Abstract
Pathogenic germline variants in telomerase (TERT) cause telomere biology disorders (TBDs) and are associated with bone marrow failure, pulmonary fibrosis, and other complications. TERT c.3150 G > C (p.K1050N) is frequent in the Ashkenazi Jewish (ASH) population and has been identified in ASH families with TBDs. Whole-genome sequencing of 96 p.K1050N heterozygotes from the UK Biobank and All of Us databases revealed a shared haplotype block, supporting a founder effect. Analyses of 15 additional p.K1050N cases validated this haplotype and identified mitochondrial and Y-STR haplogroups consistent with ASH ancestry. Clinical assessments showed that p.K1050N contributes to TBD phenotypes and shortened telomeres, while population data suggest incomplete penetrance. p.K1050N reduces telomerase activity and processivity, and decreases PCNA expression and BrdU incorporation, impairing cell proliferation. Our findings establish TERT p.K1050N as an ASH founder variant associated with TBDs, underscoring the need for genetic screening and long-term clinical studies.
Similar content being viewed by others
Introduction
Telomere biology disorders (TBDs) are a spectrum of illnesses associated with high rates of bone marrow failure, pulmonary fibrosis, liver disease, cancer, and other complications. They are caused by pathogenic or likely pathogenic (P/LP) germline variants in genes essential in telomere maintenance resulting in short and/or dysfunctional telomeres1. Heterozygous P/LP variants in telomerase (encoded by TERT) are a common cause of adult-onset TBDs and rare biallelic variants in patients with childhood-onset disease have been reported2.
The TERT single nucleotide variant (SNV) c.3150 G > C (p.K1050N, rs373400596 – hereafter referred to as “p.K1050N”) was first identified in an Ashkenazi Jewish (ASH) family affected by TBDs3. Although rare in the general population—with a global minor allele frequency (MAF) of 0.006% in gnomAD (v4.1.0)4—p.K1050N is ~40-fold more frequent in the ASH population (MAF: 0.236%). This elevated frequency, coupled with its presence in additional TBD families, suggests that p.K1050N is a founder ASH variant associated with TBDs. In this study, we explored the genetic origin of the p.K1050N variant and its biological consequences.
Results
Defining the p.K1050N haplotype
Whole genome sequencing (WGS) data from the UK Biobank (UKB) and the All of Us databases were used to characterize the p.K1050N haplotype. Phased WGS data from 13 UKB p.K1050N heterozygotes revealed shared haplotype blocks of varying lengths. Notably, all cases shared a common 497,929 base pair region (chr5:776,805–1,274,733, hg38) containing six rare single nucleotide variants (SNVs) in cis with p.K1050N (Fig. 1a). This full haplotype, which also encompasses the polymorphic microsatellite marker D5S1981, co-segregates with the p.K1050N variant in the three families included in this study (NCI-258, F339, and F552). The six rare SNVs were also present in six independent clinical cohort cases (ID 4310355 and Geisinger, Table 1) and in 75 of 83 p.K1050N heterozygotes with unphased WGS data from the population sequencing databases (Fig. 1b). The remaining eight heterozygotes retained at least the two SNVs flanking p.K1050N, indicating partial haplotype conservation. Neither the full nor partial haplotypes were detected among the 199,975 phased WGS controls in the UKB database. Additionally, none of the 290,524 unphased WGS controls carried all six rare SNVs or the two flanking ones (Fig. 1b).
a Characterization of the region (chr5: 776,805–1,274,733; hg38) shared among all 13 UKB heterozygotes with phased WGS. b Allele frequency of the rare SNVs identified within the common haplotype in cases and controls. Abbreviations: SNV single-nucleotide variant, UKB UK Biobank, WGS whole-genome sequencing, WT wild-type, AF allele frequency. Created in BioRender. De Andrade, K. (2025) https://BioRender.com/n76x380.
Ancestry analysis
Mitochondrial DNA (mtDNA) analysis showed that the 15 individuals with the TERT p.K1050N variant from the clinical cohorts belonged to either macrohaplogroup R (which includes haplogroups H, HV, K and T) or macrohaplogroup N (which includes haplogroup I) (Table 1). Y-STR analysis identified macrohaplogroups J, R, and G. Notably, male family members from two unrelated families (F339 and F552) share the same Y-STR subclade, R1aM198 (Table 1).
Clinical manifestations and telomere length
Among individuals in the clinical cohorts (Table 1), the earliest TBD-associated diagnoses were observed in the homozygous proband (NCI-258-1), whereas five heterozygotes (F339, F552, and ID 4310355) exhibited a later onset of TBD-associated conditions. All individuals with available clinical telomere length (TL) measurements (n = 10) had TL near or below the 10th percentile compared with age-matched populations (Table 1).
