Abstract
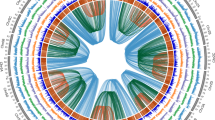
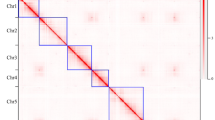
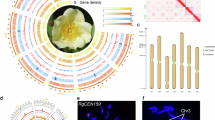
The cultivation and domestication of roses reflects cultural exchanges and shifts in aesthetics that have resulted in today’s most popular ornamental plant group. However, the narrow genetic foundation of cultivated roses limits their further improvement. Wild Rosa species harbour vast genetic diversity, yet their utilization is impeded by taxonomic confusion. Here we generated a phased and gap-free reference genome of Rosa persica for phylogenetic and population genomic analyses of a large collection of Rosa samples. The robust nuclear and plastid phylogenies support most of the morphology-based traditional taxonomy of Rosa. Population genomic analyses disclosed potential genetic exchanges among sections, indicating the northwest and southwest of China as two independent centres of diversity for Rosa. Analyses of domestication traits provide insights into selection processes related to flower colour, fragrance, double flower and resistance. This study provides a comprehensive understanding of rose domestication and lays a solid foundation for future re-domestication and innovative breeding efforts using wild resources.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
Raw sequencing data have been deposited in the National Center for Biotechnology Information (NCBI) BioProject database with the BioProject PRJNA1224503. The phased genome assembly of R. persica has been deposited in the NCBI database under accession numbers JBLIYO000000000 (Hap 1) and JBLIYP000000000 (Hap 2). The previously published reads used in this study are available from NCBI and public GDR database at https://www.rosaceae.org. Specific source data are listed in Supplementary Tables 10 and 13.
References
Altman, A., Shennan, S. & Odling-Smee, J. Ornamental plant domestication by aesthetics-driven human cultural niche construction. Trends Plant Sci. 27, 124–138 (2022).
Dempewolf, H. et al. Past and future use of wild relatives in crop breeding. Crop Sci. 57, 1070–1082 (2017).
Cheng, S. et al. Harnessing landrace diversity empowers wheat breeding. Nature 632, 823–831 (2024).
Bohra, A. et al. Reap the crop wild relatives for breeding future crops. Trends Biotechnol. 40, 412–431 (2022).
Smulders, M. J. M., et al. In Wild Crop Relatives: Genomic and Breeding Resources: Plantation and Ornamental Crops (ed. Kole, C.) 243–275 (Springer, 2011).
Krüssmann, G. The Complete Book of Roses (Timber, 1981).
Thibault, L., et al. Dark side of the honeymoon: reconstructing the Asian x European rose breeding history through the lens of genomics. Preprint at bioRxiv, https://doi.org/10.1101/2023.06.22.546162 (2024).
Purugganan, M. D. What is domestication? Trends Ecol. Evol. 37, 663–671 (2022).
Wyile, A. The history of garden roses. Part I. J. Roy. Hort. Soc. 79, 555–571 (1954).
Raymond, O. et al. The Rosa genome provides new insights into the domestication of modern roses. Nat. Genet. 50, 772–777 (2018).
Tan, J. et al. Genetic relationships and evolution of old Chinese garden roses based on SSRs and chromosome diversity. Sci. Rep. 7, 15437 (2017).
Vukosavljev, M. et al. Genetic diversity and differentiation in roses: a garden rose perspective. Sci. Hortic. 162, 320–332 (2013).
Matsumoto, S. et al. Phylogenetic analyses of the genus Rosa using the matK sequence: molecular evidence for the narrow genetic background of modern roses. Sci. Hortic. 77, 73–82 (1998).
Koopman, W. J. M. et al. AFLP markers as a tool to reconstruct complex relationships: a case study in Rosa (Rosaceae). Am. J. Bot. 95, 353–366 (2008).
Fougere-Danezan, M., Joly, S., Bruneau, A., Gao, X. F. & Zhang, L. B. Phylogeny and biogeography of wild roses with specific attention to polyploids. Ann. Bot. 115, 275–291 (2015).
Luo, L. Genus Rosa L. in China (China Forestry Publishing House, 2024).
Rehder, A. Bibliography of Cultivated Trees and Shrubs Hardy in the Cooler Temperate Regions of the Northern Hemisphere (Arnold Arboretum of Harvard University, 1949).
Wissemann, V. Reference Module in Life Sciences (Elsevier, 2017).
