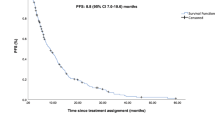
Abstract
The management of HER2-negative metastatic breast cancer (MBC) in the second-line or later setting remains challenging, due to the absence of standardized regimens and the limited efficacy of chemotherapy. Here, we report a prospectively, single-arm, phase II study evaluating anlotinib plus chemotherapy in patients with HER2-negative MBC (n = 33) who had progressed after at least one prior line of systemic therapy for metastatic disease. The primary endpoints were median progression-free survival (mPFS) and overall survival (OS), while secondary endpoints included objective response rate (ORR), clinical benefit rate (CBR), disease control rate (DCR), and safety. Exploratory proteomic profiling using the Olink Target 96 Immuno-Oncology panel was performed on baseline serum samples to identify potential predictors of response. After a median follow-up of 25.9 months and the median number of prior systemic therapy lines was 2 (range, 1–4). The mPFS was 8.3 (95% CI: 6.3–10.3) months, and the mOS was 22.2(95% CI: 13.1–31.3) months. The ORR was 33.3%, DCR reached 90.9% and CBR stood at 60.6%. Proteomic analysis indicated that higher baseline serum levels of proteins including CSF-1 were associated with shorter PFS (P < 0.05). No treatment-related fatalities were observed. This trial is registered with www.chictr.org.cn (ChiCTR2400081835) on 13 March 2024.
Similar content being viewed by others
Introduction
Breast cancer (BC) stands as one of the most common malignancies among women, and the primary contributor to cancer-related fatalities1. Human epidermal growth factor receptor 2-negative metastatic breast cancer (HER2-negative MBC) accounts for 70–80% of cases2, comprising hormone receptor-positive (HR + )/HER2-negative and triple-negative breast cancer (TNBC). For HR + /HER2-negative MBC, endocrine therapy combined with cyclin-dependent kinase (CDK) 4/6 inhibitors represents the standard first-line treatment, tremendously improving progression-free survival (PFS) and overall survival (OS)3,4. In metastatic TNBC (mTNBC), immunotherapy has shown promising first-line clinical activity in PD-L1-positive disease5, whereas chemotherapy remains an option for PD-L1-negative or ineligible populations6. Despite these advances, disease progression is almost inevitable, necessitating effective later-line therapeutic approaches.
Following first-line treatment, therapeutic options for HER2-negative MBC remain limited. Although agents such as PI3K/AKT/mTOR inhibitors have confirmed the encouraging efficacy for HR + /HER2-negative MBC patients, their accessibility is constrained in China7,8,9. Similarly, the antibody-drug conjugate sacituzumab govitecan (SG) has demonstrated superior efficacy in mTNBC but is not routinely available in China due to cost and reimbursement barriers10. Consequently, chemotherapy remains the recommendation for previously treated (beyond first-line) HER2-negative MBC11,12, despite its modest efficacy and limited duration of response13,14. Therefore, there is a clear need for novel, accessible, and effective regimens for pretreated HER2-negative MBC.
Anlotinib is a novel, orally administered multi-targeting tyrosine kinase inhibitor that targets vascular endothelial growth factor receptor (VEGFR), fibroblast growth factor receptor (FGFR), platelet-derived growth factor receptors (PDGFR), and c-kit15. Currently, anlotinib has been approved for several malignant tumors such as lung cancer, renal clear cell cancer, thyroid cancer, and soft tissue sarcoma in China16,17,18,19. In our previous retrospective studies, anlotinib in combination with chemotherapy exhibited encouraging efficacy and a manageable safety profile in heavily pretreated MBC20. This benefit was also observed in patients with HR + /HER2-negative disease who had progressed on CDK4/6 inhibitors21.
To further evaluate this combination, we conducted a phase II clinical trial to prospectively investigate the efficacy and safety of anlotinib in combination with chemotherapy as the second-line or later therapy in HER2-negative MBC patients. Simultaneously, we performed exploratory proteomic profiling of 92 immuno-oncology-related proteins in baseline serum samples to identify potential predictors of response to anlotinib-based treatment.
Results
Patient enrollment and baseline characteristics
Between August 4, 2022 and August 2, 2023, a total of 33 HER2-negative MBC patients were enrolled (Fig. 1). Baseline characteristics are summarized in Table 1. The median age was 52 (range: 32–79) years, and all patients had a grade 1 ECOG performance status. The represented IHC-based subtypes included HR + /HER2-negative (23 of 33, 69.7%) and TNBC (10 of 33, 30.3%). Visceral metastases were present in 72.7% of the cohort.
a Participant flow diagram. b Trial plan.
The median number of prior lines of systemic therapy for metastatic disease was 2 (range, 1-4). The most common chemotherapy partners combined with anlotinib was taxanes (33.3%) and eribulin(33.3%), followed by utidelone (18.2%), capecitabine (15.2%).
