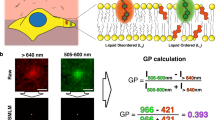
Abstract
Biological membranes play a crucial role in actively hosting, modulating and coordinating a wide range of molecular events essential for cellular function. Membranes are organized into diverse domains giving rise to dynamic molecular patchworks. However, the very definition of membrane domains has been the subject of continuous debate. For example, in the plant field, membrane domains are often referred to as nanodomains, nanoclusters, microdomains, lipid rafts, membrane rafts, signalling platforms, foci or liquid-ordered membranes without any clear rationale. In the context of plant–microbe interactions, microdomains have sometimes been used to refer to the large area at the plant–microbe interface. Some of these terms have partially overlapping meanings at best, but they are often used interchangeably in the literature. This situation generates much confusion and limits conceptual progress. There is thus an urgent need for us as a scientific community to resolve these semantic and conceptual controversies by defining an unambiguous nomenclature of membrane domains. In this Review, experts in the field get together to provide explicit definitions of plasma membrane domains in plant systems and experimental guidelines for their study. We propose that plasma membrane domains should not be considered on the basis of their size alone but rather according to the biological system being considered, such as the local membrane environment or the entire cell.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Singer, S. J. & Nicolson, G. L. The fluid mosaic model of the structure of cell membranes. Science 175, 720–731 (1972).
Suzuki, K. G. N. & Kusumi, A. Refinement of Singer–Nicolson fluid-mosaic model by microscopy imaging: lipid rafts and actin-induced membrane compartmentalization. Biochim. Biophys. Acta Biomembr. 1865, 184093 (2023).
Nicolson, G. L. & de Mattos, G. F. The fluid–mosaic model of cell membranes: a brief introduction, historical features, some general principles, and its adaptation to current information. Biochim. Biophys. Acta Biomembr. https://doi.org/10.1016/j.bbamem.2023.184135 (2023).
Nicolson, G. L. The fluid-mosaic model of membrane structure: still relevant to understanding the structure, function and dynamics of biological membranes after more than 40 years. Biochim. Biophys. Acta Biomembr. 1838, 1451–1466 (2014).
Nicolson, G. L. Update of the 1972 Singer–Nicolson fluid-mosaic model of membrane structure. Discoveries (Craiova) 1, e3 (2013).
Gorelova, V., Sprakel, J. & Weijers, D. Plant cell polarity as the nexus of tissue mechanics and morphogenesis. Nat. Plants https://doi.org/10.1038/s41477-021-01021-w (2021).
Muroyama, A. & Bergmann, D. Plant cell polarity: creating diversity from inside the box. Annu. Rev. Cell Dev. Biol. 35, 309–336 (2019).
Hutten, S. J. et al. Visualization of BRI1 and SERK3/BAK1 nanoclusters in Arabidopsis roots. PLoS ONE 12, e0169905 (2017).
Bucherl, C. A. et al. Visualization of BRI1 and BAK1(SERK3) membrane receptor heterooligomers during brassinosteroid signaling. Plant Physiol. 162, 1911–1925 (2013).
Platre, M. P. et al. Developmental control of plant Rho GTPase nano-organization by the lipid phosphatidylserine. Science 364, 57–62 (2019).
Pan, X. et al. Auxin-induced signaling protein nanoclustering contributes to cell polarity formation. Nat. Commun. 11, 3914 (2020).
Miao, Y., Guo, X., Zhu, K. & Zhao, W. Biomolecular condensates tunes immune signaling at the host–pathogen interface. Curr. Opin. Plant Biol. 74, 102374 (2023).
Jarsch, I. K. et al. Plasma membranes are subcompartmentalized into a plethora of coexisting and diverse microdomains in Arabidopsis and Nicotiana benthamiana. Plant Cell 26, 1698–1711 (2014).
Jaillais, Y. & Ott, T. The nanoscale organization of the plasma membrane and its importance in signaling: a proteolipid perspective. Plant Physiol. 182, 1682–1696 (2020).
Malinsky, J., Opekarová, M., Grossmann, G. & Tanner, W. Membrane microdomains, rafts, and detergent-resistant membranes in plants and fungi. Annu. Rev. Plant Biol. 64, 501–529 (2013).
