Abstract
Parkinson’s disease (PD) is a neurodegenerative disorder with complex motor and non-motor symptoms. This network meta-analysis evaluated the combined effects of medication and 20 rehabilitation therapies on motor function, neuro-psychiatric health, and quality of life in 8202 PD patients across 186 randomized controlled trials. Traditional Chinese Rehabilitation Therapy (TCRT), Exoskeleton-Assisted Rehabilitation Therapy (EART), Hydrotherapy Rehabilitation Therapy (HRT), and Conventional Kinesitherapy (CKT) significantly improved balance, while Mind-Body Exercise Therapy (MBET) and Non-Invasive Brain Stimulation Therapy (NIBST) enhanced overall motor capacity and reduced freezing of gait (FOG). Resistance Training Rehabilitation Therapy (RTRT) and Non-Invasive Brain Stimulation Therapy (NIBST) improved cognitive function, and Mind-Body Exercise Therapy (MBET) alleviated negative mood. Upper Limb Rehabilitation Therapy (ULRT) and Resistance Training Rehabilitation Therapy (RTRT) showed notable quality-of-life benefits. However, confidence in outcomes was often low due to risk of bias and imprecision. Meta-regression indicated that intervention duration was negatively correlated with cognitive gains. These findings highlight the need for precise, integrated rehabilitation strategies targeting specific symptoms to optimize PD management. Future research should explore individualized, mechanism-driven approaches to advance precision rehabilitation.
Similar content being viewed by others
Introduction
Parkinson’s disease (PD) is the second most common neurodegenerative disease after Alzheimer’s disease1, and its core pathology is characterized by the progressive loss of dopaminergic neurons in the midbrain substantia nigra and the abnormal aggregation of α-synuclein in Lewy bodies, with a complex pathogenesis and a high degree of heterogeneity in clinical manifestations2,3,4,5. As a multidimensional systemic disease, PD is not only characterized by motor symptoms such as resting tremor, muscle tonus, bradykinesia, and postural instability, but also a wide range of non-motor symptoms such as olfactory hyperalgesia, REM sleep behavioral abnormalities, autonomic dysfunction, and cognitive-emotional deficits6,7,8,9. Clinical interventions should focus on the integrated management of motor function, neuropsychological health, and quality of life.
Motor dysfunction in PD, particularly balance impairment, freezing of gait, and bradykinesia, markedly increases fall risk (annual fall rate of 60% for those with a Tinetti score <19)10. Such deficits substantially compromise patients’ independence, elevate caregiver burden, and diminish overall quality of life11. Rehabilitation strategies, including gait and balance training, can mitigate these effects by enhancing sensorimotor integration and are now considered essential components of PD management12.
Concurrently, neuro-psychiatric symptoms are highly prevalent in PD. Approximately 40% of patients experience statistically significant depression or anxiety (Hospital Anxiety Depression Scale score, HADS ≥11)13, while about 30% exhibit cognitive impairment (Montreal Cognitive Assessment Scale score, MoCA <26)14. These symptoms not only affect patients’ mental health but also further exacerbate motor dysfunction. Cognitive behavioral therapy (CBT) and dual-task training have been shown to improve mood and cognitive levels and play an important role in enhancing the overall health of patients15,16.
Ultimately, both motor and non-motor manifestations converge to impact quality of life—a key outcome in evaluating PD interventions. Multidimensional interventions, such as comprehensive rehabilitation, have been shown to significantly improve patients’ daily participation and subjective well-being (39-item Parkinson’s Disease Quality of Life Questionnaire, PDQ-39 total score reduction)17. These interventions are important in improving the overall health and quality of life of patients.
