Abstract
Twenty-four hours continuous intraocular pressure (IOP) monitoring is beneficial for glaucoma care. Contact lens sensors using LC technology can achieve non-invasive continuous IOP measuring around the clock and are relatively simple in principle and structure, thus dominating the mainstream. Among them, stretchable inductive LC contact lens sensors constructed with liquid metal have advantages in signal quality as well as wearing comfort, but currently its sensitivity is slightly insufficient. Here, we propose an LC sensor that constructed with liquid metal to form the stretchable inductance and capacitance further. The capacitive plate and inductive coil can response to IOP changes simultaneously, thus able to enhance the sensitivity in principle. We modeled the sensing mechanism and conducted design, fabrication, as well as various tests. The device exhibits good characteristics, including reliability, good signal quality, etc. Especially, it has a threefold increase in sensitivity, exceeding the current state-of-the-art contact lens sensors.
Similar content being viewed by others

Introduction
Glaucoma, a serious disease that is asymptomatic in the early stages but causes a gradual and irreversible loss of vision until blindness sets in refs. 1,2. According to the World Health Organization, glaucoma affects 76 million people globally in 2020 and is expected to grow to 112 million by 20403,4. Glaucoma is usually caused by obstruction of aqueous humor circulation, leading to high IOP that compresses the optic nerve5. Currently, lowering IOP through medication or surgery is effective in reducing the risk of glaucoma worsening6. However, IOP varies from person to person, fluctuates around the clock, and is affected by daily activities (e.g., sports) as well as circadian rhythms, especially that nighttime IOP is usually higher than daytime IOP7,8. Therefore, long-term IOP monitoring is needed to analyze the critical IOP fluctuations to determine the optimal treatment plan. However, conventional ocular tonometers (e.g., the Goldman tonometer and Dynamic contour tonometer in the clinic, Icare tonometer for home use, etc.) can only be used intermittently to provide silhouettes and cannot provide sufficient information for glaucoma care9,10,11. Therefore, there is an urgent need to develop a new type of tonometer for efficient, portable, 24-h continuous IOP measurement7.
Recently, implantable or wearable sensors constructed on the basis of cutting-edge technologies such as flexible electronics and integrated circuits have been investigated for continuous IOP monitoring12,13,14. Implantable sensors have the advantages of high accuracy and good interference resistance, but their use is limited by the risks associated with surgery and the questionable safety and reliability of devices under long-term use15,16,17,18,19. Contrarily, contact lens-based wearable sensors are highly regarded for their non-invasive and detachable features, they currently include three types in principle: microfluidic20,21,22, optical23,24, and electrical. Microfluidic and optical sensors are unable to detect IOP at night with the eyelids closed, owing to their visual or optical signal detection format12,20,21,22,23,24. Whereas, electrical sensors are able to achieve 24-h IOP measurements around the clock and thus dominate the mainstream. Electrical sensors for IOP monitoring are schematically comprised of two types: radio frequency integrated circuit (RFIC)25,26,27,28,29,30 and inductive-capacitive (LC) resonant31,32,33,34,35,36,37,38. For the sensors containing FRIC (e.g., the TriggerFish lens), the RFIC chip guarantees the system functionality, but its rigidity and high stiffness lead to a significant increase in lens thickness, poor in wearing comfort, and susceptibility to corneal abrasion, especially during nighttime sleep39,40,41.
Contact lens sensors based on LC technology are relatively simpler in principle and structure12. They are usually constructed from functional conductive materials (e.g., conventional metal Cu or Au, silver composites, room temperature liquid metal, etc.) to form the flexible LC sensors and integrated with biocompatible polymers (e.g., hydrogel, silicones, and PI)42,43. Among them, capacitive IOP sensors constructed from Cu, Au, PI, etc., usually use the gap-variable method, they have highly sensitive but low usability for human wearing, which mainly due to the high thickness of lens and the hardness of the electronic much higher than the biological cornea31,32,33. Inductive IOP sensors constructed from Cu, Au, PI, etc., usually use the coil-diameter variable method, they have low thickness and enough wearing comfort, but lower sensitivity34,35. Despite further there is a method that design the inductive coils with the serpentine-shape to improve the flexibility, the increase in sensitivity is still limited36. Contact lens sensor using silver composites (AgSEBS) currently utilize the principle of capacitive and inductive simultaneous response to IOPs, the device is highly sensitive and suitable for human eye wearing. But, the low conductivity of the material produces a low-quality, inconspicuous LC signal, causing a less easy signal acquisition37. Contact lens sensor constructed from liquid metal was previously proposed by the author, the device consists of a stretchable liquid metal inductive coil and a rigid chip capacitor that forms an LC loop and responds to the IOP changes through the inductive coil diameter. The liquid metal has high conductivity and softness, thus brings about an improvement in the signal quality and the device wearing comfort. However, the device is compared to be a bit lacking in sensitivity38.
Here, we further propose an LC contact lens sensor that is constructed by using liquid metal to form the stretchable inductance and capacitance. The capacitive pole plate and the inductive coil can response to IOP changes simultaneously, thus able to enhance the sensitivity in principle. We model the IOP sensing mechanism of the contact lens sensor, and conducted design, fabrication, as well as usability and reliability tests. We evaluate the performances of the contact lens sensor on biomimetic and biological eyeballs. The results show that the proposed contact lens sensor has good performances, including wear comfortable, non-destructive, high reliable, et al. In particular, the device has a threefold increase in sensitivity, which exceeds the current state-of-the-art contact lens sensors.
Results
Principle, design, and analysis
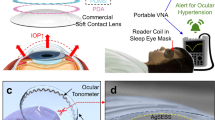
Figure 1a illustrates the structure of the contact lens sensor. The sensor consists of room temperature liquid metal and silicone, with silicone on both sides wrapping and sealing the inner liquid metal structure. The liquid metal is designed as an upper and lower double-layer shape inside the lens. The upper coil is a single-rotation shape, exhibiting inductive characteristics. While the lower coil is a round-trip shape, its inductive characteristic is less. But since it is directly opposite to the upper coil, the upper and lower corresponding areas form a capacitor through the middle silicone as a dielectric layer. The capacitor and inductor build the LC resonant circuit. Also, since the silicone and liquid metal are soft materials, the capacitor and inductor are flexible and stretchable, thereby both can respond to IOP changes. The two main characteristic parameters of the LC resonant circuit (resonant frequency and quality factor) are related to the capacitance, inductance, and resistance, calculated as follows44.
a 3D structural model and the layered schematic view of the contact lens sensor. b Data acquisition scheme of IOP monitoring. c Cross-sectional SEM of the microchannels, scale bar: 100 μm. d Photograph of contact lens sensor, scale bar: 3 mm. Figure 1a, b were created by author Hongbin An using the SoliderWorks and 3D Max software, respectively. The figures were created to describe the structure of the contact lens sensor, as well as the components and principles of LC IOP measurement system, respectively. Figure 1c was created by author Hongbin An using a scanning electron microscope (Helios NanoLab G3 CX, FEI company, America). The photograph was created to show how the contact lens sensor was manufactured. A channel cross-section containing liquid metal was not used because the liquid fluidity would have resulted in a dirty cutout, which was unsightly, interfered with observation, and did not visually represent our ideas. While, the green line in the photograph represents the scale bar as 100 μm. Figure 1d was created by author Xian Wang using a smartphone (OnePlus Ace 2 Pro, OPPO Guangdong Mobile Communications Co., Ltd, China). While, the white line in the photograph represents the scale bar as 3 mm.
