Abstract
Tumor biologic risk has replaced anatomic disease burden for guiding chemotherapy use in HR-positive, early-stage breast cancer. Recent surgical trials support less frequent axillary lymph node dissection, potentially impacting incidence of N2-3 diagnoses. As the field considers applying genomic risk assessment tools for locally advanced, operable HR-positive breast cancer, we estimated current incidence of these cancers, focusing on HR-positive/HER2-negative disease. Of 486,031 cases recorded with Stage I-III HR-positive/HER2-negative disease in the U.S. National Cancer Institute Surveillance, Epidemiology, and End Results-17 database (2010–2021), 28,585 (5.9%) and 23,307 (4.8%) had N2-3M0(Any T) and T3-4N0-1M0 disease, respectively. Invasive lobular cancer, observed across all disease stages and receptor-based subtypes, was highest in HR-positive/HER2-negative locally advanced disease. Incidence of N2-3M0(Any T) decreased for each subtype. Incidence of T3-4N0-1M0 increased for HR-positive/HER2-negative disease but not for the other subtypes. Defining chemotherapy benefit for patients with locally advanced, operable HR-positive breast cancer remains an important clinical question.
Similar content being viewed by others
Introduction
Anatomic disease burden of hormone receptor (HR)-positive breast cancer, as measured by tumor size and regional lymph node involvement, has historically been used to assess recurrence risk and to estimate the relative benefit of systemic chemotherapy. Over the last two decades, genomic assays have increasingly allowed for the incorporation of tumor biologic risk into the calculation of potential chemotherapy benefit1,2,3. Results from the TAILORx (lymph node negative disease), RxPONDER (N1 disease) and the WSG-ADAPT (N0-1) trials provide guidance on using the 21 gene recurrence score assay, OncotypeDX (Exact Sciences, Madison WI), to identify the approximately 80% of patients with early-stage breast cancer whose tumor signature indicated that administration of adjuvant chemotherapy is unlikely to result in a meaningful reduction in recurrence risk1,2,4,5. These assays have transformed the field of breast medical oncology, allowing for more precise delivery of chemotherapy to those most likely to benefit and preventing exposure in those unlikely to benefit from this lifesaving but often toxic therapy.
With the availability of genomic testing, an inherent next question is: Are there also locally advanced, operable HR-positive tumors for which chemotherapy is of limited or no benefit? The current, accepted treatment standard remains offering chemotherapy to those with N2-3 nodal disease, despite available data suggesting that only a small minority of these tumors have high genomic risk assay scores6,7. Patients with larger (T3-T4) tumors or high disease nodal burden (N2-3) were ineligible or underrepresented in most genomic assay trials, limiting information on outcomes by genomic risk for these patients. An important, related advance for those with locally advanced, operable HR-positive breast cancer is the availability of adjuvant cyclin-dependent kinase inhibitor therapy (CDK4/6i), which has demonstrated invasive disease-free survival benefit for this population when added to adjuvant endocrine therapy8,9. It is not currently known how the absolute benefit in risk reduction gained with the use of adjuvant CDK4/6i is modulated by prior receipt (or not) of chemotherapy. Moreover, the question of using genomic assay-based risk stratification for locally advanced tumors in the era of CDK4/6i arises concurrently with findings from several phase III surgical trials showing limited benefit from complete axillary lymph node dissection in those presenting with clinically node negative disease but with positive sentinel lymph nodes at surgery10,11,12. Has adoption of findings from surgical de-escalation trials changed practice and resulted in reduced identification of locally advanced, operable HR-positive breast cancers? Additionally, preoperative therapy and the related down-staging of disease could impact trends in pathologic staging. Over the last two decades, rates of neoadjuvant systemic therapy have increased, albeit more for human epidermal growth factor receptor-2 (HER2)-positive disease and HR-negative/HER2-negative (henceforth, triple negative breast cancer [TNBC]) than for HR-positive/HER2-negative disease13,14,15.
Improved understanding of current estimates and temporal trends in incidence of locally advanced, operable HR-positive breast cancer can provide a contemporary framework for considering these questions and inform clinical trial design. As such, in this study we identified Stage I-III breast cancer diagnoses recorded in the U.S. National Cancer Institute Surveillance, Epidemiology and End Results (SEER) 17 database during 2010–2021, with a focus on N2-3 and T3-4, HR-positive diagnoses. Using these data, we estimated overall incidence as well as changes in incidence over the study period for locally advanced operable breast cancer by receptor-based subtype. Our annual estimates also provide insights into incidence patterns across the years of the COVID-19 pandemic and the related healthcare delivery disruptions.