Motor function, neuro-psychiatric health, and quality of life are core targets for multidimensional interventions in Parkinson’s disease. Although existing rehabilitation approaches have been successful in improving single symptoms, challenges remain in the integrated management of multidimensional symptoms17,18 Current treatment strategies tend to focus on single symptom improvement and lack integrated management of multidimensional symptoms. Therefore, exploring the combined application of medications and rehabilitation therapies to achieve a more comprehensive intervention effect is an important direction in current clinical practice. Modern PD rehabilitation needs to integrate the synergistic effects of drugs and rehabilitation. Drugs provide a neurochemical basis for functional training by modulating basal ganglia dopaminergic transmitter levels, and rehabilitation means to enhance the synaptic plasticity of cortico-striatal loops and slow down disease progression19. This study included 20 drug-combination rehabilitation interventions, including common methods such as physical training, balance training, and gait training, as well as emerging technological tools such as virtual reality, exoskeletons, sensory stimulation movement therapy, and non-invasive brain stimulation, and traditional Chinese rehabilitation methods represented by Tai Chi, Baduanjin, and acupuncture. These methods have shown different degrees of efficacy in improving motor and cognitive functions and quality of life20,21,22. Faced with a spectrum of rehabilitation options for Parkinson’s disease, clinicians lack clear guidance on their comparative benefits, as robust head-to-head trials are scarce. While previous systematic reviews and pairwise meta-analyses have established the efficacy of individual therapies against standard care, a critical knowledge gap remains: the absence of a comprehensive, hierarchical comparison that ranks the diverse range of modern rehabilitation strategies within a unified analytical framework. To bridge this gap, this study employs a novel application of network meta-analysis (NMA). This methodology is uniquely suited to synthesize both direct and indirect evidence, enabling us to estimate the relative efficacy of interventions that have never been directly compared yet all aim to alleviate the core symptoms of PD.
This study is the first systematic integration of a large-scale systematic evaluation of pharmacotherapy combined with 20 rehabilitation therapies on an international scale, including 186 randomized controlled trials (n = 8202). Based on NMA, we conducted a multilevel, all-encompassing comparative quantitative assessment of the efficacy of these 20 interventions in the three core dimensions of motor function, neurological-mental health, and quality of life, with endpoint indicators covering balance, gait freezing, cognitive function, emotional state, and subjective quality of life. The aim of this study was to assess the comprehensive effects of medication combined with 20 rehabilitation therapies on motor function, neuro-psychiatric health, and quality of life in Parkinson’s disease patients through a Network Meta-Analysis (NMA) and to provide more precise clinical guidance for treatment. It provides evidence-based personalized rehabilitation decision support for clinicians and patients to develop more precise intervention prescriptions, which has significant clinical translational value and public health guidance.
Results
Study selection
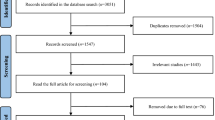
The study selection process is detailed in the PRISMA flowchart (Fig. 1). Initial searches across databases yielded 17,943 potentially eligible records. After removing 11,026 duplicates through automated and manual checking, 6917 unique records remained for title and abstract screening. This screening resulted in 1706 articles for full-text review. After assessing the full texts, 155 articles reporting 186 randomized controlled trials (RCTs) met the pre-defined eligibility criteria and were included in the network meta-analysis.
Literature search flowchart.
Features of the included studies
The 186 included RCTs, detailed in Table S1, were conducted across 20 countries. These trials enrolled a total of 8202 patients with Parkinson’s disease. The mean age, sample size, gender distribution, disease duration, cognitive level, and intervention duration and frequency for each study are reported in Table S1. The experimental interventions evaluated were medication combined with one of 20 rehabilitation therapies: CKT, BTT, GTT, MBET, TCRT, SSMT, XRRT, EART, CMDT, ULRT, HRT, AOT, RTRT, CBRT, ART, OT, NIBST, and H-NIBST. The control interventions were medication combined with CRT or CNT. The mean duration across all intervention arms was 17.78 weeks.
Risk of bias assessment
The risk of bias for the 186 RCTs was assessed using the Cochrane RoB 2.0 tool. Of these, 185 studies reported random sequence generation, 69 reported allocation concealment, and 186 described blinding of outcome assessment. All studies were judged as low risk for selective reporting and other biases. Based on the overall assessment, 63 studies were rated as low risk of bias, 116 as having some concerns, and 7 as high risk. The detailed assessment for each study is presented in Fig. S1.
Network diagram of included studies
Circular nodes represent interventions included in the analysis, and their diameter is proportional to the number of independent studies participating in that intervention. The connecting line between nodes indicates evidence of direct comparison between the two interventions, and the thickness of the edge reflects the number of independent studies directly compared. The experimental group interventions included 18 active intervention strategies, specifically CKT, BTT, GTT, MBET, TCRT, SSMT, XRRT, EART, CMDT, ULRT, HRT, AOT, RTRT, CBRT, ART, OT, NIBST, and H-NIBST; and in the control group, CRT and CNT. The network for the outcome indicators is shown in Fig. 2.
