Abstract
Wetlands, among Earth’s most carbon-dense ecosystems, are vital for climate change mitigation. While plant diversity has been widely shown to increase soil carbon storage in terrestrial ecosystems, its influence in natural wetlands remains unclear. Here, using data from 1,268 natural wetlands surveyed in the US National Wetland Condition Assessment (NWCA), we examined how trait-based plant diversity (functional diversity) and composition (functional identity) affect soil carbon storage. We show that functional diversity had a minimal effect on carbon stocks, and its influence was weakened by elevated soil nutrient availability and non-native plant stress. In contrast, soil carbon storage was generally greater in wetlands dominated by larger, slow-growing and highly hydrophytic plants. Moreover, the benefits of functional identity were contingent on higher water levels and lower human disturbance. These findings suggest that the conservation and restoration of wetlands dominated by large, conservative and hydrophytic species under hydric conditions could help achieve climate change mitigation goals.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The wetland vegetation and soil factor data were downloaded from the NWCA database (https://www.epa.gov/national-aquatic-resource-surveys/nwca). The leaf trait data were downloaded from the TRY database (https://www.try-db.org) and the BIEN 4 database (https://bien.nceas.ucsb.edu/bien/). The Landsat surface reflectance data are available from the Google Earth Engine Data Catalog (https://developers.google.com/earth-engine/datasets/catalog/landsat). The climate data were collected from the CHELSA database (https://chelsa-climate.org). The data that support the findings of this study are available in figshare at https://doi.org/10.6084/m9.figshare.29097845 (ref. 113). Source data are provided with this paper.
Code availability
All codes are archived in figshare at https://doi.org/10.6084/m9.figshare.29097845 (ref. 113).
References
Poulter, B. et al. in Wetland Carbon and Environmental Management (eds Krauss, K. W. et al.) Ch. 1 (Wiley, 2021).
Were, D., Kansiime, F., Fetahi, T., Cooper, A. & Jjuuko, C. Carbon sequestration by wetlands: a critical review of enhancement measures for climate change mitigation. Earth Syst. Environ. 3, 327–340 (2019).
IPCC. Climate Change 2022: Mitigation of Climate Change (Cambridge Univ. Press, 2022).
Fluet-Chouinard, E. et al. Extensive global wetland loss over the past three centuries. Nature 614, 281–286 (2023).
Global Wetland Outlook: Special Edition 2021 (Convention on Wetlands, 2021).
FAO, IUCN CEM & SER. Principles for Ecosystem Restoration to Guide the United Nations Decade 2021–2030 (FAO, 2021).
Temmink, R. J. M. et al. Recovering wetland biogeomorphic feedbacks to restore the world’s biotic carbon hotspots. Science 376, eabn1479 (2022).
Hulvey, K. B. et al. Benefits of tree mixes in carbon plantings. Nat. Clim. Change 3, 869–874 (2013).
Chen, X. et al. Effects of plant diversity on soil carbon in diverse ecosystems: a global meta-analysis. Biol. Rev. 95, 167–183 (2020).
Chen, X. et al. Tree diversity increases decadal forest soil carbon and nitrogen accrual. Nature 618, 94–101 (2023).
Chen, S. et al. Plant diversity enhances productivity and soil carbon storage. Proc. Natl Acad. Sci. USA 115, 4027–4032 (2018).
Gopal, B. The Wetlands Handbook (Blackwell Publishing, 2009).
Sobral, M., Schleuning, M. & Martinez Cortizas, A. Trait diversity shapes the carbon cycle. Trends Ecol. Evol. 38, 602–604 (2023).
Lange, M., Eisenhauer, N., Chen, H. & Gleixner, G. Increased soil carbon storage through plant diversity strengthens with time and extends into the subsoil. Glob. Change Biol. 29, 2627–2639 (2023).
Spohn, M. et al. The positive effect of plant diversity on soil carbon depends on climate. Nat. Commun. 14, 6624 (2023).
Lange, M. et al. Plant diversity increases soil microbial activity and soil carbon storage. Nat. Commun. 6, 6707 (2015).
Mokany, K., Ash, J. & Roxburgh, S. Functional identity is more important than diversity in influencing ecosystem processes in a temperate native grassland. J. Ecol. 96, 884–893 (2008).