Wissemann, V. & Ritz, C. M. The genus Rosa (Rosoideae, Rosaceae) revisited: molecular analysis of nrITS-1 and atp B-rbc L intergenic spacer (IGS) versus conventional taxonomy. Bot. J. Linn. Soc. 147, 275–290 (2005).
Zhang, Z. et al. Haplotype-resolved genome assembly and resequencing provide insights into the origin and breeding of modern rose. Nat. Plants 10, 1659–1671 (2024).
Zhang, X. et al. Haplotype-resolved genome assembly of the diploid Rosa chinensis provides insight into the mechanisms underlying key ornamental traits. Mol. Hortic. 4, 14 (2024).
Zhong, M. et al. Rose without prickle: genomic insights linked to moisture adaptation. Natl Sci. Rev. 8, nwab092 (2021).
Chen, F. et al. A chromosome-level genome assembly of rugged rose (Rosa rugosa) provides insights into its evolution, ecology, and floral characteristics. Hortic. Res. 8, 141 (2021).
Hibrand Saint-Oyant, L. et al. A high-quality genome sequence of Rosa chinensis to elucidate ornamental traits. Nat. Plants 4, 473–484 (2018).
Ku, T. R. K. Rosa (Rosaceae). In Flora of China 9 (eds Wu, Z.Y. & Raven, P.H.) (Missouri Botanical Garden, 2003).
Schanzer, I. A., Fedorova, A. V. & Meschersky, I. G. A haplotype network approach to reconstruct the phylogeny of Rosa L. (Rosaceae). Biol. Bull. 51, 331–345 (2024).
Lin, Y. Z. et al. quarTeT: a telomere-to-telomere toolkit for gap-free genome assembly and centromeric repeat identification. Hortic. Res. 10, uhad127 (2023).
Springer, N. M. Transposable elements: microbiomes in the genomes. Nat. Plants 1, 15004 (2015).
Bennetzen, J. L. & Wang, H. The contributions of transposable elements to the structure, function, and evolution of plant genomes. Annu. Rev. Plant Biol. 65, 505–530 (2014).
Tang, Y.-W. et al. Rosa forrestiana var. maculata, a new variety of Rosa (Rosaceae) from Yunnan, China. Phytotaxa 652, 4 (2024).
Zheng, L.-N. et al. Rosa funingensis (Rosaceae), a new species from Yunnan, China. PhytoKeys 229, 61–70 (2023).
Lyu, P. et al. Rosa yangii (Rosaceae), a new species from China. Kew Bull. 78, 663–671 (2023).
Deng, T. et al. Rosa tomurensis, a new species of Rosa (Rosaceae) from China. Phytotaxa 556, 169–177 (2022).
Liorzou, M. et al. Nineteenth century French rose Rosa sp. germplasm shows a shift over time from a European to an Asian genetic background. J. Exp. Bot. 67, 4711–4725 (2016).
Zang, F. et al. Resequencing of Rosa rugosa accessions revealed the history of population dynamics, breed origin, and domestication pathways. BMC Plant Biol. 23, 235 (2023).
Ao, H. et al. Global warming-induced Asian hydrological climate transition across the Miocene–Pliocene boundary. Nat. Commun. 12, 6935 (2021).
Clark, P. U. et al. The Last Glacial Maximum. Science 325, 710–714 (2009).
Ogata, J., Kanno, Y., Itoh, Y., Tsugawa, H. & Suzuki, M. Anthocyanin biosynthesis in roses. Nature 435, 757–758 (2005).
Fukuchi-Mizutani, M. et al. Biochemical and molecular characterization of anthocyanidin/flavonol 3-glucosylation pathways in Rosa× hybrida. Plant Biotechnol. 28, 239–244 (2011).
He, G., Zhang, R., Jiang, S., Wang, H. & Ming, F. The MYB transcription factor RcMYB1 plays a central role in rose anthocyanin biosynthesis. Hortic. Res. 10, uhad080 (2023).
Li, M. et al. Rosa1, a transposable element-like insertion, produces red petal coloration in rose through altering RcMYB114 transcription. Front. Plant Sci. 13, 857684 (2022).
Shen, Y. et al. RrMYB5- and RrMYB10-regulated flavonoid biosynthesis plays a pivotal role in feedback loop responding to wounding and oxidation in Rosa rugosa. Plant Biotechnol. J. 17, 2078–2095 (2019).
Guterman, I. et al. Rose scent: genomics approach to discovering novel floral fragrance-related genes. Plant Cell 14, 2325–2338 (2002).
