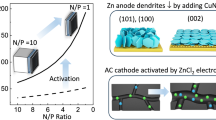
Abstract
Zinc anode-based electrochromic devices (ZECDs) represent a new generation of multifunctional electrochromic (EC) platforms, offering cost-effectiveness and high round-trip efficiency. However, their practical application remains limited due to the electric field inhomogeneity and the growth of Zn dendrites, issues primarily caused by the use of opaque peripheral Zinc (Zn) foils. Herein, we rationally designed a transparent (T = 71.4% @633 nm), durable, and flexible Ag-PVDF (polyvinylidene difluoride) coated Zinc (AP@Zn) mesh electrode. The AP@Zn mesh promotes a homogeneous electric field and potential distribution within ZECDs, exhibits excellent corrosion resistance, and possesses a low activation energy (47.59 kJ mol−1). Furthermore, it demonstrates broad compatibility with various EC electrodes. As a result, a 5 cm × 5 cm Prussian blue (PB)//AP@Zn achieved fast switching times (tc/tb 2.8 s/2.6 s), high coloration efficiency (157.44 cm2 C−1), outstanding cycling stability (93.7% ΔT retention after 500 cycles), and integrated energy storage functionalities (32.89 mA h m−2 at 0.02 mA cm−2). A large, scalable 10 cm × 10 cm PB//AP@Zn device showed significantly faster switching times (tc/tb 6.6 s/5.4 s) compared to the PB//Zn foil counterpart (tc/tb 15 s/11.4 s). Importantly, we also demonstrated devices based on Nb18W16O93 (NWO)//AP@Zn, which exhibited fast switching (tc/tb 18.5 s/20 s) and high durability (77.7% ΔT retention after 1200 cycles), as well as potassium vanadate (KVO)//AP@Zn featuring multicolor capabilities. Stacked PB//AP@Zn//KVO electrochromic displays exhibited a six-color palette including olive green1, tawny, bronzing, olive green2, deep blue-green, and cool grayish green. This work underscores the critical role of electrode design in advancing ZECDs towards multifunctional and flexible electronics.
Similar content being viewed by others
Introduction
In the era of Internet of Things, electrochromic devices (ECDs) featuring dynamic light modulation and energy efficiency have garnered significant attention across optoelectronics, flexible electronics, and wearables technologies1,2, enabling a wide range of applications, spanning smart windows3, dimmable glasses4,5, non-emissive displays6,7, camouflage, etc. Among these, EC smart windows can intelligently modulate visible (VIS) and infrared (IR) spectra on demand, substantially reducing heating and cooling loads in buildings and thereby enhancing overall energy efficiency8,9.
Conventional ECDs typically consist of transparent conductive electrodes (TCEs, e.g., fluorine-doped tin oxide FTO or indium tin oxide ITO-coated glasses), an EC layer (ECL), an electrolyte, and an ion storage layer (ISL)10. Similar to rechargeable batteries, the coloration and bleaching processes in ECDs rely on redox reactions at the electrode/electrolyte interface, often allowing the integration of energy storage functions11,12. However, due to charge imbalance and electrochemical potential mismatch between the ECL and ISL, conventional ECDs generally require energy input for both coloration and bleaching, leading to low round-trip energy efficiency13.
To address this limitation, metal anode-based ECDs, especially zinc anode-based electrochromic devices (ZECDs), have emerged, featuring inherent energy recovery capabilities, excellent charge balance between the Zn anode and ECL, and high round-trip efficiency14,15,16. Zn anode offers numerous advantages, including a low redox potential (−0.76 V vs. standard hydrogen electrode [SHE]), high theoretical gravimetric capacity (820 mAh/g), natural abundance, and cost-effectiveness15,17. More importantly, the electrochemical potential difference between the Zn anode and ECL allows for spontaneous coloration or bleaching with effective energy recovery, greatly improving overall energy efficiency in ZECDs18. Li et al. first demonstrated dual-functional WO3//Zn ZECDs with high optical contrast (∆T% = 88%), fast-switching times (tc/tb 3.9 s/5.1 s), and a large areal capacity (185.6 mAh/m2 at 0.5 mA/cm2)19. Subsequently, ZECDs employing Prussian blue (PB)//Zn20, transition metal oxide//Zn21, and conductive polymer electrodes such as polypyrrole22 and polyaniline23 have been developed for flexible display24, smart windows, and wearable electronics.
Notably, to maintain high optical contrast, opaque zinc foil anodes are commonly positioned at the device periphery, which results in a non-uniform electric field, current density, and ion flux distribution across the active area, leading to blooming effects, prolonged response times, and diminished cycling stability, particularly in large-area ZECDs25,26. As a solution, optically transparent metal mesh anodes have been proposed. For example, Wu et al. fabricated a conductive, transparent copper mesh (T% = 75% at 550 nm) via inkjet printing, achieving uniform coloration within 1 s in 100 cm2 ECDs27. Similarly, Li et al. developed a flexible zinc mesh anode with high transparency (T% = 89.3% at 550 nm) via chemical electrodeposition28,29, enabling uniform coloration in 80 cm2 ZECDs. Despite these advancements, devices with transparent Zn meshes still face limitations in cycling stability (e.g., ∆T retention of 77% after 1000 cycles)28, primarily due to Zn’s susceptibility to side reactions such as corrosion and hydrogen evolution reactions (HER), as well as the formation of zinc dendrites in aqueous electrolytes30,31,32,33,34,35,36. Thus, achieving both optical transparency and electrochemical durability in Zn anodes remains a critical bottleneck in the development of advanced ZECDs.
In this study, we fabricated a flexible, transparent, and durable Ag-polyvinylidene difluoride (PVDF) coated zinc mesh (AP@Zn) anode to address these challenges and ensure uniform electric field and potential distributions across ZECDs. By optimizing electrodeposition and chemical displacement conditions, the AP@Zn mesh achieved high transparency (T% = 71.4% at 633 nm), strong corrosion resistance, and low activation energy (47.59 kJ mol−1). To validate the practicality and universality of AP@Zn anode, we assembled ZECDs using AP@Zn in combination with Prussian blue (PB), Nb18W16O93 (NWO), and potassium vanadate (KVO) cathodes. The resulting 25 cm2 PB//AP@Zn devices exhibited fast-switching times (tc/tb 2.8 s/tb 2.6 s), high coloration efficiency (CE = 157.44 cm2 C−1), excellent cycling stability (ΔT retention of 93.7% after 500 cycles), and energy storage capability. Notably, a scaled-up 100 cm2 PB//AP@Zn achieved significantly faster switching times (tc/tb 6.6 s/5.4 s) compared to devices using peripheral Zn strip anodes (tc/tb 15 s/11.4 s). Furthermore, the NWO//AP@Zn exhibited fast switching (tc/tb 18.5 s/tb 20 s) and excellent durability (ΔT retention of 77.7% after 1200 cycles), while KVO//AP@Zn demonstrated multicolor EC behavior. A stacked PB//AP@Zn//KVO display achieved a six-color palette including olive green1, tawny, bronzing, olive green2, deep blue-green, and cool grayish green. This study highlights the importance of transparent, durable electrode design in advancing next-generation ZECDs for multifunctional, fast switching, and flexible electronic applications.