Lin, D. et al. Traits of dominant tree species predict local scale variation in forest aboveground and topsoil carbon stocks. Plant Soil 409, 435–446 (2016).
Engel, T. et al. Traits of dominant plant species drive normalized difference vegetation index in grasslands globally. Glob. Ecol. Biogeogr. 32, 695–706 (2023).
Plazas-Jiménez, D. & Cianciaruso, M. V. Leaf decomposition depends on nutritional trait values but increasing trait variability does not always increase decomposition efficiency. Oikos 130, 1171–1179 (2021).
Casanova, M. T. & Brock, M. A. How do depth, duration and frequency of flooding influence the establishment of wetland plant communities? Plant Ecol. 147, 237–250 (2000).
Merlin, A., Bonis, A., Damgaard, C. F. & Mesleard, F. Competition is a strong driving factor in wetlands, peaking during drying out periods. PLoS ONE 10, e0130152 (2015).
Engelhardt, K. A. M. & Ritchie, M. E. The effect of aquatic plant species richness on wetland ecosystem processes. Ecology 83, 2911–2924 (2002).
Rahman, M. M. et al. Functional composition outweighs taxonomic and functional diversity in maintaining ecosystem properties and processes of mangrove forests. Glob. Change Biol. 30, e17152 (2024).
Tobner, C. M. et al. Functional identity is the main driver of diversity effects in young tree communities. Ecol. Lett. 19, 638–647 (2016).
Weigel, B., Blenckner, T. & Bonsdorff, E. Maintained functional diversity in benthic communities in spite of diverging functional identities. Oikos 125, 1421–1433 (2016).
Diaz, S. et al. The global spectrum of plant form and function. Nature 529, 167–171 (2016).
Pan, Y., Cieraad, E., Bodegom, P. M. & Alahuhta, J. Are ecophysiological adaptive traits decoupled from leaf economics traits in wetlands? Funct. Ecol. 33, 1202–1210 (2019).
Pan, Y. et al. Leading trait dimensions in flood-tolerant plants. Ann. Bot. 130, 383–392 (2022).
De Deyn, G. B., Cornelissen, J. H. & Bardgett, R. D. Plant functional traits and soil carbon sequestration in contrasting biomes. Ecol. Lett. 11, 516–531 (2008).
Mueller, P. & Megonigal, J. P. Redox control on rhizosphere priming in wetlands. Nat. Geosci. 17, 1209–1217 (2024).
Fenner, N. & Freeman, C. Drought-induced carbon loss in peatlands. Nat. Geosci. 4, 895–900 (2011).
Moi, D. A. et al. Human pressure drives biodiversity–multifunctionality relationships in large Neotropical wetlands. Nat. Ecol. Evol. 6, 1279–1289 (2022).
Maestre, F. T., Callaway, R. M., Valladares, F. & Lortie, C. J. Refining the stress-gradient hypothesis for competition and facilitation in plant communities. J. Ecol. 97, 199–205 (2009).
Hou, Y. et al. Drought weakens the positive effect of plant diversity on community biomass. J. Plant Ecol. 17, rtae059 (2024).
Chen, X., Reich, P. B., Taylor, A. R., An, Z. & Chang, S. X. Resource availability enhances positive tree functional diversity effects on carbon and nitrogen accrual in natural forests. Nat. Commun. 15, 8615 (2024).
Wilsey, B., Martin, L., Xu, X., Isbell, F. & Polley, H. W. Biodiversity: net primary productivity relationships are eliminated by invasive species dominance. Ecol. Lett. 27, e14342 (2024).
Wang, X. et al. Effects of combined pollution of organic pollutants and heavy metals on biodiversity and soil multifunctionality in e-waste contaminated soil. J. Hazard. Mater. 440, 129727 (2022).
National Wetland Condition Assessment: 2011 Technical Report Technical Report EPA-843-R-15-006 (US Environmental Protection Agency, 2016).
National Wetland Condition Assessment: 2016 Technical Support Document Technical Report EPA-841-B-23-001 (US Environmental Protection Agency, 2023).
Chen, C., Xiao, W. & Chen, H. Y. H. Meta-analysis reveals global variations in plant diversity effects on productivity. Nature 638, 435–440 (2025).