Efficacy
At the data cutoff date (February 2025), the median follow‑up duration was 25.9 months (range, 5.6–32.2). Disease progression led to treatment discontinuation in 30 patients (90.9%), and three patients (9.1%) were lost to follow‑up. The mPFS was 8.3 (95% CI: 6.3–10.3) months, and the mOS was 22.2 (95% CI: 13.1–31.3) months (Fig. 2). In subgroup analyses, the mPFS of HR + /HER2-negative and TNBC was 7.4 months (95% CI, 4.5-10.2) and 10.6 months (95% CI, 7.7-13.5) respectively(Fig. 2a). Corresponding median OS values were 15.3 months (95% CI: 3.2–27.5) and 24.2 months (95% CI: 15.5–32.8), respectively (Fig. 2b). Among the 33 patients, no patient achieved CR, 11 exhibited a confirmed partial response (PR) and 19 had stable disease (SD), resulting in an ORR of 33.3% (95% CI: 18.0–51.8), CBR of 60.6% (95% CI: 42.1–77.1), and the DCR of 90.9% (95% CI: 75.7–98.1) (Fig. 3). In multivariate Cox regression analysis, no factors were identified as significantly associated with PFS, consistent with the results of univariate log‑rank tests (Fig. 3c).
Kaplan–Meier curves of (a) Progression-free survival (PFS) (b) Overall survival (OS).
a Anti-tumor activity over time (b)A waterfall plot for best percentage change in the sum of the diameters of target lesions at any time in patients with measurable disease at baseline (c) Forest plot showing HR results for the patient characteristics indicated. ▿ Means the patients had new lesions. * Represents TNBC patients.
In this study, 19 of the 23 HR + /HER2-negative MBC patients (82.6%) had received previous CDK4/6i treatment (including 11 patients who were treated with CDK4/6i in the first line). Among these first‑line CDK4/6i‑treated patients, the median progression‑free survival (PFS) with subsequent anlotinib plus chemotherapy was 6.8 months (95% CI: 2.8–10.8), and the median overall survival (OS) was 15.3 months (95% CI: 2.4–28.2) (Fig. 4). Rapid disease progression was defined as clinical benefit lasting ≤ 6 months from the start of first‑line CDK4/6i‑based therapy according to RECIST v1.1. In this subgroup, the efficacy of subsequent anlotinib‑based therapy was comparable between patients with or without rapid progression (p = 0.66).
Kaplan–Meier curves of (a) PFS (b) OS.
Exploratory Analysis of Baseline Serum Protein Levels and Treatment Response
To discover potential serum biomarkers associated with clinical response to anlotinib plus chemotherapy, we profiled the levels of 92 proteins in baseline serum samples using the Olink Target 96 Immuno‑Oncology panel. A total of 32 blood samples were included in this analysis.
Proteomic analysis revealed significant baseline differences between patients who subsequently experienced short PFS (non‑sensitive group) and those with long PFS (sensitive group) (P < 0.05). Specifically, elevated baseline levels of five proteins including colony‑stimulating factor‑1 (CSF‑1), vascular endothelial growth factor (VEGF), interleukin‑6 (IL‑6), IL‑10, and IL‑12 were associated with significantly shorter PFS (Fig. 5). These differential expression patterns were further validated in serum samples using enzyme‑linked immunosorbent assay (ELISA) (Supplementary Fig. 1).
a Heatmap showing the distribution and frequency of key proteins expression across the study cohort (b) Differential immune-oncology related proteins expression as described by volcano plot (c) ELISA experiments demonstrating protein expression levels in the serum of patients in the not sensitive group were significantly higher than those in the sensitive group at baseline.
Safety
Treatment-related adverse events (TRAEs) in all patients are detailed in Table 2. The most common any‑grade TRAEs included hypertriglyceridemia (81.8%), anemia (60.6%), hyperglycemia (60.6%), hypercholesterolemia (51.5%), leukopenia (48.5%), elevated alanine aminotransferase (48.5%), and neutropenia (42.4%). Grade 3–4 TRAEs occurred predominantly as neutropenia (21.2%) and hypertriglyceridemia (21.2%); other grade 3–4 events included leukopenia (15.2%), anemia (6.1%), hyperglycemia (3.0%), and hand‑foot syndrome (3.0%). Adverse reactions leading to dose reduction of anlotinib occurred in six patients (18.1%). There were no treatment-related deaths or instances of drug discontinuation and treatment interruption due to adverse effects recorded in the study.
Discussion
In China, chemotherapy remains the primary option for patients with HER2-negative MBC following progression on first-line standard therapy, particularly in cases of rapid progression or visceral crisis22. Unfortunately, the survival benefit of chemotherapy is limited, where the mPFS of single-agent therapy was just 3–4 months23. Therefore, we look forward to exploring new therapeutic strategies.