Results
Preparation of transparent zinc mesh
Conventional ECDs are assembled with pre-deposited ECL and ISL on TCEs (e.g., FTO glass), while the optical properties and charge balance between ECL and ISL can greatly affect the performance. Additionally, the commonly used FTO glasses in ECDs can cause a large voltage drop from the edge to the center (0.3 V, Supplementary Fig. 1), causing a blooming effect, especially in large-area ECDs. Furthermore, the continuous external electrical energy supply during both the coloring and bleaching processes of conventional ECDs, referred to as “bidirectional energy consumption”, not only increases the operating cost but also limits its application scenarios. In comparison, the inherent potential difference between the EC cathodes and Zn anodes in ZECDs can achieve “self-coloration” or “self-bleaching”, significantly reducing energy consumption and increasing round-trip efficiency via energy retrieval functionality18,37.
However, optically opaque zinc foils placed at the periphery still result in uneven electric field distribution and non-uniform ion flux28. As depicted in Fig. 1a (top) and Supplementary Fig. 2a, this asymmetric electrode layout leads to an exponential potential and electric field strength decay from the edge to the center (Fig. 1b and Supplementary Fig. 2b)38 and imbalanced Zn2+ ion flux distribution, leading to tinting inhomogeneity, particularly prominent in large-area devices. The transparent zinc mesh is regarded as the ideal anode for ZECDs (Fig. 1a bottom), as it ensures high energy efficiency and uniform distributions of potential, electric field (Fig. 1c; Supplementary Fig. 2c), and ion concentration, thereby enabling superior EC performance in ZECDs.
a Schematic of ZECDs based on zinc strip (top) and zinc mesh (bottom) anodes. Internal electric field distribution in ZECDs with b Zn strip and c Zn mesh anode. Scanning electron microscope (SEM) images of d the transparent copper mesh and e the transparent Zn mesh. SEM images of a single wire in f the transparent copper mesh and g the transparent Zn mesh. h Changes in transmittance and photographs of transparent copper mesh before and after electrodeposition.
To fabricate a transparent zinc mesh, a commercially available transparent copper mesh (Fig. 1d) was selected as the conductive substrate for Zn electrodeposition. Electrodeposition offers advantages in cost-effectiveness and scalability, making it an ideal technique for preparing large-area Zn meshes suitable for use in scalable ZECDs29. The Cu mesh, with a wire diameter of ~30 μm (Fig. 1d, f), is uniformly coated with a metallic Zn layer during deposition, forming a core-shell structured transparent Zn mesh electrode (Fig. 1e). After deposition, the wire diameter increases to ~35 μm, indicating that the thickness of the deposited zinc layer is ~2.5 μm. High magnification scanning electron microscopy (SEM) images clearly reveal the micro/nano-structured morphology of the Zn layer (Fig. 1g), which consists of tightly stacked lamellar grains. This highly ordered two-dimensional assembly pattern creates a continuous cladding on the surface of the copper mesh, significantly reducing the light scattering effect and thus maintaining optical transparency38. The amount of deposited Zn, calculated based on Faraday’s law (Supplementary Fig. 3), is estimated to be ~1.28 mg cm−2. Transmittance spectra (Fig. 1h) show a slight decrease in average transmittance from 76.3% (bare Cu mesh) to 70.8% after Zn deposition. Despite this reduction, the Rubik’s cube placed underneath the Zn mesh remains clearly visible (insets in Fig. 1h), further demonstrating the high optical transmittance of Zn mesh. In addition, the transparent Zn mesh exhibits excellent mechanical deformability (Supplementary Fig. 4), enabling conformal integration onto various surfaces, and showing strong potential for applications in flexible electronics.
Preparation of modified transparent zinc mesh
Although ZECDs with Zn mesh anodes effectively address the issue of uneven electric field distribution in bulk zinc foil-based ZECDs, Zn mesh anodes still face challenges related to limited electrochemical stability, particularly in aqueous electrolytes. In aqueous environments, Zn metal promotes inevitable water decomposition accompanied by HER, which raises the local pH, accelerates Zn corrosion, and leads to the formation of insulating by-products. These effects significantly reduce Zn utilization and cycling life39,40. Therefore, developing a durable, transparent zinc mesh is essential to the practical implementation of stable, fast-switching ZECDs.
To fabricate a durable zinc mesh anode, we employed a simple yet effective interface modification strategy. PVDF, consisting of alternately linked –CF2– and –CH2– has been widely used in batteries due to its low cost, chemical stability, and high-density properties containing polar fluorine groups41. However, its limited ionic conductivity can hinder efficiency during long-term cycling42. In this work, the durability of transparent Zn mesh anodes was enhanced by introducing ultra-thin metal-modified PVDF (M-PVDF, where M = Ag, Cu, or Sn) coatings.
The M-PVDF coatings are obtained via a facile one-step chemical displacement method (Fig. 2a), where three types of metals with high standard redox potentials were investigated for Zn mesh protection, namely Ag (+0.80 V vs. SHE), Cu (+0.34 V vs. SHE), and Sn (−0.15 V vs. SHE). Taking Ag as an example, the durable and transparent Ag-PVDF-coated zinc mesh (AP@Zn) was prepared simply by immersing the as-obtained Zn mesh into an ethylene glycol solution containing AgNO3 and PVDF with continuous stirring. The strong reducing nature of Zn drives a spontaneous displacement reaction (2Ag+ + Zn → 2Ag + Zn2+), during which PVDF molecules form a continuous film via in situ attachment to the metal surface through C–F bonding and van der Waals interactions, resulting in the formation of AP@Zn meshes.
a Preparation process of M-PVDF@Zn meshes. b The corrosion potentials and Tafel slopes of different electrodes. c Change in transmittance of transparent zinc mesh before and after displacement. d XRD plots and e High-resolution F 1s XPS spectra of different zinc mesh samples. f SEM of AP@Zn mesh. g The self-corrosion potential and Tafel slope of different samples. h Nyquist plots of the AP@Zn electrode under different temperatures. i Arrhenius curves for Ea calculation of different Zn electrodes.