Figure 1b illustrates the signal acquisition setup for IOP monitoring, it is implemented by connecting a vector network analyzer (VNA) via an external reader coil. The reader coil can be integrated within a frame eyeglass or an eye mask, providing sufficient electromagnetic induction coupling with the sensor coil at close range and without interfering with the user's daily life. The VNA can monitor either the impedance parameters or the scattering parameters (S-parameters) of the LC loop19,44. Here, based on the analysis of the LC signal (Supplementary Note 1), we choose the impedance real part spectra to be acquired during IOP measurements.
The contact lens sensor was fabricated by using a microchannel liquid metal injection method. The specific fabrication process flow of the device is demonstrated in Supplementary Fig. 3, including soft lithography, surface-modified bonding, punching and liquid injection, dicing and mold integration. Figure 1c shows the Cross-sectional SEM of the microchannels before liquid metal injection, with scale bar of 100 μm. Figure 1d illustrates a physical photograph of the contact lens sensor, scale bar: 3 mm.
The use of a contact lens to detect IOP is a spacing method since IOP is not directly applied to the contact lens but to the cornea. The contact lens is connected to the cornea by tear film (Supplementary Fig. 4). The tear film is composed of a lipid layer, an aqueous layer, and a mucin layer45. The compression of contact lens (caused by eyelid compression or IOP increase) will drive the lipid and aqueous layers in tear film to overflow, then the mucin layer will connect cornea to contact lens. The mucin layer is a mixture of protein and water, with viscoelasticity, and can transfer stress46,47. For the overall structure, the IOP sensing process can be described as follows: the cornea expands and deforms (curvature radius increase and tangential stretching) with IOP increase, then the deformation transmits to contact lens through the action of tear film stress, causing the contact lens to stretch and deform (Supplementary Fig. 5). The stretchable liquid metal coils will increase in diameter and width, leading to the increase of inductance and capacitance, as well as the decrease of resonance frequency. In the process, stress interactions exist between the interfaces of the cornea with tear film as well as tear film with the contact lens.
We analytically modeled the IOP sensing mechanism (Supplementary Note 2) to investigate the effect of parameters on sensitivity and thus guided the design of the contact lens sensor. The modeling process was mainly based on shell mechanics and shear-lag theory, wherein we adopted many assumptions, such as the corneal was a thin shell, the cornea and tear film were linear-elastic materials, etc., these were mainly to simplify the modeling difficulty48. We also carried out a finite element model (FEM) and compared the results with those of the analytical model to verify the correctness.
Figure 2a, b show the displacement and stress nephograms under the pressure of 40 mmHg from FEM. The maximum displacement and stress occur in the corneoscleral transition zone, while the stress nephogram shows a significant shear hysteresis in the tear film. Figure 2c shows the results of the coil diameter and width varying with IOP, while Fig. 2d shows the related variation of resonant frequency induced by capacitance and inductance under different IOPs. The FEM and analytical model show similar results, with each resultant parameter varying linearly with IOP. In addition, the modeling results intuitively show that the new LC sensor will be nearly three times more sensitive than the existing liquid metal inductive sensor, owing to its new principle that capacitance and inductance can simultaneously sense IOP changes.
a, b Displacement nephogram and stress nephogram under IOP of 40 mmHg. c The change rate of coil diameter and width with IOP. d Variation of resonant frequency induced by capacitance and inductance under different IOPs. e The influence of corneal parameters (include elastic modulus and thickness) on sensitivity is calculated by the analytical model. f The results comparison between the analytical model with FEM. g The influence of tear film parameters (include elastic modulus and thickness) on sensitivity and h the results comparison between the analytical model with FEM. i The influence of elastic modulus and thickness of contact lens on sensitivity and j the comparison of the results between the analytical model with FEM. k The influence of lens diameter and coil distribution radius on sensitivity, and l the comparison of the results between the analytical model with FEM. Figure 2a–l were all created by author Hongbin An. The data were calculated by using the mathematical software, Matlab, and FEM simulation software, COMSOL Multiphysics. The figures were drawn using the graphing & analysis software, Origin Pro (in 2019 vision).
Figure 2e demonstrates the effect of corneal parameters on sensitivity that calculated by the analytical model. Figure 2f shows the results comparison between the analytical model with FEM. The two result graphs indicate that, with corneal elastic modulus as well as corneal thickness increase, the sensitivity decreases. This is mainly due to the corneal stiffness increase resulting in the decrease of device deformation under pressure. Similarly, Fig. 2g demonstrates the effect of tear film parameters on performance that calculated by the analytical model and Fig. 2h shows the comparison. The results show that the sensitivity increases rapidly with the tear film elastic modulus and then decreases slowly after reaching the peak value. While, the tear film thickness has a small effect on sensitivity. Figure 2i, k demonstrate the effect of structural parameters of contact lens sensor on sensitivity, Fig. 2j, l shows the comparison results comparison between the analytical model with FEM, wherein an increase in elasticity modulus and thickness of contact lens will decrease the sensor sensitivity. Whereas, a decrease in the diameter of the lens as well as an increase in radius of the coil distribution facilitate an increase in sensitivity. In addition, Fig. 2f, h, j, l show that the results of FEM and analytical model are basically the same and the relative error is within 17%, this verifies the correctness of analytical model. The error is mainly caused by the thin-shell assumption in the modeling and the neglect of shear lag in the latitudinal direction of the cornea and contact lens.
Referring to the results of the modeling analysis, we designed the structure of the contact lens sensor. Reducing the stiffness of the contact lens sensor is conducive to improve the sensitivity, so we chose the transparent and soft silicone rubber (MED-4286; Nusil, Inc.) as the matrix material, it had a low elasticity modulus of 0.08 ± 0.01 Mpa (as shown in Supplementary Fig. 6). In terms of lens thickness, we adopted a design of thin in the middle and thick on the outer of the contact lens (with a thickness of 0.2 mm in middle and 0.45 mm outer), this guaranteed the sensitivity while also allowing the integration of coil loop on the outer. In addition, drawing on the size of a commercial contact lens, the diameter of the contact lens sensor was set to 14.0 mm. We designed the distribution diameters of liquid metal coils to be 12 and 10 mm, this would not affect the pupil vision (>10 mm). The contact lens sensor was prepared by microchannel injection method, we choose the liquid metal material as GaInSn (Ga68.5%, In21.5%, Sn10%), the material had an excellent conductivity49,50. The cross-sectional width of the microchannel and its thickness were designed to be 300 × 100 um. These can achieve a low resistance and guarantee the quality coefficient of the LC loop to obtain a high-quality spectral with a sharp peak51. The final structural design parameters of the sensor are demonstrated in Supplementary Table 1. Specifically, the lens sensor constructed from soft materials, had a good stretchability that can withstand at least 120% of tensile, as shown in Supplementary Fig. 7.