In the present study, anlotinib combination with chemotherapy achieved encouraging anti-tumor activity and controllable toxicity, with an overall mPFS was 8.3 months. Among the HR+ and TNBC populations, the mPFS was 7.4 months and 10.6 months, respectively. In addition, exploratory analyses contributed to the identification of potential serum biomarkers that may predict efficacy.
The rationale for combining anlotinib with chemotherapy is supported by the established role of angiogenesis in breast cancer progression. Prior key trials have evaluated anti-angiogenic strategies in MBC. The E2100 trial demonstrated that adding bevacizumab (a monoclonal antibody against VEGF-A) to paclitaxel significantly improved PFS in first-line HER2-negative MBC24. Subsequent studies like RIBBON-1/2 and IMELDA further confirmed the PFS benefit of bevacizumab-chemotherapy combinations, albeit with variable effects on OS and increased toxicity concerns25,26,27.The SOLTI-0701 trial explored the oral multi-kinase inhibitor sorafenib (targeting VEGFR, PDGFR, RAF) with capecitabine, showing improved PFS but also notable toxicity28.More recently, the ATRACTIB trial investigated the combination of bevacizumab plus paclitaxel and atezolizumab exhibits encouraging antitumor activity as a first-line treatment for patients with mTNBC, highlighting the ongoing interest in this therapeutic class with anti-angiogenic activity29. However, the clinical utility of these agents has been limited by tolerability and modest survival gains. Anlotinib is an oral multi-target TKI inhibiting VEGFR, FGFR, PDGFR, and c-Kit. Its approval in other solid tumors in China underscores its manageable safety16,17,18,19. Our study builds upon this historical landscape by evaluating anlotinib with flexible chemotherapy partners in the later-line setting, where effective and tolerable options are urgently needed.
Emerging therapeutic strategies for HR + /HER2-negative MBC patients, who progressed after the CDK4/6i-based treatment, typically failed to achieve satisfactory efficacy. On the one hand, a series of studies applying re-challenge with CDK4/6 inhibitors have not achieved consistent conclusions30,31,32,33. Therefore, it remains unclear whether there is a benefit to replacing CDK4/6 inhibitors in patients who have progressed on CDK4/6i. Other target drugs, such as mTOR inhibitors, histone deacetylase (HDAC) inhibitors were alternative options after CDK4/6 inhibitor progression34,35. However, the efficiency was not satisfactory, with an mPFS of around 5 months. In our study, 52.2% HR+ patients had 1-2 prior lines of chemotherapy for metastatic disease, the observed mPFS of 7.4 months compares favorably with efficacy in similar later-line settings. For example, in the DESTINY-Breast04 trial, the mPFS for physician’s choice chemotherapy just was 5.4 months36. On the other hand, although previous studies have demonstrated that PI3K inhibitors, AKT inhibitors, and oral selective estrogen receptor down regulator (SERD) have improved patient survival37,38,39. Unfortunately, these haven’t been approved in China and cost expensively, making the drug unavailable for most patients. We noted that the shorter mPFS and mOS in the HR+ subgroup in this study compared to the TNBC subgroup is intriguing but should be interpreted with extreme caution. This may be attributed to the small sample size (n = 10 for TNBC), and potential imbalances in prior therapy lines within these subgroups; secondly, only 11 HR+ patients (47.8%) received CDK4/6i as first-line standard treatment, and among all patients receiving CDK4/6i (n = 19), the PFS duration was less than 6 months in 13 patients (13/19, 68.4%). PARSIFAL-LOMG study has illustrated that rapid disease progression patients on regimens with first-line CDK4/6i therapy tended to have a worse prognosis40. Real-world data suggests a preference for chemotherapy, probably because physicians thought that these patients may benefit more from chemotherapy22,41. In this study, the efficacy of subsequent anlotinib–chemotherapy was comparable in patients with or without rapid progression on first-line CDK4/6 inhibitors, and the regimen provided clinically meaningful PFS improvement with a manageable safety profile. These findings are exploratory and require validation in larger, dedicated cohorts.
For mTNBC, the ASCENT trial established SG as a standard second-line option, demonstrating superior PFS (5.6 months) over chemotherapy42. More recently, sacituzumab tirumotecan (SKB264) has also shown promising PFS benefits in this setting43. However, the access to these novel antibody-drug conjugates is limited in China due to high cost and lack of insurance coverage. In our small exploratory cohort of only 10 mTNBC patients the observed mPFS was 10.6 months. Given the very small sample size, cross-trial comparisons with SG or SKB264 are not appropriate. Nonetheless, the observed activity, combined with the established accessibility and manageable cost of anlotinib and chemotherapy in China, suggests that this combination may represent a viable treatment alternative in resource-constrained settings where standard novel therapies are unavailable. Overall, anlotinib combined with chemotherapy achieved a clinically therapeutic effect. Although lacking head-to-head comparative trials, the preliminary efficacy and safety data from this study suggest that anlotinib combined with chemotherapy represents a potential treatment option within the evolving treatment landscape for second-line or later therapy in HER2-negative MBC in China.
