Abstract
Growing food crops for space missions requires significant improvements in technical competence. Many issues remain, including ensuring that roots grow vertically in low gravity. Although plant roots grow towards gravity, they also perceive and bend away from light, allowing for light to substitute for gravity. To explore this issue, we designed a 3D-printed mini-phytotron with adjustable light-emitting diodes to use with a random positioning machine (RPM). Simulated microgravity in the RPM, together with darkness, caused Arabidopsis roots to lose vertical perception, resulting in significantly altered root morphology parameters consistent with gravity loss. This validated the method as an Earth-based analogue and allowed us to test the addition of light. White light as low as 10 μmol m−2 s−1 compensated for simulated microgravity in the RPM. Red light was less effective than white, and white light at 1 μmol m−2 s−1 was much less effective. A dwarf variant of Arabidopsis responded similarly to the wild type, and lettuce roots also responded to light. Food plants in space will require much higher levels than 10 μmol m−2 s−1 for photosynthesis, so there are good prospects that light in growth facilities in space will replace gravity for normal root growth, as long as roots can be exposed to some light. The RPM combined with the mini-phytotron was developed here as an inexpensive Earth-based analogue to analyse root growth behaviour to changing light levels under varying gravity conditions and will serve as a valuable experimental platform for further dissection of light responses in roots.
Similar content being viewed by others
Introduction
One of the greatest challenges to long-term space missions is providing cost-effective, nutritious, and appetising food to astronauts1,2. Growing and eating fresh food is likely to be needed to supplement stored, processed foods, and may help to improve mood during a space mission or in future Moon or Mars bases3. In addition to positive psychology, plants can supply a range of benefits such as biosynthesis of nutrients, fibres and medicines, and bioremediation of waste products1. Plants can grow in relatively low air pressure and high carbon dioxide, which may provide versatility for integrating plant growth areas into space habitation design2,4.
A range of plant species have been grown in the NASA Vegetable Production System and Advanced Plant Habitat, including lettuce, radish and dwarf wheat1. These trials have confirmed that space food crops are possible and edible. However, little is known about yield and efficiency, and many engineering issues need to be solved, such as providing enough room, developing highly efficient and compact plant varieties, recycling plant waste and the influence of radiation on plants and seeds5. Banks of Light Emitting Diodes (LEDs) growing compact crop varieties may be required, and these may need to be incorporated in human-use zones to maximise area use. The impact of gravity (g) that is low or zero (microgravity) is not yet fully understood, in particular, the effect of light wavelengths on root bending (tropism) in microgravity. Moreover, more information about any deficiencies in the growth of compact dwarf plants would be useful.
Plants in space may need to be entirely reliant on light for external growth cues. If plants, in particular roots, do not respond well to light in zero g, then they may not properly access the growth medium and nutrients, which could be detrimental to crop productivity. This may also be relevant for growth on reduced g on Mars or the Moon. Encouragingly, shoot and root growth does not seem to be greatly different from Earth in trials in the International Space Station (ISS)6. Yet, many questions remain. For instance, can roots still grow away from light when they become densely covered by the leaves? What LED colours work best? Is it useful to add lighting to the root zone to influence directional growth? Clinostats and RPMs have been used over the last two centuries to nullify the effects of gravity7. Therefore, we designed a small, 3D-printed greenhouse (mini-phytotron) with variable intensity LEDs combined with an RPM, and explored some of the limits of root sensitivity to light. The specific question we addressed here is: can light compensate for roots in simulated loss of g, and if so, how much light of what wavelength is required? Such questions are also relevant to crops on Earth for improved understanding of shoot and root shading responses in densely planted crops, and in protected cropping and vertical farming industries.
Results
Random Positioning Machine simulated microgravity environment alters Arabidopsis root growth
In order to create a microgravity environment in which to grow plants, we adopted the use of a commercially available desktop Random Positioning Machine (RPM) (version 2.0 Airbus Defense and Space, the Netherlands B.V., Leiden, the Netherlands). The RPM uses dedicated software to control the RPM motors that change the speed and axes angles relative to Earth’s g vector. Simulated microgravity is created inside the device by three dimensional rotation of biological samples and has been extensively validated particularly in plant systems, and has been demonstrated to induce cellular changes in plants similar to space flight experiments8 (Fig. 1). Although earlier studies have reported the use of RPM, less emphasis has been given to the effects of light levels on modulating root growth in an RPM, as an Earth-based analogue for microgravity conditions.
a RPM and mini-phytotron for simulated microgravity experiments containing five slots to test vertical plant growth on sterile agar plates with variable intensity LED light. The plates were transferred from the growth room, the root tip positions were marked (with a horizontal scratch on the plate surface) and growth was measured after 48 h. b Roots metrics were measured as per published figure9 using ImageJ. Primary root length was calculated by tracing along each root length (L), VGI was calculated as the ratio of root displacement along the y-axis (Ly/L), HGI as the ratio of root tip displacement along the x-axis (Lx/L) and root straightness as the length of chord connecting the starting position to the primary root tip (Lc) normalised to length of the root (L). Angle B was calculated as the angle of root displacement from the vertical being zero degrees. c Example of a plate with Arabidopsis mutant and wild-type Col-0 plants growing in Earth 1 g in darkenss. d Example of Arabidopsis mutant and wild-type Col-0 plants in simulated zero g in darkness. Roots can be seen to lose directionality beyond each horizontal scratch mark on the plate surface (scale bar = 0.5 cm). (Roots grow and bend at the root tip, which is beyond the scratch mark).
