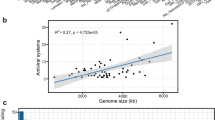
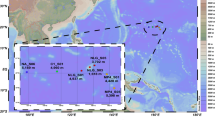
Abstract
Continental shelves are important areas for global biogeochemical cycling, yet the roles of sediment viruses in these areas remain poorly understood. Here, 48 surface sediment samples from the Chinese continental shelf were analyzed, and 12,540 viral operational taxonomic units (vOTUs) were identified. Taxonomic classification found that 93.6% of the vOTUs could not be assigned at the family level, and protein-sharing networks showed that 60.4% were singletons. Viral community structure was shaped primarily by temperature and water depth. A total of 557 auxiliary metabolic genes (AMGs) were identified, including those involved in sulfur reduction and phosphorus acquisition (Pho-family proteins). Additionally, diverse antibiotic resistance genes (ARGs) were detected, suggesting anthropogenic influence. This study reveals the diversity, ecological function, and environmental drivers of viral communities in continental shelf sediments, providing new insights into viral contributions to microbial ecology and biogeochemical processes.
Similar content being viewed by others
Data availability
The raw sequence data reported in this paper have been deposited in the Genome Sequence Archive in the National Genomics Data Center, China National Center for Bioinformation/Beijing Institute of Genomics67,68, Chinese Academy of Sciences (GSA: CRA020828), which are publicly accessible at https://ngdc.cncb.ac.cn/gsa/browse/CRA020828.
Code availability
All computational tools used in this study are publicly available through the referenced publications or their associated GitHub repositories. The custom Perl script used for quality filtering based on Phred scores (Q20 and Q30 thresholds) is provided in Supplementary Note 1. No other custom code was developed.
References
Borges, A. V. Present day carbon dioxide fluxes in the coastal ocean and possible feedbacks under global change. In Oceans and the Atmospheric Carbon Content (Duarte, P. & Santana-Casiano, J. M. eds) 47–77 (Springer Netherlands, Dordrecht, 2011).
Shi, X. et al. Distribution, burial fluxes and carbon sink effect of sedimentary organic carbon in the eastern China seas. Sci. China Earth Sci. 67, 3062–3082 (2024).
Lalzar, M. et al. Sediment microbiota as a proxy of environmental health: discovering inter- and intrakingdom dynamics along the Eastern Mediterranean Continental Shelf. Microbiol. Spectr. 11, e02242-22 (2023).
Breitbart, M., Bonnain, C., Malki, K. & Sawaya, N. A. Phage puppet masters of the marine microbial realm. Nat. Microbiol. 3, 754–766 (2018).
Coutinho, F. H., Gregoracci, G. B., Walter, J. M., Thompson, C. C. & Thompson, F. L. Metagenomics sheds light on the ecology of marine microbes and their viruses. Trends Microbiol. 26, 955–965 (2018).
Zhou, Y. et al. Variations of water transparency and impact factors in the Bohai and Yellow Seas from satellite observations. Remote Sens. 13, 514 (2021).
Wang, B., Xin, M., Wei, Q. & Xie, L. A historical overview of coastal eutrophication in the China Seas. Mar. Pollut. Bull. 136, 394–400 (2018).
Zhang, Y., Lu, J., Wu, J., Wang, J. & Lin, Y. Occurrence and distribution of antibiotic resistance genes in sediments in a semi-enclosed continental shelf sea. Sci. Total Environ. 720, 137712 (2020).
McConnell, M. M. et al. Removal of antibiotic resistance genes in two tertiary level municipal wastewater treatment plants. Sci. Total Environ. 643, 292–300 (2018).
Moon, K. et al. Freshwater viral metagenome reveals novel and functional phage-borne antibiotic resistance genes. Microbiome 8, 75 (2020).
Yu, S. X., Pang, Y. L., Wang, Y. C., Li, J. L. & Qin, S. Distribution of bacterial communities along the spatial and environmental gradients from Bohai Sea to northern Yellow Sea. PeerJ 6, e4272 (2018).
Huang, D., Zhang, Z., Sun, M., Feng, Z. & Ye, M. Characterization and ecological function of bacterial communities in seabed sediments of the southwestern Yellow Sea and northwestern East China Sea, Western Pacific. Sci. Total Environ. 761, 143233 (2021).