Among the 96 p.K1050N heterozygotes identified in the population sequencing databases, one male individual (AoU-1), who also carried the RTEL1 c.3791 G > A founder ASH variant, developed TBD-related diagnoses in his 40s. Other participants reported various cancers and clinical symptoms potentially associated with TBDs (Table 1). Unfortunately, electronic health records (EHR) were unavailable for 17 out of 64 All of Us participants. In the UKB, four out of 32 heterozygotes had no diagnoses reported in the data fields assessed (Methods). TL varied widely among UKB heterozygotes, with a notable tendency toward shorter TL compared with the median of the control population. Among 27 individuals with available TL data, seven had TL below the 10th percentile for their age (Table 1, Supplementary Fig. 1).
Structural and functional impact of the TERT p.K1050N variant
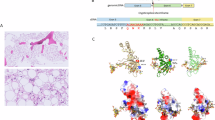
Residue p.K1050 is located in the C-terminal extension (CTE) domain of TERT. FoldX calculations predicted that substitution of lysine with asparagine at this position would be tolerated (Fig. 2). Our analysis demonstrates that p.K1050N does not impact hTERT expression or stability, or hTR association with hTERT (Supplementary Fig. 2). We next characterized the functional impact of p.K1050N on telomerase activity and repeat addition processivity. Compared with wild-type (WT) levels, the p.K1050N variant reduced telomerase activity (Fig. 3a) and processivity (Fig. 3b) by 24% and 33%, respectively. When co-expressed with WT telomerase to mimic a heterozygous state, the reduction in activity was no longer statistically significant (Fig. 3b); however, a significant 12% decrease in processivity remained (Fig. 3c). These reductions were comparable to or greater than those observed for the TERT p.V694M variant (Supplementary Fig. 3). Moreover, the p.K1050N variant reduced both PCNA expression and bromodeoxyuridine (BrdU) incorporation in primary patient-derived fibroblasts, particularly in the homozygous state, indicating impaired cell proliferation (Supplementary Fig. 4).
a The location of residue position 1050 in the cryo-EM structure of telomerase in complex with the RNA:DNA hybrid is shown (PDB code 5BG9)44. Residue 1050 and the RNA strand are shown in a surface representation with the experimental structure of K1050. b Lowest energy rotamer of asparagine as determined by PyMol40.
a Direct telomerase activity assay of immunopurified telomerase expressed in HEK293T cells, using plasmid encoding WT TERT, the p.K1050N variant, or an equimolar mixture of WT and p.K1050N. Each prominent band represents the addition of a TTAGGG repeat to the DNA substrate, with the number of nucleotides added indicated on the left. LC: labelled, biotinylated 30nt oligonucleotide recovery and loading control. b Specific activity of K1050N and K1050N + WT, compared to WT telomerase. c Processivity of WT, K1050N and K1050N + WT, relative to WT telomerase. Processivity is defined as the average number of DNA repeats synthesized before the enzyme dissociation from DNA. Data points represent independent immunopurified telomerase samples, with error bars indicating mean + SEM (n = 3). Abbreviations: WT wild-type. Plots created with GraphPad Prism version 10.0.0 (www.graphpad.com).
Discussion
In this study, we present evidence that TERT c.3150 G > C (p.K1050N) is a founder variant in the ASH population. The variant’s pathogenic potential is demonstrated by impaired telomerase activity and processivity, as well as reduced cell proliferation, highlighting its clinical relevance in TBDs.
The founder effect in the ASH population is supported by the identification of a haplotype block shared exclusively among individuals with the p.K1050N variant and by our mtDNA and Y-STR findings. Both mtDNA macrohaplogroups identified (R and N) include haplogroups that are commonly observed in ASH maternal lineages throughout Europe5. For instance, haplogroup K is the most prevalent among the ASH population, found in approximately 32% of individuals6, compared with only ~6% in Near Eastern and European non-Jewish populations7. The Y-STR macrohaplogroups J, R, and G account for approximately 38%, 19%, and 10% of all ASH paternal lineages in Europe, respectively8. The presence of the same Y-STR subclade in unrelated families reinforces a shared genetic origin. The high frequency of the p.K1050N variant in the ASH population, along with these mtDNA and Y-STR findings, strongly supports p.K1050N as a distinct founder variant within this population.
Data from our clinical databases demonstrate that the p.K1050N variant can lead to short telomeres and to the subsequent development of TBD-associated phenotypes, consistent with previous reports3,9. Although only a single p.K1050N homozygote was identified, our results suggest that the homozygous state may result in earlier or more aggressive phenotypes. Members of family F552 were also heterozygous for the RTEL1 c.3791 G > A (p.R1264H) variant, previously identified as a recessive ASH founder variant in TBD families10. While the clinical and functional consequences of RTEL1 p.R1264H heterozygosity have not been well studied, it is possible that both variants may have contributed to the clinical manifestations observed in family F552. Longitudinal studies of individuals with TERT p.K1050N, particularly homozygotes, with or without RTEL1 p.R1264H are essential to better characterize the phenotypic spectrum and refine risk estimates.
