Abstract
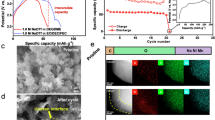
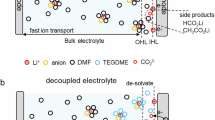
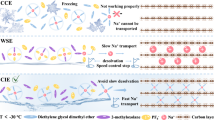
Sodium-ion batteries hold significant promise for sustainable energy storage by addressing resource scarcity and safety concerns. Elevating the upper-cut-off voltage is crucial for sodium-ion batteries to maximize electrode capacity and narrow their energy-density gap with commercial lithium-ion batteries. However, conventional electrolytes exhibit electrochemical instability on the highly desodiated positive electrode surface, undergoing strongly electrophilic attack and continuous decomposition that results in oligomer-rich interphases and rapid capacity fading. Here, we design a solvent-locked carbonate electrolyte creating electrochemically stable solvent-reinforced solvation structure and anion-rich interfacial shield-derived boride-/fluoride-rich interphase on positive electrode, thereby suppressing current leakage and parasitic reactions. The tailored electrolyte exhibits good compatibility with commercialized oxides and polyanionic positive electrodes. As-assembled Na||Na2.26Fe1.87(SO4)3 cells deliver extended lifespans operating to 4.5 V, retaining 88.2% capacity after 16,500 cycles at 1000 mA g–1 (coin cell) and 93.9% after 500 cycles at 100 mA g–1 (pouch cell). This work will inspire durable electrolyte and interphase design for high-energy batteries and beyond.
Similar content being viewed by others
Acknowledgements
The authors gratefully acknowledge the Center of Advanced Analysis & Gene Sequencing at Zhengzhou University for their support with physical characterizations, and the National Supercomputing Center in Zhengzhou for providing computational resources for the theoretical simulations and calculations. W.C. discloses support for the research of this work from National Natural Science Foundation of China [grant number U24A20566], the Joint Fund of Scientific and Technological Research and Development Program of Henan Province [grant number 222301420009] and Science Technology and Innovation Team in University of Henan Province [grant number 24IRTSTHN002]. E.W. discloses support for the research of this work from the Open Project Program of Yaoshan Laboratory [grant number 2024002]. J.Z. discloses support for the research of this work from the China Postdoctoral Science Foundation [grant number 2025M771097].
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhang, J., Tang, G., Ma, S. et al. Dual-domain solvent-locked electrolyte enabled durable 4.5 V-class sodium batteries. Nat Commun (2026). https://doi.org/10.1038/s41467-026-72849-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-72849-z