Abstract
Adding immune checkpoint inhibitors to neoadjuvant chemotherapy improves outcomes in early-stage triple-negative breast cancer (TNBC), but a fraction of patients derive benefit. Tumor-specific MHC-II (tsMHC-II) expression has been shown to be a predictive biomarker of pathological complete response (pCR) to neoadjuvant chemo-immunotherapy in early-stage TNBC. We performed biomarker analysis of the phase III NeoTRIP trial where patients were randomized to neoadjuvant carboplatin and nab-paclitaxel±atezolizumab. Imaging mass cytometry was used to assess tsMHC-II expression in tumor samples. TsMHC-II positivity was predefined as ≥5% of tumor cells expressing MHC-II, and at an 80th percentile exploratory cutoff. TsMHC-II positivity was associated with a higher pCR rate in the atezolizumab arm (OR:2.58; P = 0.016), but not in the chemotherapy-only arm (OR:1.37; P = 0.34) and these results were stronger using the exploratory cutoff. TsMHC-II expression is associated with improved response to neoadjuvant chemo-immunotherapy in early TNBC and could represent a clinically useful predictive biomarker for treatment personalization.
Similar content being viewed by others
Introduction
The anti-PD-1 monoclonal antibody pembrolizumab, an immune checkpoint inhibitor (ICI), is approved for treatment of high-risk early triple-negative breast cancer (TNBC)1,2 in combination with chemotherapy and broadly recognized as standard of care. However, less than 10% of the patients derive individual benefit from the addition of pembrolizumab to neoadjuvant chemotherapy (NAC). ICIs can cause chronic immune related adverse events, including severe or even fatal toxicities such as myocarditis, pneumonitis and encephalitis3. Therefore, biomarkers that can identify patients that benefit from the addition of ICIs are needed4.
Despite various promising biomarkers, none have been validated to demonstrate clinical utility in early stage TNBC5,. Tumor-specific major histocompatibility complex-II (tsMHC-II) has been identified as a potential biomarker for PD-1/PD-L1 targeted immunotherapy in other cancer types including melanoma6,7, Hodgkin lymphoma8 and bladder cancer9. MHC-II has multiple isotypes: HLA-DR, HLA-DP, and HLA-DQ, each with alpha and beta subunits that form heterodimers. MHC-II is constitutively expressed on the surface of antigen presenting cells and loaded with exogenously derived peptides to be presented to CD4 + T-cells to regulate immune responses. However, MHC-II can also be expressed on normal and cancer cells including breast epithelial cells and tumors10, although its function in this setting is not well-established.
TsMHC-II has been independently identified as a predictor of molecular response to single-agent anti-PD-1 in a small window trial in the early breast cancer setting using a biomarker screening approach11. TsMHC-II expression was assessed retrospectively on samples from patients with high-risk early-stage HR + HER2- and TNBC that participated in one of two Phase II clinical trials evaluating the addition of pembrolizumab12 or durvalumab13 to NAC. Using a pre-defined cutoff of ≥5% of tumor cells by immunohistochemistry, or a harmonized equivalent cutoff by an orthogonal method (reverse phase protein array/RPPA), tsMHC-II was confirmed to be predictive of pCR and event-free survival (EFS) to PD-1/PD-L1 inhibitors in addition to NAC but not to NAC alone14. Here, using the pre-defined 5% tumor positivity cutoff we tested the predictive value of tsMHC-II to predict pCR in the Phase III NeoTRIPaPDL1 trial15.
Results
Tumor-specific MHC-II expression predicts ICI benefit
A total of 220 FFPE tissues at baseline were available for the present analysis: 109 in the chemotherapy arm and 111 in the chemotherapy plus immunotherapy arm. Using the predefined cutoff of 5% tumor-cell positivity, we found that 41% of all breast cancers assessed expressed ≥5% MHC-II, with no significant differences between treatment arms (42.2% in the chemotherapy alone group and 40.5% in the chemotherapy plus immunotherapy group, respectively; p = 0.68) (Table 1).
The application of this cutoff point demonstrated predictive capacity for pCR in the immunotherapy-treated patients (one-tailed chi-square test, p = 0.016, OR: 2.58) but not in the chemotherapy alone arm (p = 0.336, OR: 1.45). The test for interaction between MHC-II status and treatment arm on pCR was not significant (p = 0.292) (Fig. 1A).