To explore the ways that light can compensate for the loss of g in plants, we built a small greenhouse using commonplace 3D-printing that we called a mini-phytotron to grow seedlings within the RPM. The mini-phytotron was developed to be mounted on the RPM platform and included white LED light strips for the plants growing inside. Translucent materials were included to diffuse the points of LED light. The entire apparatus could then be run in darkness (using black opaque cloth) to eliminate the influence of external ambient light. The mini-phytotron has five slots to allow the simultaneous growth of up to 60 Arabidopsis seedlings in square petri dishes that contain solid agar media. The plates are placed in a vertical orientation within the growing chamber to allow the roots to grow along the surface without penetrating the agar medium. This allows the study of root growth response to various microgravity and lighting conditions. In parallel, an identical mini-phytotron containing the same number of seedlings was set up as 1 g control condition alongside RPM. The skewing of roots away from the g vector has been extensively studied in agravitropic Arabidopsis mutants using morphometric measurements such as vertical growth index (VGI), horizontal growth index (HGI) and root straightness (Fig. 1b)9. In particular, HGI and VGI are two independent parameters describing different root developmental processes, and together with root straightness, provide a complete description of root behaviour to g response10. Consistent with previous microgravity studies in Arabidopsis, our novel experimental setup was able to alter HGI, VGI and straightness values in a similar range as observed earlier for Arabidopsis seedlings (ecotype Columbia) (Table 1; Fig. 2)9,10,11,12,13. Compared to control seedlings grown at 1 g, plants grown in the RPM simulated zero g, significantly increased the HGI or horizontal displacement of the roots and significantly decreased VGI and straightness, which indicates deviation from linear growth (Table 1; Fig. 2). The combination of RPM and mini-phyptron growing chamber provides a useful tool for simulating and studying microgravity effects in plants.
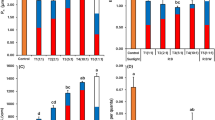
Root responses 48 h after transfer to the combination of zero simulated g and control Earth g (1 g) under two light fluence level, FluR10 (10 μmol m−2 s−1), FluR1 (1 μmol m−2 s−1) and darkness. HGI (a, d), VGI (b, e) and root straightness (c, f) values for WT and d14-1 htl-3 dwarf mutant for different light and g exposure is shown. Representative plants are shown above (scale bar = 0.5 cm). Data are mean ± SEM, n = 24–30. P values were calculated with a Student’s t-test. Asterisk denotes P value < 0.05.
Effect of light fluence rate on simulated microgravity sensing in roots
The RPM provided simulated zero g environment and the white LEDs in the mini-phytotron allowed us to test the effects of light fluence levels on plant seedling root growth behaviour.
We germinated and grew Arabidopsis plants (Arabidopsis thaliana wild-type (WT) Columbia (Col-0) ecotype) vertically on agar plates in a growth room for two days. The position of the root tip was marked and the petri dishes containing the seedlings were then transferred to the mini-phytotron chamber placed within the RPM microgravity simulation platform or 1 g control condition as described in the previous section.
In order to test whether light is adequate to compensate for the loss in g in Arabidopsis, we compared root tropism responses to white light measured at two fluence rates, 10 μmol m−2 s−1 (FluR10) and 1 μmol m−2 s−1 (FluR1) (Fig. 2). Fluence rate of 10 μmol m−2 s−1 was near the lower limit of root sensitivity (Fig. 2). This aligns with previously reported plant sensitivities14, including in the ISS15. Seedlings were also grown in darkness, both at zero g and 1 g conditions as controls. The resultant root growth in the RPM was measured using the HGI, VGI and root straightness as described earlier (Fig. 1b). A dwarf mutant strain of Arabidopsis (d14-1 htl-3) was also included in the experiment (Fig. 2). This is defective in two phytohormone-related pathways, strigolactone and karrikin, which are involved in dwarf and bushy plant architecture and branch angle gravitropism16,17. Dwarf varieties of crops that are compact in stature but still produce high yields may be required in space habitation to increase efficiency in high-density controlled facilities1.
Under 1 g control conditions, Arabidopsis seedlings exhibit characteristic root waving behaviour in both light (FluR10) and in darkness, growing vertically in response to Earth g and showing HGI values close to zero (Fig. 2a), VGI and root straightness values close to 1 (Fig. 2b, c). The dwarf d14-1 htl-3 mutant responded similarly to WT under 1 g light and dark conditions, indicating no altered root gravitropic behaviour.