Data from the population sequencing databases demonstrated variability in clinical presentation of p.K1050N heterozygotes, suggesting that disease expression may be influenced by additional genetic modifiers or other risk factors, such as aging and smoking. There was no clear association between TL and the development of TBD-associated conditions in the UKB, likely due to the small sample size. Importantly, while qPCR is a cost-effective approach for TL measurement in large populations, it is not sensitive enough for clinical diagnoses of individual patients11,12. The low prevalence of TBD manifestations in the population sequencing databases could be attributed to the lack of complete clinical data for a subset of individuals, and database-related intrinsic survival biases, exacerbated by the “healthy volunteer effect” documented in the UKB13. Consequently, the clinical findings reported in these databases likely underestimate the full TBD phenotypic spectrum associated with the p.K1050N variant.
A previously identified TBD-associated lysine-to-glutamate substitution (p.K1050E) impaired the CTE domain’s nucleic acid binding affinity, reducing both telomerase activity and processivity by ~2-fold14,15, and reducing binding of the CTE domain to an RNA:DNA hybrid in vitro14. The p.K1050N mutation likely disrupts interactions with the nucleic acid phosphate backbone due to the absence of a positive charge in the asparagine side chain. While our computational modeling suggests that p.K1050N may impair these interactions, direct experimental evidence for p.K1050N requires further investigation.
The p.K1050N variant reduced telomerase activity and processivity to levels similar to those of the TERT p.V694M variant, which served as a benchmark for P/LP-associated deficits in our functional assay16. Of note, in vitro telomerase activity for variants with processivity defects can be influenced by enzyme turnover and may not accurately reflect in vivo activity17, as observed with another variant affecting the same codon, TERT p.K1050E15. Nonetheless, defects in telomerase processivity have been shown to impact telomere length maintenance in vivo17,18. The impact of the p.K1050N variant was further substantiated by its reductions in PCNA expression and BrdU incorporation in primary patient-derived fibroblasts, particularly in the homozygous state. These results provide evidence that p.K1050N impairs telomerase function and cellular processes critical for telomere maintenance, underscoring the pathogenic nature of the p.K1050N variant and its significant role in TBDs.
Current variant classification guidelines from the American College of Medical Genomics and the Association for Molecular Pathology (ACMG/AMP)19 do not fully address curation of variants with incomplete penetrance or founder effects. Based on adapted criteria for TBD-associated genes, the p.K1050N variant was recently classified as likely pathogenic1. Our data corroborate this classification and provide additional clinical, in-silico, and functional evidence linking it to relevant TBD phenotypes. Therefore, we recommend genetic screening in at-risk populations and following TBD clinical surveillance guidelines20 for individuals with the p.K1050N variant.
Our study identifies TERT c.3150 G > C (p.K1050N) as a founder variant in the ASH population. The variant’s pathogenic potential is supported by reduced telomerase activity, processivity, and cell proliferation, along with shortened telomeres and association with typical TBD phenotypes. Longitudinal studies are encouraged to fully characterize the variant’s phenotypic spectrum and refine clinical management strategies.
Methods
Subjects (clinical cohorts)
Members of the NCI-258 family (n = 4, one homozygote, three heterozygotes), self-reported as Ashkenazi Jewish, were enrolled in the National Institutes of Health (NIH) institutional review board-approved Cancer in Inherited Bone Marrow Failure Syndromes (IBMFS) study (clinicaltrials.gov, Identifier: NCT00027274, registration date: November 30th, 2001)21 sponsored by the National Cancer Institute (NCI). Each individual underwent comprehensive evaluations at the NIH Clinical Center, including diagnostic imaging and blood tests per study protocol. The TERT c.3150 G > C (p.K1050N) variant was identified via Sanger sequencing at the NCI’s Cancer Genomic Research Laboratory and confirmed in a Clinical Laboratory Improvement Amendments (CLIA)-certified facility. Variants in TERC, MPL, and RTEL1 p.R1264H were not identified in family NCI-258 via Sanger sequencing.
Members of families F339 (n = 3 heterozygotes) and F552 (n = 2 heterozygotes), and one unrelated heterozygote (ID 4310355), participated in the IRB-AAAS0753 and IRB-AAAR1916 studies at Columbia University Medical Center. All individuals self-reported Non-Hispanic White ancestry. The proband of family F339 had previously been identified with the TERT c.3150 G > C variant in an interstitial lung disease study22. Variants in TERT, TERC, and RTEL1 p.R1264H were not identified in family F339 via Sanger sequencing.
Whole-genome sequencing (WGS)23 was performed on the proband of family F552 and ID 4310355, assessing 28 telomere biology disorder (TBD)-associated genes (ACD, ACYP2, CBX3, CTC1, DKC1, GAR1, NAF1, NHP2, NOP10, OBFC1, PARN, POT1, RAP1, RPA1, RTEL1, STN1, TEN1, TEP1, TERC, TERF1, TERF2, TERF2IP, TERT, TINF2, TNKS, WRAP53, ZCCHC8, and ZNF208). In addition to the TERT c.3150 G > C variant, the RTEL1 c.3791 G > A (p.R1264H) was also identified in the family F552 proband and later confirmed in his brother via Sanger sequencing. No other TBD-related variants were identified in ID 4310355.
