Abstract
Bone density loss is a major concern for astronauts in space, largely due to altered mechanical stimuli in microgravity. These changes are thought to impact bone cells by directly affecting musculoskeletal cell physiology and disrupting mechanosensing and mechanotransduction pathways. This review focuses on the role of the primary cilium, a small, non-motile cellular structure, involved in these processes. Previously underestimated, the primary cilium is now known to act as a mechano- and chemo-sensor on the surface of most vertebrate cells, transmitting signals via multiple intracellular pathways. The primary cilium senses the extracellular fluid flow and its dynamic changes in physiological and pathological conditions, which may include exposure to microgravity, connecting its inactivation to bone density loss. This systematic review will compile and analyze current data on how weightlessness affects the mechanosensing functions of the primary cilium and its role in bone homeostasis disruption.
Similar content being viewed by others
Introduction
Gravity, as a universal force, has played a pivotal role throughout evolution, profoundly influencing life’s development. Dynamic tissue homeostasis is strongly subject to the effects that gravity exerts on cells, and in turn on the intracellular mechanisms through which cells adapt to the extracellular environment. Cells indeed exhibit noticeable alterations when exposed to microgravity (μG), or weightless conditions. Bone, being a highly complex tissue undergoing remodeling throughout postnatal life to adjust to mechanical stressors, is among the tissues that are most directly affected by μG1. The continuous bone remodeling is crucial for maintaining structure, strength, and functionality in response to daily loading variations that, under physiological conditions, ensure homeostasis and structural integrity through the coordinated activity of osteoblasts and osteoclasts2. By nullifying the direction of the gravitational vector and reducing the load, μG significantly impacts bone, acting directly on the remodeling process. The resulting adaptive changes in cellular homeostasis increase bone resorption, inhibiting bone formation3,4,5.
Mechanobiology studies the mechanical properties and adaptation of cells to their mechanical environment. This emerging field merges engineering, biology, physics, and chemistry to understand all the small facets of cell homeostasis signaling pathways.
By mechanically interacting with the extracellular environment through highly complex mechanisms and structures, all cells can sense and respond to cues from the local extracellular matrix (ECM), influencing their own physiology to regulate tissue viability6.
This sequence of events is the consequence of the recruitment of specific mechano-molecular players capable of activating, interconnecting, and reorganizing themselves, following mechanical stimuli from the surrounding microenvironment (mechanosensing) to transduce these stimuli into intracellular biochemical signals (mechanotransduction)7,8.
The maintenance of cell functions heavily relies on the physiological mechanical stimulation, and disruption in tissue mechanics and cellular mechano-signaling can lead to severe alteration of homeostasis, triggering tissue injury in diverse pathological conditions, such as fibrosis, cancer, cardiovascular diseases, and aging9.
Physiologically, mechanical homeostasis is sustained through negative feedback mechanisms that sense extracellular environmental changes and restore them to their baseline levels. Altered mechanosensing leads to loss of this negative feedback regulation that, in turn, causes upregulation of signaling cascades leading to pathological conditions10.
Plasma membrane can sense diverse physical cues, including hydrostatic pressure (HP), tensile force (TF), fluid shear stress (FSS), ECM stiffness, tissue elasticity, and extracellular fluid (ECF) viscosity, through discrete structural domains involved in cell–ECM and cell–cell interactions and cross-talk9.
In this regard, the primary cilium (PC), which has recently emerged as a key cell surface compartment, has been found to be involved in both chemosensing and mechanosensing. Specifically, PC-mediated sensing transduces intracellular signaling pathways11, including Hedgehog12, Wingless-Type (WNT)12, Hippo12, Yes-associated protein 1/Tafazzin (Yap/Taz)13 cascades, thus playing a pivotal role in regulating cell proliferation, survival, and differentiation.
Recent data proposed PC as a gravity sensor, as changes in Earth’s gravitational field (like tides) could affect gene expression through changes in extracellular FSS sensed by the PC14. Consistently, under μG conditions, the decrease in osteoblastic differentiation was restricted to cells bearing primary cilia, further underscoring their central role as mediators of the gravitational response15.
Because of the load-related activity of osteoblasts and osteocytes, strictly associated with the peculiar nature of the calcified ECM that surrounds them, the PC is emerging as an essential player in bone tissue physiopathology. Nonetheless, its implication in the altered mechanobiology induced by μG is yet poorly explored and clarified, with new pieces of evidence being provided in the most recent scientific literature.
Bone tissue supports body weight through a specialized mineralized ECM that provides structural integrity, mechanical signals, and nutrients to resident bone cells. Depending on the kind of support role the bone has, it exists in two macroscopic forms: cortical bone, a dense outer layer that provides strength and protection16, and cancellous (or spongy) bone, a porous structure composed of trabeculae arranged to absorb stress and resist forces from multiple directions. The ECM is composed of about 60% inorganic minerals, mainly hydroxyapatite (Ca)10(PO4)6(OH)2, and 30% organic components17, such as type I collagen, forming a rigid scaffold that supports and regulates bone cell behavior.
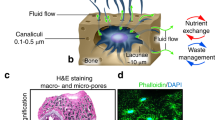
Osteocytes are the most abundant and long-lived cells in bone, making up over 90% of the cellular population18. They originate from mesenchymal stromal cells that differentiate into mature osteoblasts, which, once embedded in the mineralized matrix during bone formation, become osteocytes. As they mature into osteocytes, they reduce in size and extend dendritic processes that pass through tiny channels called canaliculi, forming an interconnected network known as the lacuno-canalicular system (LCS)19. This system allows interstitial fluid flow and cellular communication, enabling osteocytes to function as mechanosensors. In contrast, osteoclasts are the least abundant bone cells, comprising about 1%, and play a crucial role in bone resorption. Derived from the monocyte lineage, they secrete acidic vesicles and enzymes that degrade hydroxyapatite and the organic matrix, contributing to bone turnover17.
Bone is a dynamic tissue that continuously remodels in response to environmental cues, with mechanical stimulation being one of the most potent drivers20. This remodeling process is activated by both mechanical and biochemical signals, which are sensed by osteocytes and transduced through pathways involving WNT, integrins, estrogen, and calcium21,22.
According to Roux’s “use it or lose it” principle and Wolff’s law, bones adapt structurally to the loads they experience23,24 (Fig. 1). Everyday activities generate varying degrees of strain, promoting new bone formation when load increases. In this context, Frost introduced the concept of “windows of mechanical usage,” where low strain (Disuse Window) leads to resorption, optimal strain (Overuse Window) promotes bone formation, and excessive strain (above 3500 µɛ—Harmful Window) can result in bone damage25,26,27,28 (Fig. 1).
The efficiency of mechanical signal transmission is crucial for bone health. Disruptions in load transfer—caused by changes in bone stiffness or extracellular fluid flow—can impair mechanotransduction and lead to bone loss16,29. Stiffness, dependent on bone elasticity, is essential for translating mechanical signals into cellular responses16.
With aging, skeletal properties like material strength and stiffness decline, prompting compensatory changes in bone architecture16. However, this often results in increased bone resorption, especially in weight-bearing bones. Similar effects are seen in astronauts, where μG impairs remodeling, particularly in load-dependent bones30. Cortical bone, known for its load-bearing capacity and mechanical anisotropy, also shows age-related changes, further impacting its mechanical function30.
Cells can sense mechanical signals and transduce them through their membrane to the intracellular compartment; this sequence of events defines the mechanism called mechanosensing31,32.
A four-step model has been proposed to explain mechanosensing: (i) mechanopresentation: the mechanical cue is presented to the cell; (ii) mechanoreception: the cell catches the input; (iii) mechanotransmission: the signal is transferred from the outside of the cell to the inside through a transmitter; (iv) mechanotransduction: the mechanical force is transduced into a biochemical signal8.
Eukaryotic cells orchestrate these processes through specialized surface domains and structures enabling membrane/cytoskeleton physical interactions, called mechanosensors. Among these mechanosensors, focal adhesions (FA) and glycocalyx transduce mechanical cues through molecules anchored to neighboring cells or stimuli arising from the surrounding ECM33. The PC, a mechano-sensory organelle that protrudes like an antenna from the cell surface, is able to sense both mechanical and chemical stimuli provided in the extracellular environment and transduce them into intracellular molecular cascades34.
The first observations of the PC were made by the anatomist Karl Wilhelm Zimmerman in 1898, who described it as a vestigial organelle in renal tubule cells without any clear role for cellular functions35. The occurrence of this organelle in several other tissues, including bone, was demonstrated only 70 years later, though its function remained unclear for a long time36.
Currently, we know that the PC is a single, non-motile microtubule-based organelle that projects from the surface of most non-proliferating mammalian cells37. The ciliary structure comprises different portions and components, namely: basal body, axoneme, transition zone, ciliary membrane, cilioplasm, and ciliary pocket (Fig. 2).
Schematic representation of the PC structure: PC microtubules arrangement changes from the bottom, where they are arranged in doublets and in a ring conformation, to the top, where they are disorganized and in a singlet conformation42. The basal feet are connections from the basal body to the cytoskeletal microtubules38. Cytoskeletal structures are reduced in number and in length for simplicity purposes. Image not to scale. Created with BioRender.com.
The basal body is an apical microtubule-organizing centre, with the classical centriole structure made of nine triplets of microtubules organized in a circle and localized at the base of the cilium. This structure is connected to the cell membrane by fibrous proteins called transition fibers or alar sheets38 that anchor it in place and create a filter zone that separates the cellular cytoplasm from the ciliary cytoplasm (cilioplasm) (Fig. 2).
Close to these alar sheets, there are other conical structures that connect the basal body to the cytoskeletal microtubules, called basal feet39. Whereas in a motile beating cilium, there is always one basal foot oriented in the direction of the cilia beating38, the PC can have from 1 to 5 basal feet projecting in all directions.