Ma, Z. & Chen, H. Y. H. Effects of species diversity on fine root productivity in diverse ecosystems: a global meta-analysis. Glob. Ecol. Biogeogr. 25, 1387–1396 (2016).
Tiner, R. W. Lists of potential hydrophytes for the United States: a regional review and their use in wetland identification. Wetlands 26, 624–634 (2006).
Sulman, B. N., Desai, A. R. & Mladenoff, D. J. Modeling soil and biomass carbon responses to declining water table in a wetland-rich landscape. Ecosystems 16, 491–507 (2012).
Deegan, L. A. et al. Coastal eutrophication as a driver of salt marsh loss. Nature 490, 388–392 (2012).
Li, J. et al. Convergence of carbon sink magnitude and water table depth in global wetlands. Ecol. Lett. 26, 797–804 (2023).
Augusto, L. & Boca, A. Tree functional traits, forest biomass, and tree species diversity interact with site properties to drive forest soil carbon. Nat. Commun. 13, 1097 (2022).
Huang, J. et al. Influences of plant functional traits on soil organic carbon stocks: the roles of carbon input quality and diversity. Ecology 106, e70148 (2025).
Chen, X., Hisano, M., Taylor, A. R. & Chen, H. Y. H. The effects of functional diversity and identity (acquisitive versus conservative strategies) on soil carbon stocks are dependent on environmental contexts. For. Ecol. Manage. 503, 119820 (2022).
Henneron, L., Cros, C., Picon-Cochard, C., Rahimian, V. & Fontaine, S. Plant economic strategies of grassland species control soil carbon dynamics through rhizodeposition. J. Ecol. 108, 528–545 (2020).
Han, M. et al. Linking rhizosphere soil microbial activity and plant resource acquisition strategy. J. Ecol. 111, 875–888 (2023).
Bakker, M. A., Carreño-Rocabado, G. & Poorter, L. Leaf economics traits predict litter decomposition of tropical plants and differ among land use types. Funct. Ecol. 25, 473–483 (2011).
Bernal, B. & Mitsch, W. J. Comparing carbon sequestration in temperate freshwater wetland communities. Glob. Change Biol. 18, 1636–1647 (2012).
Happonen, K., Virkkala, A. M., Kemppinen, J., Niittynen, P. & Luoto, M. Relationships between above-ground plant traits and carbon cycling in tundra plant communities. J. Ecol. 110, 700–716 (2022).
Sokol, N. W., Kuebbing, S. E., Karlsen-Ayala, E. & Bradford, M. A. Evidence for the primacy of living root inputs, not root or shoot litter, in forming soil organic carbon. New Phytol. 221, 233–246 (2019).
Li, Y. et al. Leaf size of woody dicots predicts ecosystem primary productivity. Ecol. Lett. 23, 1003–1013 (2020).
Battin, T. J. et al. River ecosystem metabolism and carbon biogeochemistry in a changing world. Nature 613, 449–459 (2023).
Šímová, I. et al. The relationship of woody plant size and leaf nutrient content to large-scale productivity for forests across the Americas. J. Ecol. 107, 2278–2290 (2019).
Quan, Q. et al. Plant height as an indicator for alpine carbon sequestration and ecosystem response to warming. Nat. Plants 10, 890–900 (2024).
Li, J. et al. Two decades of improved wetland carbon sequestration in northern mid-to-high latitudes are offset by tropical and southern declines. Nat. Ecol. Evol. 9, 1861–1872 (2025).
Huang, M. et al. Nitrogen and CO2 enrichment interact to decrease biodiversity impact on complementarity and selection effects. Nat. Commun. 16, 7445 (2025).
Isbell, F. et al. Nutrient enrichment, biodiversity loss, and consequent declines in ecosystem productivity. Proc. Natl Acad. Sci. USA 110, 11911–11916 (2013).
Eskelinen, A., Harpole, W. S., Jessen, M. T., Virtanen, R. & Hautier, Y. Light competition drives herbivore and nutrient effects on plant diversity. Nature 611, 301–305 (2022).
Carboni, M., Livingstone, S. W., Isaac, M. E. & Cadotte, M. W. Invasion drives plant diversity loss through competition and ecosystem modification. J. Ecol. 109, 3587–3601 (2021).