Results
Overall, 660,451 diagnoses of stage I-III breast cancer, 486,031 (73.6%) with HR-positive/HER2-negative cancer, were identified from the SEER 17 database. Of these, 28,585 (5.9%) had N2-3M0(Any T), and 23,307 (4.8%) had T3-4N0-1M0 cancer. Proportions of ILC were higher among those with N2-3M0(Any T) and T3-4N0-1M0 diagnoses compared to all diagnoses combined for each receptor-based subtype, with proportions being highest for HR-positive/HER2-negative cancers, particularly for T3-4N0-1M0 diagnoses (Table 1 and Fig. 1).
N2-3M0(Any T) cancers (A); T3-4N0-1M0 cancers (B). HER2 human epidermal growth factor receptor-2, HR hormone receptor, ILC invasive lobular carcinoma.
Within N2-3M0(Any T) diagnoses, HR-positive cancer was more often N2 than HR-negative cancer (Fig. 2A and Table 2). Within T3-T4N0-1M0 diagnoses, the distribution of T3-4 stage tumors demonstrated a higher proportion of T4 tumors in HER2-positive and TNBC cancers, with the highest proportion of T4d (inflammatory breast cancer) seen in those with HR-negative/HER2-positive tumors (Fig. 2B and Table 3).
Distribution of N stages in N2-3M0(Any T) cancers (A); Distribution of T stages in T3-4N0-1M0 cancers (B). HER2 human epidermal growth factor receptor-2, HR hormone receptor.
During 2010-2021, incidence of N2-3M0(Any T) decreased for all receptor-based breast cancer subtypes (Table 4 and Fig. 3). Incidence for T3-4N0-1M0 increased during the study period for HR-positive/HER2-negative cancers and was stable or decreasing for the other receptor-based subtypes. The decrease in N2-3M0(Any T) was contrasted by a modest increase in N0M0(Any T) and relatively stable incidence of N1M0(Any T) HR-positive/HER2-negative cancers; incidence of N0M0(Any T) and N1M0(Any T) was stable or decreasing in the other receptor-based subtypes.
N2-3M0 (Any T) cancers (A); T3-4N0-1M0 cancers (B). HER2 human epidermal growth factor receptor-2, HR hormone receptor.
Data from 2020, relative to years immediately prior to the COVID-19 pandemic (2018–2019), showed a decrease in the number of diagnoses for N2-3M0(Any T) and stable to decreasing numbers of T3-4N0-1M0 diagnoses (Table 5). Data for 2021 were consistent with those for study years prior to the COVID-19 pandemic, with decreased N2-3M0(Any T) and increased T3-4N0-1M0 diagnoses.
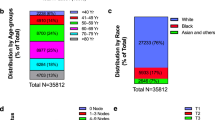
Proportions of N2-3M0(Any T) and T3-4N0-1M0 diagnoses that were HR-positive/HER2-negative were higher for those aged ≥50 years than those <50 years, although the difference was more pronounced for T3-4N0-1M0 diagnoses (Table 6). Proportions were higher for TNBC and lower for HR-positive/HER2-negative for non-Hispanic Blacks compared to other racial/ethnic groups. For N2-3M0(Any T) and T3-4N0-1M0 diagnoses and each receptor-based subtype, proportions ILC were higher for those aged ≥50 years than those <50 years. For HR-positive/HER2-negative and HR-positive/HER2-positive subtypes, proportions ILC were higher for non-Hispanic Whites compared to other racial/ethnic groups. The highest proportions ILC were observed for T3-4N0-1M0 HR-positive/HER2-negative diagnoses among those aged ≥50 years or of non-Hispanic White race/ethnicity, for which nearly one-third were ILC. Descriptive, incidence, and demographic analyses restricted to those with diagnoses of first primary breast cancers are presented in Supplementary Tables 1–6.