Usability and reliability tests
Safety in use is a prerequisite for contact lens sensors to be applied. Thus, the liquid metal sealed within the silicone channel cannot leak, as well as the silicone should be suitable for corneal wear and do not interfere with daily life. Figure 3a shows that the microchannel loop of the device can withstand a maximum pressure of more than 36 psi under a burst test52 (experimental setup showed in Supplementary Fig. 8), this exceeds the irreversible bonding requirement of a microfluidic chip (30 psi)53, demonstrating that the encapsulation of liquid metal within the silicone is sufficiently strong. Figure 3b–d show the physical and chemical characterizations of contact lens sensors. Its oxygen permeability has been tested through the Coulomb method and was greater than 300 × 10−11(cm2/s) [mLO2/(mL·hPa)], far exceeding the standard for commercial contact lenses (~50 × 10−11(cm2/s) [mLO2/(mL·hPa)])54. Its light transmittance was of greater than 93%, meeting the requirements for commercial use (>90%)55. The contact angle of the surface (after corona treatment) was 49°, hydrophilic and suitable for corneal contact. Figure 3e demonstrates observation of the ocular surface through the slit-lamp fluorescein stain after a long-term in vivo wear (>16 h), during which the rabbit was used owing to their common, small, clean, and easily controlled characteristics. The picture shows that the rabbit corneal surface is free of fluorescent residue, smooth and without abrasions, indicating the lens sensor’s good wearability.
a Sealing strength characterization of the channel through the water-filled swelling test. b Oxygen permeability of the contact lens sensors, error bars indicate Mean ± standard deviations (SD), n = 3. c Light transmittance. d Aqueous contact angle. e Photograph of sodium fluorescence staining in a rabbit eye after prolonged wear (>16 h). f Normalized baseline resonant frequency (Mean ± SD, measured three times) of the sensor following 4000 cycles of stretching with 25 and 50% rates, as well as folding. g Resonant frequency shift (Mean ± SD, measured three times) under ten vibration cleaning cycles. h Frequency shift (Mean ± SD, measured three times) of the sensor following storing in a contact lens care solution for 30 days. Figure 3a–c were created by author Hongbin An. Figure 3d was created by authors Hongbin An and Xian Wang. Figure 3e was created by authors Liang Zhang and Xian Wang. In which, Liang Zhang photographed a rabbit wearing the contact lens sensor and fluorescent staining of the corneal surface. The photograph of the rabbit wearing the sensor was created by using a smartphone. The corneal fluorescence staining photograph was obtained by using a slit-lamp microscope (SL-D7; Topcon Medical System, Tokyo, Japan), with a cobalt-blue filter at 10x magnification. Figure 3f–h were created by authors Hongbin An and Xian Wang.
Reliability is another imperative consideration of contact lens sensors, especially for long-term use. Throughout the life cycle, the contact lens sensor typically undergoes in vivo eye wearing, in vitro cleaning, and storing. Figure 3f shows that the resonance frequency shift of the contact lens sensor is less than 0.3 MHz for 4000 cycles of stretching (with 25 and 50% tensile rate) and folding (Supplementary Fig. 9a), verifying that the contact lens sensor has good mechanical reliability and should be sufficient to withstand the mechanical deformations that may be experienced during human in vivo wearing, measurement, and removal processes. Figure 3g,h demonstrates that the resonant frequency shift of the contact lens sensor is less than 0.1 MHz for undergoing ten vibration cleanings and 30 days of storage in a contact lens care solution (Biotrue; Bausch & Lomb, Inc.), Supplementary Fig. 9b, these proves that the contact lens sensor has sufficient mechanical as well as chemical reliability to cope with daily maintenance.
Sensing performances in the biomimetic eyeballs
The wireless sensing performance of the contact lens sensor was first tested under the biomimetic eyeballs. Figure 4a shows the experimental setup, where a customized pedestal held the biomimetic cornea made of silicone through adhesive, as well as a syringe pump and pressure gauge, were connected on two sides through a pagoda connector for internal pressurization and recording. The structural design of silicone cornea is shown in Supplementary Fig. 10. We constructed two biomimetic eyeballs with different mechanical properties for testing, they were manufactured using a 25:1 and 35:1 blend of PDMS (Sylgard 184; DowCorning; USA), respectively. The elastic modulus of the two PDMS materials was tested through uniaxial tensile (Supplementary Fig. 6) to be 0.49 and 0.34 Mpa, in between the elastic modulus of human cornea and also porcine cornea56,57. In practice, it was not easy to simulate the real-use environment by applying tears or artificial tears, etc., to the surface of the biomimetic eyeball in a uniform and low-thickness manner, so we conducted a performance test with a contact lens sensor and the biomimetic eyeballs conformal contact directly. In addition, the reading coil for signal acquisition was a two-turn PCB with inner and outer diameters of 19.5 and 20.5 mm, respectively, and the hardware device we used was a handheld VNA (36211; CECC Siyi Technology, Inc.; China).
a Experimental setup. b Spectral signals at different IOPs (using the 35:1 PDMS eyeball). c, d Results of resonant frequency dynamic tracking IOP under two eyeballs, respectively. e Calibration of resonant frequencies versus IOPs. f Comparison of the IOP sensing sensitivity between the experimental, theoretical, and FEM. g, h Spectral signals and the frequency shifts (Mean ± SD) at different distances and angles between the contact lens sensor and reader coil. Figure 4a–h were all created by authors Hongbin An and Xian Wang.
Figure 4b shows the spectral signals of contact lens sensor under different IOPs (test in the 35:1 PDMS eyeball), with IOP increases, the spectral signal of impedance real part shifts to the left as well as the resonant frequency decreases (Supplementary Video 1), consistent with the changing trend in previous theoretical model. In addition, it is shown that the impedance peak increases with pressure up, mainly caused by the corneal expansion leading to a decrease in the distance between the reader coil and the contact lens sensor as well as an increase in their mutual inductance coupling coefficient. Figure 4c, d show the follows of resonance frequencies with IOPs in two biomimetic eyeballs, the resonance frequency can all respond in real time with the pressure varied alternately in the range of 3–48 mmHg and 3–40 mmHg in increments of 3.5 mmHg and 2 mmHg each time, respectively. Figure 4e demonstrates the calibrations that the resonant frequencies vary linearly with IOPs (with R2 > 0.99) in two biomimetic eyeballs. The IOP responses are 0.733 MHz/mmHg and 0.972 MHz/mmHg, respectively, the sensitivities are 1665 ppm/mmHg and 2209 ppm/mmHg. The biomimetic eye with a low elastic modulus has a higher IOP response and sensitivity, consistent with the theory.
We brought the measured mechanical properties of biomimetic eyeballs into the analytical model as well as FEM for calculations, and quantitatively compared the solved sensitivity results with the experimental. In the analytical model analysis, the lens was in direct contact with the biomimetic eyeball without a tear film in the middle, so the thickness of the tear film in the model could be set as 0 \(({{\rm{t}}}_{2}=0\mu m)\). According to Equation 27, 28, 33 in Supplementary Note 2, it can be calculated that the constant k tends to infinity and the constant b equal to 1, as well as the efficiency of the strain transfer \(\varphi \approx 1\), indicating that the corneal strains are all transferred to contact lens with no loss. While in the construction of FEM, no tear film is added to the model structure, the interface between the contact lens and cornea interface is set to no slip. The sensor responses analyzed by the analytical model as well as FEM is demonstrated in Supplementary Fig. 11, the results are also presented as linear. Figure 4f demonstrates the comparison of sensitivities under the three forms of experimental model, analytical model and FEM, they are close in size and within 22% error, confirming the correctness of analytical model.
Figure 4g, h show the effect of distance and angle between the contact lens sensor and the reader coil on the signal spectrum and resonance frequency shift. The distance and angle of deflection were set from 4 to 12 mm and from 0 to 45°, respectively, on accord of the possible eye movements of humans. The results show that the impedance peak decreases with the increase of distance and angle due to the effect of mutual inductive coupling. While, the peak frequency of the signal remains unchanged, consistent with the theoretical result of Equation 4 in Supplementary Note 1, the peak frequency is not affected by the difference in coupling coefficients and only related to the characteristic parameters of LC circuit. These results indicate that the contact lens sensor can adapt to misalignment and displacement during use.
Ex vivo and in vivo evaluations in pig eyes
Since the pig eye is close in size, shape and mechanical properties to human eye and is readily available58, we chose it to evaluate the wireless sensing performances of contact lens sensor on biological eye, both ex vivo and in vivo.