Association of tumor-specific MHC-II expression with outcome to atezolizumab and chemotherapy or chemotherapy alone using the cutoffs of 5% (A) and the 80th percentile (B) tumor-cell positivity. C tsMHC-II measured continuously in treatment/response arms.
Since the predefined 5% cutoff identified significantly more positive patients using IMC than expected with immunofluorescence or the reverse phase protein array quantification equivalent (15–20%)14, we conducted an exploratory analysis to test the association between tsMHC-II status and pCR rates for each treatment, using the cutoff of the 80th percentile of tumor-cell positivity, to mirror a similar split proportion. With this cutoff point, we found that 17.4% and 22.5% of breast cancers in the chemotherapy alone and chemotherapy plus immunotherapy arms, respectively, were tsMHC-II+ (p = 0.35) (Table 1). TsMHC-II status continued to demonstrate its predictive capacity for pCR in the immunotherapy-treated patients (p = 0.006, OR: 4.19), but not in the chemotherapy alone arm (p = 0.956, OR: 1.03). Moreover, when testing the interaction between MHC-II status using this cutoff and treatment arm on pCR, we observed a trend toward significance (p = 0.052) (Fig. 1B). Measuring tsMHC-II continuously demonstrated a statistically significant interaction between treatment arms and outcome groups (p = 0.006) (Fig. 1C). Receiver-operator characteristic analysis also demonstrated an improved area-under-the-curve across cut points in the chemo-immunotherapy arm over chemotherapy alone (Supplementary Fig. 1).
Discussion
Here, we have demonstrated continued support for the predictive value of tsMHC-II to identify an early-stage TNBC patient population who seems to benefit the most from immunotherapy, in the context of a randomized, controlled Phase III trial. While the antibody used for MHC-II detection in situ is consistent with prior studies, the IMC method utilized herein is more sensitive that traditional IHC, immunofluorescence, and RPPA methodologies used in previous analyses6,14. Thus, a modified cut-off identifying a similar percentage of patients as ‘positive’ for the biomarker (20%) by IHC was also tested. Both analyses demonstrated the clinical validity of tsMHC-II as predictive biomarker of immunotherapy benefit in this setting. The planned IMC approach for assessing multiple parameters, including MHC-II, preceded the reporting of tsMHC-II as an immunotherapy biomarker in breast cancer, and remaining tissue resources were too limited to reasonably conduct direct IHC on the cohort, and thus this analysis can be considered an “analysis of convenience”.
The results from this study, and prior validations of tsMHC-II, suggest its utility as a specific predictor of immunotherapy benefit in early-stage TNBC. This is in contrast to stromal TILs and PD-L1, which have instead shown clear prognostic value or predictive value to chemotherapy16,17,18,19,20,21,22,23, and have shown lack of utility in selecting immunotherapy-specific benefit in randomized controlled trials1,24,25. Studies suggesting otherwise often lack a chemotherapy-backbone control, making such an assessment difficult to parse from prognostic or broadly predictive effects.
As additional limitations, atezolizumab and the chemotherapy regimen used in the NeoTRIP trial are different from the current approved regimen, and EFS is not available yet for translational studies. However, the consistency of these results support this as a promising biomarker with potential clinical utility to be demonstrated in correlative analyses of ongoing clinical trials. TsMHC-II is currently under evaluation as a planned integrated biomarker on multiple prospective phase III clinical trials evaluating the addition of pembrolizumab to chemotherapy in the adjuvant setting for patients with TNBC (S1418; NCT02954874) and in the neoadjuvant settings (S2206; NCT06058377) for patients with high-risk ER+ early breast cancer. We are continuing to work on harmonizing tsMHC-II assays to define the most practical approach that can be prospectively validated in a pre-planned and defined fashion to demonstrate true clinical utility, with careful consideration of establishing the most widely-available assay that can be performed in the most technically rigorous way.