Spiller, M., Berger, R. G. & Debener, T. Genetic dissection of scent metabolic profiles in diploid rose populations. Theor. Appl. Genet. 120, 1461–1471 (2010).
Sakai, M. et al. Production of 2-phenylethanol in roses as the dominant floral scent compound from l-phenylalanine by two key enzymes, a PLP-dependent decarboxylase and a phenylacetaldehyde reductase. Biosci. Biotechnol. Biochem. 71, 2408–2419 (2007).
Li, Y. et al. The coordinated interaction or regulation between floral pigments and volatile organic compounds. Hortic. Plant J. https://doi.org/10.1016/j.hpj.2024.01.002 (2024).
Li, H. et al. The complexity of volatile terpene biosynthesis in roses: particular insights into β-citronellol production. Plant Physiol. 196, 1908–1922 (2024).
Soufflet-Freslon, V. et al. Diversity and selection of the continuous-flowering gene, RoKSN, in rose. Hortic. Res. 8, 76 (2021).
Iwata, H. et al. The TFL1 homologue KSN is a regulator of continuous flowering in rose and strawberry. Plant J. 69, 116–125 (2012).
Dugo, M. L. et al. Genetic mapping of QTLs controlling horticultural traits in diploid roses. Theor. Appl. Genet. 111, 511–520 (2005).
Shubin, L. et al. Inheritance of perpetual blooming in Rosa chinensis ‘Old Blush’. Hortic. Plant J. 1, 108–112 (2015).
Kania, T., Russenberger, D., Peng, S., Apel, K. & Melzer, S. FPF1 promotes flowering in Arabidopsis. Plant Cell 9, 1327–1338 (1997).
Lei, Y. et al. Woodland strawberry WRKY71 acts as a promoter of flowering via a transcriptional regulatory cascade. Hortic. Res. 7, 137 (2020).
Kim, H.-J. et al. A genetic link between cold responses and flowering time through FVE in Arabidopsis thaliana. Nat. Genet. 36, 167–171 (2004).
Roman, H. et al. Genetic analysis of the flowering date and number of petals in rose. Tree Genet. Genomes 11, 85 (2015).
François, L. et al. A miR172 target-deficient AP2-like gene correlates with the double flower phenotype in roses. Sci. Rep. 8, 12912 (2018).
Gattolin, S. et al. Deletion of the miR172 target site in a TOE-type gene is a strong candidate variant for dominant double-flower trait in Rosaceae. Plant J. 96, 358–371 (2018).
Lopez Arias, D. C. et al. Characterization of black spot resistance in diploid roses with QTL detection, meta-analysis and candidate-gene identification. Theor. Appl. Genet. 133, 3299–3321 (2020).
Li, X. R. et al. Function of two splicing variants of RcCPR5 in the resistance of Rosa chinensis to powdery mildew. Plant Sci. 335, 111678 (2023).
Rawandoozi, Z. J. et al. QTL mapping and characterization of black spot disease resistance using two multi-parental diploid rose populations. Hortic. Res. 10, uhad059 (2023).
Radauer, C., Lackner, P. & Breiteneder, H. The Bet v 1 fold: an ancient, versatile scaffold for binding of large, hydrophobic ligands. BMC Evol. Biol. 8, 286 (2008).
Li, R. et al. Integrated proteomic analysis reveals interactions between phosphorylation and ubiquitination in rose response to Botrytis infection. Hortic. Res. 11, uhad238 (2024).
Linnaeus, C. Species Plantarum, Holmiae: Impensis Laurentii Salvii, Vol. 1, 491–492 (1753).
Debray, K. et al. Unveiling the patterns of reticulated evolutionary processes with phylogenomics: hybridization and polyploidy in the genus Rosa. Syst. Biol. 71, 547–569 (2022).
Cui, W. H. et al. Complex and reticulate origin of edible roses (Rosa, Rosaceae) in China. Hortic. Res. 9, uhab051 (2022).
Zhang, C., Li, S.-Q., Xie, H.-H., Liu, J.-Q. & Gao, X.-F. Comparative plastid genome analyses of Rosa: insights into the phylogeny and gene divergence. Tree Genet. Genomes 18, 20 (2022).
Bruneau, A., Starr, J. R. & Joly, S. Phylogenetic relationships in the genus Rosa: new evidence from chloroplast DNA sequences and an appraisal of current knowledge. Syst. Bot. 32, 366–378 (2007).