The axoneme is the core longitudinal shaft of the cilium, composed of nine peripheral microtubule doublets, extending from the nine triplets of the basal body and projecting towards the tip of the PC (Fig. 2). The doublets are asymmetric, having one microtubule made of 13 protofilaments and one made of 10 protofilaments40.
The PC axoneme differs from that of motile cilia in the lack of the central pair of singlet microtubules. This “9 + 0” conformation makes the PC devoid of active motility while being more flexible and resilient to cell surface shear stresses induced by fluid flow. Nevertheless, recent studies have shown that the 9 + 0 circle architecture is consistent only at the base of the cilium, and then it gradually becomes disorganized when approaching the ciliary tip. Microtubules transition to singlets and shift toward the center of the axoneme, forming a 7 + 2 configuration; as the PC tapers toward its distal end, its diameter decreases, leading to a further reduction in microtubule number, reaching a 3 + 0 arrangement41,42 (Fig. 2). Collectively, these irregularities increase the bending proprieties of the organelle and partially explain the variable flexibility throughout the PC length43,44.
The transition zone is a specialized region of the PC located between the basal body and the axoneme. It regulates the entry and exit of molecules in the cilium through protein structures called Y-links, thereby controlling its signaling and function (Fig. 2)45.
The ciliary membrane differs from the rest of the plasma membrane46 in its lipid composition, featuring a higher presence of sterols and sphingolipids47. This composition allows for increased fluidity and balance underlying higher resistance to flow shear stress, in particular at the base of the cilium, where they are more abundant48.
The amount of cytoplasm surrounding the axoneme represents another specific PC sub-compartment, referred to as the cilioplasm. This is segregated from the cytoplasm by the transition zone and basal body. The cilioplasm has a higher concentration of calcium, indeed, compared to the cytoplasm, due to the selective membrane properties of the transition zone and the continuous influx of calcium through mechanosensitive ion channels. This calcium concentration difference is crucial for activating mechanotransduction49.
Finally, the ciliary pocket (CiPo) is another noteworthy PC domain (Fig. 2). The CiPo is an introflexion of the membrane on the extracellular side, supported by the actin filament network50. Although its function remains largely unclear, the clustering of clathrin-coated pits (CCPs) at this level suggests a role in vesicle trafficking, which may be important in the early phases of cilium assembly50.
The anterograde and retrograde transport known as intraflagellar transport (IFT) is the mechanism that enables the transport of multisubunit protein complexes, called IFT particles, along axonemal microtubules (MTs) beneath the ciliary membrane, exploiting the microtubule-associated motor proteins kinesins and dyneins. IFT is therefore crucial for ciliary structural assembly and maintenance, but also for ciliary signaling (see next section)51.
Results
Primary cilium signaling in mechanotransduction
The PC is currently recognized as a vital signaling hub that translates external signals into internal responses, playing a crucial role in maintaining the proper balance and growth of most cells, ensuring their overall stability. It is estimated that at least 1200 genes are expressed within the PC and involved in both its structure and function52.
The PC can function as both a chemosensor and a mechanosensor, which in both cases entail an intense signaling acting in the ciliary and subsequently transmitted to the rest of the cell. For the purpose of this review, only key molecular signaling involved in PC mechanotransduction will be explored.
PC-mediated mechanosensing signaling begins in every cell when an external mechanical input, as fluid flow, bends the cell membrane. This deformation activates multiple mechanisms that transduce the mechanical stimuli into specific biochemical responses. Among these mechanisms, the PC activates various signaling pathways crucial to mechanotransduction.
SACs are ion channels that respond to membrane deformation caused by mechanical inputs from inside and outside the cells. They play pivotal roles in sensing vibrations, pressure (osmotic, fluidic, and atmospheric), and touch53, being localized in areas subjected to high strains of fluid flow, like FA and PC itself.
SACs belong to many different protein families with specific functions and ion selectivity, as summarized in Table 1. The largest and most important among them is the transient receptor (TRP) channel family, which comprises 9 subfamilies, with the most studied being subfamilies C, P, and V.
They are vital for PC function and have been found to be highly correlated with bone osteogenesis and formation, hence it has been seen that various ciliopathies are associated with loss-of-function or other mutations in these channels, like polycystic disease or craniosynostosis (see Table 1).
SACs cluster on the ciliary membrane likely due to the cilium’s elastic properties through which energy is transmitted to open these channels and facilitate specific calcium ion accumulation to act as second messengers in PC-mediated transduction48,54,55.
The cilium can indeed be considered a significant storage site for an intracellular calcium reservoir due to its high calcium concentration ([Ca²⁺] of 500 nM, compared with 100–200 nM found in mitochondria)31,56.
Lastly, the cell cortex, mostly composed of cytoskeletal actin microfilaments located in close proximity to the plasma membrane, has a role in mechanotransduction. Through specific protein domains, it enables crosstalk between actin and SACs, such as Potassium channel subfamily K member 2 (KCNK2, or TREK1)57 and Transient receptor potential Vanilloid Cation Channel Subfamily V Member 6 (TRPV6)32, creating a bridge from mechanical stimuli to the cytoskeleton and sometimes even to the nucleus, inducing epigenetic and transcriptional changes58.
Also, intermediate filaments are other cytoskeletal components involved in mechanotransduction. Owing to their flexibility and resilience to tensile and compressive forces, they mediate interactions among cells (in adherent junctions) and with the ECM (in desmosomes). They are also directly implicated in nuclear mechanotransduction as they establish indirect contact with the nuclear lamina through the LINC complex59. Due to their high sensitivity to shear stresses, they can modify their own stiffness as well as that of nearby cells60,61.
The PC is implicated in the Hippo signaling pathway (Fig. 3a; for a comprehensive review of this pathway, see ref. 62). One of the core components, the mammalian sterile 20-like kinase 1/2 (MST1/2), is localized at the basal body of PC, where it interacts with the nephronophthisis (NPHP) complex. This complex binds MST1/2 and the large tumor suppressor kinase 1/2 (LATS1/2), inhibiting their phosphorylation activity. This prevents the inactivation of the transcriptional co-regulators Yes-associated protein and Tafazzin family protein (YAP/TAZ) complex, allowing it to activate target gene transcription63.
Schematic representation of primary cilium-related signaling pathways: Hippo pathway in the OFF state (left) does not present PC, thus leading to phosphorylation of MST1/2 by Rassf and subsequently degradation of YAP/TAZ, and in the ON state (right) where the presence of PC blocks the phosphorylation of MST1/2 complex permitting the localization of YAP/TAZ protein in the nuclear compartment62 (a), the NOTCH pathway is regulated the Presenlin, present in the basal body of the PC, cleaving the NCID domain from the NOTCH membrane protein, leading it to migrate to the nucleus and activating target genes expression64,65 (b); the non-canonical WNT/Ca2+ pathway is activated by fluid flow that by bending primary cilium permit the entrance of Ca2+ (green circles) from TRPP1/2, this then increase the expression of Inversin, present on the primary cilium base. Inversin then migrates to the cytoskeleton and blocks β-catenin translocation to the nucleus. Meanwhile, calcium is stored inside the cilioplasm71 (c). Created with BioRender.com.
The PC is also essential for the development and fate determination of neuroepithelial cells through the activation of Notch signaling (Fig. 3b; for a comprehensive review of this signaling pathway, see refs. 64,65), which leads to the accumulation of Sonic hedgehog (SHH) in the cilium and determines ventral cell fate66. Presenilin, a key component of the Notch pathway, is also localized at the basal body of the PC. When a Notch receptor on the PC membrane binds its ligand—jagged or delta on a neighboring cell—Presenilin is activated, enabling the Notch intracellular domain (NICD) to translocate to the nucleus and activate downstream target genes67.
Unlike other pathways, the Hedgehog pathway relies on the PC’s chemosensory function and will not be delved into. The movement of Hedgehog and Hippo signaling key factors can be helped by the IFT proteins, and when IFT proteins are mutated, or the IFT mechanism is impaired, these signaling pathways are compromised68,69. Although major evidence of the exact mechanism by which this happens is still lacking, PC's role in modulating the WNT pathway by non-canonical WNT/Ca2+ activation (Fig. 3c) as a specialized calcium signaling compartment is widely accepted12,70.
Supporting evidence for the connection between the WNT/Ca²⁺ pathway and the PC includes findings in rats and mice models (Choi and Robling reviewed the role of the Wnt pathway in bone mechanotransduction71). Zhou and colleagues showed that the exposure of PC-expressing osteoblasts to a sinusoidal electromagnetic field (SEMF) led to increased expression of WNT10b, thereby activating downstream osteogenic signaling pathways, including Disheveled, Collagen-1α1, and β-catenin72. Moreover, WNT9b has been shown to interact specifically with the TRPP1/TRPP2 complex, acting as a ligand that activates the channel and enhances intracellular Ca²⁺ transport through a β-catenin-independent pathway73.
Additionally, He and colleagues observed that a pulsatile electromagnetic field (PEMF) primarily activated iNOS and eNOS—the main producers of nitric oxide (NO)—within the PC (for a review of the NO/cGMP signaling pathway, see ref. 74). They further demonstrated a strong correlation between the PC and the NO/cGMP signaling pathway, as inhibition of either one abolished the effects of PEMF75. It has also been suggested that this signaling pathway was regulated by the TRPP1/TRPP2 complex (also referred to as PC1/PC2)75.