Methods
Study design and prospective tissue collection
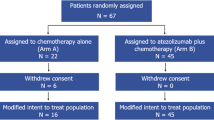
Breast tumor samples were obtained from patients enrolled in the multi-center, randomized, open-label, phase III NeoTRIP trial15. In NeoTRIP, 280 patients with early high-risk TNBC were randomized to receive eight cycles of neoadjuvant carboplatin and nab-paclitaxel on days 1 and 8 every 3 weeks, with or without atezolizumab on day 1. Following neoadjuvant therapy, patients underwent surgery, to be followed by four cycles of post-surgery anthracyclines at the treating clinician’s discretion. The Per-Protocol Population (n = 258) was used for all translational studies. Core biopsies for research were obtained at baseline, after one cycle of therapy, and at surgery. Only baseline biopsies were used for the present analysis. The study was undertaken in accordance with Good Clinical Practice guidelines and the Declaration of Helsinki. All patients provided written informed consent. Approvals for the study protocol (and any modifications thereof) were obtained from independent ethics committees at each participating institution and relevant competent authorities. The following institutions and ethics committees participated: Brustgesundheitzentrum Tirol, Univ. Frauenklinik Innsbruck, Innsbruck, Austria 6020; Universitätsklinik für Innere Medizin III, mit Hämatologie, internistischer Onkologie, Hämostaseologie, Infektiologie, Rheumatologie und Onkologisches Zentrum Salzburg, Austria 5020; Klinikum Augsburg International Patient Service Augsburg, Germany, 86156; Frauenarzt-Zentrum-Zehlendorf, Berlin, Germany, 14169;Augusta-Kranken-Anstalt gGmbH Klinik für Hämatologie, Onkologie & Palliativmedizin, Bochum, Germany, 447891;Bethanien-Krankenhaus Onkologisches Zentrum, Frankfurt, Germany, 60389;Markus Krankenhaus Klinik für Gynäkologie und Geburtshilfe, Frankfurt, Germany, 60431;Gynäkologisch-Onkologische Praxis, Hannover, Germany, 30177;NCT Nationales Centrum für Tumorerkrankungen, Heidelberg, Germany, 69120;Uniklinik Köln Klinik und Poliklinic für Frauenheilkunde und Geburtshilfe Brestzentrum, Köln, Germany, 50931;Brustzentrum St. Elisabeth-Krankenhaus, Köln, Germany, 50935;Interdisciplinary Oncology Center (IOZ), Munchen, Germany, 80336;Cork University Hospital, Cork, Ireland;Beaumont Hospital, Dublin, Ireland;Mater Misericordiae University Hospital, Dublin, Ireland;St. James’s Hospital, Dublin, Ireland;University Hospital Waterford, Waterford, Ireland;Policlinico S. Orsola Malpoghi, Bologna, Italy, 40138;Istituto per la Ricerca sul Cancro, Candiolo, Italy, 10060;IST San Martino, Genova, Italy, 16132;Istituto Toscano Tumori Ospedale Misericordia, Grosseto, Italy, 58100;Ospedale San Raffaele, Milano, Italy, 20132;Fondazione IRCCS Istituto nazionale dei Tumori, Milano, Italy, 20133;Istituto Europeo di Oncologia, Milano, Italy, 20141;Ospedale Luigi Sacco, Milano, Italy, 20160;Arcispedale Santa Maria Nuova - A.O. Reggio Emilia, Reggio Emilia, Italy, 42123;Ospedale Santa Maria della Misericordia, Udine, Italy, 33100;Russian Cancer Research Center named after N.N.Blokhin, Moscow, Russian Federation; Petrov Research Institute of Oncology, Department of Breast Cancer, Saint Petersburg, Russian Federation;Road clinical hospital of OJSC “Russian Railways”, Saint Petersburg, Russian Federation;Hospital Duran i Reynal Institut Català d’Oncologia, Hospitalet de Llobregat, Spain, 08908; Hospital Clínico San Carlos, Madrid, Spain, 28040; Hospital Universitario 12 de octubre, Madrid, Spain, 28041; Hospital Universitario HM Sanchinarro, Centro Integral Oncologico Clara Campal (CIOCC), Madrid, Spain, 28050; Hospital Clinico Universitario de Valencia Servicio de Onco-Hematologia, Valencia, Spain, 46010; Hospital Miguel Servet Zaragoza, Spain, 59009; C. Christian Hospital Taiwan, Changhua City, Taiwan; Kaohsiung Medical University Hospital, Kaohsiung, Taiwan; China Medical University Hospital No.2, Taichung City, Taiwan; National Taiwan University Hospital, Taipei, Taiwan;Veteran General Hospital Taipei, Taipei, Taiwan
Imaging mass cytometry
Imaging mass cytometry (IMC) was used to profile the expression of 43 proteins at subcellular resolution in formalin-fixed paraffin-embedded (FFPE) tumor samples. Detailed protocols of IMC analysis have been reported elsewhere26. Briefly, three ROIs measuring 500 × 500 μm2 were identified in core biopsies by a breast pathologist using the Aperio eSlideManager web application (Leica Biosystems).
