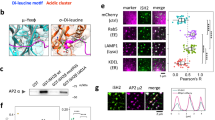
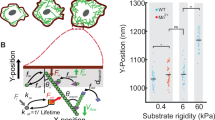
Abstract
AAK1 and BMP2K are serine/threonine kinases traditionally known for phosphorylating AP2 during clathrin-mediated endocytosis (CME), but their broader roles remained incompletely defined. Here, using motif-guided in silico, biochemical, and phosphoproteomic screens, we identify PDLIM5 and Talin1 as direct AAK1/BMP2K substrates. Despite high kinase-domain similarity, only AAK1 promotes cell migration and potentiates focal adhesion (FA) turnover. Live-cell imaging shows that AAK1 recruitment to FAs peaks as disassembly begins. The conserved AAK1 C-terminal PDZ-binding motif mediates direct, low-affinity binding to PDLIM5, providing a plausible mechanism for localized substrate access. Dynamic analyses of phospho-mimetic and phospho-null mutants support a model in which AAK1-dependent phosphorylation promotes timely release of PDLIM5 and Talin1 during FA disassembly. These findings reveal a kinase-driven contribution to FA turnover distinct from protease- and phosphatase-based mechanisms and suggest that functional divergence between AAK1 and BMP2K may provide a strategy to modulate cell migration with reduced impact on CME.
Similar content being viewed by others
Acknowledgements
We thank Laura Adamkiewicz and Jana Sebestikova for technical assistance and Sally R. Gray for cloning and M.S. Robinson for insightful discussions on NAK evolution. We thank Professor Paul Luzio for scientific discussion and feedback on the manuscript. We thank Eva Pyrihova and Tereza Nepokojova for recombinant protein production. We thank the CIMR Flow Cytometry Core Facility team (Dr Reiner Schulte and Dr Gabriela Grondys-Kotarba), the CIMR light microscopy core facility team (Dr Matthew Gratian and Mark Bowen) and CELLIM team (Dr Jakub Pospisil and Dr Milan Esner) for technical support. We thank Petr Gintar from the Proteomics Core Facility at CEITEC for his valuable help with sample preparation and advice on proteomics data analysis. Funding: this study was funded by GAČR 21-16786 M and Wellcome Trust Henry Dale Fellowship 220597/Z/20/Z. The work of V.B. and E.B. was funded by the project New Technologies for Translational Research in Pharmaceutical Sciences /NETPHARM, project ID CZ.02.01.01/00/22_008/0004607, co-funded by the European Union. A.H. was supported by the Charles University, project GA UK No. 104223. M.A. and Recombinant protein production at the Biocev CF Protein Production Facility was supported CIISB, Instruct-CZ Center of Instruct-ERIC EU consortium, funded by MEYS CR infrastructure project LM2023042 and OP JAK project “Innovation of Czech Infrastructure for Integrative Structural Biology” (no. CZ.02.01.01/00/23_015/0008175). J.H. and BIC Core facility was supported by CF Biomolecular Interactions and Crystallography of CIISB, Instruct-CZ Center, supported by MEYS CR (LM2023042) and European Regional Development Fund-Project „Innovation of Czech Infrastructure for Integrative Structural Biology“ (No. CZ.02.01.01/00/23_015/0008175). The work of Z.Z, D.P, P.M, P.R and K.T was supported by CIISB, Instruct-CZ Center of Instruct-ERIC EU consortium, funded by MEYS CR infrastructure project LM2023042 and European Regional Development Fund-Project „Innovation of Czech Infrastructure for Integrative Structural Biology“ (No. CZ.02.01.01/00/23_015/0008175), is gratefully acknowledged for the financial support of the measurements at the CEITEC Proteomics Core Facility. Computational resources were provided by the e-INFRA CZ project (ID:90254), supported by MEYS CR. J.F and V.B was supported by Internal Pilot Research Projects of the St. Anne’s University Hospital in Brno, European Union - Next Generation EU project nr. LX22NPO5107, Ministry of Health of the Czech Republic-DRO (Institute of Hematology and Blood Transfusion- 00023736). A.G.W. was supported by the Francis Crick Institute which receives its core funding from Cancer Research UK (CC2060), the UK Medical Research Council (CC2060), and the Wellcome Trust (CC2060). A.G.W. has been supported by Wellcome Trust grants 097040/Z/11/Z and 303026/Z/23/Z. D.K was supported by funds of the Faculty of Medicine MU (MUNI/A/1738/2024). D.J.O, B.T.K and J.K. were funded by Wellcome Trust Principal Research Fellowship 207455/Z/17/Z.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Krocianova, D., Dagg, A.D., Clayton, R.A. et al. AAK1-mediated phosphorylation of PDLIM5 and Talin1 promotes focal adhesion disassembly to accelerate cell migration. Nat Commun (2026). https://doi.org/10.1038/s41467-026-72501-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-72501-w