However, in simulated zero g, root growth varied depending on light fluence levels. In darkness together with simulated zero g, plant roots lacked tropism signals and showed altered growth directions as indicated by the deviation of HGI, VGI and straightness values compared to control 1 g light-grown seedlings (Fig. 2d, e). When seedlings were exposed to the two different light conditions in zero g conditions, root growth behaviour varied. With exposure to 10 μmol m−2 s−1 (FluR10), roots of WT showed significant restoration of g responses, compared to zero g dark-grown seedlings, but with exposure to reduced light levels of 1 μmol m−2 s−1 (FluR1), almost no g response was observed (Fig. 2d, e). This suggests that 10 μmol m−2 s−1 (FluR10) is near the limit of root light responses under microgravity conditions. The d14-1 htl-3 mutant displayed slightly increased microgravity-responsive root behaviour in the zero g dark for both HGI and VGI indexes and exposure to light fluence levels, FluR1 and FluR10 significantly restored root g responses. Together, this suggests that white light within a certain intensity range is sufficient to mostly compensate for g loss in Arabidopsis roots and that dwarf mutants used in this study showed minor root tropism differences compared to WT.
The simulated microgravity caused a small decrease in root straightness compared to 1 g control. However, no differences in root straightness were observed between the two light fluence rates and dark-grown seedlings in either Col-0 or d14-1 htl-3 (Fig. 2f). This suggests that the trait of straightness is less sensitive to light than HGI and VGI, hinting that root waving is regulated in a different way.
Red-light induced gravitropic responses of Arabidopsis roots in simulated microgravity
Extra red light is routinely used in indoor vertical farming systems for improving shoot biomass and delivering photosynthetically active radiation (PAR) to the plants. Red light application may also be incorporated in the design of controlled environment agriculture (CEA) facilities as part of future space exploration missions1. Earlier observations have demonstrated that red light affects both shoot and root growth. We next tested red-light-based photoresponse in roots under simulated microgravity conditions. Using the RPM and mini-phytotron microgavity set up, we exposed Arabidopsis WT and the double mutant to filtered red light measured at two fluence rates, 10 μmol m−2 s−1 (Red-FluR10) and 1 μmol m−2 s−1 (Red-FluR1), matching the rates of white light used earlier.
For wild type Arabidopsis, red light was only partially effective compared to white light in compensating for root tropism in zero g conditions (the intensity was adjusted to match white by using a light meter) (Fig. 3). While HGI values of root growth were similar for both white and red light, VGI and straightness values was lower under red light than white light at the same fluence rates. This indicates the lower sensitivity of WT roots to red light compared to white light. Interestingly, at the lower Red-FluR1 conditions, the d14-1 htl-3 mutant showed significantly higher VGI values and straightness values compared to WT (Fig. 3). This may indicate altered phytochrome response. Given that strigolactones have been linked to normal phytochrome function18,19, it will be important to unravel this further in our system using a range of single mutants, hormone treatments, and LED wavelengths.
Root responses 48 h after transfer to the combination of zero simulated g and red filtered light. Root responses 48 h after transfer to zero simulated g under two red light fluence level, Red-FluR10 (10 μmol m−2 s−1), Red-FluR1 (1 μmol m−2 s−1). HGI (a), VGI (b) and root straightness (c) values for WT and d14-1 htl-3 dwarf mutant is shown. Representative plants are shown above (scale bar = 0.5 cm). Data are mean ± SEM, n = 24–30. P values were calculated with a Student’s t test. Asterisk denotes P value < 0.05.
Low light is adequate to compensate for gravity loss in lettuce
The influence of light intensity at 10 μmol m−2 s−1 on phototropism is similar to previous reports. This is low compared to the requirement of Arabidopsis plants (leaves) to receive 100 μmol m−2 s−1 as a minimum for photosynthesis20. The need to provide much higher light fluence for photosynthesis may allow enough light to compensate for g loss in roots. We questioned whether roots of a food species that is unrelated to Arabidopsis might also be able to be tested in the mini-phytotron. Lettuce (Lactuca sativa variant Butterhead) showed straight roots in all situations and a response to 10 μmol m−2 s−1 (Fig. 4). Lettuce shows that various plant species can be tested in our mini-phytotron. Straight roots after transfer to zero g may indicate a residual ‘memory’ effect of Earth g (see next). The testing of plants from germination in zero g may be useful in distinguishing between tropism responses and a ‘memory’ effect.
Root responses 48 h after transfer to the combination of zero simulated g and control Earth g (1 g). Seedlings were exposed to two light fluence levels, FluR10 (10 μmol m−2 s−1) and FluR1 (1 μmol m−2 s−1) and dark control (1 g). HGI (a, d), VGI (b, e) and root straightness (c, f) values for different light and g exposure is shown. Representative plants are shown above (scale bar = 0.5 cm). Data are mean ± SEM, n = 45–50. P values were calculated with a Student’s t-test. Asterisk denotes P value < 0.05.