To optimize the electrochemical performance of durable and transparent Zn meshes, the parameters influencing the chemical displacement process were systematically investigated, including PVDF concentration, metal species, metal ion amount, and reaction duration. First, the PVDF concentration was optimized to 0.1 mg mL−1, as this condition exhibited a lower hydrogen evolution current density (JHER), higher corrosion potential (Ecorr), and reduced corrosion current density (Jcorr) (Supplementary Fig. 5). Next, various metal species were evaluated, revealing that CP@Zn, SP@Zn, and AP@Zn all show higher Ecorr than unmodified zinc mesh (Supplementary Fig. 6), with AP@Zn exhibiting the highest Ecorr (−0.95 V vs. Ag/AgCl), and the lowest JHER. However, both SP@Zn and CP@Zn display noticeable surface inhomogeneity (Supplementary Fig. 7), and dendritic structures are observed on CP@Zn, significantly compromising the protective capability of the CP layer. In contrast, AP@Zn demonstrates not only a higher Ecorr but also a larger Tafel slope for HER compared to SP@Zn and CP@Zn (Fig. 2b). To conduct a more thorough assessment of the corrosion resistance, SP@Zn, CP@Zn, and AP@Zn electrodes were immersed in a hybrid 1 M (OTF) + 0.5 M Zn(OTF)2 electrolyte for 12 h. As shown in Supplementary Fig. 8, SP@Zn and CP@Zn exhibited pronounced surface roughening, corrosion pits, and partial cracking, whereas AP@Zn retained a smooth and intact morphology without coating detachment. These results confirm the superior interfacial stability and corrosion resistance of the AP@Zn layer, indicating its superior anti-corrosion properties. The Ag+ amount in the displacement solution was then tuned from 5 mmol to 30 mmol. A low Ag+ amount resulted in incomplete surface coverage, while a high amount led to excessive deposition. An intermediate amount of 15 mmol provided optimal coating uniformity and corrosion resistance (Supplementary Fig. 9). Finally, the reaction time for the chemical displacement was varied from 0.5 to 10 min (Supplementary Fig. 10), with a duration of 2 min yielding the best anti-corrosion performance for AP@Zn. These optimized parameters were adopted for the synthesis of modified Zn meshes and are used in all subsequent discussions unless otherwise specified.
Interestingly, the transmittance of AP@Zn is slightly higher than that of the bare Zn mesh (Fig. 2c), likely due to partial dissolution of zinc during the displacement reaction and the formation of a more compact and smoother AP coating. The XRD pattern of AP@Zn (Fig. 2d) displays sharp diffraction peaks at 2θ = 38.1°and 44.3°, corresponding to the (111) and (200) planes of face-centered cubic silver, while PVDF does not produce Bragg diffraction peaks due to its amorphous nature.
The chemical composition of Zn, Ag@Zn, and AP@Zn was further analyzed by X-ray photoelectron spectroscopy (XPS). As expected, no Ag 3d or F 1s peaks were detected in the pristine Zn mesh (Supplementary Fig. 11), and no F 1s peaks were observed in Ag@Zn (Supplementary Fig. 12). In contrast, both Ag 3d and F 1s peaks appeared in AP@Zn (Fig. 2e and Supplementary Fig. 13). As shown in Supplementary Fig. 14, cross-sectional SEM reveals a compact and uniform coating layer on the Zn substrate, while EDS mapping confirms the homogeneous distribution of Ag, F, and Zn elements. Raman spectroscopy further exhibits characteristic CF2 and C–F stretching bands (~839, 1070, and 1280 cm−1) corresponding to the β-phase PVDF, collectively confirming the successful formation of a uniform Ag-PVDF composite layer on Zn mesh surface.
High-resolution SEM imaging (Fig. 2f) reveals that Ag nanoparticles (50–80 nm) are uniformly anchored on the surface of AP@Zn. Corresponding SEM-EDS elemental mapping (Fig. 2f) confirms a homogeneous distribution of Ag and F elements, contributing to improved surface hydrophilicity (Supplementary Fig. 15). In contrast, Ag@Zn exhibits inhomogeneous Ag nanoparticle growth (Supplementary Fig. 16), suggesting that the PVDF molecular chains (–CH2–CF2–) regulate Ag nucleation and growth, leading to a more uniform Ag-PVDF coating. Notably, PVDF not only binds to the Zn substrate, but also exhibits strong dipolar interactions between its C–F polar bonds and the Ag surface, which enhance the adhesion of Ag particles during cycling43. To further evaluate the mechanical robustness of the AP@Zn electrode, continuous bending tests (10 mm radius, 1000 cycles) confirm the remarkable mechanical robustness of the AP@Zn electrode (Supplementary Fig. 17). Both the morphology and optical transmittance remain essentially unchanged after repeated deformation. The PB//AP@Zn device also retains nearly identical ΔT and switching kinetics. These results collectively confirm the excellent mechanical robustness and interfacial adhesion of the AP@Zn coating under repeated deformation.
The necessity of PVDF incorporation is further validated by evaluating the Ecorr and Tafel slope of Zn, Ag@Zn, and AP@Zn meshes. As shown in Fig. 2g, AP@Zn exhibited a more positive Ecorr (−0.95 V) compared to Ag@Zn (−0.981 V) and bare Zn mesh (−0.987 V), indicating enhanced resistance to side reactions. Moreover, the AP@Zn electrode shows a higher Tafel slope (584.05 mV dec−1) than Ag@Zn (580.46 mV dec−1) and bare Zn (439.57 mV dec−1), further confirming its superior corrosion resistance.
To assess reaction kinetics, the activation energies (Ea) of different Zn electrodes were calculated from temperature-dependent charge transfer resistance (Rct) derived from Nyquist plots, (Fig. 3h, i; Supplementary Fig. 18). AP@Zn exhibits the lowest Ea (47.59 kJ mol−1), significantly lower than that of Ag@Zn (61.89 kJ mol−1) and bare Zn mesh (75.73 kJ mol−1), demonstrating that the AP@Zn layer effectively enhances electrode reaction kinetics.
a Schematic diagram of the working principle of PB//AP@Zn mesh-based ZECDs. b Transmission spectra of PB electrodes in colored (1.8 V) and bleached (0.6 V) states. c Dynamic transmittance change of PB electrodes at alternating applied voltages (1.8 V and 0.4 V). d Color changes of the PB electrode at different voltages. e Cycling stability of the PB electrode. f Coloration efficiency of PB//AP@Zn mesh. g CV curves and h GCD curves of PB//AP@Zn mesh. i Performance comparison with the reported ZECDs.
Analysis of electrochromic performance
After successfully preparing transparent and durable AP@Zn mesh anodes, ZECDs were assembled using various EC materials (ECMs). Among them, Prussian Blue (PB), a classic anodically colored ECM, was selected due to its numerous advantages, including low raw material cost, facile synthesis, pronounced color change (from transparent to deep blue), excellent chemical stability, suitable redox potential, and fast electrochemical response44. PB films were prepared via the hydrothermal method, which offers strong film-substrate adhesion—superior to other common approaches such as electrochemical and chemical deposition44,45. The resulting PB films, ~1 μm thick, consist of densely packed, regular nanoparticles (Supplementary Fig. 19a), which enhances both mechanical adhesion and cycling stability.