In an exploratory biomarker analysis using the Olink Immuno‑Oncology panel, we found that elevated baseline serum levels of CSF‑1, VEGFA, IL‑6, IL‑10, and IL‑12 were associated with insensitivity to anlotinib‑based therapy. We acknowledge that these elevated serum levels could reflect greater tumor burden or a state of systemic inflammation, both hallmarks of advanced disease rather than being mechanistically specific to anlotinib resistance. In the absence of adjustment for comprehensive tumor load metrics (such as burden of liver metastases, number of metastatic sites, or LDH levels), it is difficult to determine whether these proteins are drivers of resistance or are elevated as a consequence of more extensive disease. These proteins are, however, biologically implicated in processes pertinent to therapy resistance, such as angiogenesis and immunosuppression within the tumor microenvironment. The upregulation of VEGFA aligns with its established role in aberrant vascular remodeling, a known mechanism for limiting drug delivery and efficacy44,45. Concurrent elevation of CSF-1, IL-6, IL-10, and IL-12 may indicate a tumor-promoting, immunosuppressive milieu often orchestrated by tumor-associated macrophages (TAMs)46,47,48,49.Thus, while not providing causal proof, these findings generate the hypothesis that baseline activation of antiangiogenic and immunosuppressive pathways may identify tumors less likely to respond to anlotinib-based therapy. These proteomic findings are preliminary and lack of an independent validation cohort. Therefore, the predictive value of these candidate biomarkers remains unconfirmed.
Our study has several main limitations that should be considered when interpreting the results. Firstly, the single-arm, non-randomized design precludes a definitive assessment of the incremental benefit of adding anlotinib to chemotherapy. While the observed efficacy is promising, we cannot rule out that a portion of the effect may be attributable to chemotherapy alone, nor can we directly quantify the specific contribution of anlotinib. Secondly, the small overall sample size (n = 33) limits the statistical power and generalizability of our findings. This is particularly pertinent for the TNBC subgroup, which included only 10 patients. The encouraging PFS and OS observed in this small TNBC cohort should be viewed as preliminary; the results are vulnerable to chance variation and require validation in larger, dedicated studies. Thirdly, our biomarker analysis is constrained by the lack of longitudinal plasma sampling. We only assessed baseline protein levels, which prevents us from examining dynamic changes associated with treatment response or the emergence of resistance. Consequently, the identified biomarkers may be influenced by baseline tumor biology or burden.
Despite these limitations, our study provides valuable preliminary evidence supporting further investigation of this combination. Currently, we are conducting a prospective multicenter clinical trial (ChiCTR2500103534) based on this study, which aims to further explore optimal anlotinib-based combinations and to incorporate longitudinal biomarker sampling to address some of these constraints.
In conclusion, our study suggests that anlotinib combined with chemotherapy may be an optional second-line or later strategy for patients with HER2-negative MBC. Furthermore, this study offers crucial identification of potential biomarkers for resistance to this combination therapy. However, its results need further confirmation through prospective studies with larger sample sizes.
Methods
Study design and participants
This was a prospective, single‑arm, phase II clinical trial conducted at the Affiliated Cancer Hospital of Nanjing Medical University. The study evaluated anlotinib combined with chemotherapy as second‑line or later therapy in patients with HER2‑negative MBC. Key eligibility criteria included: females aged 18-75 years; Eastern Cooperative Oncology Group performance status score of 0–1; histologically confirmed HER2-negative MBC patients, which was defined as immunohistochemistry (IHC) score for HER2 staining was 0 or 1 + , or no HER2 gene amplification detected by fluorescence in situ hybridization (FISH) when the IHC score was 2 + ; participants were required to have one or more measurable lesions according to the Response Evaluation Criteria in Solid Tumors version 1.1(RECIST 1.1); experienced a minimum of one prior systemic treatment for MBC in the metastatic setting. Main exclusion criteria included prior treatment with anti-angiogenic drugs, including small molecules (e.g., anlotinib, apatinib) and large molecules (e.g., bevacizumab), uncontrollable hypertension or arrhythmias, and a history of cardiac insufficiency of grade 2 or greater.
Procedures
Eligible patients received anlotinib combined with chemotherapy. Based on prior retrospective data demonstrating both anticancer activity and safety, anlotinib (recommended initial dose was 12 mg) was administered orally once daily (days 1–14) in 21-day cycles. Adjustment of the dose, permanent discontinuation, or delays were allowed based on patients’ side effects50,51, although the duration of drug interruption could not exceed 14 days. Depending on the survival benefit of patient’s previous treatment regimens, the chemotherapy partners of anlotinib were determined by the physician. Nab-paclitaxel was administered at 125 mg/m2 on days 1 and 8, every 21 days per cycle; paclitaxel at 80 mg/m2 on days 1 and 8, every 21 days per cycle; eribulin at 1.4 mg/m² on days 1 and 8, every 21 days per cycle. Utidelone was given at 30 mg/m² for 5 days, every 21 days per cycle. Capecitabine was orally given at 1250 mg/m², twice a day (days 1–14) in 21-day cycles.