Five p.K1050N heterozygotes (ages at recruitment: 20, 61, 65, 67, and 85 years) were identified in the Geisinger DiscovEHR cohort using whole-exome sequencing (WES)24. None of them carried the RTEL1 c.3791 G > A variant. These individuals were determined to be of European ancestry through principal component (PC) analysis of WES data25 (Supplementary Fig. 5). Electronic health records (EHR) and cancer registry data were retrieved, and a thorough review of clinical charts was conducted to capture all relevant diagnoses affecting the blood, gastrointestinal tract, lung, liver, or skin.
Genomic DNA from all individuals was available for haplotype and ancestry analyses. Informed consent was obtained from all participants. This study was conducted in accordance with all relevant ethical regulations, including the Declaration of Helsinki26.
Population sequencing databases
UK Biobank (UKB) WGS data27,28,29 were accessed via the UKB DNA Nexus Research Platform. WGS was available for 490,499 individuals (data field 23374)30; with a subset of 199,975 individuals also having phased WGS data (data field 20279)31. We identified 32 TERT c.3150 G > C heterozygotes (median age at recruitment: 55 years, range 40–68 years), including 13 with also phased WGS data. No co-occurrence with the RTEL1 c.3791 G > A variant was identified. Disease-related data fields queried included 40001 (underlying cause of death), 40006 (type of cancer ICD10), 40008 (age at cancer diagnosis), 40011 (histology of cancer tumor), 40012 (behavior of cancer tumor), 40013 (type of cancer ICD9), 41270 (diagnoses ICD10), 41271 (diagnoses ICD9), and 41280 (Date of first in-patient diagnosis - ICD10). Phenotype data were extracted in July 2024. Ancestry information from the UKB allele variant browser, classified 18 heterozygotes as ASH and 14 as Non-Finnish European. Participants who withdrew from the UKB cohort (n = 351; as of July 2024) were excluded from all analyses.
Additionally, WGS was available for 414,822 individuals from the All of Us Research Program database (version 8)32,33. We identified 64 TERT c.3150 G > C heterozygotes (35 males and 29 females), including 48 of European ancestry and 16 with ancestry categorized as “Remaining”, according to the All of Us public data browser. Median age at recruitment was 66.2 years (range 21.6–92.3 years). Two individuals were also heterozygous for the RTEL1 c.3791 G > A variant. Clinical and genetic data for these individuals were obtained through approved access to the Controlled Tier (CDRv8), in accordance with the program’s data use policies. All analyses adhered to the ethical and regulatory standards set by the All of Us Research Program. Participants provided informed consent to use their de-identified genetic and clinical information for research purposes. We received an exception to the Data and Statistics Dissemination Policy from the All of Us Resource Access Board.
For both databases, phenotype analyses included primary malignant neoplasms (excluding non-melanoma skin cancer and uterine cervix cancer) and TBD-related diseases affecting the blood, gastrointestinal tract, lung, liver, or skin.
Genetic profiling
Short tandem repeat (STR) profiling was performed using the PowerPlex Fusion STR system (Promega, Madison, WI, USA), which simultaneously amplifies 24 STR loci from DNA. This system utilizes five fluorescent dyes attached to primers to label the amplified PCR fragments. The dye-labeled PCR products were separated and detected with a 3500XL Genetic Analyzer with POP-7 polymer. Data analysis was conducted using GeneMapper 6.0 (ThermoFisher Scientific).
Relatedness evaluation
STR profiling found no evidence of relatedness among participants from the clinical cohorts (data not shown to preserve participants’ confidentiality). In Geisinger DiscovEHR, identity-by-descent analysis (PLINK 1.934) further confirmed no relatedness, up to the third degree, based on pairwise pi-hat values. Kinship coefficients in the UKB28 and All of Us33 identified two pairs of p.K1050N heterozygotes in each database with kinship scores above 0.1, indicating first- or second-degree relationships.
Microsatellites
Fluorescent-labelled PCR products for four polymorphic microsatellite markers (D5S392, D5S1981, D5S2005, and D5S678; Supplementary Table 1) located within chr5:302,025–1,365,914 (hg38) were genotyped using the ABI 3500 sequencer (ThermoFisher Scientific). Fragment sizes were assigned using GeneMapper v6.0 (ThermoFisher Scientific).
Defining a founder haplotype
Haplotype profiles of chromosome 5p, covering the region 13,600–10,400,000 (GRCh38/hg38), were analyzed in 13 TERT c.3150 G > C heterozygotes from the UKB with phased WGS. The smallest shared haplotype on chromosome 5p, encompassing c.3150 G > C, was identified across all these individuals. To define a founder haplotype, we filtered this region for single nucleotide variants (SNVs) with a global minor allele frequency (MAF) < 1% in gnomAD v.4.1 non-UKB genomes4, resulting in six rare SNVs. These SNVs were subsequently genotyped by Sanger sequencing (Supplementary Table 1) in all 15 individuals. Furthermore, these SNVs were investigated in the unphased WGS data from the UKB and All of Us population databases.