Gouguet, P. et al. Connecting the dots: from nanodomains to physiological functions of REMORINs. Plant Physiol. 85, 632–649 (2020).
Tapken, W. & Murphy, A. S. Membrane nanodomains in plants: capturing form, function, and movement. J. Exp. Bot. 66, 1573–1586 (2015).
Daněk, M. et al. Cell wall contributes to the stability of plasma membrane nanodomain organization of Arabidopsis thaliana FLOTILLIN2 and HYPERSENSITIVE INDUCED REACTION1 proteins. Plant J. Cell Mol. Biol. 101, 619–636 (2019).
Bücherl, C. A. et al. Plant immune and growth receptors share common signalling components but localise to distinct plasma membrane nanodomains. eLife 6, e25114 (2017).
Malínská, K., Malínský, J., Opekarová, M. & Tanner, W. Visualization of protein compartmentation within the plasma membrane of living yeast cells. Mol. Biol. Cell 14, 4427–4436 (2003).
Wang, P. et al. The plant cytoskeleton, NET3C, and VAP27 mediate the link between the plasma membrane and endoplasmic reticulum. Curr. Biol. 24, 1397–1405 (2014).
Bayle, V. et al. Single-particle tracking photoactivated localization microscopy of membrane proteins in living plant tissues. Nat. Protoc. 6, 1600–1628 (2021).
Fratini, M. et al. Plasma membrane nano-organization specifies phosphoinositide effects on Rho-GTPases and actin dynamics in tobacco pollen tubes. Plant Cell 33, 642–670 (2021).
Lv, X. et al. Membrane microdomains and the cytoskeleton constrain AtHIR1 dynamics and facilitate the formation of an AtHIR1-associated immune complex. Plant J. 90, 3–16 (2017).
Béziat, C. & Jaillais, Y. Should I stay or should I go: the functional importance and regulation of lipid diffusion in biological membranes. J. Exp. Bot. 74, 2479–2488 (2023).
Raghupathy, R. et al. Transbilayer lipid interactions mediate nanoclustering of lipid-anchored proteins. Cell 161, 581–594 (2015).
Róg, T. et al. Interdigitation of long-chain sphingomyelin induces coupling of membrane leaflets in a cholesterol dependent manner. Biochim. Biophys. Acta Biomembr. 1858, 281–288 (2016).
Grosjean, K., Mongrand, S., Beney, L., Simon-Plas, F. & Gerbeau-Pissot, P. Differential effect of plant lipids on membrane organization. J. Biol. Chem. 290, 5810–5825 (2015).
Noack, L. C. & Jaillais, Y. Functions of anionic lipids in plants. Annu. Rev. Plant Biol. 71, 71–102 (2020).
Smokvarska, M. et al. The receptor kinase FERONIA regulates phosphatidylserine localization at the cell surface to modulate ROP signaling. Sci. Adv. 9, eadd4791 (2023).
Simons, K. & Ikonen, E. Functional rafts in cell membranes. Nature 387, 569–572 (1997).
Smokvarska, M. et al. A plasma membrane nanodomain ensures signal specificity during osmotic signaling in plants. Curr. Biol. 30, 4654–4664 (2020).
Ma, Z. et al. Membrane nanodomains modulate formin condensation for actin remodeling in Arabidopsis innate immune responses. Plant Cell 34, 374–394 (2022).
Liang, P. et al. Symbiotic root infections in Medicago truncatula require remorin-mediated receptor stabilization in membrane nanodomains. Proc. Natl Acad. Sci. USA 40, 201721868 (2018).
Heilmann, M. & Heilmann, I. Regulators regulated: different layers of control for plasma membrane phosphoinositides in plants. Curr. Opin. Plant Biol. 67, 102218 (2022).
Fuchs, V. A. F. et al. Nanodomain-mediated lateral sorting drives polarization of the small GTPase ROP2 in the plasma membrane of root hair cells. Preprint at bioRxiv https://doi.org/10.1101/2021.09.10.459822 (2021).
Gronnier, J. et al. Regulation of immune receptor kinase plasma membrane nanoscale organization by a plant peptide hormone and its receptors. eLife 11, e74162 (2022).