Yu, M. et al. Diversity and potential host-interactions of viruses inhabiting deep-sea seamount sediments. Nat. Commun. 15, 3228 (2024).
Li, Z. et al. Deep sea sediments associated with cold seeps are a subsurface reservoir of viral diversity. ISME J. 15, 2366–2378 (2021).
Guo, J. et al. VirSorter2: a multi-classifier, expert-guided approach to detect diverse DNA and RNA viruses. Microbiome 9, 37 (2021).
Ren, J. et al. Identifying viruses from metagenomic data using deep learning. Quant. Biol. 8, 64–77 (2020).
Kieft, K., Zhou, Z. & Anantharaman, K. VIBRANT: automated recovery, annotation and curation of microbial viruses, and evaluation of viral community function from genomic sequences. Microbiome 8, 90 (2020).
Chen, L. & Banfield, J. F. COBRA improves the completeness and contiguity of viral genomes assembled from metagenomes. Nat. Microbiol. 9, 737–750 (2024).
Nayfach, S. et al. CheckV assesses the quality and completeness of metagenome-assembled viral genomes. Nat. Biotechnol. 39, 578–585 (2021).
Bin Jang, H. et al. Taxonomic assignment of uncultivated prokaryotic virus genomes is enabled by gene-sharing networks. Nat. Biotechnol. 37, 632–639 (2019).
Camargo, A. P. et al. IMG/VR v4: an expanded database of uncultivated virus genomes within a framework of extensive functional, taxonomic, and ecological metadata. Nucleic Acids Res. 51, D733–D743 (2023).
Zheng, K. et al. VITAP: a high precision tool for DNA and RNA viral classification based on meta-omic data [Internet]. https://www.researchsquare.com/article/rs-4406120/v1 (2024).
Camargo, A. P. et al. Identification of mobile genetic elements with geNomad. Nat. Biotechnol. 42, 1303–1312 (2024).
Hamdan, L. J. et al. Ocean currents shape the microbiome of Arctic marine sediments. ISME J. 7, 685–696 (2013).
Fei, Y., Qiang, R., Xinyuan, D., Chuanjie, W. & Yibo, H. The sandwich structure of the Southern Yellow Sea cold water mass and Yellow Sea warm current. Front. Mar. Sci. 8, 767850 (2022).
Liu, Z. et al. Progress of studies on circulation dynamics in the East China Sea: The Kuroshio exchanges with the shelf currents. Front. Mar. Sci. 8, 620910 (2021).
Welander, P. Thermohaline effects in the ocean circulation and related simple models. In Large-Scale Transport Processes in Oceans and Atmosphere (Willebrand, J & Anderson, DLT eds) 163–200 (Springer Netherlands, Dordrecht, 1986).
Uritskiy, G. V., DiRuggiero, J. & Taylor, J. MetaWRAP—a flexible pipeline for genome-resolved metagenomic data analysis. Microbiome 6, 158 (2018).
Grubba, D. & Majtacz, J. The influence of sulfate on anaerobic ammonium oxidation in a sequencing batch reactor. Water 12, 3004 (2020).
Chaumeil, P. A., Mussig, A. J., Hugenholtz, P. & Parks, D. H. GTDB-Tk: a toolkit to classify genomes with the Genome Taxonomy Database. Bioinformatics 36, 1925–1927 (2020).
Suttle, C. A. Marine viruses — major players in the global ecosystem. Nat. Rev. Microbiol. 5, 801–812 (2007).
Baker, B. J., Lazar, C. S., Teske, A. P. & Dick, G. J. Genomic resolution of linkages in carbon, nitrogen, and sulfur cycling among widespread estuary sediment bacteria. Microbiome 3, 14 (2015).
Bao, P. et al. The role of sulfate-reducing prokaryotes in the coupling of element biogeochemical cycling. Sci. Total Environ. 613-614, 398–408 (2018).
Zhou, Z. et al. METABOLIC: high-throughput profiling of microbial genomes for functional traits, metabolism, biogeochemistry, and community-scale functional networks. Microbiome 10, 33 (2022).
Guessous, G. et al. Inherited chitinases enable sustained growth and rapid dispersal of bacteria from chitin particles. Nat. Microbiol. 8, 1695–1705 (2023).