For ex vivo evaluations in enucleated pig eyes (n = 3), Figure 5a shows the experimental setup that includes a needle inserted into an eyeball. The needle was connected to the syringe pump and pressure gauge through a three-way connection. The syringe pump adjusted the IOP of the enucleated pig eye by injecting or suctioning physiological saline into the anterior chamber. The pressure gauge measured the pressure of the closed loop (IOP) to calibrate. The contact lens sensor was attached to the cornea surface of the enucleated pig eye, while the reader coil was placed above the contact lens sensor, the distance between the two is set to 6 mm to form wireless coupling, Fig. 5b.
a Schematic diagram of the experimental setup on an enucleated porcine eye. b Photographs of contact lens sensor in an enucleated porcine eye during measuring. c Spectra signals of contact lens sensor under different IOP. d Calibration results of resonant frequencies (Mean ± SD) versus IOPs on three enucleated porcine eyes. e In vivo photograph of contact lens sensor being worn on the pig eye. f The time-varying results of IOPs and resonance frequencies for 6 h test in the live pig eye. g Calibration results of resonant frequencies versus IOPs in the live pig. Figure 5a–d were created by authors Hongbin An and Xian Wang. Figure 5e was created by authors Hongbin An, Zhikang Liao, Hui Zhao and Yingyun Yang. Where, Hui Zhao and Yingyun Yang designed the experimental program, Hongbin An and Zhikang Liao executed it and Zhikang Liao photographed the live pig’s eye wearing the sensor. Figure 5f, g were created by authors Hongbin An, and the results were discussed with Hui Zhao as well as Yingyun Yang.
Figure 5c demonstrates the spectral signals of the sensor at different IOPs during experiments on an enucleated porcine eye. With an IOP increase, the spectral signal shifts to the left as well as the resonant frequency decreases. Whereas as IOP decreases, the signal recovers to the right, and the resonant frequency increases. The phenomenon is similar to the result of the biomimetic eyeball. Supplementary Video 2 representatively shows the process of the sensor signal shifting with IOP changes (regulated by the lifting of a bottle) of an enucleated porcine eye. However, unlike the phenomenon that the IOP response of the sensor on biomimetic eyeball remains largely consistent, the IOP response of the sensor under the enucleated porcine eye is not consistent. It shows a larger response at low IOPs, i.e., the signal shift is more pronounced when the pressure is elevated at low IOPs, mainly due to the nonlinear mechanical properties of the biological eye that, its elastic modulus increases with the increase of stress caused by IOP rising56,58,59,60.
Figure 5d shows the calibration results of resonant frequencies versus IOPs on three enucleated porcine eyes. In the pressure range of 9–30 mmHg, the IOP responses on the three eyes were obtained by linear fitting as −0.533 MHz/mmHg (R2 = 0.981), −0.484 MHz/mmHg (R2 = 0.976), and −0.551 MHz/mmHg (R2 = 0.982), respectively. The sensitivities of IOP sensing were 1211 ppm/mmHg, 1100 ppm/mmHg, and 1252 ppm/mmHg, respectively. The differences in IOP responses and sensitivities were mainly due to the differences in the corneal curvature radius and thickness as well as the biomechanical properties of the pig eyes. Besides, the mechanical hysteresis and creep characteristics of the biological eyes56,61 (mainly soft tissue materials) also caused the standard deviation of resonance frequency measurements to be larger than that of the biomimetic eyeballs.
For in vivo evaluations, Fig. 5e shows a photograph of the contact lens sensor being worn on a pig’s eye. Before the experiment, the pig was anesthetized so as to prevent it from being in a state of shock hyperactivity that might interfere with subsequent test operations. The pig was acclimatized to the contact lens for 30 min after lens wear to achieve optimal conformal adhesion to its cornea. After that, the collection of spectral signals from the sensor was carried out. Since goggles could not be easily worn on the pig’s head, we used a medical dressing to adhere the reading coil fixed on the eye socket, this was similar to the approach of using an eye mask. Supplementary Video 3 representatively demonstrates the continuous acquisition of the sensor spectral signals in the live pig.
The in vivo experiment under the pig eye lasted for 6 h, during which we carried out a set of signal acquisitions and IOP measurements every half hour. The signal acquisitions lasted for 1 min in each set, and within, we measured the IOP by three times through an animal-specific tonometer (Tonovet plus; Icare Inc.). It should be mentioned that the signal acquisitions and Icare IOP measurements were simultaneously performed on both eyes of the pig. Before the experiment, we tested the two eyes with similar IOPs of 25 ± 0.67 mmHg and 25.7 ± 1.2 mmHg (Mean ± SD, n = 3), respectively. Generally, the variation tends of IOPs in response to the environment to be the same in both eyes of normal animals. So, the separate measurements through the two eyeballs could avoid the need to disassemble the contact lens. While the disassembling could bring a positive effect on maintaining a consistent position between the contact lens and the cornea throughout the process. As is known, the biological cornea is not perfectly spherical (approximately elliptical)62,63, so if a single eye is periodically worn and removed with a contact lens to collect signals separately, the relative position of the contact lens to the cornea and the state of their fit will change each time the lens is re-worn, affecting the resonance frequency and IOP response.
Figure 5f shows the time-varying results of IOPs and resonance frequencies for 6 h test in the live pig. The IOPs of the pig firstly increased slowly and then stabilized from 8:30 to 10:00. At 10:05, we shock-damaged the thoracic cavity of the pig through the Bioshock tube64, the IOPs began to have a large drop until to 11:00. The pressure drop should be caused by the shock damage that affected the atrial water circulation. After 11:00, the IOPs of the pig eye stabilized and showed small fluctuating changes. Throughout the process, the trend of the sensor resonant frequencies over time was consistent with the trend of IOP changes. Figure 5g demonstrates the IOPs-resonant frequencies calibration results based on the 6 h test data, the IOP response of the sensor under linear fitting was −0.529 MHz/mmHg (R2 = 0.993) and the sensitivity was 1213 ppm/mmHg, close to the results under the enucleated pig eyes. In addition, the standard deviation of the resonant frequency measurements in each set was very small, mainly since that the pig was under anesthesia and its eye movement was small, having little interference with the device state and signal.
In vivo evaluations in human eyes
Wearability adaptation testing of the contact lens sensor was first conducted under human eyes and two adults who have experience in commercial contact lens use volunteered to take the test. The contact lens sensors underwent a surface corona treatment prior to wear, this improved the surface hydrophilicity while disinfecting and sterilizing. Figure 6a shows a photograph of one volunteer wearing the contact lens sensor, the contact lens sensor fitted well against the cornea. The LC circuit of the contact lens sensor was located at the corneal limbus (corneoscleral junction), much larger than the pupil diameter, and so had no effect on the vision field of the user.
a Photograph of the human eye wearing the contact lens sensor. b Slit-lamp fluorescence examination of the corneal surface after 6 h wear of the contact lens sensor. c Time-varying changes in resonance frequencies and Icare IOPs under the head-down bed-rest experiment. d Calibration of the sensor resonant frequencies (Mean ± SD) versus with IOPs in the human eye. e Time-varying changes in IOPs (Mean ± SD) obtained with the contact lens sensor and Icare tonometer, respectively, for 5 h. Figure 6a was created by authors Hongbin An, Xian Wang, Zhikang Liao, Hui Zhao, and Yingyun Yang. In which, Hongbin An wore the contact lens sensor, Xian Wang and Zhikang Liao performed the LC signal acquisitions and IOP measurement, Yingyun Yang and Hui Zhao assisted and took the photograph. Figure 6b was created by author Yingyun Yang using the slit-lamp microscope (SL-D7; Topcon Medical System, Tokyo, Japan). Figure 6c–e were created by author Hongbin An and the results were discussed with Hui Zhao, Yingyun Yang, Jizhou Song, and Yinji Ma.