Hurst, C. H. et al. S-acylation stabilizes ligand-induced receptor kinase complex formation during plant pattern-triggered immune signaling. Curr. Biol. 33, 1588–1596 (2023).
McKenna, J. F. et al. The cell wall regulates dynamics and size of plasma-membrane nanodomains in Arabidopsis. Proc. Natl Acad. Sci. USA 4, 201819077 (2019).
Tran, T. M. et al. The bacterial quorum sensing signal DSF hijacks Arabidopsis thaliana sterol biosynthesis to suppress plant innate immunity. Life Sci. Alliance 3, e202000720 (2020).
Gronnier, J. et al. Structural basis for plant plasma membrane protein dynamics and organization into functional nanodomains. eLife 6, e26404 (2017).
Perraki, A. et al. Plasma membrane localization of Solanum tuberosum Remorin from Group 1, Homolog 3 is mediated by conformational changes in a novel C-terminal anchor and required for the restriction of potato virus X movement. Plant Physiol. 160, 624–637 (2012).
Sun, H. et al. Xanthomonas effector XopR hijacks host actin cytoskeleton via complex coacervation. Nat. Commun. 12, 4064 (2021).
Ott, T. Membrane nanodomains and microdomains in plant–microbe interactions. Curr. Opin. Plant Biol. 40, 82–88 (2017).
Martin, S. W., Glover, B. J. & Davies, J. M. Lipid microdomains—plant membranes get organized. Trends Plant Sci. 10, 263–265 (2005).
Zappel, N. F. & Panstruga, R. Heterogeneity and lateral compartmentalization of plant plasma membranes. Curr. Opin. Plant Biol. 11, 632–640 (2008).
Yu, M., Cui, Y., Zhang, X., Li, R. & Lin, J. Organization and dynamics of functional plant membrane microdomains. Cell. Mol. Life Sci. 77, 275–287 (2020).
Nakamura, M. & Grebe, M. Outer, inner and planar polarity in the Arabidopsis root. Curr. Opin. Plant Biol. 41, 46–53 (2017).
Łangowski et al. Cellular mechanisms for cargo delivery and polarity maintenance at different polar domains in plant cells. Cell Discov. 2, 16018 (2016).
Langowski, L., Růzicka, K., Naramoto, S., Kleine-Vehn, J. & Friml, J. Trafficking to the outer polar domain defines the root–soil interface. Curr. Biol. 20, 904–908 (2010).
Kleine-Vehn, J. et al. Recycling, clustering, and endocytosis jointly maintain PIN auxin carrier polarity at the plasma membrane. Mol. Syst. Biol. 7, 540 (2011).
Barberon, M. et al. Polarization of IRON-REGULATED TRANSPORTER 1 (IRT1) to the plant–soil interface plays crucial role in metal homeostasis. Proc. Natl Acad. Sci. USA 111, 8293–8298 (2014).
Rodriguez-Furlan, C., Emami, A. & Van Norman, J. M. Distinct ADP–ribosylation factor–GTP exchange factors govern the opposite polarity of 2 receptor kinases. Plant Physiol. 194, 673–683 (2024).
Denninger, P. et al. Distinct RopGEFs successively drive polarization and outgrowth of root hairs. Curr. Biol. 29, 1854–1865 (2019).
Elliott, L. & Kirchhelle, C. The importance of being edgy: cell geometric edges as an emerging polar domain in plant cells. J. Microsc. 278, 123–131 (2020).
Lace, B. et al. RPG acts as a central determinant for infectosome formation and cellular polarization during intracellular rhizobial infections. eLife 12, e80741 (2023).
Scholz, P., Anstatt, J., Krawczyk, H. E. & Ischebeck, T. Signalling pinpointed to the tip: the complex regulatory network that allows pollen tube growth. Plants 9, 1098 (2020).
Müller, S. Update: on selected ROP cell polarity mechanisms in plant cell morphogenesis. Plant Physiol. 193, 26–41 (2023).
Oda, Y. & Fukuda, H. Initiation of cell wall pattern by a Rho- and microtubule-driven symmetry breaking. Science 337, 1333–1336 (2012).