Garbowski, M. et al. Naturalized species drive functional trait shifts in plant communities. Proc. Natl Acad. Sci. USA 121, e2403120121 (2024).
Liao, C. et al. Altered ecosystem carbon and nitrogen cycles by plant invasion: a meta-analysis. New Phytol. 177, 706–714 (2008).
Ferreira, V. et al. A meta-analysis of the effects of nutrient enrichment on litter decomposition in streams. Biol. Rev. 90, 669–688 (2015).
Kushwaha, A., Rani, R., Kumar, S. & Gautam, A. Heavy metal detoxification and tolerance mechanisms in plants: implications for phytoremediation. Environ. Rev. 24, 39–51 (2015).
Garssen, A. G., Baattrup-Pedersen, A., Voesenek, L. A. C. J., Verhoeven, J. T. A. & Soons, M. B. Riparian plant community responses to increased flooding: a meta-analysis. Glob. Change Biol. 21, 2881–2890 (2015).
Schück, M. & Greger, M. Plant traits related to the heavy metal removal capacities of wetland plants. Int. J. Phytoremediation 22, 427–435 (2020).
Rosentreter, J. A. et al. Half of global methane emissions come from highly variable aquatic ecosystem sources. Nat. Geosci. 14, 225–230 (2021).
McCleery, R. et al. Uniting experiments and big data to advance ecology and conservation. Trends Ecol. Evol. 38, 970–979 (2023).
Ma, Y. et al. Global crop production increase by soil organic carbon. Nat. Geosci. 16, 1159–1165 (2023).
Noe, G. B., Hupp, C. R. & Rybicki, N. B. Hydrogeomorphology influences soil nitrogen and phosphorus mineralization in floodplain wetlands. Ecosystems 16, 75–94 (2012).
Dee, L. E. et al. Clarifying the effect of biodiversity on productivity in natural ecosystems with longitudinal data and methods for causal inference. Nat. Commun. 14, 2607 (2023).
Díaz, S. & Malhi, Y. Biodiversity: concepts, patterns, trends, and perspectives. Annu. Rev. Environ. Resour. 47, 31–63 (2022).
Wang, G. et al. Biotic and abiotic drivers of ecosystem temporal stability in herbaceous wetlands in China. Glob. Change Biol. 31, e70056 (2025).
Zhang, C., Cai, M., Ndungu, C. N., Ma, L. & Liu, W. Plant diversity promotes soil nitrogen retention and removal processes in wetlands. New Phytol. 248, 587–599 (2025).
Ferreira, J. et al. Carbon-focused conservation may fail to protect the most biodiverse tropical forests. Nat. Clim. Change 8, 744–749 (2018).
National Aquatic Resource Surveys. National Wetland Condition Assessment 2016 (data and metadata files) (US EPA, 2023); https://www.epa.gov/national-aquatic-resource-surveys/data-national-aquatic-resource-surveys
National Aquatic Resource Surveys. National Wetland Condition Assessment 2011 (data and metadata files) (US EPA, 2016); https://www.epa.gov/national-aquatic-resource-surveys/data-national-aquatic-resource-surveys
National Wetland Condition Assessment 2011: Field Operations Manual (US EPA, 2011).
National Wetland Condition Assessment 2016: Field Operations Manual (US EPA, 2016).
Nahlik, A. M. & Fennessy, M. S. Carbon storage in US wetlands. Nat. Commun. 7, 13835 (2016).
Ridgeway, G. et al. gbm: Generalized Boosted Regression Models. R package version 2.2.2 (2024).
Jalabert, S. S. M. et al. Estimating forest soil bulk density using boosted regression modelling. Soil Use Manage. 26, 516–528 (2010).
Martin, M. P. et al. Optimizing pedotransfer functions for estimating soil bulk density using boosted regression trees. Soil Sci. Soc. Am. J. 73, 485–493 (2009).
Balesdent, J. et al. Atmosphere–soil carbon transfer as a function of soil depth. Nature 559, 599–602 (2018).
Kattge, J. et al. TRY plant trait database – enhanced coverage and open access. Glob. Change Biol. 26, 119–188 (2020).
Maitner, B. S. et al. The bien r package: a tool to access the Botanical Information and Ecology Network (BIEN) database. Methods Ecol. Evol. 9, 373–379 (2017).