Zhu, Z.-M., Gao, X.-F. & Fougère-Danezan, M. Phylogeny of Rosa sections Chinenses and Synstylae (Rosaceae) based on chloroplast and nuclear markers. Mol. Phylogenet. Evol. 87, 50–64 (2015).
Liu, C. Y. et al. Phylogenetic relationships in the genus Rosa revisited based on rpl16, trnL-F, and atpB-rbcL sequences. Hortscience 50, 1618–1624 (2015).
Zhang, C. et al. Molecular and morphological evidence for hybrid origin and matroclinal inheritance of an endangered wild rose, Rosa × pseudobanksiae (Rosaceae) from China. Conserv. Genet. 21, 1–11 (2020).
Meng, J., Fougère-Danezan, M., Zhang, L.-B., Li, D.-Z. & Yi, T.-S. Untangling the hybrid origin of the Chinese tea roses: evidence from DNA sequences of single-copy nuclear and chloroplast genes. Plant Syst. Evol. 297, 157–170 (2011).
Debray, K. et al. Identification and assessment of variable single-copy orthologous (SCO) nuclear loci for low-level phylogenomics: a case study in the genus Rosa (Rosaceae). BMC Evol. Biol. 19, 152 (2019).
Zuntini, A. R. et al. Phylogenomics and the rise of the angiosperms. Nature 629, 843–850 (2024).
Wang, Q., Guo, Q., Chi, X., Zhu, S. & Tang, Z. Evolutionary history and climate conditions constrain the flower colours of woody plants in China. J. Plant Ecol. 15, 196–207 (2021).
Gao, Y.-D., Zhang, Y., Gao, X.-F. & Zhu, Z.-M. Pleistocene glaciations, demographic expansion and subsequent isolation promoted morphological heterogeneity: a phylogeographic study of the alpine Rosa sericea complex (Rosaceae). Sci. Rep. 5, 11698 (2015).
Xing, Y. & Ree, R. H. Uplift-driven diversification in the Hengduan Mountains, a temperate biodiversity hotspot. Proc. Natl Acad. Sci. USA 114, E3444–E3451 (2017).
Gao, Y.-D., Gao, X.-F. & Harris, A. Species boundaries and parapatric speciation in the complex of alpine shrubs, Rosa sericea (Rosaceae), based on population genetics and ecological tolerances. Front. Plant Sci. 10, 321 (2019).
Su, T. et al. A Miocene leaf fossil record of Rosa (R. fortuita n. sp.) from its modern diversity center in SW China. Palaeoworld 25, 104–115 (2016).
Caissard, J. C., Adrar, I., Conart, C., Paramita, S. N. & Baudino, S. Do we really know the scent of roses? Bot. Lett. https://doi.org/10.1080/23818107.2022.2160807 (2022).
Magnard, J. L. et al. Biosynthesis of monoterpene scent compounds in roses. Science 349, 81–83 (2015).
Sun, P., Schuurink, R. C., Caissard, J. C., Hugueney, P. & Baudino, S. My way: noncanonical biosynthesis pathways for plant volatiles. Trends Plant Sci. 21, 884–894 (2016).
Zvi, M. M. et al. PAP1 transcription factor enhances production of phenylpropanoid and terpenoid scent compounds in rose flowers. New Phytol. 195, 335–345 (2012).
Liu, J. Y., Osbourn, A. & Ma, P. D. MYB transcription factors as regulators of phenylpropanoid metabolism in plants. Mol. Plant 8, 689–708 (2015).
Zhai, Y. et al. APETALA2/ethylene responsive factor in fruit ripening: roles, interactions and expression regulation. Front. Plant Sci. 13, 979348 (2022).
Dang, Q. Y. et al. An apple (Malus domestica) AP2/ERF transcription factor modulates carotenoid accumulation. Hortic. Res. 8, 223 (2021).
Shen, S. L. et al. CitAP2.10 activation of the terpene synthase CsTPS1 is associated with the synthesis of (+)-valencene in ‘Newhall’ orange. J. Exp. Bot. 67, 4105–4115 (2016).
Wei, C. Y. et al. Linalool synthesis related PpTPS1 and PpTPS3 are activated by transcription factor PpERF61 whose expression is associated with DNA methylation during peach fruit ripening. Plant Sci. 317, 111200 (2022).