Network diagram of included studies (A is balance function, B is motor ability, C is improvement of gait freezing, D is cognitive function, E is negative emotions, F is QoL).
Network meta-analysis of drugs combined with 20 rehabilitation therapies
This NMA evaluated the comparative efficacy of 20 rehabilitation therapies combined with medication on motor function, neuropsychological health, and quality of life in Parkinson’s disease patients. Prior to interpretation, consistency between direct and indirect evidence was assessed. Global inconsistency tests revealed no significant deviation across outcomes (all p > 0.05), and node-splitting analysis confirmed no local inconsistency (all P > 0.05), supporting the reliability of the network estimates. Detailed results are provided in Table S2. Figure S2 presents the forest plots for pairwise comparisons, with I² values indicating considerable heterogeneity across domains: balance function (I² = 99.8%), motor function (I² = 99.8%), Improvement of gait freezing (I² = 99.7%), cognitive function (I² = 99.1%), negative emotion (I² = 98.9%), and quality of life (I² = 98.6%). Treatment ranking summaries are presented in Table S3 and visualized in Figs. 3 and 4. Data with significant statistical significance for each indicator are presented in Table S4.
Effects of medication combined with rehabilitation therapies on outcomes versus active controls (e.g., CNT), with 95% credible intervals.
(A is balance function, B is motor ability, C is improvement of gait freezing, D is cognitive function, E is negative emotions, F is QoL).
Motor function
The meta-analysis showed that in terms of improving balance function, several interventions demonstrated a significant effect compared to the control group. Specifically, TCRT, HRT, EART, BT, CMDT, and AOT demonstrated statistically significant effects. For Motor ability, MBET and NIBST demonstrated a statistically significant improvement compared to the control. For Improvement of gait freezing, no single intervention demonstrated a statistically significant effect compared to the control. See Fig. 3 for details.
Neuro-psychiatric health
In terms of improving cognitive function, RTRT, NIBST and ART demonstrated statistically significant efficacy compared to the control. CMDT and GTT showed point estimates indicating improvement, though the results were not statistically significant. For the improvement of negative mood, MBET demonstrated statistically significant efficacy. See Fig. 3 for details.
Quality of life
In terms of improving quality of life, ULRT, RTRT and CBRT demonstrated statistically significant improvements compared to the control. Interventions such as OT, BTT, SSMT, XRRT, TCRT, CKT, GTT, and ART suggested potential improvement, though their confidence intervals included zero. See Fig. 3 for details.
Credibility assessment
The credibility of NMA findings was assessed using the CINeMA framework. Overall confidence in the evidence was low to very low across outcomes, primarily due to imprecision and within-study bias (e.g., challenges in blinding and allocation concealment in rehabilitation trials). Although clinical heterogeneity existed (e.g., in disease duration, severity, and age), direct and indirect evidence showed no significant inconsistency. Confidence was low for motor ability, freezing of gait, cognitive function, and negative mood, and very low for balance function and quality of life. CINeMA summaries are presented in Figs. 5, 6.
Summary table of CINeMA (A is balance function, B is motor ability, C is improvement of gait freezing, D is cognitive function, E is negative emotions, F is QoL).
CINeMA net plots (A is balance function, B is motor ability, C is improvement of gait freezing, D is cognitive function, E is negative emotions, F is QoL).
Small-sample effects or publication bias tests
For studies included in the reticulated meta-analysis, small-sample effect estimates and publication bias tests were performed using a corrected comparison funnel plot (comparison-adjusted funnel plot). The included studies were largely symmetrical, indicating that there was no small-sample effect in the current study, and no significant publication bias was found. See Fig. 7.
Comparison-adjusted funnel plot (A is balance function, B is motor ability, C is improvement of gait freezing, D is cognitive function, E is negative emotions, F is QoL).
Sensitivity analysis
We performed a sensitivity analysis to assess the robustness of the pooled effect sizes by iteratively removing individual included studies one at a time. The results demonstrated that the point estimates (Estimate) and the lower and upper limits of the 95% confidence intervals (CI) for the pooled effect sizes remained relatively consistent without significant shifts. Notably, after the removal of any single study, the confidence intervals consistently fluctuated around the overall estimate, and no substantial widening of the interval ranges was observed. This indicates that the results of this meta-analysis possess good robustness, are minimally influenced by any single study, and that the conclusions drawn are highly reliable. Please refer to Fig. S3 and Table S5.