A significant mechanosensing mechanism also localized to the PC of bone cells is cAMP/PKA signaling pathway76 (reviewed by Sassone-Corsi, 201277), mediated by adenylyl cyclase isoform 6 (AC6)78. RNA interference against Polaris—an important component of IFT—has shown that the removal of PC prevented the fluid flow-dependent cAMP level reduction, and osteocytes lacking AC6 exhibited similar behavior to those depleted of the PC, demonstrating the importance of cAMP compartmentalization as a second messenger for intracellular responses, such as cell proliferation, differentiation, and apoptosis79. cAMP negatively regulates Hedgehog signaling by phosphorylating Gli2, blocking its activation, while SHH reduces cAMP and PKA activity, affecting Ca²⁺-dependent signal transduction80.
Interestingly, Polycystin-1 (TRPP1) and Polycystin-2 (TRPP2), key proteins implicated in ciliopathies, such as autosomal dominant polycystic kidney disease (ADPKD), localize to the PC. Within this structure, they form a heterodimeric polycystin complex, which plays a crucial role in mechanotransduction processes, particularly in renal and hepatic tissues81,82,83. Although skeletal abnormalities remain a relatively underexplored aspect of ciliopathies, emerging evidence suggests that mutations in PKD1/2-encoded polycystin (TRPP) proteins also affect PC expression in bone cells. Indeed, its abnormal expression alters TRPP1/TRPP2-related ciliary mechanosensing in these cells, contributing to the skeletal phenotype observed in ADPKD, independently of renal dysfunction84,85,86.
In this regard, Xiao et al. demonstrated that mouse models with mutations in Pkd1 displayed reduced bone mineral density, trabecular bone volume, and cortical thickness87. These mice also exhibited decreased expression of osteogenic markers, such as Runx2-II, Osterix, and Osteocalcin, along with a reduced receptor activator of nuclear factor kappa-Β ligand/osteoprotegerin (RANKL/OPG) ratio87. Notably, these defects appear in a gene-dose-dependent manner, underscoring a direct role for TRPP1 in osteoblast-mediated bone formation, likely via downregulation of key osteogenic pathways.
Microgravity effects on bone cells and mechanotransduction
In recent years, we have been witnessing an increased interest and demand for longer space travel. Yet, the space environment is inherently inhospitable to human life without proper protective gear, and even with the correct equipment, there are numerous threats that can severely disrupt the body’s homeostasis.
The most problematic space condition affecting the human body is weightlessness, commonly referred to as μG, which occurs when individuals or objects experience an extremely weak gravitational field (10-6 times Earth’s gravity), making them appear to be weightless. A prominent example is the experience of astronauts aboard the International Space Station (ISS), where, despite being subjected to a gravity vector that is 90% of that on Earth, they float due to the free-fall movement of the space station orbiting the planet. Prolonged exposure to weightlessness during spaceflight induces notable changes in the human body. Initially, the redistribution of internal fluids triggers motion sickness with symptoms such as headaches and nausea, resulting from increased blood pressure on the skull88. By only one month, the immune system and the musculoskeletal system start suffering the effects of μG too, which lead to immunosuppression89, muscle atrophy, and loss of bone density90.
Astronauts experiencing μG or weightlessness undergo a proportional reduction in bone and muscle density that correlates linearly with the duration of their space journey91. It may be reasonably hypothesized that the reduced mechanical load on bones leads to a gradual deactivation of the bone remodeling unit, hence disrupting the delicate balance between bone resorption and formation.
To support the hypothesis that μG has a specific impact on weight-bearing bones—because they are constantly subjected to mechanical loading92, while having minimal-to-no effects on non-weight-bearing bones93—bone density data from 148 astronauts have been evaluated pre- and post-spaceflight. This analysis showed a greater bone density loss on the lumbar spine, lower limbs, and pelvis; meanwhile, the skull presented an increase in bone density because of the increase in intracranial pressure21. These results were also strengthened by an increase in biochemical bone resorption markers, confirming that μG exerts region-specific effects on the skeletal system30.
Despite astronauts engaging in rigorous physical activity (2.5 h of daily exercise) to mitigate bone and muscle density loss, the magnitude of loss remains substantial94.
Another hypothesis regarding how μG affects bone homeostasis stems from physics. Water cohesion and gravity work in concert to maintain water persistence on the Earth's surface. In the absence of a consistent gravitational vector, smaller forces, such as surface tension and cohesion, influence the behavior of fluids95. This alteration can impact fluids within the human body, altering ECF dynamics, particularly near the cell membrane. This abnormal flow behavior, characterized by local vortexes, changes in diffusivity, etc., may affect fluid–structure interaction and subsequently influence the proper bending of PC and mechanotransduction. Bone represents one of the most remarkable examples of organ adaptation to Earth’s gravity, and consequently, bone cells are presumed to have evolved in response to this gravitational force.
The main effect of μG on bone is a reduction of bone mass, suggesting a direct influence on osteoclastogenesis96 and osteoblastogenesis97. While spaceflight provides the most physiologically relevant model for studying μG (µG)-induced bone loss, it is limited by infrequent missions, small sample sizes, and high costs. Due to logistical constraints, many studies now rely on simulated µG models on Earth using cellular and animal models, which have significantly advanced our understanding of the molecular mechanisms underlying bone loss in μG. Small animal models like rodents and fish help mitigate these issues, enabling larger cohorts and revealing bone loss patterns comparable to those seen in astronauts.
Different studies, briefly discussed in the following sections, have demonstrated that bone loss due to µG exposure can be explained by alterations in the survival and differentiation of osteoblasts and osteocytes, along with increased osteoclastic activity98.
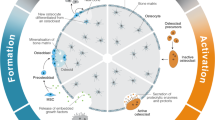
Osteoblasts/Osteocytes impairment and reduced osteoblastogenesis due to microgravity
Microgravity appears to exert negative effects on osteoblasts, leading to the downregulation of key osteogenic markers and impairing their biological function. As previously described, μG diminishes the expression of Tumor necrosis factor (TNF) receptor superfamily member 11b (TNFRSF11B)4, which codes for osteoprotegerin (OPG) that plays a vital role in inhibiting osteoclastogenesis by sequestering RANKL and is typically produced by osteoprogenitors and osteoblasts during excessive bone resorption99. Another gene affected by µG in osteoblasts is Alkaline Phosphatase, Biomineralization Associated (ALPL)—encoding for a hydrolytic enzyme playing a key role in matrix remodeling—representing a crucial marker for osteoblast differentiation and bone formation100. The expression of ALPL is reduced in osteoblasts subjected to μG, compared with those in standard conditions4.
Osteoblast differentiation is further hindered through the downregulation of focal adhesion kinase (FAK)101, mammalian target of rapamycin complex 1 (mTORC1), adenosine monophosphate-activated protein kinase (AMPK), and WNT/β-catenin. Specifically, the downregulation of the WNT/β-catenin pathway affects the expression of Bone Morphogenetic Protein 2 (BMP2) and collagen type I alpha 1 chain COL1A1—both vital markers for osteoblast differentiation101. These effects are believed to be the consequence of the reduced expression of the Runt-related transcription factor 2 (RUNX2)102, a critical regulator of osteogenic differentiation103. In addition, when accompanied by the µG-induced increase in Mitogen-activated protein kinase (p38 MAPK) and Peroxisome proliferator-activated receptor γ2 (PPARγ2) expression, human mesenchymal stromal cells (hMSCs) are induced to differentiate into adipocytes, reducing the osteoprogenitor pool104.
Moreover, researchers have also explored whether simulated µG influences and enhances cell apoptosis as a potential contributor to bone loss. Intriguingly, in vitro studies on bone cells have shown that µG upregulates both antiapoptotic genes—including cyclin-dependent kinase inhibitor 1A (CDKN1A), B-cell lymphoma 2 (BCL2), and p21—as well as apoptotic genes—p53 and BCL2 Associated X (BAX)105. All these effects result in a reduction in osteoblast differentiation and activity.
These findings were observed in short-duration spaceflight, suggesting that apoptosis is temporarily inhibited during brief space missions in in vitro osteoblast culture. However, during extended spaceflights, cells become more predisposed to programmed cell death106.
Once osteoblasts remain trapped within the osteoid, they terminally differentiate into osteocytes, which cease the matrix deposition and take on their mechanosensory role within bone lamellae71. When exposed to a μG environment, osteocytes have also been observed to undergo apoptosis. Histological analysis of postflight samples has revealed a doubling of empty bone lacunae, indicating osteocyte death107. Moreover, spaceflight induces alterations in lacunar shape and volume, causing osteocytes to gradually assume a more spherical and smaller shape107. A computational analysis of the 3D structure of osteocyte lacunae, aimed at explaining the activation of apoptosis, revealed that in μG, the velocity of solute transport is approximately 600 times slower29. This implies that osteocytes receive fewer nutrients and experience compromised waste removal, ultimately driving them towards programmed cell death29.
To substantiate the hypothesis that osteocytes undergo apoptosis in μG, osteocyte cultures were subjected to μG and concurrently treated with Irisin, a myokine known for its antiapoptotic function on bone cells108. As expected, Irisin increased the expression of Activating Transcription Factor 4 (Atf4) mRNA, thereby mitigating bone loss in μG108.
Unfortunately, osteocytes remain the least studied bone cell type in μG, primarily due to their inaccessibility within bone lacunae and the difficulty of replicating their native 3D matrix environment in experimental settings and function.
Osteoclastogenesis increases and osteoclasts’ functional activation due to microgravity
Mechanosensitive osteoclasts contribute to space-related bone loss by disrupting normal bone homeostasis. In response to μG exposure, osteoclasts increase their resorptive activity when compared to control cells exposed to gravity on Earth4. This evidence has been further supported by the effects of μG on the molecular regulation of pro-osteoclastogenic genes. The expression of the Tumor Necrosis Factor-Related Apoptosis-Inducing Ligand (TRAIL) increased in osteoclast precursors subjected to μG109, increasing their differentiation into a mature form110. The upregulation of TRAIL indeed activated the TNF receptor-associated factor (TRAF6)-signaling pathway, leading to increased osteoclast formation111. Furthermore, TRAIL can weakly bind to OPG, a protein that inhibits osteoclast differentiation, thereby reducing its availability and elevating the RANKL/OPG ratio4.