Cell phenotypes were assigned by semi-supervised clustering. Cells were first classified as epithelial or tumor microenvironment (TME) cells using multiple classification methods. Thresholds were identified for assigning a cell as ‘positive’ for a given marker by inspecting a random selection of at least 50 images for which cells passing a quantile threshold (calculated using all data) were highlighted. This procedure was repeated at differing quantile thresholds until the value that most closely aligned with marker positivity was identified. For this study, MHC-II-positive tumor cells were identified as the cells expressing the HLA-DR antigen, detected by using the anti-mouse monoclonal antibody TAL1B5 (Abcam) at concentration of 0.5 μg/mL. Although TAL1B5 is labeled to recognize only HLA-DR, prior studies by our group have shown extremely high concordance across tumors with pan-MHC-II (HLA-DR/DP/DQ/DX) antibodies6, and the TAL1B5 antibody is widely used and provides extremely robust signals at high dilutions. Moreover, MHC-II gene products are widely accepted to be co-regulated by CIITA activation, and therefore are nearly always expressed coordinately with one another, albeit at different levels27.
Statistical analysis
We aimed to evaluate whether the proportion of tumor cells with positive MHC-II status predicts the benefit of adding immunotherapy to NAC. We evaluated pCR rates15 as outcome measure, because the EFS results for the NeoTRIP trial have not been published yet. Consistently with previous studies, MHC-II status was initially defined according to a cutoff of 5% tumor cell-positivity14. A modified cutoff was also tested to account for the differences in detection method sensitivity (imaging mass cytometry versus IHC) and antibodies. Association between MHC-II status and pCR rates for each treatment arm and test of interaction were assessed using logistic regression. The statistical analyses included herein were not preplanned per study protocol but were planned as exploratory correlative endpoints. Statistical analyses were performed in R version 4.2.1.
Data availability
All imaging mass cytometry and clinical response data can be accessed via a Zenodo data repository (https://doi.org/10.5281/zenodo.7990870) for academic non-commercial research.
Code availability
Not applicable.
References
Schmid, P. et al. Pembrolizumab for early triple-negative breast cancer. N. Engl. J. Med. 382, 810–821 (2020).
Schmid, P. et al. Event-free survival with pembrolizumab in early triple-negative breast cancer. N. Engl. J. Med. 386, 556–567 (2022).
Johnson, D. B., Nebhan, C. A., Moslehi, J. J. & Balko, J. M. Immune-checkpoint inhibitors: Long-term implications of toxicity. Nat. Rev. Clin. Oncol. 19, 254–267 (2022).
Bianchini, G., De Angelis, C., Licata, L. & Gianni, L. Treatment landscape of triple-negative breast cancer - expanded options, evolving needs. Nat. Rev. Clin. Oncol. 19, 91–113 (2022).
Licata, L. et al. Tissue- and liquid biopsy-based biomarkers for immunotherapy in breast cancer. Breast 69, 330–341 (2023).
Johnson, D. B. et al. Melanoma-specific MHC-II expression represents a tumour-autonomous phenotype and predicts response to anti-PD-1/PD-L1 therapy. Nat. Commun. 7, 10582 (2016).
Rodig, S. J. et al. MHC proteins confer differential sensitivity to CTLA-4 and PD-1 blockade in untreated metastatic melanoma. Sci. Transl. Med. 10 https://doi.org/10.1126/scitranslmed.aar3342 (2018).
Roemer, M. G. et al. Major histocompatibility complex class II and programmed death ligand 1 expression predict outcome after programmed death 1 blockade in classic Hodgkin lymphoma. J. Clin. Oncol. 36, 942 (2018).