ZECDs were then assembled by pairing the AP@Zn anode with a PB cathode (Fig. 3a), using a hybrid K(OTF)- Zn(OTF)2 electrolyte. The electrochemical reaction of the as-assembled ZECDs follows a typical redox mechanism. During the discharge (bleaching) process, the Zn anode undergoes oxidation (Zn → Zn2+ + 2e−), while the PB cathode undergoes a reduction reaction, accompanied by the insertion of cations and electrons (K4Feᴵᴵ4[Feᴵᴵ(CN)6]3). Upon charging (coloration), the reverse reaction occurs: the PB electrode is oxidized (K4FeII4[FeII(CN)6]3 → 4 K++ FeIII4[FeII(CN)6]3 + 4e−), while Zn2+ ions are reduced and deposited at the anode (Zn2+ + 2e− → Zn). To rationalize the hybrid electrolyte design, dynamic transmittance and cycling tests were also performed in single-ion electrolytes (Supplementary Fig. 20). Devices operated in single 1 M K(OTF) or 0.1 M Zn(OTF)2 electrolytes exhibited rapid ΔT attenuation and poor reversibility, confirming that neither K+ nor Zn2+ alone can ensure good reversibility in the PB//AP@Zn device. Further optimization of Zn2+ concentration (Supplementary Fig. 21) revealed that excessive Zn(OTF)2 (≥0.5 M) caused reduced optical modulation and faster degradation, whereas 1 M K(OTF) + 0.1 M Zn(OTF)2 provided optimal response speed, optical contrast, and cycling stability. Therefore, this hybrid electrolyte was adopted to enable fast K+ intercalation in PB and stable Zn2+ deposition/dissolution at the AP@Zn anode, ensuring charge balance and long-term device durability.
As shown in Fig. 3b, the PB electrode exhibits efficient and reversible coloration/bleaching under alternating potential steps of 1.8 V (30 s) and 0.6 V (30 s), achieving a high transmittance modulation (ΔT) of 63.8% at 633 nm. Additionally, the PB electrode also manifests fast-switching kinetics (tc/tb 3.0 s/2.8 s, Fig. 3c) under alternating voltages of 1.8 V–0.4 V. This ultra-fast response time is attributed to the uniquely uniform and conductive AP@Zn network, which ensures a homogeneous spatial electric field distribution. This prevents local polarization, thereby accelerating the insertion/extraction of K+ ions and facilitating rapid electrochromic transitions of the PB films (Fig. 3d).
The PB electrode also demonstrated excellent long-term electrochemical stability, retaining 96.3% of its initial ΔT after 1000 consecutive cycles (Fig. 3e), meanwhile, it still maintains a relatively fast response time (Fig. 3e inset), outperforming devices using zinc mesh and SP@Zn mesh counter electrodes (Supplementary Fig. 22). The surface morphology of the cycled zinc mesh showed obvious dendrite evolution and irregular aggregation (Supplementary Fig. 23). However, the cycled AP@Zn electrode remained smooth and intact, without obvious dendrite growth or structural degradation. Additionally, it was noticed that the electrolyte from the cycled PB//AP@Zn contained significantly less dissolved Ag compared to that from the cycled PB//Ag@Zn (Supplementary Fig. 24), indicating that the PVDF matrix effectively suppresses Ag dissolution. This enhanced durability is credited to the Ag-PVDF composite layer on the AP@Zn meshes. The Ag nanoparticles enhance electrical conductivity and suppress the formation of zinc dendrites and side reactions, while the PVDF matrix regulates Ag nanoparticle distribution and protects the zinc mesh from corrosion by the electrolyte.
A high coloration efficiency (CE) of 157.44 cm2 C−1 was achieved by fitting the optical density-charge density curve (Fig. 3f), indicating that large optical modulation can be obtained with minimum charge input (Fig. 3f). Collectively, the PB//AP@Zn device demonstrates high optical contrast (ΔT = 63.8%@633 nm), ultra-fast-switching times (tc = 3.0 s, tb = 2.8 s), excellent cycling stability (ΔT retention of 96.3% after 1000 cycles), and ultra-high CE (157.44 cm2 C−1), alongside a self-bleaching capability.
The energy storage performance of the device was also evaluated via cyclic voltammetry (CV, Fig. 3g) and galvanostatic charge/discharge (GCD, Fig. 3h) tests. Well-defined redox peaks and distinct charge/discharge plateaus are observed, corresponding to the intercalation/deintercalation of K+ ions into/from the PB film. The clearly visible redox peaks, even at the high scan rate of 50 mV s−1 highlight the fast reaction kinetics at both electrodes.
The areal capacity of the PB//AP@Zn device, determined from the GCD curves, reached 32.89 mA h m−2 at a low current density of 0.02 mA cm−2 and remained at 28.17 mA h m−2 when the current density increased to 0.2 mA cm−2, demonstrating outstanding rate capability. A comparative analysis (Fig. 3i and Supplementary Table 1) further underscores the superior switching speed and high CE of the PB//AP@Zn device17,25,46,47,48, positioning it as a promising EC platform for next-generation smart energy-saving technologies.
To further evaluate the practical applicability of the AP@Zn mesh electrode, large-area ZECDs were assembled using PB cathodes. The resulting 5 × 5 cm2 PB//AP@Zn device exhibited a transmittance of 59.4% at 633 nm in its transparent state (Fig. 4a), and can be electrochemically colored to a low transmittance (T%) of ~5%, achieving a transmittance modulation (ΔT) of 55.3%, comparable to that observed in cuvette-scale tests (Fig. 3b). The switching kinetics of this large-area device remained fast, with a bleaching time (tb) of 2.6 s and a coloration time (tc) of 2.8 s (Fig. 4b), despite the increased device size.
a Transmittance spectra, b dynamic transmittance variation at alternating step voltages (1.8 V and 0.4 V), c open-circuit potential, and d optical photographs during bleaching (top) and coloration (bottom) of a 5 × 5 cm2 PB//AP@Zn device. Dynamic transmittance variation at alternating step voltages (1.8 V and 0.4 V) of PB-based 10×10 cm2 ZECDs with e Zn strips anode, and f AP@Zn anode. g Energy storage effect demonstration of a 10 × 10 cm2 PB//AP@Zn device.
Additionally, the 5 × 5 cm2 PB//AP@Zn device maintained excellent cycling stability, retaining 93.7% of its initial ΔT after 500 cycles (Supplementary Fig. 25). It also exhibited a high open-circuit potential of 1.269 V in the colored state, along with excellent uniformity in coloration and bleaching processes (Fig. 4c, d).