Discussion
Our analysis for locally advanced, operable breast cancer during 2010–2021 showed decreasing incidence of N2-3 breast cancer for all tumor types. An increase in T3-4N0-1M0 was observed for HR-positive/HER2-negative tumors, which may partially be explained by modest increases in N0-1M0(Any T) cancers in this subtype. Incidence of T3-4N0-1M0 disease was stable or decreasing for other receptor-based breast cancer subtypes over our study period. These locally advanced, operable disease presentations, particularly HR-positive/HER2-negative breast cancer, continue to be observed with high frequency in the clinic. Our analysis also showed that ILC was more common in the high anatomic disease burden groups than all diagnoses combined, not only in HR-positive/HER2-negative cancers but across all breast cancer subtypes. Collectively, these data provide contemporary information on occurrence of locally advanced, operable breast cancer.
Our finding of a temporal decrease in disease presentations with high nodal burden may be reflective of the surgical trials challenging the benefit of full axillary nodal dissection. In 2011, the American College of Surgeons Oncology Group Z0011 trial and subsequently the After Mapping of the Axilla: Radiotherapy or Surgery? (AMAROS) trial in 2014 reported no benefit to axillary lymph node dissection in patients with positive sentinel lymph nodes at surgery who were clinically node negative at presentation10,11. More recently, the Sentinel Node Negative Breast Cancer with Macrometastasis (SENOMAC) trial, which addressed some limitations from the aforementioned trials (including early closure, large non-inferiority margin, uncertain nodal irradiation volumes, and low event rates), showed similar findings12. Additionally, recent large cohort studies have observed decreasing rates of axillary lymph node dissection16,17. Practice changes related to these findings bring about a new clinical question: What is the optimal medical management of patients without full axillary staging information? Additionally, based on our findings a sizeable population appears to still undergo complete axillary lymph node evaluation, perhaps including those with T3-4 tumors or those with clinically apparent axillary lymph nodes at presentation who were generally not included in the aforementioned trials.
Increased use of neoadjuvant systemic therapy, which could lead to down-staged nodal burden, may also account for some of the observed decline in N2-3 disease, particularly for the more chemotherapy-sensitive HER2-positive and TNBC breast cancers. The increased use of neoadjuvant systemic therapy has been largely for HER2-positive cancers and TNBC13, where our data suggest the greatest declines in incidence of N2-3 disease. Marked responses to neoadjuvant therapy, pathological complete response (pCR) or residual cancer burden (RCB)-1, have been observed less frequently for HR-positive/HER2-negative cancer than for other breast cancer subtypes18,19, indicating a reduced impact of neoadjuvant therapy in this group.
Study of locally advanced, operable breast cancer offers an opportunity to improve disease outcomes for patients with ILC. In our dataset, these cancers, characterized by the loss of E-cadherin, were overrepresented among high anatomic disease stage presentations, particularly in those with T3-4N0-1M0 HR-positive/HER2-negative disease. Although numbers are smaller for those with ILC in other breast cancer receptor-based subtypes, given the overall high incidence of breast cancer, these groups represent a sizeable number of patients. Series reporting outcomes for “atypical” HER2-positive or triple negative ILC suggest inferior survival compared to the more common HR-positive/HER2-negative ILC or triple negative invasive ductal carcinoma20,21,22. Although ILC is phenotypically less sensitive to chemotherapy than breast cancer not otherwise specified, enhanced understanding of the molecular vulnerabilities of ILC, such as frequent HER2 mutations or androgen receptor expression20, suggests possible pathways to address this unmet need.
We did not observe a marked change in the number of diagnoses of locally advanced, operable breast cancer in 2020 when access to healthcare was most impacted by the COVID-19 pandemic. Although a slight decrease in N2-3M0(Any T) presentations was observed in 2020 compared to 2018 and 2019, we also observed declining presentations from 2010–2019. It is possible that by late 2020, many patients who had delayed care had sought attention. For T3-4N0-1M0 disease, our incidence estimates for 2020 did not stand apart from those for 2018 and 2019. In 2021, we observed an increase in T3-4N0-1M0 disease; however, we also observed an increasing trend prior to 2020. Our findings around the number of diagnoses from 2018–2021 are consistent with reports that showed a marked decrease in cancer screening during spring 2020 that had largely, if not fully, rebounded by late 202023,24, as well as a report showing that in 2021, incidence of localized breast cancer in the US matched or very slightly exceeded expected pre-pandemic estimates25.
Our findings for age and race/ethnicity of those with a locally advanced, operable breast cancer are consistent with receptor-based subtype patterns reported elsewhere, with proportionally more patients <50 years of age with HER2-positive disease or TNBC than seen in older patients26, as well as greater proportions of TNBC in Black patients and greater proportions of non-Hispanic White patients HR-positive/HER2-negative breast cancer. Recent data, however, show that incidence patterns are evolving with the incidence of HR-positive/HER2-negative breast cancer increasing in women aged ≤50 years27,28.