Meta-regression
Meta-regression analyses examined country, intervention duration, and mean participant age as potential moderators (see Table S6). A significant moderating effect of country was found only for improvement in freezing of gait (β = –0.96, 95% CI [–1.63, –0.28], P < 0.05), with studies conducted in Asian countries generally reporting larger effect estimates. For cognitive function, a significant negative association was identified with intervention duration (β = –0.10, 95% CI [–0.19, –0.08], P < 0.05). No other moderators reached statistical significance for any of the examined outcomes (balance function, locomotor ability, quality of life; all P > 0.05).
Discussion
This study conducted a large-scale network meta-analysis (NMA) to estimate the comparative efficacy of medications combined with 20 rehabilitation therapies for Parkinson’s disease (PD), based on 186 randomized controlled trials (RCTs) involving 8,202 patients. It is imperative to interpret the following comparative rankings and effect estimates within the context that the CINeMA framework rated the certainty of evidence for most outcomes as “low” to “very low,” primarily due to risks of bias and imprecision. Given the low confidence rating per CINeMA assessment, this study should be regarded as exploratory. Its findings are intended primarily to generate hypotheses for future research and to offer preliminary insights for clinical consideration, rather than to serve as a definitive basis for clinical decision-making. In practice, any application of these results should involve careful integration with individual patient factors, clinical expertise, and higher-level evidence.
For motor function, the analysis provided the following estimates, all of which are supported by low-certainty evidence. Regarding balance, TCRT, EART, HRT, and BTT were estimated to be significantly better than controls, a finding consistent with prior studies23,24,25,26,27. For overall motor ability, MBET and NIBST showed the largest effect estimates, though the low certainty of evidence precludes definitive conclusions. MBET was associated with an exceptionally large effect size. In the presence of sparse connections and high heterogeneity within the evidence network, the random-effects model may have produced an extreme or inflated effect estimate. Therefore, this substantial effect size may reflect not only a potential clinical benefit but also methodological uncertainty. Concerning freezing of gait, while NIBST and HRT had favorable point estimates, their confidence intervals included zero, indicating that no statistically significant benefit was demonstrated and underscoring the need for more robust evidence.
For neuropsychiatric symptoms, the analysis yielded the following patterns, also derived from low-certainty evidence. In cognitive function, RTRT and NIBST were associated with the greatest estimated improvement, aligning with literature on physical activity and neuromodulation28,29,30,31,32,33. RTRT was associated with a large effect estimate; however, this finding requires cautious interpretation, as it may reflect methodological amplification rather than a true clinical effect. For negative mood, MBET was estimated to be superior, and trends were observed for other interventions, corresponding with known benefits of mind-body practices34,35,36,37.
In terms of quality of life, which is supported by very low‑certainty evidence, ULRT and RTRT showed the largest point estimates for improvement. While this may hint at a possible link between functional gains and perceived well-being38, the extremely wide confidence intervals and substantial heterogeneity urge extreme caution in interpretation. Importantly, interventions that were effective in single domains (e.g., TCRT for balance) did not demonstrate significant overall quality‑of‑life benefits in this analysis, possibly due to the multidimensional nature of the construct or insufficient follow‑up time.
In summary, this network meta‑analysis offers a structured yet highly preliminary ranking of rehabilitation approaches for Parkinson’s disease across key domains. All findings are derived from evidence of low to very low overall certainty. A central limitation is the substantial and pervasive heterogeneity across studies, stemming from variations in intervention delivery (intensity, frequency, duration) and considerable clinical diversity in patient populations (disease stage, symptom severity). Parkinson’s disease itself is inherently heterogeneous, and such variability is reflected in the exceptionally wide confidence intervals observed for many comparisons. It must be explicitly acknowledged that some of the large effect sizes reported (e.g., Hedges’ g > 4) are unusual in clinical rehabilitation research and may reflect methodological artefacts—such as residual scale incompatibility, influential small studies, or imbalanced network structure—rather than true clinical effects of that magnitude. Consequently, the efficacy hierarchy presented here should be interpreted strictly as hypothesis‑generating. Its primary utility lies in identifying candidates for future high‑certainty research, not in guiding clinical decision‑making. Future work should therefore prioritize larger, rigorously designed, and standardized head‑to‑head randomized controlled trials to verify these comparative estimates, alongside mechanistic studies to advance personalized rehabilitation.