To further underscore the advantages of AP@Zn meshes for scalable applications, 10 × 10 cm2 ZECDs were assembled using either Zn strips or AP@Zn mesh anodes, respectively. It should be noted that PB electrodes for these large-area ZECDs were synthesized via electrodeposition. To evaluate the scalability of PB films, large-area electrodeposition was performed on a 15 × 25 cm2 FTO substrate. PB films exhibiting uniform appearance without cracks or defects (Supplementary Fig. 26). SEM analysis confirmed the typical valley-like morphology of electrodeposited PB. Comparable transmittance (between 1.7% and 5.6%@633 nm) across different regions verifies the reproducibility, highlighting the scalability of this deposition method. As shown in Fig. 4e, f and Supplementary Fig. 27, the response times in ZECDs utilizing the AP@Zn mesh anode (tc/tb 6.6 s/5.4 s) are generally faster than those of ZECDs assembled with Zn mesh anode (tc/tb 6.2 s/7.4 s) or zinc foil anode (tc/tb 15 s/11.4 s). This clearly validates the effectiveness of the transparent and conductive Zinc meshes geometry in enabling fast-switching behavior in large-area ZECDs.
Moreover, the 10 × 10 cm2 PB//AP@Zn device was capable of simultaneously powering a digital hygrometer for over one hour and undergoing color switching from the colored to the bleached state during discharge (Fig. 4g), thereby demonstrating its dual functionality in both electrochromic modulation and energy storage. Despite these promising results, the aqueous Zn2+/K+ electrolyte may still face challenges such as evaporation, leakage, and parasitic reactions during extended operation. To address these issues, future work could focus on developing quasi-solid or gel-based electrolytes, which can effectively prevent the occurrence of side reactions and avoid problems such as electrolyte leakage and evaporation24.
To further validate the universality of the as-prepared AP@Zn electrode beyond pairing with anodically coloring Prussian Blue (PB) cathodes, we explored its compatibility with other electrochromic materials (ECMs), including cathodically coloring Nb18W16O93 (NWO) and multicolored potassium vanadate (KVO). The NWO layer was uniformly deposited onto FTO glass, with a thickness of approximately 593 nm (Supplementary Fig. 28), and its successful synthesis was confirmed via XPS analysis (Supplementary Fig. 29).
As shown in Fig. 5a, the assembled NWO//AP@Zn device, using a K(OTF)-Zn(OTF)2 electrolyte, exhibited reversible color switching behavior. During discharging, Zn2+ and K+ ions are intercalated into the NWO framework, leading to coloration, while ion deintercalation during charging results in bleaching. The device achieved a notable ∆T of ~44.3% at 633 nm, with switching times tc/tb of 18.5 s/20 s (Fig. 5b, c). Furthermore, it retained 77.7% of its initial ∆T after 1200 cycles (Supplementary Fig. 30a), along with a CE of 16.2 cm2 C−1 (Supplementary Fig. 30b).
a Schematic diagram, b Transmission spectra in colored (0 V) and bleached (2.0 V) states, and c Dynamic Transmittance variation at alternating applied voltages (0 V and 2.0 V) of the Nb18W16O93//AP@Zn device. d Schematic diagram, e Transmission spectra and optical photographs, and f CV plot at 20 mV s−1 of the KVO//AP@Zn device. g Photographs of a 5 × 5 cm2 NWO//AP@Zn device in the colored and bleached states. h Photographs of 5 × 5 cm2 KVO//AP@Zn devices at different voltages. i Schematic illustration of the stacked PB//AP@Zn//KVO ZECDs offering six colors. j The schematic diagram illustrating the color overlay effect produced by the combination of KVO and PB. The color codes on the top represent the color of the top KVO electrode, while the color codes on the bottom represent the color of the bottom PB electrode. k Digital photographs of the PB//AP@Zn//KVO display showing six colors.
In addition, an EC display was fabricated by pairing the AP@Zn mesh anode with a KVO cathode (Fig. 5d and Supplementary Fig. 31). The KVO film displayed a nanoparticle morphology and a thickness of ~200 nm. This device exhibited multicolor behavior—transitioning among orange, yellow, and green—driven by Zn2+ intercalation/deintercalation, as evidenced by the voltage-dependent transmittance spectra (Fig. 5e) and corresponding redox peaks (~0.3 V/1.2 V) in the CV curves (Fig. 5f). Notably, the KVO//AP@Zn multicolor EC display showed excellent cycling stability, maintaining a stable current response over 500 cycles (Supplementary Fig. 32).
To further demonstrate the scalability and universality, 5 cm × 5 cm EC devices were assembled using both NWO and KVO cathodes with the AP@Zn mesh anode. The large-area NWO//AP@Zn device (Fig. 5g) showed full bleaching from a dark blue state, while the KVO//AP@Zn device (Fig. 5h) exhibited clear and reversible multicolor changes from green to orange upon varying the applied voltage.
Inspired by the demonstrated compatibility of the AP@Zn electrode with cathodically, anodically, and multicolored ECMs, a stacked multicolor PB//AP@Zn//KVO EC display was designed by leveraging a color overlay effect. As illustrated in Fig. 5i, the PB//AP@Zn//KVO device was assembled with a KVO top electrode, a PB bottom electrode, and the AP@Zn electrode sandwiched in between. In this stacked configuration, the optical states of the PB and KVO electrodes can be independently modulated, enabling multicolor display through color superimposition (Fig. 5j). Specifically, the KVO electrode demonstrates three-color behavior (orange ⇄ yellow ⇄ green; Fig. 5h), while the PB electrode exhibits two distinct states (transparent ⇄ blue). Consequently, the PB//AP@Zn//KVO device can produce six distinct colors via controlling the applied voltage: olive green1, tawny, bronzing, olive green2, deep blue-green, cool grayish green (Fig. 5k). These colors result from the independent and combinatorial modulation of the two electrodes. For example, when the PB electrode is in its bleached (transparent) state, and KVO is in the green state, the device appears olive green1. Further oxidation of KVO produces tawny and bronzing colors. Alternatively, when the PB electrode is switched to its colored (mazarine blue) state and combined with the varying KVO states, additional composite colors such as olive green2, deep blue-green, and cool grayish green emerge. As can be seen in Supplementary Fig. 33, the transmittance spectra of the stacked electrochromic display in six different color states have been collected, clearly demonstrating the tunable multicolor performance. Thus, through precise voltage control, the device achieves versatile multicolor display functionality via dynamic optical layering of individual ECMs.
Discussion
In summary, to address the challenges of non-uniform electric field and ion concentration distributions in ZECDs caused by conventional peripheral Zn foils and to further enhance the overall durability of Zn anodes, we developed a durable, transparent, flexible, and scalable Zn mesh electrode for high-performance ZECDs. This Zn mesh electrode was fabricated through a combined electrodeposition and displacement reaction strategy, followed by the application of a Ag-PVDF coating, resulting in the optimized AP@Zn mesh electrode.