Interestingly, the promotion of osteoclastogenesis through the increase in expression of sclerostin and RANKL also has a negative effect on osteocytes112. The increased expression of sclerostin can impair osteoblastic-osteoid deposition and osteocyte differentiation by negatively regulating the BMP/WNT signaling pathway113 while RANKL promotes directly osteoclastogenesis and osteoid resorption.
Additionally, μG has been demonstrated to enhance autophagy processes in pre-osteoclast cells, leading to increased expression of autophagy-related proteins such as Autophagy Related 5 (ATG5), light chain protein 3 (LC3), and autophagy-related protein 16L (Atg16L)114. Autophagy, as a recycling process for nutrients and energy, gives a boost to pre-osteoclasts to differentiate and activate their bone resorption activity114. This enhancement is also facilitated by a higher presence of pro-inflammatory cytokines115, including Chemokine Tumor Necrosis Factor Ligand Superfamily Member 10 (Tnfsf10). Sambandam et al. demonstrated that the administration of autophagy inhibitors, like 3’-metyladenine, greatly decreased Cathepsin K expression (marker for bone resorption116), suggesting a possible target to modulate osteoclastogenesis in μG114.
Collectively, these data suggest the potential for abnormal osteoclast differentiation and overgrowth, which—if sustained over time—could lead to the significant bone resorption observed in astronauts. Notably, this surge in osteoclast numbers is primarily observed during the initial 15 days of exposure to μG98, as well as during immobilization during bed-rest experiments106. In this early stage, bone loss occurs at a rate of 12–24% per month98. Subsequently, the rate decreases to the well-documented 1–2% loss per month4.
PC in bone cells’ response to microgravity
PC are significantly reduced in osteoblasts exposed to μG97 (Fig. 4). Shi and collaborators demonstrated that PC are significantly reduced in osteoblasts exposed to μG by 60.5 ± 10.4% in PC-presenting cells (reaching a maximum reduction of 85 ± 1.9% after 24 h) as soon as after 6 h of culture in simulated μG97. Additionally, they observed an average reduction in PC length from 5.11 ± 0.89 to 0.79 ± 0.18 μm97. In this regard, Shi and colleagues also demonstrated that PC abrogation may impair osteoblastogenesis97. The authors treated cultured osteoblasts with Cytochalasin D, an inhibitor of actin polymerization that blocks PC elongation, and then subjected them to simulated μG. As a result, osteoblasts’ number and differentiation capabilities were not affected anymore by μG in the absence of PC97. This was accompanied by restored expression of osteogenic marker, an increase in PC-presenting cells, and acetylated-α tubulin (a microtubule post-translational modification that is highly present in PC axoneme) levels, which were also downregulated in μG97.
Synthetic representation of microgravity effects on osteoclasts (a), osteoblasts (b), and on bone cell tensegrity127 (c). Created with BioRender.com.
By contrast, osteoclasts and osteocytes do not rely primarily on PC presence to be active and receptive to mechanical stimuli (Fig. 4).
Indeed, to fully differentiate from monocyte precursors, osteoclasts must undergo the PC disassembly. In fact, macrophages treated with fenoldopam mesylate—a dopamine D1-like receptor agonist used to increase PC length and stability—the expression of key regulators of osteoclast differentiation, including c-Fos, CTSK, and acp5, was downregulated117.
Nonetheless, μG was also shown to increase osteoclast functionality by increasing resorption pit118 (Fig. 4). Because these effects were shown to increase the osteolytic processes118 and the effect of PC reduction was seen in other cell type97, we could infer that µG affects pre-osteoclasts maturation to fully functional osteoclasts by disrupting primary cilium incidence on cells.
The contribution of the PC in bone cell signaling yet represents a topic of debate, partially due to the limited space gap between osteocyte and pericellular matrix (PCM) inside the bone lacunae, often considered too small for the PC to bend.
The osteocyte-lacunar wall gap was measured to be from 0.1 to 2 μm119 with a PCM that does not touch the cell body that is 0.5–1 μm thick54. While the PC length was measured to be 4–9 μm in vitro120 and 1–4 μm in vivo, with an average of 1.62 μm34,82,121,122. In the first case, if the PC is long as in the in vitro condition, inside the lacunae, it will be curled, or it will be touching and connected to the PCM, gaining a different behavior, more akin to the PC in the chondrocytes, hence being more prone to stretching than bending123.
The second case can be split into two: the short-length scenario and the long-length scenario. If we consider the PC to be below 1 μm, its functionality will be completely abolished or highly compromised124,125. In such cases, other mechanotransducers—such as the tethering elements within the osteocytes processes of the LCS system—may compensate for the loss of PC function in osteocytes82. This scenario may be plausible given the high fluid drag within the canaliculi; however, it may not be sufficient to transmit mechanotransduction signals as effectively as the PC.
Alternatively, a length of 1.62 μm may allow the PC to maintain its role as a mechanosensor by permitting sufficient bending and mobility in response to ECF flow34,82.
All these different measurements demonstrated how particular and unique the osteocyte lacuna is, and a 2D culture will never be totally perfect for studying certain pathways and especially for mechanosensors.
Given that, as mentioned earlier, shorter PC are less stiff and more prone to bending compared to longer ones43, it might be speculated that the PC senses the reduction of fluid flow due to μG and tries to compensate for it by depolymerizing the axoneme and the microtubules’ network to reduce its length and flexural rigidity.
Microgravity effects on bone cell tensegrity and ciliary mechanosensing
The PC represents a notable paradigm of cellular adaptation to gravity, responsible not only for establishing planar cell polarity but also for cell mechanosensing. The functionality of the PC has been correlated with cyclical changes in Earth’s gravitational field, as evidenced in zebrafish embryos, where gene expression activation was dependent on the presence of the cilium during low and high tides, representing lower and higher Earth gravity, respectively14.
Over the past two decades, researchers have intensified their investigations into the PC, especially focusing on its role in μG conditions, sparked by the reduction in the number of osteoblasts observed in μG and the understanding that μG alters mechanical stimuli.
Microgravity also significantly impacts cell shape and overall cell architecture, from FA to cell morphology, all of which undergo changes during weightlessness. Typically, osteoblasts exhibit a cuboidal cell morphology, but simulated μG in a random positioning machine (RPM) causes cells to adopt a more spindle-shaped or rounder form126,127. These changes in cellular shape are primarily a consequence of cytoskeletal reorganization. Stress fibers (actin and myosin) and FAs (Paxillin) are drastically reduced, meanwhile microtubules undergo a gradual depolymerization in cells exposed to μG15 compared to control cells grown in normal gravity101. This results in the disruption of PC and in decreased interconnectivity between cells and the ECM, which plays a key role in mechanotransduction127, connecting external stimuli to the cytoskeleton. Therefore, the reduction in Paxillin expression could alter the mechanosensitivity of osteoblasts.
Microgravity also causes an alteration of cell tensile and compressive forces, compromising cell tensegrity128 and directly inhibiting differentiation, as well as reducing ciliary length. Osteoblasts were seen as larger because of F-actin and internal stress-fiber thinning and reorganization, along with changes in nuclear morphology. This compromises IFT, which represents a critical component for mechanosensitivity and mechanotransduction129.
Although these effects were not directly correlated with the PC, previous in vitro studies on mice subjected to μG have shown a reduction in FA maturation and stabilization, which is associated with a reorganization of acetylated microtubules, leading to increased microtubule restructuring. Acetylated microtubules are highly abundant in the PC axoneme, and during μG, they have been observed to become shorter and curlier, preventing them from extending long distances from the perinuclear area. This, in turn, alters osteoblast tensegrity118.
Cell swelling leads to increased activation of phospholipase A2 (PLA2), which promotes the metabolism of arachidonic acid, thereby raising levels of epoxyeicosatrienoic acid (EET)130. These EETs can directly bind a site on TRPV4, a SAC highly localized in the PC (notably 5′,6′-EET), thereby gating the channel in response to osmotic/mechanical stress131. This binding causes increased channel opening and activity, which can result in Ca²⁺ overload and mitochondrial dysfunction125,131.
Notably, when cells are pretreated with either chloral hydrate—a sedative-hypnotic drug known to induce ciliary loss—or siRNA against IFT88—blocking IFT trafficking and ciliogenesis—the depolymerization of microtubules induced by μG conditions is attenuated15. Consistently, a recent work showed that microgravity alters ciliary dynamics by increasing the ratio of anterograde/retrograde IFT, leading to primary cilium shortening132, suggesting that simulated microgravity-induced depolymerization of microtubules requires the presence of PC. Additionally, cells treated with docetaxel trihydrate (DOC)—a microtubule stabilizer—under μG conditions exhibit reduced levels of PC expression, from 60.8% to 26.7%. However, the effects of μG on ALP activity as well as on the expression of RUNX2 and Osterix (OSX) osteospecific genes are rescued by DOC treatment, leading to enhanced osteogenesis and bone mineralization through stabilization of the microtubule network15. These results suggest the pivotal role of the PC in signaling the μG condition to the cell and reducing osteoblastic differentiation. It appears that microtubule depolymerization starts from the primary cilium tip and then is sent to the ciliary transition zone, where the PC’s microtubules are interconnected with the entire microtubule network system and then hindering the cytoskeleton structure and transducing the μG effects inside the cell body15.
Targeting the primary cilium to counteract microgravity effects
Given its key role as a bone cell’s mechanosensor and its importance in maintaining cellular homeostasis under μG, the PC can be considered a promising target for mitigating the adverse effects of spaceflights.