Haplotypes were determined by segregation analysis of SNVs and polymorphic markers in the NCI-258, F339, and F552 families, and were inferred in the independent heterozygotes without additional family members included in the study (ID 4310355 and Geisinger). Phased WGS data from 199,975 UKB controls (individuals without TERT c.3150 G > C) were evaluated to assess the frequency of the haplotype containing the alternate alleles for the six rare SNVs in cis with the wild-type (WT) allele in the chr5:1,255,294 position (A-C-T-A-A-C-T; WT allele underlined). Unphased WGS data from the remaining 290,514 UKB controls were analyzed to further assess the allele frequency of the six SNVs within the UKB database.
Mitochondrial DNA control region
The nucleotide sequences of the mitochondrial DNA (mtDNA) hypervariable segments 1 (HVI; positions 16024 and 16365), 2 (HV2; positions 73 and 340), and 3 (HV3, positions 438 to 574) within the control region of mtDNA were determined in all individuals from the clinical cohorts. The mtDNA control region was amplified using the primers L15781 (5’-CCCTTTTACCATCATTGGACA-3’) and H727 (5’-AGGGTGAACTCACTGGAACG-3’). Sequencing of the amplified segments was carried out with primers L15781, H16478, L109, H408, and H727, as previously described35,36. The resulting sequences were compared to the Homo sapiens mitochondrial genome (NCBI reference sequence: NC_012920.1), also known as the revised Cambridge Reference Sequence (rCRS). mtDNA haplogroups were identified using Haplogrep software (https://haplogrep.i-med.ac.at/), EMPOP (https://empop.online/), and visual inspection.
Y-chromosome markers
Twenty-three Y-STRs were analyzed for five males from clinical cohorts using the PowerPlex Y23 System (Promega), following the manufacturer’s instructions. The alleles were separated and detected with an ABI 3500 sequencer (ThermoFisher Scientific) and the fragment sizes were assigned using GeneMapper v 6.0 (ThermoFisher Scientific). The alleles were named according to the number of repeated units, as per the sequenced allelic ladder and the recommendations of the International Society for Forensic Genetics37. Y-STRs haplogroup were classified using the Haplogroup Predictor program FTDNA 2.0 (http://www.hprg.com/hapest5/index.html) and the Y-chromosome Haplotype Reference Database (https://yhrd.org/search).
Telomere length measurement
Lymphocyte telomere lengths were measured by flow FISH at Repeat Diagnostics, Inc. (Vancouver, BC, Canada) for the NCI-258 family members, with percentiles calculated based on clinical testing parameters38. Leukocyte telomere lengths (LTL) were measured by qPCR for members of families F339, F552, and ID 4310355, as previously described22. Telomere length measurements were not determined for Geisinger participants (n = 5).
In the UKB, LTL was determined by qPCR (data field 22192, Z-adjusted T/S log) and was available for 27 out of 32 p.K1050N heterozygotes. Percentiles were calculated based on LTL data from UK controls (n = 460,541) which excluded participants (n = 12,128) with rare coding germline TERT variants (MAF < 1% in gnomAD v4.1.0 non-UKB). Telomere length data were not available in the All of Us database.
Computational modelling of structural impact
Structural effects of lysine to asparagine (p.K1050N) in the TERT protein were predicted using FoldX39, using the reported crystal structure14, and visualized by PyMOL (https://pymol.org/2/)40.
Telomerase activity and processivity (direct assay)
The TERT c.3150 G > C variant was introduced by site-directed mutagenesis into a plasmid encoding FLAG-tagged hTERT under a CMV promoter. This plasmid was transfected into HEK293T cells along with a plasmid encoding TERC (the RNA component of telomerase, hTR) and DKC1 (dyskerin)41. Wild-type (WT) hTERT was transfected as a control, and a third transfection included equimolar amounts of WT and c.3150 G > C, to represent the heterozygous state. Telomerase was immunopurified using anti-FLAG M2 antibody affinity gel (Sigma), and hTERT levels were quantified by Western blot using an anti-FLAG antibody41 (Supplementary Table 2). Binding of hTERT to hTR (i.e. the potential impact of each variant on enzyme assembly) was assessed by measuring the amount of hTR recovered after immunopurification (IP) of hTERT by northern dot-blot41. The relative amounts of recovered hTR were normalized to the relative amounts of recovered hTERT (i.e. [% hTR relative WT]/[% hTERT relative to WT] in the IP eluate was calculated), and the result was also normalized to the initial hTR levels in the cell lysate to control for differences in expression from the plasmid encoding hTR. Equal volumes of immunopurified telomerase were used in direct telomerase activity assays, involving extension of a telomeric primer with radiolabeled nucleotides, followed by electrophoresis on a high-resolution acrylamide gel41.
Telomerase activity (the enzyme’s ability to add nucleotides to a DNA molecule) was quantified by analyzing the sample lanes and normalizing the results to the amount of hTR recovered. The activity was expressed as a percentage of WT telomerase activity. Processivity (the ability of telomerase to continuously extend a single DNA molecule) was quantified as previously described42.