Therefore, we tested root tropism when plants are germinated and grown within the mini-phytotron and on the RPM under FluR10 (10 μmol m−2 s−1) and dark conditions. Plants still partly responded to light (10 μmol m−2 s−1) under zero g conditions, but not to the levels observed when seedlings were transferred from 1 g to zero g as shown earlier (Fig. 5). This suggest that plant roots cells may have a g sensing directional ‘memory’, which continues gravitropic growth to some extent after transfer. However, there are many variables that could impact on this. For example, plants display elongated growth when germinated in low light. Also, younger seedlings may be initially less responsive to light. More experiments are required to investigate possible gravitropic memory effects. Furthermore, differences observed in root tropism behaviour between FluR10 and dark-grown conditions were not likely related to the timing of germination, as this might be expected to result in differences in total root lengths. We observed no significant differences in total root length between seedlings grown in FluR10 compared to dark-grown conditions (average root length for FluR10-grown seedlings = 6.27 cm ± SEM 0.28; and dark-grown seedlings = 5.75, ± SEM 0.20, Student’s t test P > 0.05). Taken together, the data suggest that the presence of light at varying fluence levels can influence root phototropism in simulated microgravity conditions.
a–f FluR10 white light (10 μmol m−2 s−1) was partly effective at compensating for zero g compared to dark and 1 g. However, germinated plants were not as responsive as the transferred ones in Fig. 2. Representative plants are shown above (scale bar = 0.5 cm). Data are mean ± SEM, n = 20-30.
Discussion
We show that an RPM can adequately cancel out g, and, when combined with a mini-phytotron to supply light from ‘above’, can be used to explore plant tropism, and to test the potential requirements of crops in space. Finding better ways to support space missions with fresh food is a major and urgent problem. As the setup utilises a commercially available microgravity analogue RPM device and an inexpensive 3D-printed plastic mini-phytotron with five plant growth slots (design provided in online supplemental material: https://doi.org/10.25909/26828584.v1), it facilitates experimental reproducibility and cross-study comparison.
In this study, HGI and VGI responses to low light from repeated experiments with Arabidopsis confirm that minimal light may be enough to compensate for zero g. This may mean that crops grown in space can grow similarly to Earth g, provided that roots have exposure to light. Straightness as a trait is likely to be unimportant because it represents just a small part of the whole root growth patterns.
Plants have a strong response to g on Earth. Shoots grow away from the g vector, while the roots grow towards it. This is mostly due to the displacement of starch granules that promote auxin translocation in shoot endodermal and root columella cells21. Plants also bend according to the direction of light, a response that is regulated by photoreceptor proteins. Photosynthetic pigments also absorb photons for metabolic energy production, but this is unrelated to bending and requires higher levels of light.
It has been helpful to use zero g to reveal root light responses (root phototropism) by performing experiments in space or in a clinostat, and other experiments have been undertaken on Earth g with side-directed light or with mutants that fail to perceive g well (e.g., starchless mutants). Such techniques have confirmed that root tip growth can be influenced by photoreceptors22. Although there are five known classes of photoreceptors in plants, only two of these predominate in roots: phototropins and phytochromes22. Sensing of specific wavelengths can occur via individual photoreceptors in root tips, where root bending occurs23. The most effective wavelength that repels roots is blue22. However, red either has no effect or is attractive for roots22.
A key experiment relating to root phototropism in space was conducted on the European Modular Cultivation System facility on the ISS. A centrifuge was used to add levels of simulated g and confirmed repulsion of Arabidopsis thaliana roots from white light above and attraction towards red LEDs on the side15. It also revealed attraction of roots towards blue LEDs on the side, but only at very low or absent g, and which was enhanced by red pretreatment15. Single mutants in phytochrome A or phytochrome B were partly defective in response to red light, but appeared closer to normal in response to blue light15. This revealed a hitherto unknown effect of blue.
The root meristem lies within the root tip and, along with being the site of phototropism, is also the site of the cellular building blocks of the whole root. It will be important to understand any negative effects on meristem competence of growing plants in space. A range of deleterious effects on root meristems have been observed, and transcriptomic studies have been carried out24. Spaceflight had a general impact on genes involved in oxygen status, cell defence or stress, cell polarity, and cell wall development25. Interestingly, red light seems to counteract stress in the root meristem due to spaceflight24. Thus, it could be useful to specifically illuminate roots with red light in space, not only for its attraction effect. It is important to note that while the RPM and mini-phytotron can provide new information about root phytotropism, it cannot fully replace experiments in true microgravity. For instance, Arabidopsis roots in a SciSpinner MAX RPM were not attracted to lateral blue light as was observed in the ISS, which may relate to the stress that roots experience on the ISS26.
Instruments like our RPM and mini-phytotron may be useful additions to help test and unravel the connections between light wavelengths, photoreceptors, and stress, which, in addition to improving our understanding of root tip phototropism, may be helpful in informing future strategies to engineer or manage crop roots in space for enhanced food production.
Individual plants use light and chemical signals from neighbours to detect nearby plants that may be competitors for light and nutrients. Plants then alter their growth to be more competitive, such as growing taller with less lateral branching27,28,29. This response can reduce the overall yield per plant, because it focuses plant resources into fewer progeny (seeds). It means that denser sown plants may not actually increase yield per land area. Manipulating genetics or applying hormones or altering lighting (for indoor crops) can weaken shade avoidance responses in plants, which may increase yield29,30. However, genetically altering plant hormone pathways may also result in poor phototropic responses when crops are grown in space.