The AP@Zn meshes exhibited excellent optical transmittance (71.4% at 633 nm), a favorable Ecorr (−0.95 V), low JHER (23.73 mA cm−2), and low activation energy (47.59 kJ mol−1). Critically, it enabled uniform electric field and potential distributions in ZECDs. As a result, a large-area 5 cm × 5 cm PB//AP@Zn ZECDs achieved fast-switching times (tc 2.8 s and tb 2.6 s), high CE (157.44 cm2 C−1), and excellent cycling stability (ΔT retention of 93.7% after 500 cycles), along with energy storage capability (32.89 mA h m−2 at 0.02 mA cm−2), among the best of reported ZECDs. Furthermore, the scalable 10 cm × 10 cm PB//AP@Zn showed significantly faster switching (tc 6.6 s and tb 5.4 s) than its counterpart using peripheral Zn strip anodes (tc 15 s and tb 11.4 s).
Importantly, this AP@Zn mesh anode demonstrated excellent universality, functioning effectively with various ECMs. It enabled the assembly of NWO//AP@Zn devices and with fast switching (tc/tb 18.5 s/20 s) and robust durability (ΔT retention of 77.7% after 1200 cycles), and KVO//AP@Zn device with multicolor capabilities. Moreover, a stacked PB//AP@Zn//KVO was constructed, achieving six distinct color states (olive green1, tawny, bronzing, olive green2, deep blue-green, cool grayish green) via controllable superposition of individual electrode colors. This study highlights the rational design, scalable fabrication, and multifunctional performance of a transparent and flexible Zn mesh anode, offering a viable pathway toward practical integration in large-area smart windows, flexible electronics, and wearable electrochromic devices.
Methods
Materials
All chemicals are of analytical grade and used without further purification. Acetic acid, stannous chloride, ethylene glycol (EG), zinc trifluoromethanesulfonate (Zn(OTF)2), hydroxyethyl cellulose (HEC), potassium trifluoromethanesulfonate (KOTF), and niobium oxalate were purchased from Macklin Biochemicals and Technology Co. Anhydrous ethanol, zinc sulfate heptahydrate, anhydrous sodium sulfate, boric acid, silver nitrate, hydrochloric acid (36–38 wt.%), and potassium chloride were purchased from Sinopharm Chemical Reagent Co. Polyethylene imine solution (PEI, 50% (w/v) in H2O), polyvinylidene fluoride (PVDF), potassium ferricyanide, glucose monohydrate, acrylamide (AM), ammonium persulfate (APS), cupric chloride dihydrate, N,N′-methylenebisacrylamide (MBAA), ammonium metatungstate, citric acid, and vanadium oxide were purchased from Aladdin Reagent Co. Ltd. The cooper mesh (100 Mesh, 0.0012″ Wire Diameter) was purchased from TWP Inc. Fluorine-doped tin oxide (FTO) coated glass was produced by Wuhan Jingge Solar Technology Co. Deionized (DI) water was used throughout the experiments.
Preparation of transparent zinc mesh
Preparation of transparent zinc mesh follows previously reported work28. Firstly, 15 g ZnSO4 and 15 g Na2SO4 were dissolved in 100 ml DI water, followed by adding 2.5 g boric acid, and 0.2 g PEI was then added and dissolved to obtain a clear electrodeposition solution. After that, the purchased copper mesh was cleaned with a mixture of glacial acetic acid and ethanol (V: V = 1: 1), and then rinsed with DI water before drying in air. Subsequently, with the as-formulated electrodeposition solution, zinc sheet as the counter electrode and the copper mesh as the working electrode, zinc was deposited on Cu mesh at −0.2 V for 1 h. The transparent zinc mesh was then obtained after being rinsed with DI water and dried.
Preparation of modified transparent zinc mesh
The as-obtained transparent zinc mesh was subsequently modified. The displacement solution was obtained by dissolving 15 mmol silver nitrate in 20 ml ethylene glycol, followed by adding 0.002 g PVDF and continuous stirring for 12 h. Subsequently, the prepared transparent zinc mesh was immersed in the displacement solution for 2 min to obtain Ag-PVDF@Zn (abbreviated as AP@Zn). By controlling the formulation of the displacement solution, other modified transparent zinc mesh samples, including Ag@Zn, Cu-PVDF@Zn (abbreviated as CP@Zn), and Sn-PVDF@Zn (abbreviated as SP@Zn), were prepared similarly.
Preparation of electrochromic films
Preparation of anodically colored Prussian blue (PB) films
According to the previous report49, PB films were synthesized on FTO glass substrates by the hydrothermal method. 0.5 g glucose and 1.32 g K3[Fe(CN)6] were dissolved in 120 ml DI water to form a pale yellow solution. The pH of the solution was subsequently adjusted to ~7 by adding 2 ml concentrated hydrochloric acid. The solution was then transferred to a 300 ml Teflon stainless steel autoclave. At the same time, the FTO glass was ultrasonicated with ethanol and DI water in sequence and dried in a 60 °C oven. The dried FTO was placed into the autoclave with the FTO surface facing down. Subsequently, the high-pressure hydrothermal kettle was sealed and heated in the oven at 120 °C for 4 h. After natural cooling, the FTOs were thoroughly rinsed with DI water and dried at 60°C overnight. The areal loading of the PB film obtained by this method is approximately 0.08 mg/cm2.
Due to the limited scalability of the hydrothermal method, electrodeposition was used instead to prepare large-area PB films (10 cm × 10 cm)44. A three-electrode setup was adopted with FTO glass as the working electrode, where a Pt sheet and Ag/AgCl were used as the counter electrode and reference electrode. PB films were electrodeposited on FTO glass by applying a constant current density (−50 μA cm−2) for 300 s in an aqueous solution containing 10 mM FeCl3, 10 mM K3[Fe(CN)6], and 50 mM KCl.
Preparation of cathodically colored Nb18W16O93 (NWO) films
According to the previous report50, first, the FTO-coated glass was cleaned with DI water and ethanol in an ultrasonic bath, and dried in an oven at 60 °C. 0.20 g niobium oxalate and 0.49 g ammonium metatungstate were dissolved in a mixture of 2 ml DI water and 8 ml anhydrous ethanol, and after complete dissolution, 1.0 g citric acid was added to the solution. Then, the mixed solution was magnetically stirred and heated at 60 °C for 2 h. Finally, the solution was spin-coated onto the cleaned FTO glass, which was then vacuum dried at 150 °C and then annealed in air at 600 °C for 4 h. The areal loading of the NWO film obtained by this method is approximately 0.16 mg/cm2.
Preparation of multicolored potassium vanadate (K2V6O16·1.5H2O, KVO)
KVO films were prepared following a previously reported work51. First, 2 g of commercially available V2O5 powder was added to 30 mL aqueous KCl solution (2 M) at room temperature and stirred for 5 days to form a solution containing KVO suspension. Next, KVO powder was obtained by centrifugation in DI water six times and freeze-drying. KVO electrodes were fabricated by scraping. A KVO slurry of appropriate viscosity was first prepared by dissolving 1.4 g HEC in 60 mL KVO colloid (8 mg mL−1) by stirring at 60 °C for 24 h. The FTO glass substrate was sequentially pretreated with acetone, ethanol, and DI water, and dried in ambient air. The KVO/HEC slurry was then scrape-coated onto the FTO glass to form a uniform film. After heat treatment of the KVO/HEC-coated FTO glass at 200 °C in air for 24 h, KVO electrodes with a thickness of approximately 200 nm were prepared (Supplementary Fig. 22a).