Locally advanced HR-positive/HER2-negative breast cancer remains a presentation encountered frequently in the clinic and one which is associated with poor outcomes. Developing the next generation of clinical trials to improve these outcomes and offer effective systemic therapy appropriately matched to tumor biology remains an important clinical need. This breast cancer subtype is complex and heterogenous with wide-ranging sensitivity to different therapies29. Emerging risk stratification tools, as well as new therapies, offer promise for more precision, where treatment is better matched to individual tumor characteristics. In addition, genomic assays can provide information on sensitivities to chemotherapy1,2,30,31 and emerging tools, such as endocrine response assessment by dynamic Ki67 or the SET2,3 index, could provide predictive information on anti-estrogen therapy5,32,33,34,35.
Assays for circulating tumor DNA and other markers of minimal residual disease hold promise for risk stratification, including for HR-positive/HER2-negative disease, despite the frequent dormancy, low tumor fractions, and extended time horizons which characterize this breast cancer subtype36,37,38. Novel anti-estrogen approaches are entering an era of growth with multiple classes, including selective estrogen receptor degraders, complete estrogen receptor antagonists, and proteolysis-targeting chimerics, entering late-phase trials. Further, the extent to which the benefit of adjuvant CDK4/6i, now widely used in this population, may appropriately replace any perceived benefit from chemotherapy remains unknown, offering another opportunity to study de-escalation of generally more toxic chemotherapy.
Although our findings provide contemporary information on locally advanced, operable disease presentations, they need to be interpreted with caution. We only report data from a US population, and incidence patterns may differ globally, as could the practice patterns which may underlie the observed findings. As with any retrospective database review, there may be unmeasured confounders. We were unable to report the proportion of cases where complete axillary dissection was not done, which would most impact the number of cases in the N2-3M0(Any T) group. A decrease in axillary dissection of 2.4%/year during 2007−2014 has been reported in another large dataset39. We were, however, able to look at incidence of N1 disease as a surrogate marker, although incidence changes observed may be due to multiple factors. Individual patients who had more than one breast cancer diagnosis during our study period could have been accounted for more than once in our dataset, although our analyses of only primary breast cancer diagnoses from the same SEER data showed similar trends. As stated above, changing neoadjuvant systemic therapy could have contributed to the incidence patterns observed. The rates of neoadjuvant therapy and the portion of intended therapy delivered cannot be accounted for in the SEER dataset over our full study period. Strengths of our work include that we were able to study a large, contemporary population-based cohort, which allowed for assessment of disease incidence during a time period when both surgical practice and timing of systemic therapy delivery have been evolving.
Over the past two decades, tools for assessing biologic risk relative to anatomic disease burden in HR-positive/HER2-negative breast cancer have advanced considerably. These advances now open opportunities to study the application of these tools in instances of very high anatomic burden of disease. Newer, non-chemotherapy treatment options offer further potential for optimizing care of individual patients. Data presented herein confirm that a breast cancer population with high anatomic disease burdens continues to exist, despite changes in surgical and systemic therapy practices over the same period. These data can inform the design of clinical trials exploring therapy optimization in this high-risk cohort. Additionally, these data offer a roadmap for other related areas where well-designed clinical trials could improve disease understanding and advance care, particularly for ILC, including not only HR-positive/HER2-negative disease but also the “atypical” HER2-positive and triple-negative ILC subtypes. Overall, refining treatment approaches for patients with locally advanced breast cancer, as well as understanding how to decrease the frequency of these locally advanced disease presentations, holds promise for considerable improvement of patient outcomes over the next decade.
Methods
Study population
This retrospective cohort study included malignant stage I-III breast cancer diagnoses during 2010–2021 (n = 670,747) identified from the SEER 17 database (November 2023 submission, 2000–2021) which includes diagnoses from 17 population-based registries that comprise approximately 26.5% of the total US population40. The study protocol was reviewed by the University of Iowa Institutional Review Board and determined not to meet the regulatory definition of human subjects research.