Dwarf wheat was grown in space, and a range of compact crop varieties may be needed1. However, dwarf varieties can be deficient in plant hormones that also affect tropism. For instance, some dwarf varieties are deficient in strigolactones, and strigolactones act to regulate root and seedling growth patterns in part by inhibiting auxin transport16,17,31,32. Karrikins are related to strigolactones and act in a similar way16,17,31,32. The d14-1 htl-3 dwarf mutant variety was shown to have altered root gravitropism, and the seedling shoot (hypocotyl) is elongated11. Thus, it is useful to determine if there are major deficiencies in the root phototropism of dwarf plants. We also reasoned that, if necessary, adding hormones into the agar may be a way to promote straight root growth. While the d14-1 htl-3 dwarf variety that we tested did not show deficiencies in root phototropism, it will be important to test dwarf varieties based on other plant hormones, such as gibberellins.
Note that we used agar that has minimal nutrients, and in particular lacks ammonium. Ammonium promotes straight root growth when added to agar33, and roots can be attracted to a spot of ammonium on agar34. We, therefore, used media lacking ammonium as it may confound the interpretation of root phototropic responses, and also to create a situation where we could add ammonium if necessary in the future. The medium also contained 0.5% sucrose, which improved seedling vigour.
Red-enriched light is often used in plant growth facilities because red is the main wavelength used by plant photosynthesis, and it promotes faster growth. So we also included experiments with a red filter to test the effect of red-enriched light on root tropism. This confirmed that filtered red light is not as effective in repelling roots. It has been reported that red light is poor at repelling roots, and red LEDs have even been shown to attract roots22. Therefore, we would recommend further investigation when considering using red light in space crops. Alternatively, red LEDs might be employed below plants to attract roots. The mini-phytotron can be designed with LEDs at the base to investigate this option.
White light repelled Arabidopsis roots in seedling growth in the ISS15. However, using just red or blue LEDs, exposure to the side of Arabidopsis roots was actually attractive at zero g in the ISS, particularly with red first and then blue15. Previously, studies of starchless mutant roots in Arabidopsis also observed growth towards red filtered light35. However, blue filtered light was repulsive35. It was suggested that the absence of g in the ISS revealed an attraction to blue15. It is possible that specific LED wavelengths may be attractive alone, but repulsive in combination with other wavelengths, perhaps due to molecular interactions with different light receptors. Also, root angle may be a sum of tropism responses, and the net effect of various wavelengths may be repulsive. For instance, our red filter was not strict, hence it also allows other wavelengths through, such as green and far-red. This may explain the net repulsive effect of red-filtered light that was not as strong as white light.
Green parts of plants need to grow towards light to maximise photosynthesis. However, roots are mostly underground and are guided down by g and soil signals. So, why do we find a response to light in roots? And why would roots have repulsive and attractive responses to different light wavelengths? It may be that root tips respond to different wavelengths of sunlight that penetrate the top few centimetres of soil. Red wavelengths can penetrate deeper into soil than blue. Lateral roots initially do not grow straight down like the main root. The g-sensing molecular machinery takes several days to fully develop36. The existence of a blue and red attraction in zero g may be relevant for lateral roots near the soil surface that lack g-sensing. Further studies looking at lateral roots with combinations of LEDs and mutants may help to reveal more hidden responses.
Plants detect relative levels of wavelengths to regulate phytochrome transport into the nuclei of cells. The most well-known is the red:far-red ratio, which triggers gene phytochrome-dependent gene expression changes, asymmetric growth in expanding cells and plant bending37. Light wavelengths act as neighbour proximity signals, mainly for neighbour competition and shade avoidance. Neighbour plants absorb mostly red and some blue wavelengths, while reflecting green and far-red. Hence, light that is enriched in green and far-red tells a plant that a neighbour plant is near. This triggers tall and narrow shoot growth away from the neighbour. It may also be possible that roots can sense its own canopy or a neighbour plant, and grow away from it27. Ratios of various wavelengths may allow plant roots to fine-tune growth. I.e., more red and blue indicate a region of neighbour absence, hence are attractive. Whereas more green and far-red indicate neighbour’s leaf canopy is present, hence are repulsive. Neighbour-proximity signals from the shoot that influence root architecture seem to be an important phenomenon that avoids root crowding in densely planted crops27,28. This is poorly understood. Other signals, such as root exuded hormones, move between individual plant roots and may act as neighbour sensing signals38,39. Further research in this area may help optimise crop root growth, not only in space, but also on Earth. One aim could be to discover the signalling mechanism(s) for crop root sensing of neighbour-proximity, which could then be manipulated genetically or by phytohormone treatments to improve crop root architecture for agriculture. Another aim would be to integrate other signals with root phototropism, such as allelopathy (root signalling with neighbour organisms), mechanosensing (root sensing of soil structure and density), nutritropism (root nutrient attraction) and hydrotropism (root water attraction).