Figure 6b shows a photograph of a slit-lamp biomicroscopic observation of the volunteer’s cornea after 6 h of wear. Before observation, the corneal surface was stained with sodium fluorescein. The result of the photograph shows no obvious fluorescence aggregation on the corneal surface, indicating that the wearing of a contact lens sensor causes no corneal abrasion. Also, regarding the wearing comfort of the contact lens sensor, the volunteers reported that the contact lens sensor was thicker than the commercial hydrogel contact lenses, but within the acceptable range. The foreign body sensation of the contact lens sensor was not obvious, these should be mainly attributed to the improved hydrophilicity of contact lens sensor and the low mechanical modulus as well as soft properties of the silicone material. In addition, neither of the two volunteers expressed overheating, either during continuous acquisition of LC signals (for 25 min) or during long-term intermittent acquisitions (every 30 min for 5 h). Further exploring the thermal effects, the thermographic results (Supplementary Fig. 12) after prolonged IOP measuring through the porcine eye ex vivo were the same as those at the beginning of the experiment, also indicating that there were no obvious thermal effects during the measuring that would cause damage.
The wireless sensing performance evaluations of the contact lens sensor were carried out in the human eye. The LC spectral signals from the contact lens sensor were detected through goggles with an integrated reading coil (Supplementary Fig. 13), while Supplementary Video 4 representatively demonstrates the real-time acquisition of the spectral signals from the contact lens sensor when the volunteer is in a sedentary state. We carried out the calibration of the contact lens sensor by realizing the dynamic change of IOP in volunteers through a human head-down bed-rest experiment65,66. Before starting the experiment, the volunteers wore the contact lens sensor for half an hour to achieve the best match between the lens with the cornea. After that, the volunteers laid flat on the bed, tied and secured well to start the test. The photograph of the experiment is shown in Supplementary Fig. 14. The format of the test was the same as that on the live porcine eye, with both eyes used for LC signal acquisition and IOP measurement, respectively, since the IOP change trend in the two eyes of human was usually the same67,68. The signal acquisition speed was set to about 0.33 s per frame. IOP measuring was performed with a handheld tonometer (Ic100; Icare Inc.), while the other side of the goggle was cut out to allow for easy insertion of the tonometer.
Figure 6c demonstrates the time-varying results of the sensor resonance frequencies versus Icare IOPs under a nearly 30-min head-down bed-rest experiment. In which, the volunteer underwent a state change of lying down (9:11–9:16), −30° head-down bed-rest (9:16–9:26), and returning to lying down (9:26–9:36). During the period, the IOP increased firstly and then decreased to recover, while the resonant frequency of the sensor first increased and then decreased, the trends of the two corresponded. There was noise in the resonant frequency time-varying result, this was mainly caused by human eye movements and blinking interfering with the contact lens state (pulling and pressing the lens). For the noise, it could be filtered out by a sliding evaluation algorithm (yellow line in Fig. 6c). The same phenomenological results was obtained for another volunteer under the head-down bed-rest experiment (Supplementary Fig. 15).
Figure 6d shows the calibration results of resonance frequencies versus IOPs based on the data under the head-down bed-rest experiment. The IOP response of the contact lens sensor on the volunteer was −0.702 MHz/mmHg (R2 = 0.896) with a sensitivity of 1595 ppm/mmHg for a linear fit. In particular, the new contact lens sensor has a threefold increase in sensitivity compared with the original stretchable inductive LC sensor38, it exceeds the highest sensitivity of current state-of-the-art wearable tonometers25,26,27,28,29,30,31,32,33,34,35,36,37,38, as showed in Table 1. Figure 6e shows the dynamic IOP changes in the volunteer wearing contact lenses for 5 h after the head-down bed test. Wherein the IOP was monitored every half hour using an Icare tonometer (n = 3), and the contact lens sensor signal was acquired for 1 min each set. The result show that the IOP measurements from the contact lens sensor are close to the Icare measurements, as well as the time-varying trends of the two are consistent, these verifying the sensor’s good characteristics.
Discussion
In summary, we propose an LC contact lens sensor that is constructed with liquid metal to form the stretchable capacitor and inductor. The capacitive pole plate and the inductive coil can response to IOP changes simultaneously, thus able to enhance the sensitivity in principle. We model the IOP sensing mechanism of the contact lens sensor, and conducted design, fabrication, as well as usability and reliability tests. We evaluate the performances of the contact lens sensor on biomimetic and biological eyeballs. The results show that the proposed contact lens sensor has good performances, including high reliable, good signal quality, wearing non-destructive, et al. Especially that, the device has a 3-fold increase in sensitivity, which exceeds the current state-of-the-art contact lens sensors.
Nevertheless, considering future clinical applications, there are still some issues that need to be improved: (1) we subsequently plan to use the printing methods to achieve mass-production of the sensor, as these processes are more efficient49,50; (2) the sensor response depends on the cornea stiffness and will vary from human to human, so we future intend to measure the corneal biomechanical parameters through the ocular response analyser and analyse in conjunction with the deep learning techniques to obtain the accurate calibration models for various different categories of peoples. In general, our current work is the foundation and offers a meaningful and practical solution for clinical glaucoma management.
Methods
Device fabrication
The contact lens sensor was prepared by using the microchannel injection method. Supplementary Fig. 3 shows the preparation process, which consists of the following steps: (a) pour liquid silicone on two wafer templates containing SU-8 lithography patterns, the silicone has undergone degassing previously; (b) cure the liquid silicone at 80° for 100 min, and then demold the cured silicone from template wafer to obtain the film containing channel patterns; (c) punch hole in the patterned film to connect the top and bottom channel; (d–f) scrape liquid silicone onto another flat wafer containing no patterns and cure it to prepare a flat film (with cured thickness of ~80 um), bond the flat film with the patterned film through the following steps: first using corona treatment to form polar hydroxyl (-OH) groups on surface of two silicone films, then quickly apply the film to contact and remove bubbles, heat at 80° for 100 min for treatment to drive the hydroxyl groups to polymerize and form stable covalent bonds, thereby achieving irreversible bonding of two films; (g–i) adopt same method to bond the other side of the patterned silicone to form closed channels. (j, k) punch holes in the bonding channels, use a syringe pump to connect a needle (BD-30G) to inject liquid metal into the channels, and then seal the holes with silicone dripping and heat curing; (l, m) use two customized circular cutters (with diameters of 12.6 and 10.4 mm, respectively) to cut off the inner and outer areas of film sensor, and then place the formed circular sensor into a contact lens mold; (n, o) pour liquid silicone into the mold and solidify it, then the contact lens with integrated LC sensor is prepared after detachment. The surface of contact lens can be modified to be hydrophilic by corona treatment, and then stored in contact lens care solution (Biotrue; Bausch & Lomb, Inc.; USA). During the preparation process, the structure of the bonded microchannels prior to liquid-metal injection was characterized using scanning electron microscopy (Helios NanoLab G3 CX, FEI company, America). After the preparation was completed, the device was photographed via a smartphone (OnePlus Ace 2 Pro, OPPO Guangdong Mobile Communications Co., Ltd, China). Before and after every test, the device was cleaned using a special contact lens cleaner (Shery; Danyang, Inc.; China). During each subsequent test, we photographed the contact lens sensor in use.
Tensile test
Uniaxial tensile tests were carried out to determine the mechanical properties of the silicone materials, with reference to international standard ISO 37-2005. The samples with bone shape were prepared and loaded on a universal electronic material tensile testing machine (MicroTester-5944; Instron Corporation; USA). The stretching range was controlled within 60% at the elongation rate of 25% per minute. The stress was calculated by the following equation:
Where F is the force measured by the equipment at different tensile rates; and w and h are the cross-sectional width and thickness of the specimen. For each type of silicone, three samples were used for testing.