Treatment was structured in two phases. All patients began with an induction phase consisting of 4–8 cycles of anlotinib plus chemotherapy. Transition to the maintenance phase was protocol‑specified and required meeting both of the following criteria: (1) at least stable disease after induction, and (2) acceptable toxicity (adverse events ≤ Grade 1 or deemed manageable by the investigator). Upon meeting these criteria, HR + /HER2‑negative patients continued anlotinib in combination with endocrine (addition in maintenance phase), while TNBC patients received anlotinib monotherapy. Treatment continued in the maintenance phase until disease progression, unacceptable toxicity, death, or patient withdrawal.
Treatment response was routinely assessed at baseline and every two cycles on combination therapy, every three cycles on maintenance therapy by computed tomography (CT) or magnetic resonance imaging (MRI), based on the RECIST 1.1. Follow-up for overall survival was done every 3 months until death, loss to follow-up, or completion of the study. Physical examinations, blood routine, and biochemical tests were done every 3 weeks. Echocardiography, coagulation test, and urine and faeces tests were done every 6 weeks. Treatment-related adverse events (TRAEs) were monitored before each drug administration and at each examination from the initiation of study treatment until 30 days after the last dose, and evaluated according to the Common Terminology Criteria for Adverse Events version 5.0 (CTCAE 5.0).
Outcomes
The primary endpoints of this study were PFS (defined as time from receiving anlotinib-based treatment to radiographically or clinically confirm disease progression or death from any cause, whichever occurred first) and OS (time from treatment initiation to death from any cause).
Secondary endpoints included objective response rate (ORR), which was defined as the percentage of patients who achieved a complete response (CR) or partial response (PR), according to RECIST 1.1), disease control rate (DCR, the proportion of patients who achieved CR, PR, or stable disease), clinical benefit rate (CBR, defined as the proportion of patients with a CR, PR, or SD lasting ≥ 6 months), as well as safety.
Sensitivity to anlotinib-based therapy was defined as a documented clinical benefit equal to or more than four cycles from the initiation of anlotinib treatment.
Serum proteomics profiling by Olink technology
Peripheral blood samples were collected from each study participant in EDTA tubes before treatment. The blood samples were centrifuged at 2000–2500 g for 10 min and the serum samples were then stored at -80°C until assayed.
Protein concentrations in serum were measured using the 96 Immuno-Oncology panels, including 92 protein biomarkers, according to the manufacturer’s instructions. The Immuno-Oncology panel is a high-throughput, multiplex immunoassay enabling analysis of 92 protein biomarkers52. In short, these measurements are based on proximity extension assay (PEA) technology, which enables high-throughput multiplex immunoassays of proteins using 1 μl of serum. Normalized protein expression (NPX) provided by Olink was used for data analysis.
A total of 32 serum samples at baseline were successfully detected and used for further analysis. Differential protein expression between the sensitive group and non- sensitive group were verified in serum samples using ELISA. For the ELISA experiments, VEGF-A (HM10978, Bioswamp, Wuhan, China), IL-6 (HM10205, Bioswamp, Wuhan, China), IL-10(HM10203, Bioswamp, Wuhan, China), IL-12(HM10196, Bioswamp, Wuhan, China), and M-CSF (HM10372, Bioswamp, Wuhan, China) levels were tested in serum samples. The optimal dilution was determined, and ELISA was performed according to the manufacturer’s instructions. Fitting curves were generated by CurveExpert 1.4.
Statistical analysis
Sample size estimation was based on a conservative historical benchmark for single‑agent chemotherapy efficacy in pretreated HER2‑negative MBC. The phase III EMBRACE trial, which reported an mPFS of 3.7 months for eribulin in patients with 2–5 prior chemotherapy lines, was used as a stringent reference. Although the EMBRACE population was more heavily pretreated than our cohort (which required ≥ 1 prior line), this benchmark was intentionally adopted to provide a conservative control value. We hypothesized that the combination of anlotinib with investigator-selected chemotherapy would improve the mPFS to 6.5 months. Sample size estimation was performed using a one-sample log-rank test. With an accrual period of 12 months and total study duration of 12 months, a minimum of 30 subjects are required to achieve 80% power at a two-sided significance level (α) of 0.05. Assuming a dropout rate of 10%, an accrual size of 33 patients was chosen in this trial.
Statistical analyses were conducted using R Studio. The Kaplan-Meier curves were employed for analyzing PFS and OS, and stratified log-rank tests were utilized to obtain P-values between groups. Additionally, the Cox proportional hazards model was employed to calculate hazard ratios (HRs) and corresponding 95% confidence intervals (CIs). P-value < 0.05 was considered indicative of statistical significance.