Smokvarska, M., Jaillais, Y. & Martinière, A. Function of membrane domains in Rho-of-Plant signaling. Plant Physiol. 185, 663–681 (2021).
Lin, W. & Yang, Z. Unlocking the mechanisms behind the formation of interlocking pavement cells. Curr. Opin. Plant Biol. 57, 142–154 (2020).
Kost, B. et al. Rac homologues and compartmentalized phosphatidylinositol 4,5-bisphosphate act in a common pathway to regulate polar pollen tube growth. J. Cell Biol. 145, 317–330 (1999).
Noack, L. C. & Jaillais, Y. Precision targeting by phosphoinositides: how PIs direct endomembrane trafficking in plants. Curr. Opin. Plant Biol. 40, 22–33 (2017).
Sternberg, H. et al. Formation of self-organizing functionally distinct Rho of plants domains involves a reduced mobile population. Plant Physiol. 187, 2485–2508 (2021).
Bozkurt, T. O. et al. The plant membrane-associated REMORIN1.3 accumulates in discrete perihaustorial domains and enhances susceptibility to Phytophthora infestans. Plant Physiol. 165, 1005–1018 (2014).
Qin, L. et al. Specific recruitment of phosphoinositide species to the plant–pathogen interfacial membrane underlies Arabidopsis susceptibility to fungal infection. Plant Cell 32, 1665–1688 (2020).
Kolbeck, A. et al. CASP microdomain formation requires cross cell wall stabilization of domains and non-cell autonomous action of LOTR1. eLife 11, e69602 (2022).
Barbosa, I. C. R. et al. Directed growth and fusion of membrane-wall microdomains requires CASP-mediated inhibition and displacement of secretory foci. Nat. Commun. 14, 1626 (2023).
Yoshida, S. et al. A SOSEKI-based coordinate system interprets global polarity cues in Arabidopsis. Nat. Plants 5, 160–166 (2019).
van Dop, M. et al. DIX domain polymerization drives assembly of plant cell polarity complexes. Cell 180, 427–439 (2020).
Ramalho, J. J., Jones, V. A. S., Mutte, S. & Weijers, D. Pole position: how plant cells polarize along the axes. Plant Cell 34, 174–192 (2022).
Lee, Y., Rubio, M. C., Alassimone, J. & Geldner, N. A mechanism for localized lignin deposition in the endodermis. Cell 153, 402–412 (2013).
Hosmani, P. S. et al. Dirigent domain-containing protein is part of the machinery required for formation of the lignin-based Casparian strip in the root. Proc. Natl Acad. Sci. USA 110, 14498–14503 (2013).
Pan, X., Pérez-Henríquez, P., Van Norman, J. M. & Yang, Z. Membrane nanodomains: dynamic nanobuilding blocks of polarized cell growth. Plant Physiol. 93, 83–97 (2023).
Sugiyama, Y., Wakazaki, M., Toyooka, K., Fukuda, H. & Oda, Y. A novel plasma membrane-anchored protein regulates xylem cell-wall deposition through microtubule-dependent lateral inhibition of Rho GTPase domains. Curr. Biol. 27, 2522–2528 (2017).
Alassimone, J., Naseer, S. & Geldner, N. A developmental framework for endodermal differentiation and polarity. Proc. Natl Acad. Sci. USA 107, 5214–5219 (2010).
Aliaga Fandino, A. C., Jelínková, A., Marhava, P., Petrášek, J. & Hardtke, C. S. Ectopic assembly of an auxin efflux control machinery shifts developmental trajectories. Plant Cell 36, 1791–1805 (2024).
Bloch, D. et al. Exocyst SEC3 and phosphoinositides define sites of exocytosis in pollen tube initiation and growth. Plant Physiol. 172, 980–1002 (2016).
Vukašinović, N. et al. Microtubule-dependent targeting of the exocyst complex is necessary for xylem development in Arabidopsis. N. Phytol. 213, 1052–1067 (2016).
Kusumaatmaja, H. et al. Wetting of phase-separated droplets on plant vacuole membranes leads to a competition between tonoplast budding and nanotube formation. Proc. Natl Acad. Sci. USA 118, e2024109118 (2021).