A few studies have already tested various effectors to protect the PC expression and function, primarily by mimicking normal physiological conditions, thereby providing new insights for future research.
As already introduced before, an interesting mechanical induction method used to increase PC presence or activity is the usage of an electromagnetic field (EMF), such as SEMF or PEMF133. These physical stimuli have already been recognized as safe and effective treatments for osteoporosis, also in space environments, due to their ability to increase bone density and promote osteogenic differentiation134,135. Notably, the efficacy of this mechanical stimulation required the presence of PC on bone cells to activate bone deposition. EMF exposure has been shown to directly promote elongation of the PC axoneme and enhance the activity of its associated signaling pathways, thereby facilitating bone formation75.
During μG, the whole cytoskeleton is subjected to a different load, causing MT depolymerization and impaired axoneme assembly118. To counteract these effects and stabilize the axoneme, two pharmacological strategies have been explored: Cytochalasin D, an inhibitor of actin polymerization that indirectly favors microtubule polymerization, and Docetaxel, a well-known microtubule stabilizer15,97. Although only Cytochalasin D increased PC presence and PC length on bone cells, both treatments were able to decrease bone resorption, while increasing bone formation15,97.
Lastly, μG is known to enhance free radical and oxidative stress formation that leads to bone density loss125. Recent findings suggest that PC abrogation may be one of the underlying causes. In this regard, the use of antioxidants to protect the PC has been associated with increased osteogenesis and a higher prevalence of ciliated cells125. Specifically, the proposed mechanism behind the increase in oxidative stress involves dysregulated TRPV4 activity. Following the loss of the PC, TRPV4 expression significantly increases and translocates to the cytoplasm, where it elevates intracellular calcium levels. This, in turn, leads to mitochondrial Ca²⁺ overload and a consequent rise in reactive oxygen species (ROS) production125.
To understand and evaluate if TRPV4 could be a possible therapeutic target to attenuate μG-driven oxidative stress, a TRPV4 antagonist called HC-067047 has been evaluated125. Miao and colleagues demonstrated that when administered to osteoblast cultures, this compound decreased Ca2+ influx and reduced ROS formation, increasing osteogenesis, thus confirming the importance of PC in maintaining low and localized TRPV4 expression in vitro125. Also, plant-derived molecules and extracts were screened based on their known antioxidant activity. Among these, moslosooflavone (MFL), a flavonoid isolated from natural compounds with antioxidant and anti-inflammatory properties, was identified as a promising candidate. Cell treatment with MFL increased PC length, reduced TRPV4 expression, and restored the osteogenic potential of bone cells during μG125. Interestingly, other studies using plant-derived molecules with antioxidant properties had similar effects on preventing bone cell loss during μG136,137,138,139.
Discussions
“Life as we know it” has evolved under Earth’s gravitational field, as reflected in the way cells have developed the ability to sense mechanical stimuli and transduce them into intracellular signals. Mechanotransduction is crucial for maintaining tissue homeostasis, and any alteration in mechanical input, such as in space and μG, increases the likelihood of health issues. The wide and heterogeneous pieces of scientific evidence collected and analyzed in this review focus on the modification or perturbation of bone tissue homeostasis in μG conditions due to its crucial role in sustaining other organs and to its direct correlation implication in mechanical sensing throughout the body.
Microgravity affects the movement and transport of fluids, directly impacting bone cells by reducing nutrient transport and increasing apoptosis in osteocytes, increasing osteoclasts' growth rate and differentiation speed, meanwhile also affecting osteoblasts' differentiation and reducing the mesenchymal stromal cells pool, affecting astronauts' bone healing speed when coming back on earth. Moreover, it indirectly alters the mechanical input sensed by PC or other cellular organelles, disrupting downstream signaling pathways. The PC undergoes various changes due to μG, including its disappearance from most cells and shortening in the remaining ones. μG completely alters cell shape and cytoskeletal architecture, compromising IFT within the PC and the whole cell.
In conclusion, μG detrimentally affects every aspect of bone homeostasis, from microfluidic alterations to subcellular and protein changes, ultimately accelerating bone density loss compared to the natural course of an osteoporotic patient on Earth. In all of these effects, the primary cilium-related pathways are in the majority of cases to get affected. Considering the significance of deep space exploration and the colonization of new planets for humanity, it is imperative to investigate these mechanisms fundamental to cell viability. An increasing number of emerging studies are uncovering novel mechanisms by which the PC contributes to altered mechanotransduction and bone loss under μG conditions. As a result, the PC is now considered a promising therapeutic target for mitigating the adverse effects of μG on bone cells. This has initiated the screening of molecules aimed to reduce space effects on bone tissue that are mainly directed to exert mechanical induction, enhancing PC functionality, or counteracting the oxidative stress induced by spaceflight. Among these treatment strategies, modulation of TRPV4 overexpression stands out as particularly promising, as it addresses one of the primary drivers of ROS production and mitochondrial dysfunction.
An improved knowledge in this field may lead to the development of novel potential strategies to prevent and restore correct cell homeostasis in astronauts. Moreover, understanding key concepts in extreme environments like space can propel a better understanding of bone and PC-related diseases, such as osteoporosis.
Methods
Objective of the review
This review has been conceived to delve into a deeper understanding of the μG effects that appear to act via the PC in bone tissue. We will analyze the details of PC-mediated mechanobiology to give a comprehensive picture of its impact on human bone cells' physiology and homeostasis, and deduce the implications in space biology. Collecting and critically analyzing existing scientific evidence that connects the PC to the mechanosensing and mechanotransduction governing bone homeostasis would provide new insights towards clarifying the mechanisms involved in tissue adaptation to μG. Focusing on the PC might be particularly relevant for future human space explorations, being a discrete spot on the cell surface, hence a putative druggable target. This will possibly provide novel hints paving the way to the design of innovative therapeutic interventions targeting mechanotransduction-related bone diseases, such as osteoporosis.
Study design and search strategy
The present study is a systematic review reported according to the Preferred Reporting Items for Systematic review and Meta-Analyses (PRISMA) guidelines140 PubMed, ScienceDirect & Scopus database were searched using the keywords: “Bone”, “microgravity”, “space”, “weightlessness”, “primary cilium/a”, “osteoblast”, “osteoclast”, “osteocyte”, “mechanotransduction”, “mechanosensitive”, “cytoskeleton”, “microtubules”, “actin”, “calcium”, “ion channel” and “fluid dynamics”, and their MeSH terms in any possible combination using the logical operators “AND” and “OR”.
Inclusion and exclusion criteria
All the reviews, original articles, and book chapters, which were with free open access or were available as full text versions in scientific publication, databases and open access resources (PubMed, ScienceDirect, Scopus), describing the effects of space or microgravity on bone cells/primary cilium and/or regarding the change of mechanotransduction/fluid dynamics in microgravity/primary cilium, were considered eligible. No time limits were established regarding the publication date of the articles. The studies to be included had to be written in English only.
Data extraction and analysis
The authors independently searched and collected data from all the studies found by the inclusion/exclusion criteria (Fig. 5).
The flow diagram represents the flow of information through the different phases of the systematic, with paper identification number, inclusion and exclusion criteria, following the “Prisma 2020 flow diagram chart”139. Created with BioRender.com.
The exclusion process of not relevant, duplicates, or out-of-focus articles was performed with the website “Rayyan”141.
Data availability
All data generated or analyzed during this study are included in this published article.
References
Tagliaferri, C., Wittrant, Y., Davicco, M.-J., Walrand, S. & Coxam, V. Muscle and bone, two interconnected tissues. Ageing Res. Rev. 21, 55–70 (2015).
Juhl, O. J. et al. Update on the effects of microgravity on the musculoskeletal system. NPJ Microgravity 7, 28 (2021).
Vico, L. et al. Effects of long-term microgravity exposure on cancellous and cortical weight-bearing bones of cosmonauts. Lancet Lond. Engl. 355, 1607–1611 (2000).
Man, J., Graham, T., Squires-Donelly, G. & Laslett, A. L. The effects of microgravity on bone structure and function. Npj Microgravity 8, 1–15 (2022).
Caillot-Augusseau, A. et al. Bone formation and resorption biological markers in cosmonauts during and after a 180-day space flight (Euromir 95). Clin. Chem. 44, 578–585 (1998).
Sugawara, Y. et al. The alteration of a mechanical property of bone cells during the process of changing from osteoblasts to osteocytes. Bone 43, 19–24 (2008).
Argentati, C. et al. Insight into mechanobiology: how stem cells feel mechanical forces and orchestrate biological functions. Int. J. Mol. Sci. 20, 5337 (2019).
Chen, Y., Ju, L., Rushdi, M., Ge, C. & Zhu, C. Receptor-mediated cell mechanosensing. Mol. Biol. Cell 28, 3134–3155 (2017).
Di, X. et al. Cellular mechanotransduction in health and diseases: from molecular mechanism to therapeutic targets. Signal Transduct. Target. Ther. 8, 282 (2023).
Shutova, M. S. & Boehncke, W.-H. Mechanotransduction in skin inflammation. Cells 11, 2026 (2022).
Stoufflet, J. et al. Primary cilium-dependent cAMP/PKA signaling at the centrosome regulates neuronal migration. Sci. Adv. 6, eaba3992 (2020).
Wheway, G., Nazlamova, L. & Hancock, J. T. Signaling through the primary cilium. Front. Cell Dev. Biol. 6, 8 (2018).
Jun, J. H., Lee, E. J., Park, M., Ko, J. Y. & Park, J. H. Reduced expression of TAZ inhibits primary cilium formation in renal glomeruli. Exp. Mol. Med. 54, 169–179 (2022).