Yi, R. et al. MHC-II signature correlates with anti-tumor immunity and predicts anti-PD-L1 response of bladder cancer. Front. Cell Dev. Biol. 10 (2022).
Axelrod, M. L., Cook, R. S., Johnson, D. B. & Balko, J. M. Biological consequences of MHC-II expression by tumor cells in cancer. Clin. Cancer Res. J. Am. Assoc. Cancer Res. 25, 2392–2402 (2019).
Bassez, A. et al. A single-cell map of intratumoral changes during anti-PD1 treatment of patients with breast cancer. Nat. Med. 27, 820–832 (2021).
Nanda, R. et al. Effect of pembrolizumab plus neoadjuvant chemotherapy on pathologic complete response in women with early-stage breast cancer: An analysis of the ongoing phase 2 adaptively randomized I-SPY2 trial. JAMA Oncol. 6, 676–684 (2020).
Foldi, J. et al. Neoadjuvant durvalumab plus weekly nab-paclitaxel and dose-dense doxorubicin/cyclophosphamide in triple-negative breast cancer. NPJ Breast Cancer 7, 1–7 (2021).
Gonzalez-Ericsson, P. I. et al. Tumor-specific major histocompatibility-II expression predicts benefit to anti–PD-1/L1 therapy in patients with HER2-negative primary breast cancer. Clin. Cancer Res. 27, 5299–5306 (2021).
Gianni, L. et al. Pathologic complete response (pCR) to neoadjuvant treatment with or without atezolizumab in triple-negative, early high-risk and locally advanced breast cancer: NeoTRIP Michelangelo randomized study. Ann. Oncol. 33, 534–543 (2022).
Russo, L. et al. Locally advanced breast cancer: Tumor-infiltrating lymphocytes as a predictive factor of response to neoadjuvant chemotherapy. Eur. J. Surg. Oncol. 45, 963–968 (2019).
Luen, S. J. et al. Prognostic implications of residual disease tumor-infiltrating lymphocytes and residual cancer burden in triple-negative breast cancer patients after neoadjuvant chemotherapy. Ann. Oncol. J. Eur. Soc. Med. Oncol. 30, 236–242 (2019).
Loi, S. et al. Tumor-infiltrating lymphocytes and prognosis: A pooled individual patient analysis of early-stage triple-negative breast cancers. J. Clin. Oncol. J. Am. Soc. Clin. Oncol. 37, 559–569 (2019).
Denkert, C. et al. Tumour-infiltrating lymphocytes and prognosis in different subtypes of breast cancer: a pooled analysis of 3771 patients treated with neoadjuvant therapy. Lancet Oncol. 19, 40–50 (2018).
Denkert, C. et al. Tumor-infiltrating lymphocytes and response to neoadjuvant chemotherapy with or without carboplatin in human epidermal growth factor receptor 2-positive and triple-negative primary breast cancers. J. Clin. Oncol. J. Am. Soc. Clin. Oncol. 33, 983–991 (2015).
Wimberly, H. et al. PD-L1 Expression correlates with tumor-infiltrating lymphocytes and response to neoadjuvant chemotherapy in breast cancer. Cancer Immunol Res. https://doi.org/10.1158/2326-6066.CIR-14-0133 (2014).
Loi, S. et al. Tumor infiltrating lymphocytes are prognostic in triple negative breast cancer and predictive for trastuzumab benefit in early breast cancer: results from the FinHER trial. Ann. Oncol. 25, 1544–1550 (2014).
Adams, S. et al. Prognostic value of tumor-infiltrating lymphocytes in triple-negative breast cancers from two phase III randomized adjuvant breast cancer trials: ECOG 2197 and ECOG 1199. J. Clin. Oncol. [pii]https://doi.org/10.1200/JCO.2013.55.0491 (2014).
Mittendorf, E. A. et al. Neoadjuvant atezolizumab in combination with sequential nab-paclitaxel and anthracycline-based chemotherapy versus placebo and chemotherapy in patients with early-stage triple-negative breast cancer (IMpassion031): a randomised, double-blind, phase 3 trial. Lancet 396, 1090–1100 (2020).
Loibl, S. et al. A randomised phase II study investigating durvalumab in addition to an anthracycline taxane-based neoadjuvant therapy in early triple-negative breast cancer: clinical results and biomarker analysis of GeparNuevo study. Ann. Oncol. J. Eur. Soc. Med. Oncol. 30, 1279–1288 (2019).