An important caveat for our experiments is that we have not yet determined where our light signals are perceived. It is possible that our root repulsion was a response from the shoot, which then somehow sends a directional signal to the root tip. There are examples of bending communication between plant parts. For example, phytohormone signals from the main stem impose an anti-gravitropic offset, resulting in a horizontal angle in side branches40. Phytohormone signals can also coordinate gravitropic signals between root and shoot tissues41,42. Moreover, light transmission within the stem may regulate root phototropism43. Similar responses to light wavelengths in the shoot may induce signals that flow down into roots41. Also, light may be reflected down through the translucent stem of the plant, somewhat akin to fibre optics43. Studies need to be carried out using techniques such as mutants, grafting and fibre optics to separate root and shoot-specific responses. This may lead to new ideas for how to illuminate roots in space, such as using red light below plants to attract roots.
The RPM mini-phytotron is a suitable method of trialling LEDs and root light perception. It avoids confounding issues of performing experiments in space and negative complications from starchless mutants44,45. We confirm that a low level of white light is required to repel roots and allow them to grow relatively normally in the absence of g. Further research is required to assess combinations of LEDs in the RPM to discover distinctive roles and perception mechanisms of light wavelengths in roots. It may be possible to find a combination that provides sufficient root repulsion while delivering optimal shoot growth. Further designs of the 3D-printed mini-phytotron can be easily achieved to explore light exposure directly on roots and from the sides or from above or below the plants by using opaque black material for the mini-phytotron to reduce light diffusion including ways to isolate and expose roots and shoots to different light regimes, and to test whether hormonal or nutrient additives can be used to encourage roots to grow down. The RPM can also be used to simulate Moon or Mars g. While we found that root growth responded adequately to light, even with ammonium-free nutrients and the dwarf mutant that is defective in phytohormone pathways, there are many questions that remain. The ability to explore the effects of LEDs in combination with mutants and nutrient or hormone additives in a system like ours will be useful not only for studying the interactions of nutrients and phytohormones with root phototropism for space, but also for Earth-based crops.
Methods
Plant material
Arabidopsis thaliana Col-0 ecotype and d14-1 htl-3 mutant strain were from our laboratory stocks and Lactuca sativa variant Butterhead seed was purchased from a local store.
Plant growth conditions
Seeds were surface sterilised with weak bleach solution for 10 min, then rinsed and sown on ammonium-free nutrient medium as published46 but with of 1% agar (10 g/L) and 0.5% sucrose (5 g/L). The agar was solidified in 10 × 10 cm plastic petri dishes, without an air vent. The lids were affixed with micropore tape and plates were stored at 4 °C for 2 days to promote synchronous germination. Unless otherwise specified, plates containing seeds were then placed in a plant growth room at 22 °C with 16 h day length under white LEDs (120 μmol m−2 s−1) to allow for germination and straight vertical root growth that served a baseline before being transferred to mini-phytotron placed in the RPM (zero g microgravity condition) or control mini-phytotron grown alongside (1 g control condition) as in Figs. 2–4. For root growth behaviour of Arabidopsis grown from germination under microgravity, plates containing seeds were placed directly in the mini-phytotron RPM or control condition following stratification as in Fig. 5.
A desktop Random Positioning Machine (RPM, version 2.0 Airbus Defense and Space, the Netherlands B.V., Leiden, the Netherlands) was used in standard laboratory conditions. The RPM inbuilt software controls the motors of the rotating platform to provide simulated microgravity conditions continuously throughout the experiment. The predefined RPM zero g file was chosen from the motion mode menu and was used in Random Speed mode with random interval and a maximum speed of 75°/s. Twelve seeds were sown in a line near the top of the plate, such that roots can grow down towards the centre of rotation.
Roots were measured manually using ImageJ software (https://imagej.net/software/fiji/downloads) to calculate root lengths and HGI, VGI and root straightness displacements as described earlier and in Fig. 1e.
Mini-phytotron
A small greenhouse (mini-phytotron) was designed with three compartments, as shown in Fig. 6, to grow seedlings within an RPM. The model was first designed by AutoCAD 2022 and was divided into three parts which are the top lid with integrated LED lights (warm white 50 mm strip lighting 12 V 60ma 5050 LED, RS Components, Catalogue No 153-3638) behind narrow translucent diffuser panels, the bottom body included channels to accommodate maximum 5 square petri dish plates that can be screwed to the RPM machine and a middle connector piece that secures the top LED section with the bottom base. The mini-phytotron was then fabricated through 3-D printing using high-density polyethylene (HDPE) and was connected to a DC power supply that provides power to the light. A red cellophane film (Krystal Cello) was added to the diffuser panels of the mini-phytotron to grow seedlings in filtered red light conditions. The raw 3D printing files (.stl) for the mini-phytotron and the LED strip circuitry design (.dxf) are available online (https://doi.org/10.25909/26828584.v1).
Mini-phytotron designed by AutoCAD (top-down (a); side-view (b); and internal view of compartment (c) and fabricated by a 3D printer (d).
Data availability
All data generated during this study are either included in the manuscript or available online (https://doi.org/10.25909/26828584.v1).
References
Mortimer, J. C. & Gilliham, M. SpaceHort: redesigning plants to support space exploration and on-earth sustainability. Curr. Opin. Biotechnol. 73, 246–252 (2022).