ZECDs assembly
The ZECDs used transparent zinc mesh as the anode and an EC cathode (PB, NWO, or KVO), and a Zn2+/K+ electrolyte was used for PB and NWO-based ZECDs, while a Zn2+ electrolyte was used for KVO-based ZECDs. Transparent zinc mesh was mounted on bare glass, and 1 mm-thick 3 M double-sided tape was placed around the EC electrode as the spacer. After aligning the cathode and anode, ZECDs can be assembled with electrolyte injection and sealing. Stacked PB//AP@Zn//KVO was assembled with Zn2+/K+ electrolyte.
Material characterizations
X-ray diffraction (XRD) patterns were recorded on a Bruker D8 Advance diffractometer (Cu Kα radiation, λ = 1.5406 Å). Morphological analyses were performed using scanning electron microscopy (SEM, ZEISS Gemini300). Chemical states were characterized via X-ray photoelectron spectroscopy (XPS, Thermo Scientific K-Alpha with Al Kα source). Contact angle measurements were conducted using a JC2000DM goniometer at ambient conditions.
Electrochemical and electrochromic measurements
All electrochemical tests were performed on an electrochemical workstation (CHI760E, Shanghai Chenhua Instruments, Inc.). The linear polarization curve (LSV) was conducted in a three-electrode configuration in 1 M Na2SO4 aqueous electrolyte at 5 mV s−1, where different Zn electrodes act as the working electrode, Pt foil serves as the counter electrode, and Ag/AgCl as the reference electrode. Tafel tests were performed similarly with 1 M KCl electrolyte, and temperature-dependent charge transfer resistance (Rct) was also analyzed using a three-electrode configuration in 1 M ZnSO4 electrolyte. The activation energy (Ea) of the different Zn electrodes can be obtained from the slope by linearly fitting ln(1/Rct) versus 1000/T. Galvanostatic charge-discharge curves were carried out using a two-electrode configuration with PB working electrode and AP@Zn counter electrode in 1 M K(OTF)-0.1 M Zn(OTF)2 mixed electrolyte.
The optical properties of the samples were characterized by UV-Vis spectrophotometry (UV-2600i, SHIMADZU).
COMSOL simulation
The electric field simulations were performed using the commercial finite element software COMSOL 6.3. Here, the potential distribution along the centerline of the FTO-coated glass is calculated and applied to the bottom edge of the finite element model. According to the device parameters, the distance between the FTO-coated glass and the zinc mesh is 0.2 cm. Due to its high electrical conductivity, the potential drop along the top edge (i.e., the zinc mesh) is assumed to be zero.
Data availability
The data that support the findings of this study are available upon request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
References
Zhao, F. et al. Inorganic electrochromic smart windows for advancing building energy efficiency. Nat. Rev. Clean Technol. 1, 396–412 (2025).
Shao, Z. et al. All-solid-state proton-based tandem structures for fast-switching electrochromic devices. Nat. Electron. 5, 45–52 (2022).
Zhou, Y. et al. Electrochromic smart windows with on-demand photothermal regulation for energy-saving buildings. Adv. Mater. 37, e2502706 (2025).
Chen, J. et al. Electrochromic devices for dimming and display in augmented reality smart glasses. Device 2, 100470 (2024).
Zhao, Q. et al. Three-dimensional knotting of W17O47@PEDOT:PSS nanowires enables high-performance flexible cathode for dual-functional electrochromic and electrochemical device. InfoMat 4, e12298 (2022).
Kim, J. H. et al. Meniscus-guided micro-printing of Prussian blue for smart electrochromic display. Adv. Sci. 10, 2205588 (2023).
Zhang, W., Li, H., Yu, W. W. & Elezzabi, A. Y. Transparent inorganic multicolour displays enabled by zinc-based electrochromic devices. Light Sci. Appl. 9, 121 (2020).
Xie, Y. et al. Reusing the wasted energy of electrochromic smart window for near-zero energy building. Adv. Sci. 11, 2406232 (2024).
Xie, Y., Li, M., Huang, R., Cao, N. & Chao, D. How much of the energy in the electrochromic energy storage window can be reused?. Energy Storage Mater. 67, 103321 (2024).
Li, Y. et al. Colorful electrochromic displays with high visual quality based on porous metamaterials. Adv. Mater. 35, 2300116 (2023).
Dong, H. et al. Mg-doped layered vanadates for high-energy zinc anode-based electrochromic devices. Nanoscale 17, 16646–16653 (2025).
Xu, B. et al. The progress and outlook of multivalent-ion-based electrochromism. Small Sci. 3, 2300025 (2023).
Park, J., Lee, C. & Choi, D. Enhancing the inherently limited electrochromic redox reactions via integration with a transparent planar heater. Small 21, 2411929 (2025).
Liang, Y. et al. Reversible Zn2+ insertion in tungsten ion-activated titanium dioxide nanocrystals for electrochromic windows. Nanomicro Lett. 13, 196 (2021).
Chen, J. et al. The birth of zinc anode-based electrochromic devices. Appl. Phys. Rev. 11, 011316 (2024).
Chen, J., Eh, A. L.-S., Ciou, J.-H. & Lee, P. S. Pseudocapacitive and dual-functional electrochromic Zn batteries. Mater. Today Energy 27, 101048 (2022).
Wu, W. et al. Electrochromic devices constructed with water-in-salt electrolyte enabling energy-saving and prolonged optical memory effect. Chem. Eng. J. 446, 137122 (2022).
Yun, T. G., Hwang, B. & Cheong, J. Y. Recent progress and future research directions for electrochromic zinc-ion batteries. J. Energy Chem. 90, 220–232 (2024).
Li, H., Firby, C. J. & Elezzabi, A. Y. Rechargeable aqueous hybrid Zn2+/Al3+ electrochromic batteries. Joule 3, 2268–2278 (2019).
Luo, Y. et al. Potential gradient-driven fast-switching electrochromic device. ACS Energy Lett. 7, 1880–1887 (2022).
Rao, T., Zhou, Y., Jiang, J., Yang, P. & Liao, W. Low dimensional transition metal oxide towards advanced electrochromic devices. Nano Energy 100, 107479 (2022).
Wang, J. et al. A flexible, electrochromic, rechargeable Zn//PPy battery with a short circuit chromatic warning function. J. Mater. Chem. A 6, 11113–11118 (2018).