Moorman, S. J. & Shorr, A. Z. The primary cilium as a gravitational force transducer and a regulator of transcriptional noise. Dev. Dyn. 237, 1955–1959 (2008).
Shi, W. et al. Primary cilia act as microgravity sensors by depolymerizing microtubules to inhibit osteoblastic differentiation and mineralization. Bone 136, 115346 (2020).
Martin, B. Aging and strength of bone as a structural material. Calcif. Tissue Int. 53, S34–S40 (1993).
Feng, X. Chemical and biochemical basis of cell–bone matrix interaction in health and disease. Curr. Chem. Biol. 3, 189–196 (2009).
Franz-Odendaal, T. A., Hall, B. K. & Witten, P. E. Buried alive: how osteoblasts become osteocytes. Dev. Dyn. 235, 176–190 (2006).
Palumbo, C. & Ferretti, M. The osteocyte: from “Prisoner” to “Orchestrator”. J. Funct. Morphol. Kinesiol. 6, 28 (2021).
Ma, X. et al. Functional role of vanilloid transient receptor potential 4-canonical transient receptor potential 1 complex in flow-induced Ca2+ influx. Arterioscler. Thromb. Vasc. Biol. 30, 851–858 (2010).
Robling, A. G., Castillo, A. B. & Turner, C. H. Biomechanical and molecular regulation of bone remodeling. Annu. Rev. Biomed. Eng. 8, 455–498 (2006).
Chen, N. X. et al. Ca(2+) regulates fluid shear-induced cytoskeletal reorganization and gene expression in osteoblasts. Am. J. Physiol. Cell Physiol. 278, C989–C997 (2000).
Roux, W. Beiträge zur Entwickelungsmechanik des Embryo. Anat. Hefte 2, 277–333 (1893).
Wolff, J. Concept of the law of bone remodelling. In The Law of Bone Remodelling (ed. Wolff, J.) 1–1 (Springer, 1986).
Frost, H. M. The mechanostat: a proposed pathogenic mechanism of osteoporoses and the bone mass effects of mechanical and nonmechanical agents. Bone Mineral 2, 73–85 (1987).
Burr, D. B. et al. In vivo measurement of human tibial strains during vigorous activity. Bone 18, 405–410 (1996).
Fritton, S. P., McLeod, K. J. & Rubin, C. T. Quantifying the strain history of bone: spatial uniformity and self-similarity of low-magnitude strains. J. Biomech. 33, 317–325 (2000).
Carter, D. R., Fyhrie, D. P. & Whalen, R. T. Trabecular bone density and loading history: regulation of connective tissue biology by mechanical energy. J. Biomech. 20, 785–794 (1987).
Liu, H.-Y. et al. Research on solute transport behaviors in the lacunar-canalicular system using numerical simulation in microgravity. Comput. Biol. Med. 119, 103700 (2020).
Stavnichuk, M., Mikolajewicz, N., Corlett, T., Morris, M. & Komarova, S. V. A systematic review and meta-analysis of bone loss in space travelers. Npj Microgravity 6, 1–9 (2020).
Dobner, S., Amadi, O. C. & Lee, R. T. Chapter 14 - Cardiovascular Mechanotransduction. in Muscle (eds. Hill, J. A. & Olson, E. N.) 173–186 (Academic Press, 2012).
Uray, I. P. & Uray, K. Mechanotransduction at the plasma membrane–cytoskeleton interface. Int. J. Mol. Sci. 22, 11566 (2021).
Freund, J. B., Goetz, J. G., Hill, K. L. & Vermot, J. Fluid flows and forces in development: functions, features and biophysical principles. Development 139, 1229–1245 (2012).
Qin, L., Liu, W., Cao, H. & Xiao, G. Molecular mechanosensors in osteocytes. Bone Res. 8, 1–24 (2020).
Zimmermann, K. W. Beiträge zur Kenntniss einiger Drüsen und Epithelien. Arch. Mikrosk. Anat. 52, 552–706 (1898).
Cameron, D. A. The ultrastructure of bone. In The Biology and Physiology of Bone 2nd edn (ed. Bourne, G. H.) Ch. 6, 191–236 (Academic Press, 1972).
Wheatley, D. N., Wang, A. M. & Strugnell, G. E. Expression of primary cilia in mammalian cells. Cell Biol. Int. 20, 73–81 (1996).
Boisvieux-Ulrich, E., Sandoz, D. & Allart, J.-P. Determination of ciliary polarity precedes differentiation in the epithelial cells of quail oviduct. Biol. Cell 72, 3–14 (1991).
Albrecht-Buehler, G. & Bushnell, A. The ultrastructure of primary cilia in quiescent 3T3 cells. Exp. Cell Res. 126, 427–437 (1980).
Downing, K. H. & Sui, H. Structural insights into microtubule doublet interactions in axonemes. Curr. Opin. Struct. Biol. 17, 253–259 (2007).
Saternos, H., Ley, S. & AbouAlaiwi, W. Primary cilia and calcium signaling interactions. Int. J. Mol. Sci. 21, 7109 (2020).
Kiesel, P. et al. The molecular structure of mammalian primary cilia revealed by cryo-electron tomography. Nat. Struct. Mol. Biol. 27, 1115–1124 (2020).
Flaherty, J., Feng, Z., Peng, Z., Young, Y.-N. & Resnick, A. Primary cilia have a length-dependent persistence length. Biomech. Model. Mechanobiol. 19, 445–460 (2020).
Sun, S.-Y., Zhang, L.-Y., Chen, X. & Feng, X.-Q. Biochemomechanical tensegrity model of cytoskeletons. J. Mech. Phys. Solids 175, 105288 (2023).
Garcia-Gonzalo, F. R. & Reiter, J. F. Open sesame: how transition fibers and the transition zone control ciliary composition. Cold Spring Harb. Perspect. Biol. 9, a028134 (2017).
Satir, P. & Gilula, N. B. The cell junction in a Lamellibranch Gill ciliated epithelium. J. Cell Biol. 47, 468–487 (1970).
Nechipurenko, I. V. The enigmatic role of lipids in cilia signaling. Front. Cell Dev. Biol. 8, 777 (2020).
Hoey, D. A., Tormey, S., Ramcharan, S., O’Brien, F. J. & Jacobs, C. R. Primary cilia-mediated mechanotransduction in human mesenchymal stem cells. Stem Cells 30, 2561–2570 (2012).
Jin, X. et al. Cilioplasm is a cellular compartment for calcium signaling in response to mechanical and chemical stimuli. Cell. Mol. Life Sci. 71, 2165–2178 (2014).
Molla-Herman, A. et al. The ciliary pocket: an endocytic membrane domain at the base of primary and motile cilia. J. Cell Sci. 123, 1785–1795 (2010).
Davenport, J. R. & Yoder, B. K. An incredible decade for the primary cilium: a look at a once-forgotten organelle. Am. J. Physiol. Renal Physiol. 289, F1159–F1169 (2005).
Gherman, A., Davis, E. E. & Katsanis, N. The ciliary proteome database: an integrated community resource for the genetic and functional dissection of cilia. Nat. Genet. 38, 961–962 (2006).
Li, D. & Wang, Y. Mechanobiology, tissue development, and tissue engineering. In Principles of Tissue Engineering 5th edn, Ch. 14 (eds. Lanza, R., Langer, R., Vacanti, J. P. & Atala, A.) 237–256 (Academic Press, 2020).
Subauste, M. C. et al. Vinculin modulation of paxillin–FAK interactions regulates ERK to control survival and motility. J. Cell Biol. 165, 371–381 (2004).
McNamara, L., Majeska, R., Weinbaum, S., Friedrich & Schaffler, M. Attachment of osteocyte cell processes to the bone matrix. Anat. Rec. Hoboken NJ 2007 292, 355–363 (2009).
Xu, Z., Zhang, D., He, X., Huang, Y. & Shao, H. Transport of calcium ions into mitochondria. Curr. Genom. 17, 215–219 (2016).
Lauritzen, I. et al. Cross-talk between the mechano-gated K2P channel TREK-1 and the actin cytoskeleton. EMBO Rep. 6, 642–648 (2005).
Maniotis, A. J., Chen, C. S. & Ingber, D. E. Demonstration of mechanical connections between integrins, cytoskeletal filaments, and nucleoplasm that stabilize nuclear structure. Proc. Natl. Acad. Sci. USA 94, 849–854 (1997).
Maurer, M. & Lammerding, J. The driving force: nuclear mechanotransduction in cellular function, fate, and disease. Annu. Rev. Biomed. Eng. 21, 443–468 (2019).
Helmke, B. P., Thakker, D. B., Goldman, R. D. & Davies, P. F. Spatiotemporal analysis of flow-induced intermediate filament displacement in living endothelial cells. Biophys. J. 80, 184–194 (2001).
Ackbarow, T., Sen, D., Thaulow, C. & Buehler, M. J. Alpha-helical protein networks are self-protective and flaw-tolerant. PLoS ONE 4, e6015 (2009).
Dupont, S. et al. Role of YAP/TAZ in mechanotransduction. Nature 474, 179–183 (2011).
Habbig, S. et al. NPHP4, a cilia-associated protein, negatively regulates the Hippo pathway. J. Cell Biol. 193, 633–642 (2011).
Suarez Rodriguez, F., Sanlidag, S. & Sahlgren, C. Mechanical regulation of the Notch signaling pathway. Curr. Opin. Cell Biol. 85, 102244 (2023).
Stassen, O. M. J. A., Ristori, T. & Sahlgren, C. M. Notch in mechanotransduction—from molecular mechanosensitivity to tissue mechanostasis. J. Cell Sci. 133, jcs250738 (2020).
Stasiulewicz, M. et al. A conserved role for Notch signaling in priming the cellular response to Shh through ciliary localisation of the key Shh transducer Smo. Development 142, 2291–2303 (2015).