Dragwidge, J. M. et al. Biomolecular condensation orchestrates clathrin-mediated endocytosis in plants. Nat. Cell Biol. 26, 438–449 (2024).
Liu, C. et al. SEC14-like condensate phase transitions at plasma membranes regulate root growth in Arabidopsis. PLoS Biol. 21, e3002305 (2023).
Noack, L. C. et al. A nanodomain-anchored scaffolding complex is required for the function and localization of phosphatidylinositol 4-kinase alpha in plants. Plant Cell 34, 302–332 (2022).
Gomez, R. E. et al. Phosphatidylinositol-4-phosphate controls autophagosome formation in Arabidopsis thaliana. Nat. Commun. 13, 4385 (2022).
Chambaud, C., Cookson, S. J., Ollat, N., Bayer, E. & Brocard, L. A correlative light electron microscopy approach reveals plasmodesmata ultrastructure at the graft interface. Plant Physiol. 188, 44–55 (2021).
Li, Z. P. et al. Plant plasmodesmata bridges form through ER-driven incomplete cytokinesis. Science adn4630 https://doi.org/10.1101/2023.12.12.571296 (2024 in press).
Watanabe, S. et al. Protein localization in electron micrographs using fluorescence nanoscopy. Nat. Methods 8, 80–84 (2011).
Fu, Z. et al. mEosEM withstands osmium staining and Epon embedding for super-resolution CLEM. Nat. Methods 17, 55–58 (2020).
Brown, D. A. & Rose, J. K. Sorting of GPI-anchored proteins to glycolipid-enriched membrane subdomains during transport to the apical cell surface. Cell 68, 533–544 (1992).
Renne, M. F. & Ernst, R. Membrane homeostasis beyond fluidity: control of membrane compressibility. Trends Biochem. Sci. 48, 963–977 (2023).
Marsh, D. Handbook of Lipid Bilayers (CRC, 2013).
Mair, A., Xu, S.-L., Branon, T. C., Ting, A. Y. & Bergmann, D. C. Proximity labeling of protein complexes and cell-type-specific organellar proteomes in Arabidopsis enabled by TurboID. eLife 8, e47864 (2019).
Arora, D. et al. Establishment of proximity-dependent biotinylation approaches in different plant model systems. Plant Cell 32, 3388–3407 (2020).
Wallner, E.-S. et al. Spatially resolved proteomics of the Arabidopsis stomatal lineage identifies polarity complexes for cell divisions and stomatal pores. Dev. Cell 59, 1096–1109 (2024).
Liu, C. et al. An actin remodeling role for Arabidopsis processing bodies revealed by their proximity interactome. EMBO J. 42, e111885 (2023).
Laganowsky, A. et al. Membrane proteins bind lipids selectively to modulate their structure and function. Nature 510, 172–175 (2014).
Laganowsky, A., Reading, E., Hopper, J. T. S. & Robinson, C. V. Mass spectrometry of intact membrane protein complexes. Nat. Protoc. 8, 639–651 (2013).
Liu, P. et al. Mechanism of sphingolipid homeostasis revealed by structural analysis of Arabidopsis SPT–ORM1 complex. Sci. Adv. 9, eadg0728 (2023).
Sarewicz, M. et al. High-resolution cryo-EM structures of plant cytochrome b6f at work. Sci. Adv. 9, eadd9688 (2023).
Mandala, V. S., Loftis, A. R., Shcherbakov, A. A., Pentelute, B. L. & Hong, M. Atomic structures of closed and open influenza B M2 proton channel reveal the conduction mechanism. Nat. Struct. Mol. Biol. 27, 160–167 (2020).
Xie, H. et al. Solid-state NMR structure determination of a membrane protein in E. coli cellular inner membrane. Sci. Adv. 9, eadh4168 (2023).
Marrink, S. J. et al. Computational modeling of realistic cell membranes. Chem. Rev. 119, 6184–6226 (2019).
Lee, E. H., Hsin, J., Sotomayor, M., Comellas, G. & Schulten, K. Discovery through the computational microscope. Structure 17, 1295–1306 (2009).
Sych, T., Levental, K. R. & Sezgin, E. Lipid–protein interactions in plasma membrane organization and function. Annu. Rev. Biophys. 51, 135–156 (2022).