Ali, M., Zubair, M., Rosyidi, A. & Amin, M. Screening of ammonia-degrading bacteria to reduce ammonia content in the manure of laying hens. IOP Conf. Ser. Earth Environ. Sci. 441, 012138 (2020).
Dyksma, S., Lenk, S., Sawicka, J. E. & Mußmann, M. Uncultured gammaproteobacteria and desulfobacteraceae account for major acetate assimilation in a coastal marine sediment. Front. Microbiol. 9, 3124 (2018).
Zimmerman, A. E. et al. Metabolic and biogeochemical consequences of viral infection in aquatic ecosystems. Nat. Rev. Microbiol. 18, 21–34 (2020).
Tian, F. et al. Prokaryotic-virus-encoded auxiliary metabolic genes throughout the global oceans. Microbiome 12, 159 (2024).
Epstein, G. et al. Predictive mapping of organic carbon stocks in surficial sediments of the Canadian continental margin. Earth Syst. Sci. Data 16, 2165–2195 (2024).
Kohlmeier, M. G., White, C. E., Fowler, J. E., Finan, T. M. & Oresnik, I. J. Galactitol catabolism in Sinorhizobium meliloti is dependent on a chromosomally encoded sorbitol dehydrogenase and a pSymB-encoded operon necessary for tagatose catabolism. Mol. Genet. Genomics 294, 739–755 (2019).
Grimek, T. L., Holden, H., Rayment, I. & Escalante-Semerena, J. C. Residues C123 and D58 of the 2-methylisocitrate lyase (PrpB) enzyme of Salmonella enterica are essential for catalysis. J. Bacteriol. 185, 4837–4843 (2003).
Gazitúa, M. C. et al. Potential virus-mediated nitrogen cycling in oxygen-depleted oceanic waters. ISME J. 15, 981–998 (2021).
Simon, J., Eichler, R., Pisa, R., Biel, S. & Gross, R. Modification of heme c binding motifs in the small subunit (NrfH) of the Wolinella succinogenes cytochrome c nitrite reductase complex. FEBS Lett. 522, 83–87 (2002).
Heiss, B. & Frunzke, K. Formation of the N-N Bond from nitric oxide by a membrane-bound cytochrome bc complex of nitrate-respiring (denitrifying) Pseudomonas stutzerit. J. Bacteriol. 171, 3288-3297 (1989).
Vu, H. N. & Downs, D. M. An Unexpected role for the periplasmic phosphatase PhoN in the salvage of B 6 vitamers in Salmonella enterica. Appl. Environ. Microbiol 87, e02300–e02320 (2021).
Feehily, C. & Karatzas, K. A. G. Role of glutamate metabolism in bacterial responses towards acid and other stresses. J. Appl. Microbiol. 114, 11–24 (2013).
Alcock, B. P. et al. CARD 2023: expanded curation, support for machine learning, and resistome prediction at the Comprehensive Antibiotic Resistance Database. Nucleic Acids Res. 51, D690–D699 (2023).
Imchen, M. & Kumavath, R. Metagenomic insights into the antibiotic resistome of mangrove sediments and their association to socioeconomic status. Environ. Pollut. 268, 115795 (2021).
Polianciuc, S. I., Gurzău, A. E., Kiss, B., Ștefan, M. G. & Loghin, F. Antibiotics in the environment: causes and consequences. Med. Pharm. Rep. https://medpharmareports.com/index.php/mpr/article/view/1742 (2020).
Zhang, R. et al. Antibiotics in the offshore waters of the Bohai Sea and the Yellow Sea in China: occurrence, distribution and ecological risks. Environ. Pollut. 174, 71–77 (2013).
Yang, Y. et al. Antibiotic use in China’s public healthcare institutions during the COVID-19 pandemic: an analysis of nationwide procurement data, 2018–2020. Front. Pharmacol. 13, 813213 (2022).
Su, H.-C. et al. Wastewater discharge drives ARGs spread in the coastal area: a case study in Hangzhou Bay, China. Mar. Pollut. Bull. 149, 110856, https://doi.org/10.1016/j.marpolbul.2019.110856 (2019).
Chen, Y. et al. Land-derived wastewater facilitates antibiotic resistance contamination in marine sediment of semi-closed bay: a case study in Jiaozhou Bay, China. J. Environ. Manag. 339, 117870 (2023).