Revell, L. J. phytools: an R package for phylogenetic comparative biology (and other things). Methods Ecol. Evol. 3, 217–223 (2012).
Stekhoven, D. J. & Bühlmann, P. MissForest—non-parametric missing value imputation for mixed-type data. Bioinformatics 28, 112–118 (2011).
Carmona, C. P. et al. Fine-root traits in the global spectrum of plant form and function. Nature 597, 683–687 (2021).
Barajas Barbosa, M. P. et al. Assembly of functional diversity in an oceanic island flora. Nature 619, 545–550 (2023).
Penone, C. et al. Imputation of missing data in life-history trait datasets: which approach performs the best? Methods Ecol. Evol. 5, 961–970 (2014).
Jin, Y. & Qian, H. V. PhyloMaker: an R package that can generate very large phylogenies for vascular plants. Ecography 42, 1353–1359 (2019).
Smith, S. A. & Brown, J. W. Constructing a broadly inclusive seed plant phylogeny. Am. J. Bot. 105, 302–314 (2018).
de Vries, F. T. et al. Abiotic drivers and plant traits explain landscape-scale patterns in soil microbial communities. Ecol. Lett. 15, 1230–1239 (2012).
Revelle, W. psych: Procedures for Psychological, Psychometric, and Personality Research. R package version 2.4.12 (2024).
Grenié, M. & Gruson, H. fundiversity: a modular R package to compute functional diversity indices. Ecography 2023, e06585 (2023).
Laliberte, E. & Legendre, P. A distance-based framework for measuring functional diversity from multiple traits. Ecology 91, 299–305 (2010).
Pettorelli, N. et al. Using the satellite-derived NDVI to assess ecological responses to environmental change. Trends Ecol. Evol. 20, 503–510 (2005).
Gorelick, N. et al. Google Earth Engine: planetary-scale geospatial analysis for everyone. Remote Sens. Environ. 202, 18–27 (2017).
Roy, D. P. et al. Characterization of Landsat-7 to Landsat-8 reflective wavelength and normalized difference vegetation index continuity. Remote Sens. Environ. 185, 57–70 (2016).
Karger, D. N. et al. Climatologies at high resolution for the earth’s land surface areas. Sci. Data 4, 170122 (2017).
Bourgeois, C. F. et al. Four decades of data indicate that planted mangroves stored up to 75% of the carbon stocks found in intact mature stands. Sci. Adv. 10, eadk5430 (2024).
Aguirre-Gutiérrez, J. et al. Functional susceptibility of tropical forests to climate change. Nat. Ecol. Evol. 6, 878–889 (2022).
Pinheiro, J., Bates, D., DebRoy, S., Sarkar, D. & R Core Team. nlme: Linear and Nonlinear Mixed Effects Models. R package version 3.1-167 (2022).
Kuznetsova, A., Brockhoff, P. B. & Christensen, R. H. B. lmerTest package: tests in linear mixed effects models. J. Stat. Softw. 82, 1–26 (2017).
Barton, K. MuMIn: Multi-Model Inference. R package version 1.48.4 (2024).
Lefcheck, J. S. & Freckleton, R. piecewiseSEM: piecewise structural equation modelling in R for ecology, evolution, and systematics. Methods Ecol. Evol. 7, 573–579 (2016).
R Core Team. R: A Language and Environment for Statistical Computing (R Foundation for Statistical Computing, 2020).
Liu, H., Li, J., Wu, J., Li, B. & Nie, M. Large slow-growing hydrophytes increase wetland carbon storage. figshare https://doi.org/10.6084/m9.figshare.29097845 (2025).
Acknowledgements
The National Wetland Condition Assessment 2011 and 2016 data were a result of the collective efforts of dedicated field crews, laboratory staff, data management and quality control staff, analysts and many others from EPA, states, tribes, federal agencies, universities and other organizations. We thank the TRY initiative, the BIEN database, and their contributors and administrators. This work was supported by the National Key Research and Development Program of China (2023YFF1304504 to M.N.), the National Natural Science Foundation of China (32430065 to M.N., 32501442 to H.L., 32522064 and 32471831 to J.L.), the Shanghai Pilot Program for Basic Research–Fudan University 21TQ1400100 (21TQ004 to M.N.), the Shanghai Science and Technology Innovation Action Plan (23015810100 to B.L.), and the Science and Technology Plan Project of Shanghai (23DZ1202700 to J.L.).