Ezratty, E. J., Pasolli, H. A. & Fuchs, E. A presenilin-2-ARF4 trafficking axis modulates notch signaling during epidermal differentiation. J. Cell Biol. 214, 89–101 (2016).
Huangfu, D. et al. Hedgehog signalling in the mouse requires intraflagellar transport proteins. Nature 426, 83–87 (2003).
Ezratty, E. J. et al. A role for the primary cilium in notch signaling and epidermal differentiation during skin development. Cell 145, 1129–1141 (2011).
Anvarian, Z., Mykytyn, K., Mukhopadhyay, S., Pedersen, L. B. & Christensen, S. T. Cellular signalling by primary cilia in development, organ function and disease. Nat. Rev. Nephrol. 15, 199–219 (2019).
Choi, J. U. A., Kijas, A. W., Lauko, J. & Rowan, A. E. The mechanosensory role of osteocytes and implications for bone health and disease states. Front. Cell Dev. Biol. 9, (2022).
Zhou, J. et al. Sinusoidal electromagnetic fields increase peak bone mass in rats by activating Wnt10b/β-catenin in primary cilia of osteoblasts. J. Bone Miner. Res. 34, 1336–1351 (2019).
Kim, S. et al. The polycystin complex mediates Wnt/Ca2+ signalling. Nat. Cell Biol. 18, 752–764 (2016).
Russwurm, M., Russwurm, C., Koesling, D. & Mergia, E. NO/cGMP: the past, the present, and the future. Methods Mol. Biol. 1020, 1–16 (2013).
He, W.-F. et al. The interdependent relationship between the nitric oxide signaling pathway and primary cilia in pulse electromagnetic field-stimulated osteoblastic differentiation. FASEB J. 36, e22376 (2022).
Shi, W. et al. The flavonol glycoside icariin promotes bone formation in growing rats by activating the cAMP signaling pathway in primary cilia of osteoblasts. J. Biol. Chem. 292, 20883–20896 (2017).
Sassone-Corsi, P. The cyclic AMP pathway. Cold Spring Harb. Perspect. Biol. 4, a011148 (2012).
Paolocci, E. & Zaccolo, M. Compartmentalised cAMP signalling in the primary cilium. Front. Physiol. 14, 1187134 (2023).
Kwon, R. Y., Temiyasathit, S., Tummala, P., Quah, C. C. & Jacobs, C. R. Primary cilium-dependent mechanosensing is mediated by adenylyl cyclase 6 and cyclic AMP in bone cells. FASEB J. 24, 2859–2868 (2010).
Tschaikner, P., Enzler, F., Torres-Quesada, O., Aanstad, P. & Stefan, E. Hedgehog and Gpr161: regulating cAMP signaling in the primary cilium. Cells 9, 118 (2020).
Yoder, B. K., Hou, X. & Guay-Woodford, L. M. The polycystic kidney disease proteins, polycystin-1, polycystin-2, polaris, and cystin, are co-localized in renal cilia. J. Am. Soc. Nephrol. 13, 2508 (2002).
Verbruggen, S. W., Sittichokechaiwut, A. & Reilly, G. C. Osteocytes and primary cilia. Curr. Osteoporos. Rep. 21, 719–730 (2023).
Temiyasathit, S. Osteocyte primary cilium and its role in bone mechanotransduction. Ann. N. Y. Acad. Sci. https://nyaspubs.onlinelibrary.wiley.com/doi/10.1111/j.1749-6632.2009.05243.x (2010).
Xiao, Z. et al. Cilia-like structures and polycystin-1 in osteoblasts/osteocytes and associated abnormalities in skeletogenesis and Runx2 expression. J. Biol. Chem. 281, 30884–30895 (2006).
Gargalionis, A. N., Adamopoulos, C., Vottis, C. T., Papavassiliou, A. G. & Basdra, E. K. Runx2 and polycystins in bone mechanotransduction: challenges for therapeutic opportunities. Int. J. Mol. Sci. 25, 5291 (2024).
Xiao, Z. et al. Osteoblast-specific deletion of Pkd2 leads to low-turnover osteopenia and reduced bone marrow adiposity. PLoS ONE 9, e114198 (2014).
Xiao, Z., Zhang, S., Magenheimer, B. S., Luo, J. & Quarles, L. D. Polycystin-1 regulates skeletogenesis through stimulation of the osteoblast-specific transcription factor RUNX2-II. J. Biol. Chem. 283, 12624–12634 (2008).
Grimm, D. Microgravity and space medicine. Int. J. Mol. Sci. 22, 6697 (2021).
White, R. J. & Averner, M. Humans in space. Nature 409, 1115–1118 (2001).
Moosavi, D. et al. The effects of spaceflight microgravity on the musculoskeletal system of humans and animals, with an emphasis on exercise as a countermeasure: a systematic scoping review. Physiol. Res. 70, 119–151 (2021).
Nagaraja, M. P. & Risin, D. The current state of bone loss research: data from spaceflight and microgravity simulators. J. Cell. Biochem. 114, 1001–1008 (2013).
Belavy, D. L., Beller, G., Ritter, Z. & Felsenberg, D. Bone structure and density via HR-pQCT in 60d bed-rest, 2-years recovery with and without countermeasures. J. Musculoskelet. Neuronal Interact. 11, 215–226 (2011).
Gabel, L. et al. Pre-flight exercise and bone metabolism predict unloading-induced bone loss due to spaceflight. Br. J. Sports Med. 56, 196–203 (2022).
Sibonga, J. et al. Resistive exercise in astronauts on prolonged spaceflights provides partial protection against spaceflight-induced bone loss. Bone 128, 112037 (2019).
Fox, R. W., McDonald, A. T. & Mitchell, J. W. Fox and McDonald’s Introduction to Fluid Mechanics (John Wiley & Sons, 2020).
Smith, J. K. Osteoclasts and microgravity. Life 10, 207 (2020).
Shi, W. et al. Microgravity induces inhibition of osteoblastic differentiation and mineralization through abrogating primary cilia. Sci. Rep. 7, 1866 (2017).
Blaber, E. A. et al. Microgravity induces pelvic bone loss through osteoclastic activity, osteocytic osteolysis, and osteoblastic cell cycle inhibition by CDKN1a/p21. PLoS ONE 8, e61372 (2013).
De Leon-Oliva, D. et al. The RANK–RANKL–OPG system: a multifaceted regulator of homeostasis, immunity, and cancer. Medicina (Mexico) 59, 1752 (2023).
Vimalraj, S. Alkaline phosphatase: structure, expression and its function in bone mineralization. Gene 754, 144855 (2020).
Fan, C. et al. Activation of focal adhesion kinase restores simulated microgravity-induced inhibition of osteoblast differentiation via Wnt/Β-catenin pathway. Int. J. Mol. Sci. 23, 5593 (2022).
Zayzafoon, M., Gathings, W. E. & McDonald, J. M. Modeled microgravity inhibits osteogenic differentiation of human mesenchymal stem cells and increases adipogenesis. Endocrinology 145, 2421–2432 (2004).
Ducy, P., Zhang, R., Geoffroy, V., Ridall, A. L. & Karsenty, G. Osf2/Cbfa1: a transcriptional activator of osteoblast differentiation. Cell 89, 747–754 (1997).
Lecka-Czernik, B. et al. Inhibition of Osf2/Cbfa1 expression and terminal osteoblast differentiation by PPARgamma2. J. Cell. Biochem. 74, 357–371 (1999).
Chatziravdeli, V., Katsaras, G. N. & Lambrou, G. I. Gene expression in osteoblasts and osteoclasts under microgravity conditions: a systematic review. Curr. Genom. 20, 184–198 (2019).
Baecker, N. et al. Bone resorption is induced on the second day of bed rest: results of a controlled crossover trial. J. Appl. Physiol. 95, 977–982 (2003).
Gerbaix, M. et al. One-month spaceflight compromises the bone microstructure, tissue-level mechanical properties, osteocyte survival and lacunae volume in mature mice skeletons. Sci. Rep. 7, 2659 (2017).
Colucci, S. et al. Irisin prevents microgravity-induced impairment of osteoblast differentiation in vitro during the space flight CRS-14 mission. FASEB J. 34, 10096–10106 (2020).
Sambandam, Y. et al. Microgravity induction of TRAIL expression in preosteoclast cells enhances osteoclast differentiation. Sci. Rep. 6, 25143 (2016).
Brunetti, G. et al. TRAIL effect on osteoclast formation in physiological and pathological conditions. Front. Biosci. Elite Ed. 3, 1154–1161 (2011).
Yen, M.-L., Hsu, P.-N., Liao, H.-J., Lee, B.-H. & Tsai, H.-F. TRAF-6 dependent signaling pathway is essential for TNF-Related Apoptosis-Inducing Ligand (TRAIL) induces osteoclast differentiation. PLoS ONE 7, e38048 (2012).
Spatz, J. M. et al. The Wnt inhibitor sclerostin is up-regulated by mechanical unloading in osteocytes in vitro*. J. Biol. Chem. 290, 16744–16758 (2015).
Li, X. et al. Sclerostin binds to LRP5/6 and antagonizes canonical Wnt signaling*. J. Biol. Chem. 280, 19883–19887 (2005).
Sambandam, Y. et al. Microgravity control of autophagy modulates osteoclastogenesis. Bone 61, 125–131 (2014).
Lin, N.-Y., Stefanica, A. & Distler, J. H. W. Autophagy: a key pathway of TNF-induced inflammatory bone loss. Autophagy 9, 1253–1255 (2013).
Dai, R. et al. Cathepsin K: the action in and beyond bone. Front. Cell Dev. Biol. 8, 433 (2020).