Burst test to assess the bonding and sealing strength
The bonding and sealing strength of the device containing channels was assessed by burst test52. A needle (BD-30G) was inserted into the channel port, a drop of liquid silicone was then dripped and cured to make a seal at the socket, the end of the microchannel was also sealed with the silicone. The needle was connected to a three-way pipe, and the other two ends of the three-way pipe were connected to a syringe pump (ISPLab01; Hebei Shenchen, Inc.; China) and a pressure transducer (PX273-200DI; Omega Engineering, Inc.; America). During the test, the syringe pump was controlled to pressurize the loop at a volume of 1 mL per minute until the sealing loop of the microchannel ruptured.
Evaluations of oxygen permeability and light transmittance
The oxygen permeability of the contact lens sensor was tested based on the principle of the Coulomb method. The experiment was carried out in accordance with the Chinese Standard: GB/T11417.7-2012. The equipment used was an oxygen transmission rate testing machine (ISOBARIC-404; Saicheng® instrument, Inc.; China). The humidity was controlled at 50%, and the temperature was controlled at 37 °C. Three samples were used, and each group was measured three times. The light transmittance of the contact lens sensor was tested by a double-beam spectrophotometer (U-2910; HITACHI, Inc.; Japan). The experiment was conducted in accordance with the Chinese Standard: GB/T11417.5-2012. The light wavelengths were set in the visible range of the human eye from 380 to 780 nm, and the measurement area was concentrated in the central region of the contact lens sensor. Three samples were used in the test.
Evaluations of wearability in rabbit eyes
Approval for tests on the rabbits was obtained from the Laboratory Animal Ethics Committee. The study strictly complied with the Scientific Statement on the Use of Animals in Ophthalmic and Vision Research. Three New Zealand Large White rabbits (7 months old, weighing 2.0–2.5 Kg) with normal eyeballs were included. The rabbits were anaesthetized before testing to facilitate contact lens wear. After wearing, the rabbits were anaesthetized at regular intervals to reduce their activity. The contact lens sensors were worn on rabbit eyes and photographed. The wear lasted continuously for more than >16 h, after that the lenses were removed and the eye examinations were performed. During examination, the rabbit was immobilized in a holding device and kept quiet, while a drop of 1% fluorescein sodium solution (Baiyunshan Mingxing Pharmaceutical Co., Ltd., Guangzhou, China) was applied to the surface of the eye, and then the staining degree of corneal was assessed by observation under a slit-lamp microscope (SL-D7; Topcon Medical System, Tokyo, Japan) with a cobalt-blue filter at 10x magnification.
Reliability evaluations
The mechanical reliability of the contact lens sensor was tested by cyclic stretching and folding, since the contact lens sensor would subject to various mechanical effects when used in the human eye (including lens placement, wearing, and removal). The tests were conducted by a universal electronic material tensile testing machine (MicroTester-5944, Instron Corporation, USA) with every load of 4000 cycles. The elongations were controlled at 25 and 50%, respectively, with a rate of 25% per minute. For folding, the intermittent movement between the fixtures was controlled from 7.5 to 2.5 mm at a rate of 2.5 mm per minute. During the experiments, the contact lens sensors were removed every 500 cycles to collect the signals of the devices by the VNA (36211; CECC Siyi Technology, Inc.; China) and calculate the resonance frequency shifts. To evaluate the reliability of contact lens sensors under In vitro maintenance conditions such as multiple cleaning and long-term storing, a special contact lens cleaner (Shery; Danyang, Inc.; China) was used for the vibratory-cleaning, the care solution (Biotrue; Bausch & Lomb, Inc.; USA) was used for long-term storing. The vibratory-cleaning was conducted 10 times, with each time for 20 min. The storing of the contact lens sensor lasted for a total of 30 days and the device was removed every 3 days for evaluation.
Performance evaluations on bionic eyeballs
Two biomimetic corneas were prepared using PDMS (Sylgard 184; DowCorning; USA) mixed in a ratio of 25:1 and 35:1, respectively. The bionic cornea was glued to a customized pedestal, and the two ends of the pedestal was connected to a syringe pump (ISPLab01, Hebei Shenchen Company, China) and an electronic pressure gauge (CKHTP-01, CHEKON Company, China). We developed a corresponding hardware circuitry to transmit the pressure from the gauge to the host computer via USB at ten times per second. The contact lens sensor was conformally applied to the surface of the bionic cornea, the air bubbles were removed, and there was no other medium between the lens and cornea. A 2-turn PCB was used as a reader coil and fixed above the bionic eye at a distance <8 mm. The VNA was connected to the reader coil, and the impedance spectroscopy of the contact lens sensor was collected uninterruptedly at a rate of 5 frames per second. The initial IOP of the bionic eye was controlled to be greater than 2 mmHg before the test. The pressure was regulated in an increment of 3.5 mmHg and 2 mmHg under each eye, as well as with the pressure ranging from 3 to 48 mmHg and from 3 to 40 mmHg, respectively. The IOP response and sensitivity of the sensor can be calculated, respectively.
Where \(\triangle f\) is the resonance frequency shift, ∆P is the IOP increment; \(f(P={P}_{\min })\) is the resonant frequency at the lowest measurement pressure.
Ex vivo evaluations on enucleated porcine eyes
The fresh enucleated porcine eyes (n = 3) were obtained in the slaughterhouse and kept in a cold bag to be brought back to the laboratory for testing. For the experiment, a needle was inserted into the anterior chamber of the enucleated porcine eye, also the needle end was connected to a tee joint. The other two ends of the tee joint were connected to a syringe pump as well as a pressure gauge. The syringe pump injected or removed saline to regulate the IOPs. The pressure data from the gauge was transmitted to the host computer via USB. Tear film mucus was present on the corneal surface of the porcine eye, and the contact lens sensor adhered conformally to the corneal surface. The reader coil was fixed above the eyeball at a distance <8 mm, and connected to a network analyzer to acquire the impedance spectroscopy from the sensor. During the measurement, the initial IOP of the porcine eye was stabilized at 9 mmHg, then the syringe pump regulated the IOP with an increment or decrement of 3 mmHg at each time. The pressure was controlled to range between 9 and 27 mmHg, with cycling for three times. Three measurements of the resonant frequency were taken and averaged at each pressure step.
In vivo evaluations on pig eye
In vivo porcine experiment was performed at the Army Medical University with approval from the Experimental Ethics Committee (AMUWEC20223478). The care and handling of animals was performed strictly in accordance with the guidelines of the Guide for the Care and Use of Laboratory Animals. One healthy male Panamanian miniature pig, 4–6 months old and 25.28 ± 2.95 kg weight, was taken for the test. The pig was anaesthetized during the experiment to avoid being in a hyperactive state that would interfere with the experiment. The two eyeballs of the pig were separately used for signal acquisition of the contact lens sensor and IOP measurement by an animal tonometer (Tonovet plus; Icare Inc.). The contact lens sensors were worn on the pig eye In vivo and photographed. The experiment was conducted to last for 5 h, with every half hour a set of data were detected, in which the IOP was measured three times through ICare and the impedance signal from the contact lens sensor was collected for 1 min. During the experiment, a biological shock tube was used to shock damage the chest cavity of the pig, achieving changes in IOP. The corresponding data of resonance frequency and IOP was used for calibration through curve fitting.