Singh, S. B., Tran, D. T., Jeong, K.-U., Kim, N. H. & Lee, J. H. A flexible and transparent zinc-nanofiber network electrode for wearable electrochromic, rechargeable Zn-ion battery. Small 18, 2104462 (2022).
Liu, Q. et al. Flexible Zn-ion electrochromic batteries with multiple-color variations. Angew. Chem. Int. Ed. 63, e202317944 (2024).
Wang, B. et al. A long-life battery-type electrochromic window with remarkable energy storage ability. Sol. RRL 4, 1900425 (2020).
Yin, J. et al. Integrated photoelectrochromic supercapacitor for applications in energy storage and smart windows. J. Energy Storage 51, 104460 (2022).
Hu, Z. et al. Ultra-low resistivity copper mesh as embedded current collector layer for inkjet-printed flexible electrochromic device realizing fast response and uniform coloration. Adv. Mater. Technol. 8, 2201037 (2023).
Li, H., Zhang, W. & Elezzabi, A. Y. Transparent zinc-mesh electrodes for solar-charging electrochromic windows. Adv. Mater. 32, 2003574 (2020).
Xu, Y. et al. Integrating transparent zinc mesh and anti-freezing hydrogel electrolyte toward durable zinc anode-based electrochromic devices. Adv. Mater. Technol. 10, 70023 (2025).
Liu, X. et al. A sustainable and scalable approach for in situ induction of gradient nucleation sites in biomass-derived interface layers for ultra-stable aqueous zinc metal batteries. Angew. Chem. Int. Ed. 64, e202504613 (2025).
Zhang, F. et al. Selective interface engineering with large π-conjugated molecules enables durable Zn anodes. Angew. Chem. Int. Ed. 64, e202425487 (2025).
Qu, Y. F., Liu, X., Qian, J. W., Chen, J. & Chen, L.-F. Agar-based interface for suppressing parasitic reactions toward high-performance aqueous Zn-ion batteries. Batter. Supercaps 7, e202400159 (2024).
Zhang, F. et al. Integration of confinement crosslinking and in situ grafting for constructing artificial interphases toward stabilized zinc anodes. Energy Environ. Sci. 17, e202400159 (2024).
Hu, J. et al. A hydrophobic alloy-coated Zn anode for durable electrochromic devices. Chem. Commun. 60, 566–569 (2024).
Liu, L. et al. Full-temperature all-solid-state dendrite-free Zn-ion electrochromic energy storage devices for intelligent applications. Chem. Eng. J. 468, 143837 (2023).
Jiang, Y. et al. Visualization of the key proton activities in hydrogen evolution reaction by electrochromic catalyst. Small 21, 2500631 (2025).
Zhang, Y. et al. Color-neutral smart window enabled by gradient reversible alloy deposition. ACS Energy Lett. 9, 4162–4171 (2024).
Bellchambers, P. et al. Zinc grid based transparent electrodes for organic photovoltaics. Adv. Energy Mater. 2405148 https://doi.org/10.1002/aenm.202405148 (2025).
Ma, G. et al. Reshaping the electrolyte structure and interface chemistry for stable aqueous zinc batteries. Energy Storage Mater. 47, 203–210 (2022).
Xia, C., Guo, J., Li, P., Zhang, X. & Alshareef, H. N. Highly stable aqueous zinc-ion storage using a layered calcium vanadium oxide bronze cathode. Angew. Chem. Int. Ed. 57, 3943–3948 (2018).
Wang, J. et al. Enabling stable Zn anode with PVDF/CNTs nanocomposites protective layer toward high-performance aqueous zinc-ion batteries. Adv. Funct. Mater. 34, 2316083 (2024).
Lee, H., Yanilmaz, M., Toprakci, O., Fu, K. & Zhang, X. A review of recent developments in membrane separators for rechargeable lithium-ion batteries. Energy Environ. Sci. 7, 3857–3886 (2014).
Chen, X. et al. Combining catalysis and separation on a PVDF/Ag composite membrane allows timely separation of products during reaction process. Chem. Eng. J. 295, 518–529 (2016).
Xu, B. et al. Transparent metal oxide interlayer enabling durable and fast-switching zinc anode-based electrochromic devices. Nanoscale 15, 19629–19637 (2023).
Demiri, S., Najdoski, M. & Velevska, J. A simple chemical method for deposition of electrochromic Prussian blue thin films. Mater. Res. Bull. 46, 2484–2488 (2011).
Liu, P. et al. Amorphous tungsten oxide nanodots for chromatic applications. Adv. Funct. Mater. 34, 2400760 (2024).
Ding, Y. et al. Galvanic-driven deposition of large-area Prussian blue films for flexible battery-type electrochromic devices. J. Mater. Chem. A 11, 2868–2875 (2023).
Roy, R., Greeshma, R., Basith, A., Banerjee, R. & Singh, A. K. Self-rechargeable aqueous Zn2+/K+ electrochromic energy storage device via scalable spray-coating integrated with marangoni flow. Energy Storage Mater. 71, 103680 (2024).
Zhang, W., Li, H. & Elezzabi, A. Y. A dual-mode electrochromic platform integrating zinc anode-based and rocking-chair electrochromic devices. Adv. Funct. Mater. 33, 2300155 (2023).
Wu, C. et al. High-performance aqueous Zn2+/Al3+ electrochromic batteries based on niobium tungsten oxides. Adv. Funct. Mater. 33, 2214886 (2023).
Wang, B. et al. Inhibiting vanadium dissolution of potassium vanadate for stable transparent electrochromic displays. Small Sci. 3, 2300046 (2023).
Acknowledgements
This work was supported financially by the National Natural Science Foundation of China (52572186, 52202320), National Natural Science Foundation of China–China Academy of Engineering Physics “NSAF” Joint Fund (U2230101), the National Natural Science Fund for Excellent Young Scientists Fund (Overseas) Program (GG2090007003), Shandong Excellent Young Scientists Fund (Overseas) Program (2023HWYQ-060), the Fundamental Research Funds for the Central Universities (WK2490000002), and the Joint Research Center for Multi-Energy Complementation and Conversion.
Author information
Authors and Affiliations
Contributions
J.C. and G.Z. conceived and planned the experiments. G.Z., M.Z., B.X., and Y.G. contributed to sample preparation, measurements, and formal analysis. J.C., G.Z., T.H., Q.X., Z.C., and W.W. supported the experiment. G.Z. and J.C. wrote the original draft. J.C., Z.C., L.C., W.Y., and S.L. reviewed and edited the manuscript. J.C., L.C., W.Y., and S.L. supervised the project. All authors helped shape the research and revise the manuscript. All authors have read and approved the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhou, G., Zhu, M., Xu, B. et al. Durable and flexible zinc mesh anodes for scalable and fast-switching electrochromic devices. npj Flex Electron 10, 9 (2026). https://doi.org/10.1038/s41528-025-00509-1
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41528-025-00509-1