Sutton, M. M., Duffy, M. P., Verbruggen, S. W. & Jacobs, C. R. Osteoclastogenesis requires primary cilia disassembly and can be inhibited by promoting primary cilia formation pharmacologically. Cells Tissues Organs 213, 235–244 (2024).
Nabavi, N., Khandani, A., Camirand, A. & Harrison, R. E. Effects of microgravity on osteoclast bone resorption and osteoblast cytoskeletal organization and adhesion. Bone 49, 965–974 (2011).
Wassermann, F. & Yaeger, J. A. Fine structure of the osteocyte capsule and of the wall of the lacunae in bone. Z. Zellforsch. Mikrosk. Anat. 67, 636–652 (1965).
Malone, A. M. D. et al. Primary cilia mediate mechanosensing in bone cells by a calcium-independent mechanism. Proc. Natl. Acad. Sci. USA 104, 13325–13330 (2007).
Coughlin, T. R., Voisin, M., Schaffler, M. B., Niebur, G. L. & McNamara, L. M. Primary cilia exist in a small fraction of cells in trabecular bone and marrow. Calcif. Tissue Int. 96, 65–72 (2015).
Uzbekov, R. et al. Centrosome fine ultrastructure of the osteocyte mechanosensitive primary cilium. Microsc. Microanal. 18, 1430–1441 (2012).
McGlashan, S. R., Jensen, C. G. & Poole, C. A. Localization of extracellular matrix receptors on the chondrocyte primary cilium. J. Histochem. Cytochem. 54, 1005–1014 (2006).
Vaughan, T. J., Mullen, C. A., Verbruggen, S. W. & McNamara, L. M. Bone cell mechanosensation of fluid flow stimulation: a fluid–structure interaction model characterising the role integrin attachments and primary cilia. Biomech. Model. Mechanobiol. 14, 703–718 (2015).
Miao, L.-W. et al. Simulated microgravity-induced oxidative stress and loss of osteogenic potential of osteoblasts can be prevented by protection of primary cilia. J. Cell. Physiol. 238, 2692–2709 (2023).
Hughes-Fulford, M. Physiological effects of microgravity on osteoblast morphology and cell biology. In Advances in Space Biology and Medicine, Vol. 8 (ed. Cogoli, A.) 129–157 (Elsevier, 2002).
Gioia, M. et al. Simulated microgravity induces a cellular regression of the mature phenotype in human primary osteoblasts. Cell Death Discov. 4, 1–14 (2018).
Andreeva, E. et al. Real and simulated microgravity: focus on mammalian extracellular matrix. Life 12, 1343 (2022).
Ingber, D. E. Tensegrity: the architectural basis of cellular mechanotransduction. Annu. Rev. Physiol. 59, 575–599 (1997).
Heller, S. & O’Neil, R. G. Molecular mechanisms of TRPV4 gating. In TRP Ion Channel Function in Sensory Transduction and Cellular Signaling Cascades (eds Liedtke, W. B. & Heller, S.) (CRC Press/Taylor & Francis, 2007).
Berna-Erro, A. et al. Structural determinants of 5′,6′-epoxyeicosatrienoic acid binding to and activation of TRPV4 channel. Sci. Rep. 7, 10522 (2017).
Ding, D. et al. The microgravity induces the ciliary shortening and an increased ratio of anterograde/retrograde intraflagellar transport of osteocytes. Biochem. Biophys. Res. Commun. 530, 167–172 (2020).
Lutz, K., Trevino, T. & Adrian, C. Modeling effects of electromagnetic fields on bone density on humans in microgravity. In Proc. of AIAA ASCEND 2022–4291 (American Institute of Aeronautics and Astronautics, 2022).
Zhang, T., Zhao, Z. & Wang, T. Pulsed electromagnetic fields as a promising therapy for glucocorticoid-induced osteoporosis. Front. Bioeng. Biotechnol. 11, 1103515 (2023).
Prakash, D. & Behari, J. Synergistic role of hydroxyapatite nanoparticles and pulsed electromagnetic field therapy to prevent bone loss in rats following exposure to simulated microgravity. Int. J. Nanomed. 4, 133–144 (2009).
Zwart, S. R., Morgan, J. L. L. & Smith, S. M. Iron status and its relations with oxidative damage and bone loss during long-duration space flight on the International Space Station. Am. J. Clin. Nutr. 98, 217–223 (2013).
Wang, N. et al. Salidroside alleviates simulated microgravity-induced bone loss by activating the Nrf2/HO-1 pathway. J. Orthop. Surg. 19, 531 (2024).
Xin, M. Attenuation of hind-limb suspension-induced bone loss by curcumin is associated with reduced oxidative stress and increased vitamin D receptor expression. Osteoporos. Int. https://link.springer.com/article/10.1007/s00198-015-3153-7 (2015).
Liang, X. et al. Angelicae dahuricae radix alleviates simulated microgravity induced bone loss by promoting osteoblast differentiation. Npj Microgravity 10, 1–14 (2024).
Page, M. J. et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ 372, 71 (2021).
Ouzzani, M., Hammady, H., Fedorowicz, Z. & Elmagarmid, A. Rayyan—a web and mobile app for systematic reviews. Syst. Rev. 5, 210 (2016).
Minke, B. & Cook, B. TRP channel proteins and signal transduction. Physiol. Rev. 82, 429–472 (2002).
Sinkins, W. G., Estacion, M. & Schilling, W. P. Functional expression of TrpC1: a human homologue of the Drosophila Trp channel. Biochem. J. 331, 331–339 (1998).
Tsiokas, L. et al. Specific association of the gene product of PKD2 with the TRPC1 channel. Proc. Natl. Acad. Sci. USA 96, 3934–3939 (1999).
Maroto, R. et al. TRPC1 forms the stretch-activated cation channel in vertebrate cells. Nat. Cell Biol. 7, 179–185 (2005).
Quick, K. et al. TRPC3 and TRPC6 are essential for normal mechanotransduction in subsets of sensory neurons and cochlear hair cells. Open Biol. 2, 120068 (2012).
Klein, S. et al. Modulation of transient receptor potential channels 3 and 6 regulates osteoclast function with impact on trabecular bone loss. Calcif. Tissue Int. 106, 655–664 (2020).
Shao, L. et al. Genetic reduction of cilium length by targeting intraflagellar transport 88 protein impedes kidney and liver cyst formation in mouse models of autosomal polycystic kidney disease. Kidney Int. 98, 1225–1241 (2020).
Masyuk, A. I. et al. Cholangiocyte cilia detect changes in luminal fluid flow and transmit them into intracellular Ca2+ and cAMP signaling. Gastroenterology 131, 911–920 (2006).
Lee, K. L. et al. The primary cilium functions as a mechanical and calcium signaling nexus. Cilia 4, 7 (2015).
Nomura, H. et al. Identification of PKDL, a novel polycystic kidney disease 2-like gene whose murine homologue is deleted in mice with kidney and retinal defects*. J. Biol. Chem. 273, 25967–25973 (1998).
DeCaen, P. G., Delling, M., Vien, T. N. & Clapham, D. E. Direct recording and molecular identification of the calcium channel of primary cilia. Nature 504, 315–318 (2013).
Fois, G. et al. An ultra fast detection method reveals strain-induced Ca2+ entry via TRPV2 in alveolar type II cells. Biomech. Model. Mechanobiol. 11, 959–971 (2012).
Köttgen, M. et al. TRPP2 and TRPV4 form a polymodal sensory channel complex. J. Cell Biol. 182, 437–447 (2008).
Corrigan, M. A. et al. TRPV4-mediates oscillatory fluid shear mechanotransduction in mesenchymal stem cells in part via the primary cilium. Sci. Rep. 8, 3824 (2018).
Acharya, T. K. et al. TRPV4 regulates osteoblast differentiation and mitochondrial function that are relevant for channelopathy. Front. Cell Dev. Biol. 11, 1066788 (2023).
Gayden, T. et al. Association of novel mutation in TRPV4 with familial nonsyndromic craniosynostosis with complete penetrance and variable expressivity. J. Neurosurg. Pediatr. 31, 584–592 (2023).
Cox, C. D. et al. Removal of the mechanoprotective influence of the cytoskeleton reveals PIEZO1 is gated by bilayer tension. Nat. Commun. 7, 10366 (2016).
Qin, L. et al. Roles of mechanosensitive channel Piezo1/2 proteins in skeleton and other tissues. Bone Res. 9, 44 (2021).
Lin, Y., Ren, J. & McGrath, C. Mechanosensitive Piezo1 and Piezo2 ion channels in craniofacial development and dentistry: recent advances and prospects. Front. Physiol. 13, 1039714 (2022).
Ashley, Z., Mugloo, S., McDonald, F. J. & Fronius, M. Epithelial Na+ channel differentially contributes to shear stress-mediated vascular responsiveness in carotid and mesenteric arteries from mice. Am. J. Physiol. Heart Circ. Physiol. 314, H1022–H1032 (2018).
Acknowledgements
This study was partially funded by Università Cattolica del Sacro Cuore “linea D.1 and linea D.3.1” to W.L. The authors acknowledge the Regenerative Medicine Research Center (CROME) of the Università Cattolica del Sacro Cuore.
Author information
Authors and Affiliations
Contributions
Conceptualization: D.D.T. and W.L.; writing—original draft preparation: D.D.T., F.T., L.P.; writing review and editing: W.L., F.T., A.A., A.M., and L.D.P.; funding acquisition: A.M., W.L., and O.P. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Tosi, D.D., Tiberio, F., Di Pietro, L. et al. Effects of microgravity mechanotransduction in bone tissue and cells: systematic review on primary cilium-dependent mechanisms. npj Microgravity 12, 12 (2026). https://doi.org/10.1038/s41526-025-00556-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41526-025-00556-y