In vivo evaluations in human eyes
In vivo experiments in human eyes were conducted in the presence of clinicians. Our two adults who had worn commercially available contact lenses volunteered to take the test, due to the confidence that the contact lens sensors have been proven to be safe to wear for long-term in vivo in animals. Before the test, we laser-cut the experimental goggles and affixed the reader coil to prepare a more convenient wearer. The wearer facilitated the subsequent signal acquisition and calibrated IOP measurement. For the experiment, the volunteer lied on an electric-stand-up bed (SHUT -100, Jiangsu Standley Medical Company, China) to conduct the test, its two eyes were individually used for the signal acquisition of the contact lens sensor and the IOP measurement through a handheld tonometer (Ic100; Icare Inc.). The contact lens sensors were worn on the volunteer’s eye and photographed. The experiment was conducted lasted up to 6 h per volunteer and was divided into 2 phases. The first phase was a short-range head-down bed-rest experiment, during which the contact lens sensor signals were continuously acquired at 6 frames per second as well as the IOP were measured at an average of 1 min intervals. The position of the volunteer was adjusted through the experimental from 0° to −30° for 10 min and then returning to 0°, this changed the IOP of volunteer so as to obtain data for calibration. The second phase was a long-time test, where the volunteer was in a lying position. During the second phase, the signals of the contact lens sensor were collected for 1 min every half an hour, as well as the IOP was measured three times by an Icare tonometer. The IOP results from the contact lens sensor of each set could be calculated through the measured resonance frequencies with the previous calibrated model. The Icare results were used for comparison with the IOP results from the contact lens sensor.
Heat generated in long-term monitoring
The contact lens sensor was worn on an enucleated porcine eye ex vivo. A reader coil that connected with a VNA was placed externally 4 mm above the contact lens sensor to detect the signals for real-time. An infrared thermal imager (H21PRO, HIKMICRO Company, China) was used to monitor the heating status of the porcine eye and the contact lens sensor during the 12-h long-term measurement.
Data availability
The data that support the plots in this paper and other findings of this study are available from the corresponding authors upon reasonable request.
Code availability
All the custom codes deemed central to the conclusions are available from the corresponding authors upon reasonable request.
References
Quigley, H. A. 21st century glaucoma care. Eye 33, 254–260 (2019).
Okeke, C. The Glaucoma Guidebook: Expert Advice on Maintaining Healthy Vision (John Hopkins Univ. Press, 2023).
Soh, Z. et al. The global extent of undetected glaucoma in adults: a systematic review and meta-analysis. Ophthalmology 128, 1393–1404 (2021).
Reis, T. F., Paula, J. S. & Furtado, J. M. Primary glaucomas in adults: epidemiology and public health‐a review. Clin. Exp. Ophthalmol. 50, 128–142 (2022).
Shalaby, W. S., Ahmed, O. M., Waisbourd, M. & Katz, L. J. A review of potential novel glaucoma therapeutic options independent of intraocular pressure. Surv. Ophthalmol. 67, 1062–1080 (2022).
Chintalapudi, S. R. et al. Systems genetics identifies a role for Cacna2d1 regulation in elevated intraocular pressure and glaucoma susceptibility. Nat. Commun. 8, 1755 (2017).
McMonnies, C. W. The importance of and potential for continuous monitoring of intraocular pressure. Clin. Exp. Optom. 100, 203–207 (2017).
Terauchi, R. et al. Seasonal fluctuation in intraocular pressure and its associated factors in primary open-angle glaucoma. Eye 35, 3325–3332 (2021).
Davies, L. N., Bartlett, H., Mallen, E. A. & Wolffsohn, J. S. Clinical evaluation of rebound tonometer. Acta Ophthalmol. Scand. 84, 206–209 (2006).
Willekens, K. et al. Review on dynamic contour tonometry and ocular pulse amplitude. Ophthalmic Res. 55, 91–98 (2016).
Subramaniam, A. G., Allen, P. & Toh, T. Y. Comparison of the Icare ic100 rebound tonometer and the Goldmann Applanation Tonometer in 1,000 eyes. Ophthalmic Res. 64, 321–326 (2021).
Yang, C. et al. Wearable and implantable intraocular pressure biosensors: recent progress and future prospects. Adv. Sci. 8, 2002971 (2021).
Ma, Y. et al. Flexible hybrid electronics for digital healthcare. Adv. Mater. 32, 1902062 (2020).
Farandos, N. M., Yetisen, A. K., Monteiro, M. J., Lowe, C. R. & Yun, S. H. Contact lens sensors in ocular diagnostics. Adv. Healthc. Mater. 4, 792–810 (2015).
Araci, I. E., Su, B., Quake, S. R. & Mandel, Y. An implantable microfluidic device for self-monitoring of intraocular pressure. Nat. Med. 20, 1074–1078 (2014).
Lee, J. O. et al. A microscale optical implant for continuous in vivo monitoring of intraocular pressure. Microsyst. Nanoeng. 3, 1–9 (2017).
Phan, A., Truong, P., Trumpp, J. & Talke, F. E. Design of an optical pressure measurement system for intraocular pressure monitoring. IEEE Sens. J. 18, 61–68 (2017).
Todani, A. et al. Intraocular pressure measurement by radio wave telemetry. Invest. Ophthalmol. Vis. Sci. 52, 9573–9580 (2011).
Chen, P. J., Rodger, D. C., Saati, S., Humayun, M. S. & Tai, Y. C. Microfabricated implantable parylene-based wireless passive intraocular pressure sensors. J. Microelectromech. Syst. 17, 1342–1351 (2008).
An, H. et al. Microfluidic contact lenses for unpowered, continuous and non-invasive intraocular pressure monitoring. Sens. Actuat. A Phys. 295, 177–187 (2019).
Agaoglu, S. et al. Ultra-sensitive microfluidic wearable strain sensor for intraocular pressure monitoring. Lab Chip 18, 3471–3483 (2018).
Yang, W. et al Notched-ring structured microfluidic contact lens for intraocular pressure monitoring. Appl. Phys. Lett. 119, 193701 (2021).
Maeng, B., Chang, H. K. & Park, J. Photonic crystal-based smart contact lens for continuous intraocular pressure monitoring. Lab Chip 20, 1740–1750 (2020).
Ye, Y. et al. Smart contact lens with dual-sensing platform for monitoring intraocular pressure and matrix metalloproteinase-9. Adv. Sci. 9, 2104738 (2022).
Leonardi, M., Pitchon, E. M., Bertsch, A., Renaud, P. & Mermoud, A. Wireless contact lens sensor for intraocular pressure monitoring: assessment on enucleated pig eyes. Acta Ophthalmol. 87, 433–437 (2009).
Liu, Z. et al. An ultrasensitive contact lens sensor based on self-assembly graphene for continuous intraocular pressure monitoring. Adv. Funct. Mat. 31, 2010991 (2021).
Chiou, J. C. et al. A wirelessly powered smart contact lens with reconfigurable wide range and tunable sensitivity sensor readout circuitry. Sensors 17, 108 (2017).
Xu, J. et al. Highly transparent and sensitive graphene sensors for continuous and non-invasive intraocular pressure monitoring. ACS Appl. Mater. Inter. 12, 18375–18384 (2020).
Kim, J. et al. A soft and transparent contact lens for the wireless quantitative monitoring of intraocular pressure. Nat. Biomed. Eng. 5, 772–782 (2021).
Kim, T. Y. et al. Wireless theranostic smart contact lens for monitoring and control of intraocular pressure in glaucoma. Nat. Commun. 13, 6801 (2022).
Yang, C. et al. Intelligent wireless theranostic contact lens for electrical sensing and regulation of intraocular pressure. Nat. Commun. 13, 2556 (2022).
Chen, G. Z., Chan, I. S. & Lam, D. C. Capacitive contact lens sensor for continuous non-invasive intraocular pressure monitoring. Sens. Actuat. A Phys. 203, 112–118 (2013).