Cui, L. G., Shan, J. X., Shi, M., Gao, J. P. & Lin, H. X. The miR156-SPL9-DFR pathway coordinates the relationship between development and abiotic stress tolerance in plants. Plant J. 80, 1108–1117 (2014).
Yu, Z.-X. et al. Progressive regulation of sesquiterpene biosynthesis in Arabidopsis and patchouli (Pogostemon cablin) by the miR156-targeted SPL transcription factors. Mol. Plant 8, 98–110 (2015).
Zhu, Z. Q. et al. An O-methyltransferase gene, RrCCoAOMT1, participates in the red flower color formation of Rosa rugosa. Sci. Hortic. 336, 113402 (2024).
Yu, H. & Li, J. Y. Breeding future crops to feed the world through de novo domestication. Nat. Commun. 13, 1171 (2022).
Zsögön, A. et al. De novo domestication of wild tomato using genome editing. Nat. Biotechnol. 36, 1211–1216 (2018).
Rhie, A., Walenz, B. P., Koren, S. & Phillippy, A. M. Merqury: reference-free quality, completeness, and phasing assessment for genome assemblies. Genome Biol. 21, 245 (2020).
Simao, F. A., Waterhouse, R. M., Ioannidis, P., Kriventseva, E. V. & Zdobnov, E. M. BUSCO: assessing genome assembly and annotation completeness with single-copy orthologs. Bioinformatics 31, 3210–3212 (2015).
Li, H. & Durbin, R. Fast and accurate long-read alignment with Burrows–Wheeler transform. Bioinformatics 26, 589–595 (2010).
McKenna, A. et al. The Genome Analysis Toolkit: a MapReduce framework for analyzing next-generation DNA sequencing data. Genome Res. 20, 1297–1303 (2010).
Chen, C. J. et al. TBtools: an integrative toolkit developed for interactive analyses of big biological data.Mol. Plant 13, 1194–1202 (2020).
Nguyen, L. T., Schmidt, H. A., von Haeseler, A. & Minh, B. Q. IQ-TREE: a fast and effective stochastic algorithm for estimating maximum-likelihood phylogenies. Mol. Biol. Evol. 32, 268–274 (2015).
Jin, J. J. et al. GetOrganelle: a fast and versatile toolkit for accurate de novo assembly of organelle genomes. Genome Biol. 21, 241 (2020).
Qu, X. J., Moore, M. J., Li, D. Z. & Yi, T. S. PGA: a software package for rapid, accurate, and flexible batch annotation of plastomes. Plant Methods 15, 50 (2019).
Galili, T. dendextend: an R package for visualizing, adjusting and comparing trees of hierarchical clustering. Bioinformatics 31, 3718–3720 (2015).
Maddison, W. P. and Maddison, D. R. Mesquite: a modular system for evolutionary analysis. Version 3.81. (2023); http://www.mesquiteproject.org .
Alexander, D. H., Novembre, J. & Lange, K. Fast model-based estimation of ancestry in unrelated individuals. Genome Res. 19, 1655–1664 (2009).
Purcell, S. et al. PLINK: a tool set for whole-genome association and population-based linkage analyses. Am. J. Hum. Genet. 81, 559–575 (2007).
Zhang, C., Dong, S.-S., Xu, J.-Y., He, W.-M. & Yang, T.-L. PopLDdecay: a fast and effective tool for linkage disequilibrium decay analysis based on variant call format files. Bioinformatics 35, 1786–1788 (2018).
Zhang, R. L., Jia, G. Q. & Diao, X. M. geneHapR: an R package for gene haplotypic statistics and visualization. BMC Bioinformatics 24, 199 (2023).
Rosyara, U. R., De Jong, W. S., Douches, D. S. & Endelman, J. B. Software for genome-wide association studies in autopolyploids and its application to potato. Plant Genome 9, plantgenome2015.08.0073 (2016).
Danecek, P. et al. The variant call format and VCFtools. Bioinformatics 27, 2156–2158 (2011).
Pickrell, J. K. & Pritchard, J. K. Inference of population splits and mixtures from genome-wide allele frequency data. PLoS Genet. 8, e1002967 (2012).
Malinsky, M., Matschiner, M. & Svardal, H. Dsuite - fast D-statistics and related admixture evidence from VCF files. Mol. Ecol. Resour. 21, 584–595 (2021).
Liu, X. & Fu, Y. X. Stairway Plot 2: demographic history inference with folded SNP frequency spectra. Genome Biol. 21, 280 (2020).