Wang, X. Q. et al. Spatial predictors of immunotherapy response in triple-negative breast cancer. Nature https://doi.org/10.1038/s41586-023-06498-3 (2023).
Wong, D. et al. Genomic mapping of the MHC transactivator CIITA using an integrated ChIP-seq and genetical genomics approach. Genome Biol. 15, 494 (2014).
Acknowledgements
This work has been supported has been supported in part by Fondazione Michelangelo, Fondazione AIRC per la Ricerca sul Cancro (IG 2018—ID21787 to G.B. and I.G. 2024—ID30919 to G.B.), Breast Cancer Research Foundation (BCRF 21-181 to L.G.; BCRF-22-133 to L.P.; BCRF-24-202 to J.M.B.), and (Susan G. Komen SAC220225 to L.P.).
Author information
Authors and Affiliations
Contributions
Concept and design: J.M.B., G.B. Acquisition, analysis, or interpretation of data: J.M.B., L.L., G.B. Drafting of the paper: J.M.B., L.L., G.B. Critical revision of the paper for important intellectual content: J.M.B., L.L., G.B., X.Q.W., M.D., C.H., D.E., B.B., C.Z., M.T., A.A., S.R., E.S., E.M.C., R.G., V.S., M.C., C.M.K., G.M., L.D.M., S.Z., Giulia V., M.C., Giuseppe V., L.P., L.G., H.R.A. Statistical analysis: M.D., M.C. Final approval of the paper for submission: J.M.B., L.L., G.B., X.Q.W., M.D., C.H., D.E., B.B., C.Z., M.T., A.A., S.R., E.S., E.M.C., R.G., V.S., M.C., C.M.K., G.M., L.D.M., S.Z., Giulia V., M.C., Giuseppe V., L.P., L.G., H.R.A.
Corresponding author
Ethics declarations
Competing interests
J.M.B. receives research support from Genentech/Roche and Incyte Corporation, has received advisory board payments from AstraZeneca, Eli Lilly, and Mallinckrodt and is an inventor on patents regarding immunotherapy targets and biomarkers in cancer. L.P. has received consulting fees and honoraria for advisory board participation from Pfizer, Astra Zeneca, Merck, Bristol-Myers Squibb, Stemline-Menarini, BeiGene, Personalis, Natera, Agendia, Exact Sciences, Radionetics, and institutional research funding from Pfizer, AstraZeneca, Merck, Bristol Myers Squibb, and Menarini-Stemline. Stock options in Ataraxis.GB has received consulting fees from Seagen, Roche, AstraZeneca, MSD, Daiichi Sankyo, Menarini, Tethis and Gilead Payment or honoraria for lectures, presentations, speakers bureaus, manuscript writing or educational events: Roche, AstraZeneca, Daiichi Sankyo, Eli Lilly, MSD, Gilead, Seagen, Pfizer, Novartis, Takeda; Support for attending meetings and/or travel: Roche, Pfizer, MSD, Novartis, Daiichi Sankyo, AstraZeneca, Takeda, Gilead, Eli Lilly, Menarini; and Advisory Board fees from Helsinn, Pfizer, Roche, AstraZeneca, Eli Lilly, Novartis, MSD, Daiichi Sankyo, Gilead, Seagen, Menarini, Exact Science. L.L. has received consulting fee from Exact Sciences, Helsinn, AstraZeneca, Daiichi Sankyo; honoraria for speakers’ bureaus from Gilead, Exact Sciences, Helsinn, Lilly, AstraZeneca, Daiichi Sankyo; support for attending meetings from Lilly, Gilead, Accord, Helsinn, Roche; has served on Advisory Board for Lilly, Exact Sciences, AstraZeneca, Italfarmaco, Daiichi Sankyo, Accord, Pfizer, Gilead, Novartis.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Balko, J.M., Licata, L., Wang, X.Q. et al. Tumor-specific major histocompatibility-II expression predicts pathological complete response to atezolizumab combined to chemotherapy in triple-negative breast cancer. npj Breast Cancer 11, 103 (2025). https://doi.org/10.1038/s41523-025-00828-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41523-025-00828-6