Zhu, H. et al. Hydrogel-based smart contact lens for highly sensitive wireless intraocular pressure monitoring. ACS Sens. 7, 3014–3022 (2022).
Kouhani, M. H. M., Wu, J., Tavakoli, A., Weber, A. J. & Li, W. Wireless, passive strain sensor in a doughnut-shaped contact lens for continuous non-invasive self-monitoring of intraocular pressure. Lab Chip 20, 332–342 (2020).
Chen, G. Z., Chan, I. S., Leung, L. K. & Lam, D. C. Soft wearable contact lens sensor for continuous intraocular pressure monitoring. Med. Eng. Phy. 36, 1134–1139 (2014).
Karunaratne, I. K. et al. Wearable dual-element intraocular pressure contact lens sensor. Sens. Actuat. A Phys. 321, 112580 (2021).
Zhang, J. et al. Smart soft contact lenses for continuous 24-hour monitoring of intraocular pressure in glaucoma care. Nat. Commun. 13, 5518 (2022).
An, H. et al. High-sensitivity liquid-metal-based contact lens sensor for continuous intraocular pressure monitoring. J. Micromech. Microeng. 31, 035006 (2021).
Dunbar, G. E., Shen, B. Y. & Aref, A. A. The sensimed triggerfish contact lens sensor: efficacy, safety, and patient perspectives. Clin. Ophthalmol. 11, 875–882 (2017).
Toshida, H. Topographical central island-like pattern after 24 h of continuous intraocular pressure monitoring with a contact lens sensor. Int. Med. Case Rep. J. 13, 19–26 (2020).
Morales-Fernandez, L. at al. Changes in corneal biomechanical properties after 24 h of continuous intraocular pressure monitoring using a contact lens sensor. Can. J. Ophthalmol. 53, 236–241 (2018).
Jiao, Y. et al. A multifunctional bioelectronic device with switchable rigidity and reconfigurable shapes for comprehensive diagnosis. Adv. Electron. Mat. 9, 2201343 (2023).
Chen, X. et al. Wrap-like transfer printing for three-dimensional curvy electronics. Sci. Adv. 9, eadi0357 (2023).
Lu, D. et al. Bioresorbable wireless sensors as temporary implants for in vivo measurements of pressure. Adv. Funct. Mat. 30, 2003754 (2020).
Pflugfelder, S. C. & Stern, M. E. Biological functions of tear film. Exp. Eye Res. 197, 108115 (2020).
Recchioni, A., Mocciardini, E., Ponzini, E. & Tavazzi, S. Viscoelastic properties of the human tear film. Exp. Eye Res. 219, 109083 (2022).
Day, K. D. The Mechanics of a Hydrogel Contact Lens on the Human Eye with a Lubricating Tear Layer. Thesis, Massachusetts institute of Technology (1997).
Lu, L., Leanza, S. & Zhao, R. R. Origami with rotational symmetry: a review on their mechanics and design. Appl. Mech. Rev. 75, 050801 (2023).
Deng, Y. et al. Stretchable liquid metal based biomedical devices. npj Flex. Electron. 8, 12 (2024).
Guo, R. et al. One-step liquid metal transfer printing: toward fabrication of flexible electronics on wide range of substrates. Adv. Mater. Technol. 3, 1800265 (2018).
Masud, M. et al. Measurement techniques and challenges of wireless LC resonant sensors: a review. IEEE Access 11, 95235 (2023).
Borók, A., Laboda, K. & Bonyár, A. PDMS bonding technologies for microfluidic applications: a review. Biosensors 11, 292 (2021).
Sia, S. K. & Whitesides, G. M. Microfluidic devices fabricated in poly (dimethylsiloxane) for biological studies. Electrophoresis 24, 3563–3576 (2003).
Lee, S. E., Kim, S. R. & Park, M. Oxygen permeability of soft contact lenses in different pH, osmolality and buffering solution. Int. J. Ophthalmol. 8, 1037 (2015).
Artigas, J. M., Navea, A., García-Domene, M., Gené, A. & Artigas, C. Light transmission and ultraviolet protection of contact lenses under artificial illumination. Cont. Lens Anterior Eye 39, 141–147 (2016).
Elsheikh, A., Alhasso, D. & Rama, P. Biomechanical properties of human and porcine corneas. Exp. Eye Res. 86, 783–790 (2008).
Orssengo, G. J. & Pye, D. C. Determination of the true intraocular pressure and modulus of elasticity of the human cornea in vivo. Bull. Math. Biol. 61, 551–572 (1999).
Bao, F. et al. Assessment of the ex vivo biomechanical properties of porcine cornea with inflation test for corneal xenotransplantation. J. Med. Eng. Technol. 36, 17–21 (2012).
Liu, S. et al. Regulation of cell behavior by hydrostatic pressure. Appl. Mech. Rev. 71, 040803 (2019).
Hellmich, C. et al. Hierarchical biomechanics: concepts, bone as prominent example, and perspectives beyond. Appl. Mech. Rev. 74, 030802 (2022).
Ruberti, J. W., Sinha Roy, A. & Roberts, C. J. Corneal biomechanics and biomaterials. Annu. Rev. Biomed. Eng. 13, 269–295 (2011).
Dubbelman, M., Sicam, V. A. D. P. & Van der Heijde, G. L. The shape of the anterior and posterior surface of the aging human cornea. Vis. Res. 46, 993–1001 (2006).
Pandolfi, A. & Boschetti, F. The influence of the geometry of the porcine cornea on the biomechanical response of inflation tests. Comput. Method. Biomech. Biomed. Engin. 18, 64–77 (2015).
Chen, H. in Explosive Blast Injuries: Principles and Practices (Springer Nature, 2023)
Ong, J., Lee, A. G. & Moss, H. E. Head-down tilt bed rest studies as a terrestrial analog for spaceflight associated neuro-ocular syndrome. Front. Neurol. 12, 648958 (2021).
Chiquet, C. et al. Changes in intraocular pressure during prolonged (7-day) head-down tilt bedrest. J. Glaucoma 12, 204–208 (2003).
Liu, J. H., Sit, A. J. & Weinreb, R. N. Variation of 24-hour intraocular pressure in healthy individuals: right eye versus left eye. Ophthalmology 112, 1670–1675 (2005).
Pekmezci, M., Chang, S. T., Wilson, B. S., Gordon, M. O. & Bhorade, A. M. Effect of measurement order between right and left eyes on intraocular pressure measurement. Arch. Ophthalmol. 129, 276–281 (2011).
Acknowledgements
The authors gratefully acknowledge the support from the National Basic Research Program of China (Grant No. 2023YFB3609002), the National Natural Science Foundation of China (Grant Nos. 12225209 and U21A20502), the Tsinghua Precision Medicine Foundation (Grant No. 100010104), the capital health research and development of special (Grant No. 2022-4-4019) and the Natural Science Foundation of Zhejiang Province (Grant No. LQ23H18007).
Author information
Authors and Affiliations
Contributions
H.A., J.S., and Y.M. conceived and designed the research. H.A. and X.W. conducted the theoretical analysis, the device fabrication and characterization, the measurement system construction, as well as the performance evaluation. H.A., X.W., Z.L., L.Z., H.Z., and Y.Y. designed and conducted the experiments on animals and humans. H.A., J.S., and Y.M. wrote and edited the manuscript. J.S. and Y.M. supervised the research. All the authors read, discussed, and revised the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
An, H., Wang, X., Liao, Z. et al. LC contact lens sensor for ultrasensitive intraocular pressure monitoring. npj Flex Electron 8, 53 (2024). https://doi.org/10.1038/s41528-024-00341-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41528-024-00341-z