Breast cancer diagnoses
Diagnoses were not restricted by age or sex. Stage was assigned using the derived American Joint Committee on Cancer 7th edition (2010–2015)41, derived SEER Combined Stage 7th edition (2016–2017)42, and derived Extent of Disease 8th edition (2018–2021)43. Stage M0(i+) diagnoses (n = 581) and those without an M stage classification (n = 159) were excluded, leaving 670,007 diagnoses in the analytical sample, all of which were stage M0. Reporting for SEER typically prioritizes pathologic stage over clinical stage unless neoadjuvant therapy is given, or clinical stage was more extensive.
Given the primary objective of understanding incidence trends in locally advanced, operable breast cancer defined here as N2-3M0(Any T) or T3-4N0-1M0, diagnoses were not limited to first primary diagnoses; thus, an individual could have multiple primary malignancies and be included more than once if they had more than one breast cancer diagnosis during the study period. Additional, early-stage primary breast cancer diagnoses would have been included in Table 1 (All diagnoses); those with multiple, distinct breast cancer diagnoses meeting the locally advanced, operable breast cancer cohort selection—N2-3M0(Any T) or T3-4N0-1M0—would have been included more than once in these analyses. Analyses restricted to diagnoses of first primary breast cancers only were also conducted.
Eligible diagnoses identified were classified by tumor anatomic stage and receptor status. All stage I-III diagnoses were selected. Diagnoses were classified as HR-positive if a positive ER or PR status was recorded and HR-negative if a negative ER and PR status was recorded; borderline ER or PR status was not considered to be positive. HER2-positive status was defined as a positive or borderline test result and HER2-negative status as a negative test result. Diagnoses with unclassified or borderline HR or unclassified HER2 status were excluded (n = 9556), leaving 660,451 diagnoses in the analytical sample. Given that invasive lobular carcinoma (ILC) may be overrepresented in those with locally advanced, operable breast cancer and associated with reduced chemotherapy sensitivity15, we also identified the proportion of ILC diagnoses, defined as ICD-O-3 Histology/behavior = 8520/3, by receptor-based subtypes.
Statistical analysis
Frequencies of diagnoses and proportions ILC were examined for each combination of cancer stage (N2-3M0 [Any T], T3-4N0-1M0) and receptor status (HR-positive/HER2-negative, HR-positive/HER2-positive, TNBC, HR-negative/HER2-positive). The distributions of nodal stage among N2-3M0 (Any T) diagnoses and of T stage among T3-4N0-1M0 diagnoses were examined for each receptor status. Annual percentage changes (APC) in incidence were estimated over the study period, with corresponding 95% confidence intervals estimated using weighted least squares. Incidence was estimated as the number of annual breast cancer diagnoses per 100,000 individuals. Rates were per 100,000 and age-adjusted to the 2000 US Standard Population (19 age groups). The Tiwari et al. 2006 modification was used for confidence intervals44. APCs were examined for each combination of cancer stage and receptor status, along with N0-1M0 (Any T) by receptor status for comparison. Diagnoses per year during 2018–2021 for each combination of cancer stage group and receptor-based disease subtype were examined to assess patterns before (2018–2019), during (2020), and after (2021) the COVID-19 pandemic. Frequencies and proportions ILC diagnoses over the full study period, 2010–2021, were also examined for each combination of age (<50, ≥50 years) and race/ethnicity (non-Hispanic White, non-Hispanic Black, non-Hispanic American Indian/Alaska Native, non-Hispanic Asian/Pacific Islander, Hispanic). Analyses were conducted using SEER*Stat45 software version 8.4.4.
Data availability
The datasets used the current study are available from the SEER database (https://seer.cancer.gov/).
References
Sparano, J. A. et al. Adjuvant chemotherapy guided by a 21-gene expression assay in breast cancer. N. Engl. J. Med. https://doi.org/10.1056/NEJMoa1804710 (2018).
Kalinsky, K. et al. 21-gene assay to inform chemotherapy benefit in node-positive breast cancer. N. Engl. J. Med. 385, 2336–2347 (2021).
Cardoso, F. et al. 70-gene signature as an aid to treatment decisions in early-stage breast cancer. N. Engl. J. Med. 375, 717–729 (2016).
Iles, K. et al. The impact of age and nodal status on variations in oncotype DX testing and adjuvant treatment. npj Breast Cancer 8, 27 (2022).
Nitz, U. A. et al. Endocrine therapy response and 21-gene expression assay for therapy guidance in HR+/HER2- early breast cancer. J. Clin. Oncol. 40, 2557–2567 (2022).
Amlicke, M. J. et al. Prevalence of pathologic N2/N3 disease in postmenopausal women with clinical N0 ER+/HER2- breast cancer. Ann. Surg. Oncol. 29, 7662–7669 (2022).