Hessel, V. et al. Eustress in space: opportunities for plant stressors beyond the earth ecosystem. Front. Astron. Space 9, https://doi.org/10.3389/fspas.2022.841211 (2022).
Douglas, G. L., Zwart, S. R. & Smith, S. M. Space food for thought: challenges and considerations for food and nutrition on exploration missions. J. Nutr. 150, 2242–2244 (2020).
MacDonald, J. G., Rodriguez, K. & Quirk, S. An oxygen delivery polymer enhances seed germination in a Martian-like environment. Astrobiology 20, 846–863 (2020).
Hessel, V., Stoudemire, J., Miyamoto, H. & Fisk, I. D. In-Space Manufacturing and Resources: Earth and Planetary Exploration Applications. (Wiley-VCH, 2022).
Paul, A. L., Amalfitano, C. E. & Ferl, R. J. Plant growth strategies are remodeled by spaceflight. BMC Plant Biol. 12, 232 (2012).
van Loon, J. J. Some history and use of the random positioning machine, RPM, in gravity-related research. Adv. Space Res. 39, 1161–1165 (2007).
Kraft, T. F., van Loon, J. J. & Kiss, J. Z. Plastid position in Arabidopsis columella cells is similar in microgravity and on a random-positioning machine. Planta 211, 415–422 (2000).
Vaughn, L. M. & Masson, P. H. A QTL study for regions contributing to Arabidopsis thaliana root skewing on tilted surfaces. G3 (Bethesda) 1, 105–115 (2011).
Grabov, A. et al. Morphometric analysis of root shape. New Phytol. 165, 641–651 (2005).
Swarbreck, S. M., Guerringue, Y., Matthus, E., Jamieson, F. J. C. & Davies, J. M. Impairment in karrikin but not strigolactone sensing enhances root skewing in Arabidopsis thaliana. Plant J. 98, 607–621 (2019).
Villacampa, A., Sora, L., Herranz, R., Medina, F. J. & Ciska, M. Analysis of Graviresponse and biological effects of vertical and horizontal Clinorotation in Arabidopsis thaliana Root Tip. Plants 10, https://doi.org/10.3390/plants10040734 (2021).
Xu, P. et al. Pectin methylesterase gene AtPMEPCRA contributes to physiological adaptation to simulated and spaceflight microgravity in Arabidopsis. iScience 25, 104331 (2022).
Kami, C. et al. Reduced phototropism in pks mutants may be due to altered auxin-regulated gene expression or reduced lateral auxin transport. Plant J. 77, 393–403 (2014).
Vandenbrink, J. P., Herranz, R., Medina, F. J., Edelmann, R. E. & Kiss, J. Z. A novel blue-light phototropic response is revealed in roots of Arabidopsis thaliana in microgravity. Planta 244, 1201–1215 (2016).
Sang, D. et al. Strigolactones regulate rice tiller angle by attenuating shoot gravitropism through inhibiting auxin biosynthesis. Proc. Natl Acad. Sci. USA 111, 11199–11204 (2014).
Waters, M. T. et al. Specialisation within the DWARF14 protein family confers distinct responses to karrikins and strigolactones in Arabidopsis. Development 139, 1285–1295 (2012).
Brewer, P. B., Koltai, H. & Beveridge, C. A. Diverse roles of strigolactones in plant development. Mol. Plant. 6, 18–28 (2013).
Jia, K. P., Luo, Q., He, S. B., Lu, X. D. & Yang, H. Q. Strigolactone-regulated hypocotyl elongation is dependent on cryptochrome and phytochrome signaling pathways in Arabidopsis. Mol. Plant. 7, 528–540 (2014).
Schumann, T., Paul, S., Melzer, M., Dormann, P. & Jahns, P. Plant growth under natural light conditions provides highly flexible short-term acclimation properties toward high light stress. Front. Plant Sci. 8, 681 (2017).
Vandenbrink, J. P., Kiss, J. Z., Herranz, R. & Medina, F. J. Light and gravity signals synergize in modulating plant development. Front. Plant Sci. 5, 563 (2014).
Kutschera, U. & Briggs, W. R. Root phototropism: from dogma to the mechanism of blue light perception. Planta 235, 443–452 (2012).
Mo, M., Yokawa, K., Wan, Y. & Baluška, F. How and why do root apices sense light under the soil surface?. Front. Plant Sci. 6, 775 (2015).
Medina, F. J., Manzano, A., Herranz, R. & Kiss, J. Z. Red light enhances plant adaptation to spaceflight and Mars g-Levels. Life 12, https://doi.org/10.3390/life12101484 (2022).
Correll, M. J. et al. Transcriptome analyses of Arabidopsis thaliana seedlings grown in space: implications for gravity-responsive genes. Planta 238, 519–533 (2013).
Hughes, A. M., Vandenbrink, J. P. & Kiss, J. Z. Efficacy of the random positioning machine as a terrestrial analogue to microgravity in studies of seedling phototropism. Microgravity Sci. Technol. 35, 43 (2023).
Gundel, P. E., Pierik, R., Mommer, L. & Ballare, C. L. Competing neighbors: light perception and root function. Oecologia 176, 1–10 (2014).