A control panel consisting of three TERT variants previously classified16 as pathogenic or likely pathogenic (P/LP) - p.K570N, p.R865H, and p.V694M—and four TERT variants classified as benign or likely benign (B/LB)—p.A279T, p.A1062T, p.S191T, and p.E280K - was included in the direct telomerase activity assay.
PCNA protein expression
Primary fibroblasts from NCI-258 family members were used to assess PCNA protein expression, a marker of cell proliferation43. Primary fibroblasts from NCI-258 family members and paired controls (Coriell Cell Repository) (Supplementary Table 3) were cultured in AmnioMAXTM-II Complete Medium (Thermo Fisher Scientific) supplemented with 1% Penicillin-Streptomycin. All cells were maintained in a 5% CO2 incubator at 37°C. Cultured fibroblasts were harvested and lysed in RIPA buffer (Sigma) supplemented with a dual protease/phosphatase inhibitor cocktail (Thermo Fisher Scientific) for 30 min on ice. The extracted proteins were separated on 4–12% Bis-Tris gels (Thermo Fisher Scientific) and transferred onto polyvinylidene difluoride (PVDF) membranes (MilliporeSigma). The membranes were incubated overnight at 4 °C with the indicated primary antibodies, followed by near-infrared fluorescent conjugated secondary antibodies (Supplementary Table 2). Protein expression was visualized by Odyssey DLx near-infrared imager and quantified with the Empiria Studio Software (LICORbio).
BrdU cell proliferation assay
Primary fibroblasts from NCI-258 family members and their matched controls were seeded in 96-well plates (CELLTREAT) at a density of 5000 cells/well in 100 µL/well of AmnioMAXTM-II Complete Medium, with 4 replicates/sample. The control wells contained only medium without cells. After 24 h of culture, BrdU reagent was added in the cell media at 1:500 and incubated for an additional 24 h. BrdU cell proliferation assay (Abcam) was conducted according to the manufacturer’s instructions. Absorbance was measured at dual wavelengths of 450/560 nm using a GloMax Explorer plate reader (Promega). Sample absorbance was calculated by subtracting the absorbance of the blank controls.
Data availability
UK Biobank data are available to approved researchers upon application. Access requires completion of a data access application, which includes a research proposal and approval from the UK Biobank's Ethics and Governance Council. Due to privacy and ethical considerations, data are not publicly available. Access to MyCode data is restricted and requires approval from the Geisinger Institutional Review Board (IRB) and adherence to the MyCode data access policies. These data are not publicly available due to privacy and ethical restrictions. Researchers interested in accessing the MyCode data should submit a formal request through the MyCode Research Portal. Access to “All of Us” data is available through the All of Us Research Hub, where researchers can apply for access to participant data. Data are available to approved researchers who have completed the necessary training and data use agreements. Due to the sensitive nature of the data, access is governed by the All of Us Institutional Review Board (IRB) and Data and Research Center (DRC), and the National Cancer Institute does not have authority to share any of these data. Authors have received an exception to the Data and Statistics Dissemination Policy from the All of Us Resource Access Board.
Code availability
We utilized publicly available bioinformatics tools and libraries, including BCFtools and UNIX commands for data processing. Python libraries such as Pandas for data manipulation, NumPy for numerical operations, Matplotlib and Seaborn for data visualization. All these tools are freely accessible, and detailed documentation can be found through their respective repositories or platforms. Additionally, the UK Biobank “Swiss Army knife” tool was employed for general data handling within the Research Hub. No new code was developed for this study.
References
Niewisch, M. R. et al. Disease progression and clinical outcomes in telomere biology disorders. Blood 139, 1807–1819 (2022).
Revy, P., Kannengiesser, C. & Bertuch, A. A. Genetics of human telomere biology disorders. Nat. Rev. Genet. 24, 86–108 (2023).
Collopy, L. C. et al. Triallelic and epigenetic-like inheritance in human disorders of telomerase. Blood 126, 176–184 (2015).
Chen, S. et al. A genomic mutational constraint map using variation in 76,156 human genomes. Nature 625, 92–100 (2024).
Costa, M. D. et al. A substantial prehistoric European ancestry amongst Ashkenazi maternal lineages. Nat. Commun. 4, 2543 (2013).
Behar, D. M. et al. MtDNA evidence for a genetic bottleneck in the early history of the Ashkenazi Jewish population. Eur. J. Hum. Genet. 12, 355–364 (2004).
Richards, M. et al. Tracing European founder lineages in the Near Eastern mtDNA pool. Am. J. Hum. Genet 67, 1251–1276 (2000).
Behar, D. M. et al. Contrasting patterns of Y chromosome variation in Ashkenazi Jewish and host non-Jewish European populations. Hum. Genet 114, 354–365 (2004).
Gerbing, R. B. et al. Shorter remission telomere length predicts delayed neutrophil recovery after acute myeloid leukemia therapy: a report from the Children’s Oncology Group. J. Clin. Oncol. 34, 3766–3772 (2016).