Ethical approval of the study protocol
Ethical approval was obtained from the Ethics Committee of Jiangsu Cancer Hospital (\({\rm{No}}.\,2022-049)\).This study was undertaken in accordance with the Declaration of Helsinki and all applicable local regulations. All patients provided written informed consent prior to participation. This trial is registered with www.chictr.org.cn (ChiCTR2400081835) on 13 March 2024.
Data availability
Anonymized patient data, the research protocol, and the analysis methodology can be made available for sharing upon reasonable request and provision of a comprehensive protocol and analysis plan to the corresponding author.
References
Bray, F. et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries [J/OL]. CA: A Cancer J. Clinicians 74, 229–263 (2024).
Łukasiewicz, S. et al. Breast Cancer—Epidemiology, Risk Factors, Classification, Prognostic Markers, and Current Treatment Strategies—An Updated Review [J/OL]. Cancers 13, 4287 (2021).
Hortobagyi, G. N. et al. Updated results from MONALEESA-2, a phase III trial of first-line ribociclib plus letrozole versus placebo plus letrozole in hormone receptor-positive, HER2-negative advanced breast cancer[J/OL]. Ann. Oncol. 29, 1541–1547 (2018).
Finn, R. S. et al. Palbociclib and Letrozole in Advanced Breast Cancer. N. Engl. J. Med 375, 1925–1936 (2016).
Cortes, J. et al. KEYNOTE-355 Investigators. Pembrolizumab plus chemotherapy versus placebo plus chemotherapy for previously untreated locally recurrent inoperable or metastatic triple-negative breast cancer (KEYNOTE-355): a randomised, placebo-controlled, double-blind, phase 3 clinical trial. Lancet 396, 1817–1828 (2020).
Gradishar, W. J. et al. Breast Cancer, Version 3.2024, NCCN Clinical Practice Guidelines in Oncology[J/OL]. J. Natl. Compr. Cancer Netw. 22, 331–357 (2024).
Rugo, H. S. et al. Alpelisib plus fulvestrant in PIK3CA-mutated, hormone receptor-positive advanced breast cancer after a CDK4/6 inhibitor (BYLieve): one cohort of a phase 2, multicentre, open-label, non-comparative study[J/OL]. Lancet Oncol. 22, 489–498 (2021).
Turner, N. C. et al. Capivasertib in Hormone Receptor–Positive Advanced Breast Cancer [J/OL]. N. Engl. J. Med. 388, 2058–2070 (2023).
Cook, M. M. et al. Everolimus Plus Exemestane Treatment in Patients with Metastatic Hormone Receptor-Positive Breast Cancer Previously Treated with CDK4/6 Inhibitor Therapy [J/OL]. Oncologist 26, 101–106 (2021).
Bardia, A. et al. Sacituzumab Govitecan in Metastatic Triple-Negative Breast Cancer [J/OL]. N. Engl. J. Med. 384, 1529–1541 (2021).
Huppert, L. A. et al. Systemic therapy for hormone receptor-positive/human epidermal growth factor receptor 2-negative early stage and metastatic breast cancer[J/OL]. CA: A Cancer J. Clinicians 73, 480–515 (2023).
Pivot, X. et al. Pooled analyses of eribulin in metastatic breast cancer patients with at least one prior chemotherapy [J/OL]. Ann. Oncol. 27, 1525–1531 (2016).
Cardoso, F. et al. 5th ESO-ESMO international consensus guidelines for advanced breast cancer (ABC 5). Ann. Oncol. 31, 1623–1649 (2020).
Li, C. H., Karantza, V., Aktan, G. & Lala, M. Current treatment landscape for patients with locally recurrent inoperable or metastatic triple-negative breast cancer: a systematic literature review. Breast Cancer Res 21, 143 (2019).
Shen, G. et al. Anlotinib: a novel multi-targeting tyrosine kinase inhibitor in clinical development [J/OL]. J. Hematol. Oncol. 11, 120 (2018).
Han, B. et al. Effect of Anlotinib as a Third-Line or Further Treatment on Overall Survival of Patients With Advanced Non–Small Cell Lung Cancer: The ALTER 0303 Phase 3 Randomized Clinical Trial[J/OL]. JAMA Oncol. 4, 1569 (2018).
Zhou, A. P. et al. Anlotinib Versus Sunitinib as First-Line Treatment for Metastatic Renal Cell Carcinoma: A Randomized Phase II Clinical Trial [J/OL]. Oncologist 24, e702–e708 (2019).
Li, D. et al. Anlotinib in Locally Advanced or Metastatic Medullary Thyroid Carcinoma: A Randomized, Double-Blind Phase IIB Trial[J/OL]. Clin. Cancer Res. 27, 3567–3575 (2021).