Jumper, J. et al. Highly accurate protein structure prediction with AlphaFold. Nature 596, 583–589 (2021).
König, S., Mosblech, A. & Heilmann, I. Stress-inducible and constitutive phosphoinositide pools have distinctive fatty acid patterns in Arabidopsis thaliana. FASEB J. 21, 1958–1967 (2007).
Boutté, Y. & Jaillais, Y. Metabolic cellular communications: feedback mechanisms between membrane lipid homeostasis and plant development. Dev. Cell 54, 171–182 (2020).
Cassim, A. M. et al. Plant lipids: key players of plasma membrane organization and function. Prog. Lipid Res. 73, 1–27 (2019).
Colin, L. A. & Jaillais, Y. Phospholipids across scales: lipid patterns and plant development. Curr. Opin. Plant Biol. 53, 1–9 (2019).
Martinière, A. et al. Cell wall constrains lateral diffusion of plant plasma-membrane proteins. Proc. Natl Acad. Sci. USA 109, 12805–12810 (2012).
Liu, Z., Persson, S. & Sanchez-Rodriguez, C. At the border: the plasma membrane–cell wall continuum. J. Exp. Bot. 66, 1553–1563 (2015).
Kirchhelle, C. & Hamant, O. Discretizing the cellular bases of plant morphogenesis: emerging properties from subcellular and noisy patterning. Curr. Opin. Cell Biol. 81, 102159 (2023).
Elliott, L. et al. A self-regulatory cell-wall-sensing module at cell edges controls plant growth. Nat. Plants 10, 483–493 (2024).
Wallner, E.-S., Dolan, L. & Bergmann, D. C. Arabidopsis stomatal lineage cells establish bipolarity and segregate differential signaling capacity to regulate stem cell potential. Dev. Cell 58, 1643–1656 (2023).
Minami, A. et al. Arabidopsis dynamin-related protein 1E in sphingolipid-enriched plasma membrane domains is associated with the development of freezing tolerance. Plant J. 83, 501–514 (2015).
Laine, R. F. et al. High-fidelity 3D live-cell nanoscopy through data-driven enhanced super-resolution radial fluctuation. Nat. Methods 20, 1949–1956 (2023).
Levet, F. et al. A tessellation-based colocalization analysis approach for single-molecule localization microscopy. Nat. Commun. 10, 2379 (2019).
Wallis, T. P. et al. Super-resolved trajectory-derived nanoclustering analysis using spatiotemporal indexing. Nat. Commun. 14, 3353 (2023).
Acknowledgements
We thank C. Miège and N. Geldner for their help in preparing the manuscript. We thank M. von Arx (Fig. 4p–r), V. Markovic (Fig. 4g,h), E.-S. Wallner (Fig. 4e), D. Biermann (Fig. 4i–k), A. De Meyer (Fig. 4f), M. van Dop (Fig. 4d) and L. Elliot (Fig. 4c) for providing the microscopy images shown in Fig. 4.
Author information
Authors and Affiliations
Contributions
Y.J., E.B., Y.B., M.-C.C., A.M., S.M., F.S.-P., E.Z. and J.G. planned the project. Y.J. and J.G. wrote the draft manuscript. Y.J., E.B., D.C.B., M.A.B., Y.B., T.O.B., M.-C.C., V.G., G.G., I.H., P.A.H., C.K., A.M., Y.M., S. Mongrand, S. Müller, L.C.N., Y.O., T.O., X.P., R.P., M.P., S.R., C.S.R., F.S.-P., E.R., D.V.D., J.M.v.N., D.W., S.Y., Z.Y., E.Z. and J.G. edited and commented on the manuscript. J.G. prepared the figures. Y.B. (Fig. 4a,b), L.C.N. (Fig. 4l) and J.G. contributed to the figure panels.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Plants thanks Pengwei Wang and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jaillais, Y., Bayer, E., Bergmann, D.C. et al. Guidelines for naming and studying plasma membrane domains in plants. Nat. Plants 10, 1172–1183 (2024). https://doi.org/10.1038/s41477-024-01742-8
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41477-024-01742-8