Ballew, B. J. et al. A recessive founder mutation in regulator of telomere elongation helicase 1, RTEL1, underlies severe immunodeficiency and features of Hoyeraal Hreidarsson syndrome. PLoS Genet. 9, e1003695 (2013).
Gutierrez-Rodrigues, F., Santana-Lemos, B. A., Scheucher, P. S., Alves-Paiva, R. M. & Calado, R. T. Direct comparison of flow-FISH and qPCR as diagnostic tests for telomere length measurement in humans. PLoS One 9, e113747 (2014).
Gadalla, S. M. et al. The limitations of qPCR telomere length measurement in diagnosing dyskeratosis congenita. Mol. Genet. Genom. Med. 4, 475–479 (2016).
Fry, A. et al. Comparison of Sociodemographic and Health-Related Characteristics of UK Biobank Participants With Those of the General Population. Am. J. Epidemiol. 186, 1026–1034 (2017).
Hoffman, H., Rice, C. & Skordalakes, E. Structural analysis reveals the deleterious effects of telomerase mutations in bone marrow failure syndromes. J. Biol. Chem. 292, 4593–4601 (2017).
Niaz, A. et al. Functional interaction between compound heterozygous TERT mutations causes severe telomere biology disorder. Blood Adv. 6, 3779–3791 (2022).
Nelson, N. et al. Functional genomics for curation of variants in telomere biology disorder associated genes: a systematic review. Genet. Med. 25, 100354 (2023).
Wu, R. A., Tam, J. & Collins, K. DNA-binding determinants and cellular thresholds for human telomerase repeat addition processivity. EMBO J. 36, 1908–1927 (2017).
Alder, J. K. et al. Ancestral mutation in telomerase causes defects in repeat addition processivity and manifests as familial pulmonary fibrosis. PLoS Genet. 7, e1001352 (2011).
Richards, S. et al. Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet. Med. 17, 405–424 (2015).
Agarwal, S., Savage, S. A., Stevens, K. B., Raj, H. & Carson, H. K. Telomere biology disorders: diagnosis and management guidelines: Second Edition, (Team Telomere Inc, New York, NY, 2022).
Alter, B. P., Giri, N., Savage, S. A. & Rosenberg, P. S. Cancer in the National Cancer Institute inherited bone marrow failure syndrome cohort after fifteen years of follow-up. Haematologica 103, 30–39 (2018).
Newton, C. A. et al. Telomere-related lung fibrosis is diagnostically heterogeneous but uniformly progressive. Eur. Respir. J. 48, 1710–1720 (2016).
Zhang, D. et al. Utility of whole genome sequencing in assessing risk and clinically relevant outcomes for pulmonary fibrosis. Eur. Respir. J. 60, 2200577 (2022).
Carey, D. J. et al. The Geisinger MyCode community health initiative: an electronic health record-linked biobank for precision medicine research. Genet. Med. 18, 906–913 (2016).
Staples, J. et al. Profiling and leveraging relatedness in a precision medicine cohort of 92,455 exomes. Am. J. Hum. Genet. 102, 874–889 (2018).
World Medical, A. World Medical Association Declaration of Helsinki: ethical principles for medical research involving human subjects. JAMA 310, 2191–2194 (2013).
Sudlow, C. et al. UK biobank: an open access resource for identifying the causes of a wide range of complex diseases of middle and old age. PLoS Med. 12, e1001779 (2015).
Bycroft, C. et al. The UK Biobank resource with deep phenotyping and genomic data. Nature 562, 203–209 (2018).
Backman, J. D. et al. Exome sequencing and analysis of 454,787 UK Biobank participants. Nature 599, 628–634 (2021).
Li, S., Carss, K. J., Halldorsson, B. V. & Cortes, A. Whole-genome sequencing of half-a-million UK Biobank participants. Preprint at medRxiv, 2023.12.06.23299426 (2023).
review : Journal Type Hofmeister, R. J., Ribeiro, D. M., Rubinacci, S. & Delaneau, O. Accurate rare variant phasing of whole-genome and whole-exome sequencing data in the UK Biobank. Nat. Genet. 55, 1243–1249 (2023).
All of Us Research Program, I et al. The “All of Us” research program. N. Engl. J. Med. 381, 668–676 (2019).
All of Us Research Program Genomics, I. Genomic data in the All of Us research program. Nature 627, 340–346 (2024).
Purcell, S. et al. PLINK: a tool set for whole-genome association and population-based linkage analyses. Am. J. Hum. Genet 81, 559–575 (2007).
Goncalves, F. T. et al. The E180splice mutation in the GHR gene causing Laron syndrome: witness of a Sephardic Jewish exodus from the Iberian Peninsula to the New World?. Am. J. Med. Genet. A 164A, 1204–1208 (2014).
Pinto, E. M. et al. Multiple TP53 p.R337H haplotypes and implications for tumor susceptibility. HGG Adv. 5, 100244 (2024).