Zhang, Q. H. et al. Impact of the 21-gene recurrence score assay on chemotherapy decision making and outcomes for breast cancer patients with four or more positive lymph nodes. Ann. Transl. Med. 7, 446 (2019).
Rastogi, P. et al. Adjuvant abemaciclib plus endocrine therapy for hormone receptor-positive, human epidermal growth factor receptor 2-negative, high-risk early breast cancer: results from a preplanned monarche overall survival interim analysis, including 5-year efficacy outcomes. J. Clin. Oncol. 42, 987–993 (2024).
Slamon, D. et al. Ribociclib plus endocrine therapy in early breast cancer. N. Engl. J. Med. 390, 1080–1091 (2024).
Giuliano, A. E. et al. Axillary dissection vs no axillary dissection in women with invasive breast cancer and sentinel node metastasis: a randomized clinical trial. JAMA 305, 569–575 (2011).
Donker, M. et al. Radiotherapy or surgery of the axilla after a positive sentinel node in breast cancer (EORTC 10981-22023 AMAROS): a randomised, multicentre, open-label, phase 3 non-inferiority trial. Lancet Oncol. 15, 1303–1310 (2014).
de Boniface, J. et al. Omitting axillary dissection in breast cancer with sentinel-node metastases. N. Engl. J. Med. 390, 1163–1175 (2024).
Habermann, E. B. et al. American College of Surgeons Cancer Program Annual Report from 2021 Participant User File. J. Am. Coll. Surg. 240, 95–110 (2025).
Chiba, A. et al. Trends in neoadjuvant endocrine therapy use and impact on rates of breast conservation in hormone receptor-positive breast cancer: a national cancer data base study. Ann. Surg. Oncol. 24, 418–424 (2017).
Tausch, C. et al. Trends in use of neoadjuvant systemic therapy in patients with clinically node-positive breast cancer in Europe: prospective TAXIS study (OPBC-03, SAKK 23/16, IBCSG 57-18, ABCSG-53, GBG 101). Breast Cancer Res. Treat. 201, 215–225 (2023).
Zhao, C., Sriram, N., Hitos, K., Hughes, T. M. & Ngui, N. Clinical impact of the Z0011 trial on axillary surgical management in Australia and New Zealand from the BreastSurgANZ Quality Audit. ANZ J. Surg. 95, 503–511 (2025).
Goldhaber, N. H., O’Keefe, T., Kang, J., Douglas, S. & Blair, S. L. Is Choosing wisely wise for lobular carcinoma in patients over 70 years of age? A National Cancer Database Analysis of Sentinel Node Practice Patterns. Ann. Surg. Oncol. 30, 6024–6032 (2023).
Cortazar, P. et al. Pathological complete response and long-term clinical benefit in breast cancer: the CTNeoBC pooled analysis. Lancet 384, 164–172 (2014).
Symmans, W. F. et al. Long-term prognostic risk after neoadjuvant chemotherapy associated with residual cancer burden and breast cancer subtype. J. Clin. Oncol. 35, 1049–1060 (2017).
Conforti, F. et al. Biological and clinical features of triple negative invasive lobular carcinomas of the breast. Clinical outcome and actionable molecular alterations. Breast 59, 94–101 (2021).
Quirke, N. P. et al. Invasive lobular carcinoma of the breast; clinicopathologic profile and response to neoadjuvant chemotherapy over a 15-year period. Breast 76, 103739 (2024).
Okines, A. et al. Clinical outcomes in patients with triple negative or HER2 positive lobular breast cancer: a single institution experience. Breast Cancer Res. Treat. 192, 563–571 (2022).
Howlader, N. et al. Cancer and COVID-19: US cancer incidence rates during the first year of the pandemic. J. Natl. Cancer Inst. 116, 208–215 (2024).
Lowry, K. P. et al. Breast Biopsy Recommendations and Breast Cancers Diagnosed during the COVID-19 Pandemic. Radiology 303, 287–294 (2022).
Howlader, N. et al. Impact of COVID-19 on 2021 cancer incidence rates and potential rebound from 2020 decline. J. Natl. Cancer Inst. https://doi.org/10.1093/jnci/djae180 (2024).
DeSantis, C. E. et al. Breast cancer statistics, 2019. CA 69, 438–451 (2019).