Yang, Z. PAML: a program package for phylogenetic analysis by maximum likelihood. Comput. Appl. Biosci. 13, 555–556 (1997).
Acknowledgements
We thank Y. Yang (Kunming Yang Chinese Rose Gardening) for collecting wild Rosa resources and for helpful discussions on Rosa taxonomy. We are grateful to Y. Sui and R. Guo (Xinjiang Career Technical College) for their efforts in collecting Rosa resources in Xinjiang. We thank X. Gao (Key Laboratory of Molecular Epigenetics of Ministry of Education (MOE), Northeast Normal University) for his inspiring discussion and comments. We appreciate the valuable suggestions on the manuscript provided by R. Smulders and P. Arens (Plant Breeding, Wageningen University and Research). We thank J. Endelman (University of Wisconsin) for his instruction on GWASpoly software. We thank Man Zhang and T. Zheng for the suggestions on improving the paper. We thank J. Zhao (Boyce Thompson Institute) for his instruction on demographic analyses. We thank Yuxuan Zhang, L. Ji, Yujing Zhang, L. Jiang, C. Feng, Y. Zhuang, Z. Ou, R. Wang, J. Tang and K. Xiong for their assistance with the investigations of Rosa species. Furthermore, we thank Wuhan Dazhong Yuansheng Technology, Wuhan Feisha Genetic Information (www.frasergen.com) and BerryGenomics (www.berrygenomics.com) for providing essential sequencing service. Finally, the authors are profoundly indebted to every member of the Fishpond family, whose collective efforts were instrumental in the fruition of this work. This research was supported by the Fundamental Research Funds for the Central Universities (grant number QNTD202306 to C. Yu), National Key R&D Program of China (grant number 2019YFD1000400 to C. Yu) and National Natural Science Foundation of China (grant numbers 32471955 and 32071818 to C. Yu).
Author information
Authors and Affiliations
Contributions
C. Yu designed and managed the project. B.C. performed population genomic analyses and wrote the manuscript. K.Z. assembled the Rosa persica genome and performed genomic analyses. M.Z. performed GWAS analysis. P.M.B. provided guidance on GWAS analysis and revised the manuscript. L. Zhou and Y.S. managed the plant materials. S.W. performed population history analyses. L.G. performed selective sweep analyses. W.D. performed demographic analyses. C. Yang and J.C. constructed phylogenetic trees and performed phylogenetic analyses. R.H. designed and revised the figures. X.T. performed karyotype analyses of Rosa materials. L. Zhang performed data analyses. C. Yu, B.C., K.Z., M.Z., L.G., W.D. and S.W. wrote the paper with contributions from H.H., Y.H., L.L., H.P. and Q.Z.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Plants thanks Xingtan Zhang and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Flower traits of Rosa accessions used in this study.
The plates of accessions are sorted by scientific groups. Sample IDs correspond to Supplementary Table 13.
Extended Data Fig. 2 Leaf traits of Rosa accessions used in this study.
The plates of accessions are sorted by number of leaflets. Sample IDs correspond to Supplementary Table 13.
Extended Data Fig. 3 Comparison between plastid phylogeny (left) based on chloroplast coding sequences, and nuclear phylogeny (right) based on single-copy SNPs.
Both trees were constructed using maximum likelihood method. Bootstrap values were tested with 1,000 replicates. The colors represent accessions from different botanical groups, in correspondence with previous figures.
Extended Data Fig. 4 Geographical distributions of different botanical sections of Rosa.
The white dots represent distribution information of Rosa accessions, which was derived from field investigation, specimen information, and geographic distribution records from Flora of China and Chinese Virtual Herbarium (http://www.cvh.ac.cn).
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–12, notes and methods.
Supplementary Tables (download XLSX )
Supplementary Tables 1–20.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Cheng, B., Zhao, K., Zhou, M. et al. Phenotypic and genomic signatures across wild Rosa species open new horizons for modern rose breeding. Nat. Plants 11, 775–789 (2025). https://doi.org/10.1038/s41477-025-01955-5
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41477-025-01955-5
This article is cited by
-
RpNACs transcription factors coordinate leaf margin development in Rosa persica
BMC Genomics (2026)
-
An evolutionary bouquet for roses
Nature Plants (2025)
-
Roses are red — but their ancestors were yellow
Nature (2025)
-
From petals to potential: the multifaceted world of Rosa persica Michx. ex Juss
Genetic Resources and Crop Evolution (2025)