Jacob, C. E., Tozzi, E. & Willenborg, C. J. Neighbour presence, not identity, influences root and shoot allocation in pea. PLoS One 12, e0173758 (2017).
Huber, M., Nieuwendijk, N. M., Pantazopoulou, C. K. & Pierik, R. Light signalling shapes plant-plant interactions in dense canopies. Plant Cell Environ. 44, 1014–1029 (2021).
Xie, X., Cheng, H., Hou, C. & Ren, M. Integration of light and auxin signaling in shade plants: from mechanisms to opportunities in urban agriculture. Int. J. Mol. Sci. 23, https://doi.org/10.3390/ijms23073422 (2022).
Xu, P., Jinbo, H. & Cai, W. Karrikin signaling regulates hypocotyl shade avoidance response by modulating auxin homeostasis in Arabidopsis. New Phytol. 236, 1748–1761 (2022).
Zhang, J. et al. Strigolactones inhibit auxin feedback on PIN-dependent auxin transport canalization. Nat. Commun. 11, 3508 (2020).
Zou, N., Li, B., Dong, G., Kronzucker, H. J. & Shi, W. Ammonium-induced loss of root gravitropism is related to auxin distribution and TRH1 function, and is uncoupled from the inhibition of root elongation in Arabidopsis. J. Exp. Bot. 63, 3777–3788 (2012).
Yamazaki, K., Ohmori, Y. & Fujiwara, T. A positive tropism of rice roots toward a nutrient source. Plant Cell Physiol. 61, 546–553 (2020).
Ruppel, N. J., Hangarter, R. P. & Kiss, J. Z. Red-light-induced positive phototropism in Arabidopsis roots. Planta 212, 424–430 (2001).
Guyomarc’h, S. et al. Early development and gravitropic response of lateral roots in Arabidopsis thaliana. Philos. Trans. R. Soc. Lond. B Biol. Sci. 367, 1509–1516 (2012).
Fernandez-Milmanda, G. L. & Ballare, C. L. Shade avoidance: expanding the color and hormone palette. Trends Plant Sci. 26, 509–523 (2021).
Wheeldon, C. D., Hamon-Josse, M., Lund, H., Yoneyama, K. & Bennett, T. Environmental strigolactone drives early growth responses to neighboring plants and soil volume in pea. Curr. Biol. 32, 3593–3600.e3593 (2022).
Yoneyama, K., Xie, X., Nomura, T., Yoneyama, K. & Bennett, T. Supra-organismal regulation of strigolactone exudation and plant development in response to rhizospheric cues in rice. Curr. Biol. 32, 3601–3608.e3603 (2022).
Roychoudhry, S., Del Bianco, M., Kieffer, M. & Kepinski, S. Auxin controls gravitropic setpoint angle in higher plant lateral branches. Curr. Biol. 23, 1497–1504 (2013).
Burko, Y., Gaillochet, C., Seluzicki, A., Chory, J. & Busch, W. Local HY5 activity mediates hypocotyl growth and shoot-to-root communication. Plant Commun. 1, https://doi.org/10.1016/j.xplc.2020.100078 (2020).
Zhu, Q. et al. Root gravity response module guides differential growth determining both root bending and apical hook formation in Arabidopsis. Development 146, https://doi.org/10.1242/dev.175919 (2019).
Lee, H. J. et al. Stem-piped light activates phytochrome B to trigger light responses in Arabidopsis thaliana roots. Sci. Signal 9, ra106 (2016).
Richter, P., Strauch, S. M. & Lebert, M. Disproval of the starch-amyloplast hypothesis?. Trends Plant Sci. 24, 291–293 (2019).
Vitha, S., Zhao, L. & Sack, F. D. Interaction of root gravitropism and phototropism in Arabidopsis wild-type and starchless mutants. Plant Physiol. 122, 453–462 (2000).
Becher, M., Talke, I. N., Krall, L. & Kramer, U. Cross-species microarray transcript profiling reveals high constitutive expression of metal homeostasis genes in shoots of the zinc hyperaccumulator Arabidopsis halleri. Plant J. 37, 251–268 (2004).
Acknowledgements
We thank Matthew Gilliham for project guidance and Hien Minh Vu and Alban O’Brien for assistance in helping to design, build and optimise the RPM mini-phytotron. This work was supported by The University of Adelaide, The Waite Research Institute, the Andy Thomas Centre for Space Resources and Australian Research Council grant CE230100015.
Author information
Authors and Affiliations
Contributions
R.D., V.H. and P.B.B. developed the concept. R.D., N.N.T., V.H. and P.B.B. designed the apparatus. R.D., A.S. and T.M. performed the experiments. R.D., A.S., T.M. and P.B.B. analysed the data. R.D. and P.B.B. compiled the figures and wrote the manuscript. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
David, R., Situmorang, A., Tran, N.N. et al. Light is sufficient to compensate for random positioning machine-simulated microgravity in plant roots. npj Microgravity 11, 28 (2025). https://doi.org/10.1038/s41526-025-00493-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41526-025-00493-w