Thomas, A. et al. Incidence and survival among young women with stage I-III breast cancer: SEER 2000-2015. JNCI Cancer Spectr. 3, pkz040 (2019).
Xu, S., Murtagh, S., Han, Y., Wan, F. & Toriola, A. T. Breast cancer incidence among US women aged 20 to 49 years by race, stage, and hormone receptor status. JAMA Netw. Open 7, e2353331 (2024).
Rueda, O. M. et al. Dynamics of breast-cancer relapse reveal late-recurring ER-positive genomic subgroups. Nature 567, 399–404 (2019).
Freeman, J. Q. et al. Evaluation of multigene assays as predictors for response to neoadjuvant chemotherapy in early-stage breast cancer patients. npj Breast Cancer 9, 33 (2023).
Albain, K. S. et al. Prognostic and predictive value of the 21-gene recurrence score assay in postmenopausal women with node-positive, oestrogen-receptor-positive breast cancer on chemotherapy: a retrospective analysis of a randomised trial. Lancet Oncol. 11, 55–65 (2010).
Speers, C. W. et al. Evaluation of the sensitivity to endocrine therapy index and 21-gene breast recurrence score in the SWOG S8814 trial. J. Clin. Oncol. 41, 1841–1848 (2023).
Du, L. et al. Predicted sensitivity to endocrine therapy for stage II-III hormone receptor-positive and HER2-negative (HR+/HER2-) breast cancer before chemo-endocrine therapy. Ann. Oncol. Off. J. Eur. Soc. Med. Oncol. 32, 642–651 (2021).
Suman, V. J. et al. Evaluation of sensitivity to endocrine therapy index (SET2,3) for response to neoadjuvant endocrine therapy and longer-term breast cancer patient outcomes (Alliance Z1031). Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 28, 3287–3295 (2022).
Smith, I. et al. Long-term outcome and prognostic value of Ki67 after perioperative endocrine therapy in postmenopausal women with hormone-sensitive early breast cancer (POETIC): an open-label, multicentre, parallel-group, randomised, phase 3 trial. Lancet Oncol. 21, 1443–1454 (2020).
Garcia-Murillas, I. et al. Assessment of molecular relapse detection in early-stage breast cancer. JAMA Oncol. 5, 1473–1478 (2019).
Lipsyc-Sharf, M. et al. Circulating tumor DNA and late recurrence in high-risk hormone receptor-positive, human epidermal growth factor receptor 2-negative breast cancer. J. Clin. Oncol. 40, 2408–2419 (2022).
Ignatiadis, M., Sledge, G. W. & Jeffrey, S. S. Liquid biopsy enters the clinic - implementation issues and future challenges. Nat. Rev. Clin. Oncol. 18, 297–312 (2021).
Nocera, N. F. et al. NSQIP analysis of axillary lymph node dissection rates for breast cancer: implications for resident and fellow participation. J. Surg. Educ. 75, 1281–1286 (2018).
Surveillance, E., and End Results (SEER) Program SEER*Stat Database: Incidence - SEER Research Data, 17 Registries, Nov 2023 Sub (2000-2021) - Linked To County Attributes - Time Dependent (1990-2022) Income/Rurality, 1969-2022 Counties, National Cancer Institute, DCCPS, Surveillance Research Program, released April 2024, based on the November 2023 submission.
Edge S. B. et al. AJCC Cancer Staging Manual 7th edn (Springer, 2010).
Ruhl J, A. M. & Dickie, L. SEER Program Coding and Staging Manual 2016: Section V. (February 2016).
Ruhl JL, C. C. & Schussler, N. Extent of Disease (EOD) 2018 General Coding Instructions. (2024).
Tiwari, R. C., Clegg, L. X. & Zou, Z. Efficient interval estimation for age-adjusted cancer rates. Stat. Methods Med. Res. 15, 547–569 (2006).
Surveillance Research Program. National Cancer Institute SEER*Stat software version 84.
Acknowledgements
This analysis was conceptualized by members of the Breast International Group (BIG) and National Clinical Trials Network (NCTN) Node-Positive Luminal Disease (NPLD) Working Group established in 2023. We are grateful to the Breast Cancer Research Foundation (BCRF) for its support of the BIG-NCTN collaboration.