Liu, J. et al. Phase II Study of TQB2450, a Novel PD-L1 Antibody, in Combination with Anlotinib in Patients with Locally Advanced or Metastatic Soft Tissue Sarcoma. Clin Cancer Res. 28, 3473–3479 (2022).
Qian, Y. et al. Efficacy and safety of anlotinib-based treatment in metastatic breast cancer patients[J/OL]. Front. Oncol. 12, 1042451 (2022).
Xu, T. et al. The Effects of Anlotinib Combined with Chemotherapy following Progression on Cyclin-Dependent Kinase 4/6 Inhibitor in Hormone Receptor-Positive Metastatic Breast Cancer.
LI, Y. et al. A multicenter analysis of treatment patterns and clinical outcomes of subsequent therapies after progression on palbociclib in HR+/HER2-metastatic breast cancer [J/OL]. Therapeutic Adv. Med. Oncol. 13, 175883592110228 (2021).
Cortes, J. et al. Eribulin monotherapy versus treatment of physician’s choice in patients with metastatic breast cancer (EMBRACE): a phase 3 open-label randomised study [J/OL]. Lancet 377, 914–923 (2011).
Miller, K. et al. Paclitaxel plus bevacizumab versus paclitaxel alone for metastatic breast cancer. N. Engl. J. Med 357, 2666–2676 (2007).
Robert, N. J. et al. RIBBON-1: randomized, double-blind, placebo-controlled, phase III trial of chemotherapy with or without bevacizumab for first-line treatment of human epidermal growth factor receptor 2-negative, locally recurrent or metastatic breast cancer. J. Clin. Oncol. 29, 1252–1260 (2011).
Brufsky, A. M. et al. RIBBON-2: a randomized, double-blind, placebo-controlled, phase III trial evaluating the efficacy and safety of bevacizumab in combination with chemotherapy for second-line treatment of human epidermal growth factor receptor 2-negative metastatic breast cancer. J. Clin. Oncol. 29, 4286–4293 (2011).
Gligorov, J. et al. Maintenance capecitabine and bevacizumab versus bevacizumab alone after initial first-line bevacizumab and docetaxel for patients with HER2-negative metastatic breast cancer (IMELDA): a randomised, open-label, phase 3 trial. Lancet Oncol. 15, 1351–1360 (2014).
Baselga, J. et al. Sorafenib in combination with capecitabine: an oral regimen for patients with HER2-negative locally advanced or metastatic breast cancer. J. Clin. Oncol. 30, 1484–1491 (2012).
Gion, M. et al. Atezolizumab plus paclitaxel and bevacizumab as first-line treatment of advanced triple-negative breast cancer: the ATRACTIB phase 2 trial. Nat. Med 31, 2746–2754 (2025).
Mayer, E. L. et al. PACE:A randomized phase II study of fulvestrant, palbociclib, and avelumab after progression on cyclin-dependent kinase 4/6 inhibitor and aromatase inhibitor for hormone receptor-positive/human epidermal growth factor receptor-negative metastatic breast cancer [J]. J. Clin. Oncol. 42, 2050–2060 (2024).
Llombart-Cussac, A. et al. Second-line endocrine therapy (ET) with or without palbociclib (P) maintenance in patients (pts) with hormone receptor-positive (HR+)/human epidermal growth factor receptor2-negative (HER2-)advanced breast cancer (ABC): PALMIR Atrial [J]. J. Clin. Oncol. 41, 1001 (2023).
Kalinsky, K. et al. Randomized phase II trial of endocrine therapy with or without ribociclib after progression on cyclin-dependent kinase 4/6inhibition in hormone receptor-positive, human epidermal growth factor receptor 2-negative metastatic breast cancer: MAINTAIN trial [j]. J. Clin. Oncol. 41, 4004–4013 (2023).
Kalinsky, K. et al. Abemaciclib plus fulvestrant vs fulvestrant alone for HR+HER2-advanced breast cancer following progression on a prior CDK4/6 inhibitor plus endocrine therapy: primary outcome of the phase 3 postMONARCH trial [J]. J. Clin. Oncol. 42, LBA1001 (2024).
Dhakal, A. et al. Outcome of Everolimus-Based Therapy in Hormone-Receptor-Positive Metastatic Breast Cancer Patients After Progression on Palbociclib[J/OL]. Breast Cancer.: Basic Clin. Res. 14, 117822342094486 (2020).
Zhou, J. et al. Clinical outcomes of tucidinostat-based therapy after prior CDK4/6 inhibitor progression in hormone receptor-positive heavily pretreated metastatic breast cancer[J/OL]. Breast 66, 255–261 (2022).
Modi, S. et al. DESTINY-Breast04 Trial Investigators. Trastuzumab Deruxtecan in Previously Treated HER2-Low Advanced Breast Cancer. N. Engl. J. Med 387, 9–20 (2022).