Chen, S., Zhou, Y., Chen, Y. & Gu, J. fastp: an ultra-fast all-in-one FASTQ preprocessor. Bioinformatics 34, i884–i890 (2018).
Li, D., Liu, C. M., Luo, R., Sadakane, K. & Lam, T. W. MEGAHIT: an ultra-fast single-node solution for large and complex metagenomics assembly via succinct de Bruijn graph. Bioinformatics 31, 1674–1676 (2015).
Olm, M. R., Brown, C. T., Brooks, B. & Banfield, J. F. dRep: a tool for fast and accurate genomic comparisons that enables improved genome recovery from metagenomes through de-replication. ISME J. 11, 2864–2868 (2017).
Gruber-Vodicka HR, & Seah B. K. B., Pruesse E. phyloFlash: rapid SSU rRNA profiling and targeted assembly from metagenomes. mSystems 5, e00920 (2020).
Steinegger, M. & Söding, J. MMseqs2 enables sensitive protein sequence searching for the analysis of massive data sets. Nat. Biotechnol. 35, 1026–1028 (2017).
Hyatt, D., LoCascio, P. F., Hauser, L. J. & Uberbacher, E. C. Gene and translation initiation site prediction in metagenomic sequences. Bioinformatics 28, 2223–2230 (2012).
Ahlgren, N. A., Ren, J., Lu, Y. Y., Fuhrman, J. A. & Sun, F. Alignment-free d2∗ oligonucleotide frequency dissimilarity measure improves prediction of hosts from metagenomically-derived viral sequences. Nucleic Acids Res. 45, 39–53 (2017).
Gregory, A. C. et al. Marine DNA viral macro- and microdiversity from pole to pole. Cell 180, 1109–1123.e14 (2020).
Mihara, T. et al. Linking virus genomes with host taxonomy. Viruses 8, 66 (2016).
Edwards, R. A. et al. Computational approaches to predict bacteriophage–host relationships. FEMS Microbiol. Rev. 40, 258–272 (2016).
Roux, S. et al. Ecogenomics and potential biogeochemical impacts of globally abundant ocean viruses. Nature 537, 689–693 (2019).
Sunagawa, S. et al. Structure and function of the global ocean microbiome. Science 348, 1261359 (2015).
CNCB-NGDC Members and Partners. Database Resources of the National Genomics Data Center, China National Center for Bioinformation in 2022. Nucleic Acids Res. 50, D27–D38 (2022).
Chen, T. et al. The genome sequence archive family: toward explosive data growth and diverse data types. Genomics Proteom. Bioinform. 19, 578–583 (2021).
Acknowledgements
We thank for the data and samples collected aboard the R/V Lanhai101 during the open research cruise: NORC2020-01 and the R/V Xiangyanghong 18 during the open research cruise: NORC2021-02+NORC2021-301. We thank the support of the Marine Big Data Center of the Institute for Advanced Ocean Study of Ocean University of China, the high-performance server of the Center for High Performance Computing and System Simulation, Laoshan Laboratory (Qingdao). We appreciate the computing resources provided by IEMB-1, a high-performance computation cluster operated by the Institute of Evolution and Marine Biodiversity and the High-Performance Biological Supercomputing Center at the Ocean University of China. This work was supported by the Natural Science Foundation of China (No. 42120104006, 42176111, 42306156, 41949901, and 42176149), the Ocean Negative Carbon Emissions (ONCE), and the Fundamental Research Funds for the Central Universities (202172002, 201812002, and 202072001) and Andrew McMinn.
Author information
Authors and Affiliations
Contributions
Y.L., A.M., and M.W.: conceptualization, revision, project administration, supervision, and funding acquisition. Jinhao Shen, H.Y., Y.S., Jingwen Sun, and K.S.: methodology, formal analysis, writing, and original draft preparation.Jinhao Shen and X.C.: sample collection and expedition organization.Jingwen Sun, H.Y., Y.S., M.H., H.S., and Y.Y.: software, validation, and visualization. M.W., Y.L., A.M., J.S., and M.H.: review and editing. All authors contributed to the article and approved the submitted version.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Shen, J., Han, M., Sun, J. et al. Diversity and ecological potential of sediment viruses from Chinese continental shelf seas. npj Biofilms Microbiomes (2026). https://doi.org/10.1038/s41522-026-00940-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41522-026-00940-w