Author information
Authors and Affiliations
Contributions
A.T., A.R., E.L.M., A.D. and P.A.R. contributed to the study conception and design. A.R. and P.A.R. contributed to data collection. A.T., A.R., E.L.M., S.O., N.H., G.C., N.C., K.M.C., M.I., K.K., A.D. and P.A.R. contributed to data analysis and interpretation. A.T., A.R., E.L.M., S.O., N.H., G.C., Y.Z., V.A., N.C., M.I., K.K., A.D. and P.A.R. contributed to the drafting of the manuscript and critical revisions. All authors gave their final approval of the manuscript to be submitted.
Corresponding author
Ethics declarations
Competing interests
A.T.: Stock and Other Ownership Interests: Johnson & Johnson, Gilead Sciences, Bristol Myers Squibb, PfizerConsulting or Advisory Role: AstraZeneca, Delphi Diagnostics Research Funding, RTI International, Accord BioPharma: Sanofi (Inst), Merck (Inst) Patents, Royalties, Other Intellectual Property: Up-to-Date Royalties. E.L.M.: Consulting or Advisory Role: Genentech, Lilly, Novartis, AstraZeenca. S.O.: Honoraria: Novartis, AstraZeneca Consulting or Advisory Role: AstraZeneca Travel, Accommodations, Expenses: Novartis, Daichi Sankyo. N.H.: Stock and Other Ownership Interests: West German Study Group Honoraria: Roche, Novartis, Pfizer, AstraZeneca, Pierre Fabre, Daiichi-Sankyo, MSD, Seagen, Lilly, Viatris, Sanofi, Zuelligpharma, Gilead Sciences Consulting or Advisory Role: West German Study Group, Seagen, Gilead Sciences, Roche/Genentech Speakers’ Bureau: Medscape, Springer Healthcare, EPG Communication Research Funding: Roche/Genentech (Inst), Lilly (Inst), MSD (Inst), AstraZeneca (Inst). G.C.: has received research grants from Merck; has received honoraria from Ellipses Pharma; has received support for attending meetings and/or travel from Daiichi Sankyo; has a leadership role for the ESMO, the European Society of Breast Cancer Specialists and ESMO Open; is a speakers’ bureau member for Roche/Genentech, Novartis, Pfizer, Lilly, Foundation Medicine, Daiichi Sankyo, Seagen, Menarini, Gilead Sciences, AstraZeneca and Exact Sciences; and has held consulting or advisory roles for Roche/Genentech, Pfizer, Novartis, Lilly, Foundation Medicine, Bristol Myers Squibb, AstraZeneca, Daiichi Sankyo, Boehringer Ingelheim, GlaxoSmithKline, Seagen, Guardant Health, Veracyte, Celcuity, Menarini, Merck, Exact Sciences, Blueprint Medicines and Gilead Sciences. N.C.: Consulting or Advisory: Novartis, Daichi Research Funding: Merck (Inst.). M.I.: Consulting or Advisory Role: Seagen, Rejuveron Senescence Therapeutics, Menarini Group, Gilead Sciences, Daichi, Novartis Research Funding: Roche/Genentech (Inst), Pfizer (Inst), Natera (Inst), Inivata (Inst) Patents, Royalties, Other Intellectual Property: Patent entitled method for determining sensitivity to a CDK4/6 inh filed the 18 05 16 by Universite Libre de Bruxelles, Application No/Patent No 16170146.1-1403 Travel, Accommodations, Expenses: Roche (Inst), Gilead Sciences (Inst), AstraZeneca (Inst), Novartis. K.K.: Employment: EQRxStock and Other Ownership Interests: EQRxConsulting or Advisory Role: Lilly, Novartis, AstraZeneca, Genentech/Roche, Merck, Daiichi Sankyo/Astra Zeneca, Mersana, Menarini Silicon Biosystems, Myovant Sciences, Takeda, Prelude Therapeutics, RayzeBio, eFFECTOR Therapeutics, Cullinan Oncology, Gilead Sciences, Relay Therapeutics, Regor, Puma Biotechnology, Mersana, Pfizer, Bio Theranostics Research Funding: Novartis (Inst), Genentech/Roche (Inst), Lilly (Inst), Seagen (Inst), AstraZeneca (Inst), Daichi Sankyo (Inst), Ascentage Pharma (Inst). The other authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Thomas, A., Rhoads, A., Mayer, E.L. et al. Evolving incidence patterns for locally advanced operable breast cancer by receptor status: SEER 2010–2021. npj Breast Cancer 11, 127 (2025). https://doi.org/10.1038/s41523-025-00835-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41523-025-00835-7