Rugo, H. S. et al. Alpelisib plus fulvestrant in PIK3CAmutated, hormone receptorpositive advanced breast cancer after a CDK4/6 inhibitor (BYLieve): one cohort of a phase 2, multicentre,openlabel, noncomparative study[J]. Lancet Oncol. 22, 489498 (2021).
Hu, X. et al. Capivasertib plus fulvestrant in patients with HR-positive/HER2-negative advanced breast cancer: phase 3 CAPItello-291 study extended Chinese cohort. Nat. Commun. 16, 4324 (2025).
Bardia, A. et al. Abstract GS3-01:GS3-01 EMERALD phase 3 trial of elacestrant versus standard of care endocrine therapy in patients with ER+/HER2-metastatic breast cancer: updated results by duration of prior CDK4/6i in metastatic setting [J]. Cancer Res 83, GS3–1 (2023).
Cussac, L. A. et al. PARSIFAL-LONG: Extended follow-up of hormone receptor-positive/HER2-negative advanced breast cancer patients treated with fulvestrant and palbociclib vs letrozole and palbociclib in the PARSIFAL study. 2023 SABCS. RF01-03.
Princic, N. et al. Predictors of systemic therapy sequences following a CDK 4/6 inhibitor-based regimen in post-menopausal women with hormone receptor positive, HEGFR-2 negative metastatic breast cancer. Curr. Med Res Opin. 35, 73–80 (2019).
Carey, L. A. et al. Sacituzumab govitecan as second-line treatment for metastatic triple-negative breast cancer-phase 3 ASCENT study subanalysis. NPJ Breast Cancer 8, 72 (2022).
Yin, Y. et al. Sacituzumab tirumotecan in previously treated metastatic triple-negative breast cancer: a randomized phase 3 trial. Nat Med. 31,1969-1975 (2025).
Lee, T. H. et al. Vascular Endothelial Growth Factor Mediates Intracrine Survival in Human Breast Carcinoma Cells through Internally Expressed VEGFR1/FLT1[J/OL]. PLoS Med. 4, e186 (2007).
Linderholm, B. K. et al. Significantly higher levels of vascular endothelial growth factor (VEGF) and shorter survival times for patients with primary operable triple-negative breast cancer[J/OL]. Ann. Oncol. 20, 1639–1646 (2009).
Denardo, D. G. et al. Leukocyte Complexity Predicts Breast Cancer Survival and Functionally Regulates Response to Chemotherapy [J/OL]. Cancer Discov. 1, 54–67 (2011).
Salemme, V. et al. The role of tumor microenvironment in drug resistance: emerging technologies to unravel breast cancer heterogeneity[J/OL]. Front. Oncol. 13, 1170264 (2023).
Yang, C. et al. Increased drug resistance in breast cancer by tumor-associated macrophages through IL-10/STAT3/bcl-2 signaling pathway [J/OL]. Med. Oncol. 32, 14 (2015).
Xu, X. et al. M2 macrophage-derived IL6 mediates resistance of breast cancer cells to hedgehog inhibition [J/OL]. Toxicol. Appl. Pharmacol. 364, 77–82 (2019).
Syed, Y. Y. Anlotinib: First Global Approval[J/OL]. Drugs 78, 1057–1062 (2018).
Sun, Y. et al. Safety, pharmacokinetics, and antitumor properties of anlotinib, an oral multi-target tyrosine kinase inhibitor, in patients with advanced refractory solid tumors [J/OL]. J. Hematol. Oncol. 9, 105 (2016).
Lindgaard, S. C. et al. Circulating Protein Biomarkers for Use in Pancreatic Ductal Adenocarcinoma Identification [J/OL]. Clin. Cancer Res. 27, 2592–2603 (2021).
Acknowledgements
We gratefully acknowledge the support of all patients who participated in this study and their families, as well as the investigators and staff at each study site. This project was supported by the Beijing Xisike Clinical Oncology Research Foundation (Y-Young 2022—0072).
Author information
Authors and Affiliations
Contributions
T.X.: Collection and assembly of data, formal analysis, writing the original draft, manuscript review, and editing. Q.G.: Elisa analysis, collection, and assembly of data. S.Y.L.: formal analysis. L.L.Z.: Conceptualization, data curation, project administration, supervision; Y.Y.: Conceptualization, data curation, project administration, supervision, and writing–review and editing. Y.Y. and L.L.Z. are considered co-corresponding authors, and Y.Y. is the predominant corresponding author. All authors were responsible for the decision to submit the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Xu, T., Gu, Q., Li, S. et al. Phase II trial of anlotinib-chemotherapy combination in pretreated HER2-negative metastatic breast cancer: therapeutic efficacy and proteomic biomarker profiling. npj Breast Cancer 12, 47 (2026). https://doi.org/10.1038/s41523-026-00914-3
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41523-026-00914-3