diminished in middle-aged compared to young subjects. J. Clin. Endocrinol. Metab. 84, 3764–3769 (1999).
Kline, W. O., Panaro, F. J., Yang, H. & Bodine, S. C. Rapamycin inhibits the growth and muscle-sparing effects of clenbuterol. J. Appl. Physiol. 102, 740–747 (2007).
Rizza, R. A., Cryer, P. E., Haymond, M. W. & Gerich, J. E. Adrenergic mechanisms of catecholamine action on glucose homeostasis in man. Metabolism 29, 1155–1163 (1980).
DeFronzo, R. A. et al. The effect of insulin on the disposal of intravenous glucose. Results from indirect calorimetry and hepatic and femoral venous catheterization. Diabetes 30, 1000–1007 (1981).
Talamonti, E. et al. The novel adrenergic agonist ATR-127 targets skeletal muscle and brown adipose tissue to tackle diabesity and steatohepatitis. Mol. Metab. 85, 101931 (2024).
Orava, J. et al. Different metabolic responses of human brown adipose tissue to activation by cold and insulin. Cell Metab. 14, 272–279 (2011).
Heaton, J. M. The distribution of brown adipose tissue in the human. J. Anat. 112, 35–39 (1972).
Yoneshiro, T. et al. Impact of UCP1 and beta3AR gene polymorphisms on age-related changes in brown adipose tissue and adiposity in humans. Int. J. Obes. 37, 993–998 (2013).
Motso, A. et al. GRK-biased adrenergic agonists for the treatment of type 2 diabetes and obesity. Cell 188, 5429–5431 (2025).
Yamamoto, I., Iwata, K. & Nakashima, M. Pharmacokinetics of plasma and urine clenbuterol in man, rat, and rabbit. J. Pharmacobiodyn 8, 385–391 (1985).
Basu, R. et al. Effects of age and sex on postprandial glucose metabolism: differences in glucose turnover, insulin secretion, insulin action, and hepatic insulin extraction. Diabetes 55, 2001–2014 (2006).
Kneale, B. J., Chowienczyk, P. J., Brett, S. E., Coltart, D. J. & Ritter, J. M. Gender differences in sensitivity to adrenergic agonists of forearm resistance vasculature. J. Am. Coll. Cardiol. 36, 1233–1238 (2000).
Zhu, B., Liu, K., Yang, C., Qiao, Y. & Li, Z. Gender-related differences in beta-adrenergic receptor-mediated cardiac remodeling. Can. J. Physiol. Pharm. 94, 1349–1355 (2016).
Johansson, E. et al. Whole-body imaging of tissue-specific insulin sensitivity and body composition by using an integrated PET/MR system: a feasibility study. Radiology 286, 271–278 (2018).
Vulic, A., Pleadin, J., Durgo, K., Scortichini, G. & Stojkovic, R. Comparison of clenbuterol and salbutamol accumulation in the liver of two different mouse strains. J. Anal. Toxicol. 38, 265–271 (2014).
Baker, J. G. The selectivity of beta-adrenoceptor agonists at human beta1-, beta2- and beta3-adrenoceptors. Br. J. Pharmacol. 160, 1048–1061 (2010).
DeFronzo, R. A., Tobin, J. D. & Andres, R. Glucose clamp technique: a method for quantifying insulin secretion and resistance. Am. J. Physiol. 237, E214–E223 (1979).
Pan, L., Sachpekidis, C., Hassel, J., Christopoulos, P. & Dimitrakopoulou-Strauss, A. Impact of different parametric Patlak imaging approaches and comparison with a 2-tissue compartment pharmacokinetic model with a long axial field-of-view (LAFOV) PET/CT in oncological patients. Eur. J. Nucl. Med. Mol. Imaging 52, 623–637 (2025).
van Sluis, J. et al. Ultrashort Oncologic Whole-Body [(18)F]FDG Patlak Imaging Using LAFOV PET. J. Nucl. Med. 65, 1652–1657 (2024).
Pande, R. L. et al. Impaired skeletal muscle glucose uptake by [18F]fluorodeoxyglucose-positron emission tomography in patients with peripheral artery disease and intermittent claudication. Arterioscler. Thromb. Vasc. Biol. 31, 190–196 (2011).
Cypess, A. M. et al. Identification and importance of brown adipose tissue in adult humans. N. Engl. J. Med. 360, 1509–1517 (2009).
Hany, T. F. et al. Brown adipose tissue: a factor to consider in symmetrical tracer uptake in the neck and upper chest region. Eur. J. Nucl. Med. Mol. Imaging 29, 1393–1398 (2002).
Dempster, P. & Aitkens, S. A new air displacement method for the determination of human body composition. Med. Sci. Sports Exerc. 27, 1692–1697 (1995).
Plasqui, G., Soenen, S., Westerterp-Plantenga, M. S. & Westerterp, K. R. Measurement of longitudinal changes in body composition during weight loss and maintenance in overweight and obese subjects using air-displacement plethysmography in comparison with the deuterium dilution technique. Int J. Obes. 35, 1124–1130 (2011).
Brouwer, E. On simple formulae for calculating the heat expenditure and the quantities of carbohydrate and fat oxidized in metabolism of men and animals, from gaseous exchange (Oxygen intake and carbonic acid output) and urine-N. Acta Physiol. Pharm. Neerl. 6, 795–802 (1957).
Weir, J. B. New methods for calculating metabolic rate with special reference to protein metabolism. J. Physiol. 109, 1–9 (1949).
Dower, J. I. et al. Effects of the pure flavonoids epicatechin and quercetin on vascular function and cardiometabolic health: a randomized, double-blind, placebo-controlled, crossover trial. Am. J. Clin. Nutr. 101, 914–921 (2015).
Acknowledgements
Research relating to this manuscript was funded by the Netherlands Enterprise Agency (RVO) under the Eureka EuroStars funding program (ESTAR20215) (awarded to J.H.). The authors thank Marc Souren, Sanne Ahles, and Soraya de Kam for their assistance during the study. The authors thank all participants for their participation.
Author information
Authors and Affiliations
Contributions
S.M.M., T.B., P.S., T.W., and J.H. conceived and designed the research. P.M.G., T.W., F.V., K.B., and K.M.R. performed experiments. P.M.G., T.W., K.B., E.M., G.S., and K.M.R. analyzed data. P.M.G., V.B.S., P.J., E.P., P.S., T.W., and J.H. interpreted results. S.S. and R.W. created new software script. P.M.G. wrote the original paper draft. P.M.G., P.S., T.W., and J.H. edited and revised the paper. All authors approved the final paper version.
Corresponding author
Ethics declarations
Competing interests
Tore Bengtsson owns stock in Atrogi AB. The remaining authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Matthias Betz and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Van Lier, P.M.G., van de Weijer, T., Vanweert, F. et al. Prolonged β2-agonist treatment enhances muscle-specific glucose uptake in individuals with overweight and obesity: a randomized placebo-controlled trial. Nat Commun (2026). https://doi.org/10.1038/s41467-026-71897-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-71897-9