Roewer, L. et al. DNA commission of the International Society of Forensic Genetics (ISFG): recommendations on the interpretation of Y-STR results in forensic analysis. Forensic Sci. Int. Genet. 48, 102308 (2020).
Alter, B. P. et al. Telomere length is associated with disease severity and declines with age in dyskeratosis congenita. Haematologica 97, 353–359 (2012).
Schymkowitz, J. et al. The FoldX web server: an online force field. Nucleic Acids Res. 33, W382–W388 (2005).
Schrodinger, L. L. C. The PyMOL Molecular Graphics System, Version 3. (2015).
Tomlinson, C. G., Sasaki, N., Jurczyluk, J., Bryan, T. M. & Cohen, S. B. Quantitative assays for measuring human telomerase activity and DNA binding properties. Methods 114, 85–95 (2017).
Latrick, C. M. & Cech, T. R. POT1-TPP1 enhances telomerase processivity by slowing primer dissociation and aiding translocation. EMBO J. 29, 924–933 (2010).
Moldovan, G. L., Pfander, B. & Jentsch, S. PCNA, the maestro of the replication fork. Cell 129, 665–679 (2007).
Ghanim, G. E. et al. Structure of human telomerase holoenzyme with bound telomeric DNA. Nature 593, 449–453 (2021).
Acknowledgements
We are deeply grateful to the participants from the NCI, Columbia University, UKB, All of Us, and Geisinger MyCode research programs for their invaluable contributions that made this research possible. We also thank the Regeneron Genetics Center and the NIH’s All of Us Research Program for granting access to the clinical and genetic data utilized in this study. This research was funded by the Intramural Research Program of the Division of Cancer Epidemiology and Genetics at the NCI, NIH, Bethesda, MD. We also received additional support through the Team Telomere 2024 Micro Grant Award (to KCA). Telomerase functional analyses were supported by Luminesce Alliance Innovation for Children’s Health, a not-for-profit cooperative joint venture between the Sydney Children’s Hospitals Network, Children’s Medical Research Institute, the Children’s Cancer Institute, the University of Sydney, and the University of New South Wales, established with the support of the New South Wales (Australia) government, and donations from the Arcus and Neil and Norma Hill Foundations (to TMB). Evaluation of F339, F552, and ID 4310355 was supported by R01 HL093096 (to CKG). This study was conducted using the UK Biobank Resource under application #54389. The MyCode project operates under protocol #2006-0258, and the NCI-Geisinger collaboration is approved under IRB #2021-0818 and contract #75N91021C00006. The All of Us Research Program is supported by the National Institutes of Health, Office of the Director: Regional Medical Centers: 1 OT2 OD026549; 1 OT2 OD026554; 1 OT2 OD026557; 1 OT2 OD026556; 1 OT2 OD026550; 1 OT2 OD 026552; 1 OT2 OD026553; 1 OT2 OD026548; 1 OT2 OD026551; 1 OT2 OD026555; IAA #: AOD 16037; Federally Qualified Health Centers: HHSN 263201600085U; Data and Research Center: 5 U2C OD023196; Biobank: 1 U24 OD023121; The Participant Center: U24 OD023176; Participant Technology Systems Center: 1 U24 OD023163; Communications and Engagement: 3 OT2 OD023205; 3 OT2 OD023206; and Community Partners: 1 OT2 OD025277; 3 OT2 OD025315; 1 OT2 OD025337; 1 OT2 OD025276. This work utilized the computational resources of the NIH High-Performance Computing Biowulf cluster.
Funding
Open access funding provided by the National Institutes of Health.
Author information
Authors and Affiliations
Contributions
K.C.A., E.M.P., and S.A.S. conceptualized the study. K.C.A., E.M.P., L.P.Z., J.K., T.Z., N.G., J.S.H., O.F.V., S.A.A., D.T.S., and G.M.U. curated and verified the data. N.G., L.J.M., S.A.S., and C.K.G. managed the families involved. K.C.A., E.M.P., L.P.Z., T.Z., S.B.C., and M.R.E. conducted the formal analysis. K.C.A. and S.A.S. secured funding. K.C.A., E.M.P., T.M.B., D.J.C., C.K.G., D.R.S., and S.A.S. led the investigation. K.C.A. and E.M.P. performed the genetic/haplotype analysis. E.M.P., G.P.Z., and C.F. conducted genetic sequencing. L.P.Z., T.Z., S.B.C., M.R.E., T.M.B, A.H.P, R.W.K. conducted the functional assays. K.C.A., E.M.P., and S.A.S. administered the project. S.A.S. supervised the study. K.C.A. and E.M.P. wrote the manuscript. All authors provided comments and edited the text.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
de Andrade, K.C., Pinto, E.M., Zhao, T. et al. TERT c.3150 G > C (p.K1050N): a founder Ashkenazi Jewish variant associated with telomere biology disorders. npj Genom. Med. 10, 46 (2025). https://doi.org/10.1038/s41525-025-00501-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41525-025-00501-8