Author information
Authors and Affiliations
Contributions
M.N. conceived of and designed the study. H.L. performed the analysis and drafted the paper with assistance from M.N. J.L., J.W., B.L. and M.N. contributed to the revisions of the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Plants thanks Jay Sah and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
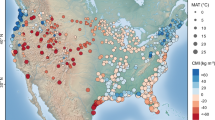
Extended Data Fig. 1 Spatial distribution and sampling design of NWCA wetland sites.
a Spatial distribution of 1,268 wetland sites surveyed during 2011 and 2016 by the NWCA, showing long-term averages of mean annual temperature (MAT) and monthly climate moisture index (CMI). b Sampling design, including a 40 m radius standard assessment area (AA) with five vegetation plots and a surrounding buffer area. Normalized difference vegetation index (NDVI) derived from nine Landsat pixels (3×3 grid) overlapping the AA.
Extended Data Fig. 2 A priori structural equation model used to assess the direct and indirect effects of climate, soil factors, functional diversity, and functional identity on wetland soil organic carbon stocks.
The variables considered in the models included functional diversity, functional identity, mean annual temperature (MAT), climate moisture index (CMI), water table depth (WTD), soil pH, productivity, root area density, and soil carbon to nitrogen (C/N) ratio. Functional identity was represented by the three principal components (PCs) from the PCA of the community-weighted means of trait values: CWMPC1 (representing plant resource economics), CWMPC2 (representing hydrophytic status), and CWMPC3 (representing plant size). Functional diversity (FD) was represented by functional dispersion. Proposed interpretations of the pathways (indexed by pathway number) are provided in Supplementary Table 3.
Extended Data Fig. 3 Structural equation models showing the effects of climate, water table depth, soil pH, functional diversity, and functional identity on the soil carbon stocks of the topsoil and subsoil layers.
a,c Path diagrams of factors influencing soil carbon stocks in the topsoil (a, n = 1,163) and subsoil (b, n = 784) layers. The final model with significant (P < 0.05, two-sided t-based Wald tests) pathways is shown, with black arrows representing positive relationships and red arrows representing negative relationships. The numbers on the arrows are standardized path coefficients, with arrow thickness proportional to their magnitude. b,d Standardized direct and indirect effects of each variable on soil carbon stocks in the topsoil (b) and subsoil (d) layers, ranked by the total effects. The goodness-of-fit statistics for the models for the topsoil and subsoil are as follows: Fisher’s C = 54.58, P = 0.132, and df = 44 and Fisher’s C = 47.13, P = 0.346, and df = 44, respectively, indicating a close model–data fit. MAT, mean annual temperature; CMI, climate moisture index; WTD, water table depth; Soil C/N, soil carbon to nitrogen ratio; and FD, functional diversity.
Extended Data Fig. 4 Environmental context-dependent responses of soil carbon stocks to functional diversity and functional identity (CWMPC1, CWMPC2, and CWMPC3).
Partial regression plots illustrating the significant interactive effects on soil carbon stocks across the entire soil profile (0–100 cm, a–c), topsoil layer (0–30 cm, d–i), and subsoil layer (30–100 cm, j–m), with bootstrapped 95% confidence intervals shaded in the corresponding colors, as fitted by the most parsimonious linear mixed-effects models (Supplementary Tables 5–7). F values, denominator degrees of freedom (df), and significance (two-sided P value) were obtained using Type III Wald F-tests with Satterthwaite’s method. For scaled MAT, CMI, WTD, soil Olsen P, and soil pH, the categories are defined as follows: low (one standard deviation (SD) below the mean, -1), moderate (mean value, 0), and high (one SD above the mean, 1). Sample sizes (n) are 1,221 (topsoil) and 814 (subsoil and entire) for MAT, CMI, WTD, and soil pH; and 1,213 (topsoil) and 810 (subsoil and entire) for soil Olsen P. For anthropogenic stressors, the colors represent low, moderate, and high levels of stress intensity. Sample sizes (n = low/moderate/high) are as follows: 629/280/305 (topsoil) and 394/197/221 (subsoil and entire) for STR_NNP; 354/465/395 (topsoil) and 226/315/271 (subsoil and entire) for STR_PALT; and 1,081/68/65 (topsoil) and 722/48/42 (subsoil and entire) for STR_HM. WTD, water table depth; Soil Olsen P, soil Olsen phosphorus; STR_NNP, non-native plants stress; STR_PALT, physical alteration stress; STR_HM, heavy metal stress; and FD, functional diversity.
