Abstract
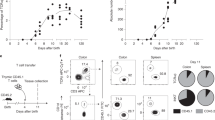
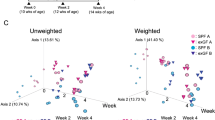
The early-life intestinal microenvironment plays a pivotal role in shaping immune cell development. Here, we identify a colonic Wnt4-expressing stromal cell, enriched during early-life, that promotes iNKT cell proliferation via BMP-MAPK signaling. These stromal cells are spatially associated with iNKT cells and macrophages and exhibit high Bmp2 expression during the neonatal period. Depletion of BMP2 in Wnt4+ stromal cells during, but not after, this time window leads to long-lasting reductions in iNKT cells. These stromal cells are shaped by microbial signals, as germ-free and early-life antibiotic-treated mice exhibit increased Wnt4+ stromal cell abundance and elevated Bmp2 expression, with excessive iNKT cell accumulation that lasts into adulthood. These persistent changes in iNKT cells due to early-life perturbations are associated with altered susceptibility to later-life mucosal disorders. Importantly, similar stromal cells are present in fetal and neonatal human colon, and human rBMP2 promotes iNKT cell growth. Together, our findings reveal a neonatal colonic stromal niche, orchestrated by microbial cues, that regulates colonic immune homeostasis in later-life.
Similar content being viewed by others
Acknowledgements
We thank the Blumberg laboratory members for their assistance with manuscript preparation, and especially Yuly Lopera for assistance with animal husbandry. We thank Dr. Dale I. Godfrey for valuable input and discussion on the manuscript. We thank Brigham and Women’s Hospital Single Cell Genomics Core for performing scRNA-seq, Harvard Medical School Flow Cytometry Core Facility for helping with the FACS experiments, Dr. Susan Hagen and the BIDMC Pathology Core Facility for performing the IF staining, Dr. Lynn Bry and the Massachusetts host-microbiome center for providing Germ-Free mice. RSB discloses support for the research and publication of this work from the NIH grants (DK044319, DK051362, DK088199, and 5P01AI073748) and the Harvard Digestive Diseases Center (P30DK034854). X.L. discloses support for the research and publication of this work from the Crohn’s and Colitis Foundation of America Research Fellow Award (1282671). A.A., C.H.L. and A.S. disclose support for the research of this work from the Chan Zuckerberg Initiative Foundation (DAF2021-237606). T.G. discloses support for the research of this work from the Fondation pour la Recherche Médicale (AJE202210016199) and the Association pour la Recherche contre le Cancer (ARCPJA2024080008675). T.Z., T.H., J.N.G., N.S.B., V.R., J.E.G. and M.K.W. declare no relevant funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Lin, X., Lee, C.HJ., Zhang, T. et al. Early-life Wnt4 expressing colon stromal cells orchestrate lifelong mucosal homeostasis via BMP-driven iNKT cell imprinting. Nat Commun (2026). https://doi.org/10.1038/s41467-026-72734-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-72734-9