Extended Data Fig. 5 Fixed effects of functional diversity and functional identity (CWMPC1, CWMPC2, and CWMPC3) on soil carbon stocks in the surface soil layer (0–10 cm).
The green dots represent the values predicted by partial regression with each explanatory variable (a), the black lines represent the mean values, and the shaded areas represent the 95% confidence intervals fitted by the linear mixed-effects model (full model, n = 1,257). F values, denominator degrees of freedom (df), and significance (two-sided P values) were obtained using Type III Wald F-tests with Satterthwaite’s method. The bar plots (b) are standardized estimates (mean and 95% confidence intervals) of predictor variable effects on soil carbon stocks derived from the most parsimonious model using two-sided t-based Wald tests. Marginal and conditional R2 values are given. MAT, mean annual temperature; CMI, climate moisture index; WTD, water table depth; Plant cover, total plant cover; and FD, functional diversity.
Extended Data Fig. 6 Correlations between species richness and functional diversity and functional identity (CWMPC1, CWMPC2, and CWMPC3).
The solid blue lines indicate ordinary least squares (OLS) linear regression fits (n = 1,268). The shaded bands represent the 95% confidence intervals of the regression predictions. R2 values and significance levels (P values, two-sided t-tests) are given. FD, functional diversity.
Extended Data Fig. 7 Environmentally mediated effects of functional diversity and functional identity (CWMPC1, CWMPC2, and CWMPC3) on productivity, root area density, and the soil C/N ratio.
Standardized coefficients from linear mixed-effects models showing the interactive effects of functional characteristics and different environmental factors on productivity, root area density, and the soil C/N ratio. The colors of the dots indicate different levels of environmental factors. For scaled MAT, CMI, WTD, soil Olsen P, and soil pH, the categories are defined as follows: low (one standard deviation (SD) below the mean, -1), moderate (mean value, 0), and high (one SD above the mean, 1). Sample sizes (n) are 1,257 (productivity), 1,171 (root area density), and 1,246 (soil C/N ratio) for MAT, CMI, WTD, and soil pH; and 1,249 (productivity), 1,163 (root area density), and 1,238 (soil C/N ratio) for soil Olsen P. For anthropogenic stressors, the colors represent low, moderate, and high levels of stress intensity. Sample sizes (n = low/moderate/high) are as follows: 655/285/310 (productivity), 597/275/294 (root area density), and 650/283/306 (soil C/N ratio) for STR_NNP; 364/480/406 (productivity), 333/447/386 (root area density), and 363/477/399 (soil C/N ratio) for STR_PALT; and 1,109/70/71 (productivity), 1,037/66/63 (root area density), and 1,098/70/71 (soil C/N ratio) for STR_HM. The mean values and 95% confidence intervals of the parameter estimate for each predictor are shown. Filled and empty symbols indicate significant (P < 0.05) and nonsignificant interactive effects between environmental factors and functional characteristics, respectively, based on two-sided Type III Wald F-tests with Satterthwaite’s method. MAT, mean annual temperature; CMI, climate moisture index; WTD, water table depth. Soil Olsen P, soil Olsen phosphorus; Soil C/N, soil carbon to nitrogen ratio; STR_NNP, non-native plant stress; STR_PALT, physical alteration stress; STR_HM, heavy metal stress; and FD, functional diversity.
Supplementary information
Supplementary Information (download PDF )
Supplementary Tables 1–8 and Figs. 1–4.
Source data
Source Data Figs. 1–4 and Extended Data Figs. 1, 3 and 4–7 (download XLSX )
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, H., Li, J., Wu, J. et al. Large slow-growing hydrophytes increase wetland carbon storage. Nat. Plants 12, 294–307 (2026). https://doi.org/10.1038/s41477-026-02221-y
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41477-026-02221-y