Methods
Guided by the PRISMA-NMA statement, Cochrane Handbook for Intervention Reviews and the International Prospective Register of Systematic Reviews (PROSPERO) with registration number CRD420251022813, this network meta-analysis strictly adhered to the pre-registered protocol without any post-hoc modifications. All analytic choices were fully consistent with the pre-registration. No substantive or technical adjustments to the analytic plan were made after data extraction and analysis initiation, ensuring methodological transparency and reproducibility. The PRISMA NMA checklist is provided in Table S7.
Eligibility criteria
This study included randomized clinical trials in people with diagnosed Parkinson’s comparing the effects of medications combined with different rehabilitation therapies in Parkinson’s patients.
Studies were eligible for inclusion if they met the following criteria: (P) Population: People with Parkinson’s Disease; (I) Interventions: Conventional Kinesitherapy, Balance Training Therapy, Gait Training Therapy, Mind-Body Exercise Therapy, Traditional Chinese Rehabilitation Therapy, Sensory Stimulation Motor Therapy, Extended Reality Rehabilitation Therapy, Exoskeleton-Active Rehabilitation Therapy, and other interventions. Therapy, exoskeleton-assisted rehabilitation therapy, cognitive-motor dual-task therapy, upper limb rehabilitation therapy, hydrotherapy rehabilitation therapy, action observation therapy, resistance training rehabilitation therapy, and cognitive behavioral rehabilitation therapy. Art Rehabilitation Therapy, Occupational Therapy, Non-Invasive Brain Stimulation Therapy, Hybrid Non-Invasive Brain Stimulation Therapy. The intervention groups were assigned according to Table S8 and Table S9, with the classification of borderline cases or multi-component interventions determined according to their primary core mechanism; (C) Control Group. Only engaged in normal physical activity; (O) Outcome: Balance indexes (including BBS, Mini-BEST, Brief BEST, Tinetti, Functional reach, OBI, BESS, PIGD, PG), motor function indexes (including UPDRS III and MDS-UPDRS III) and gait freezing improvement (including TUG); cognitive indicators (including MoCA, FAB, PDCRS, MMSE, TAPF, SCOPA-COG, ACE-R); and negative mood indicators (including BDI-II, BAI, PDQ 39, Emotion, HADS, and PDQ) were selected to assess neuropsychological health. Emotion, HADS, GDS, HAMA, HAMD, PDSS, SAS, PSQI; rated quality of life included PDQ-39, PDQ-8, PFS-16, PHQ-9, BBQ, SF-36 (S) Study type: RCTs.
Studies were excluded if they (1) were experimental animal studies, review-type literature, conference reports, case reports, letters, repetitive publications, etc.; (2) had unavailable full text, or essential outcome data could not be extracted; (4) did not report relevant metrics of interest to the study; and (5) had patients with neurologic diseases other than Parkinson’s.
Information sources
This study was systematically searched on PubMed, Web of Science, EBSCO, Embase, Cochrane, and China National Knowledge Infrastructure (CNKI), and the selection of included studies was done independently by 2 researchers (HL and XL), who cross-checked the results and included studies if the results were consistent. Searches were performed in PubMed and Cochrane using terms from MeSH. Searches were performed in Embase using terms in Emtree and in CNKI using subject terms combined with free terms. The reference lists of relevant articles were also manually screened for other studies that might be eligible. The time frame of the search was from the deadline of January 2008 to the present and was limited to human studies published in Chinese or English, and only core journals were included in Chinese.
Study selection and data collection
As an example, the PubMed database was searched using several medical search terms, including “Training” [Mesh], “Exercise Therapy” [Mesh], “Rehabilitation” [Mesh], and “Parkinson Disease, Secondary” [Mesh], to ensure that literature related to exercise, training, rehabilitation, and Parkinson’s disease was retrieved. These search terms allowed for extensive coverage of research related to the field. To further ensure the comprehensiveness and accuracy of the literature, the reference lists of relevant articles were also manually screened for relevant studies that may have been missed.
After the initial literature was obtained, a rigorous screening process was performed on these documents. First, the EndNote software was utilized to automatically perform a check for duplicate literature in order to remove duplicate records in the database that may have arisen due to different search strategies or data sources. Subsequently, duplicates that were not automatically identified during the screening process were further removed by manually reading the titles and abstracts to ensure the uniqueness and representativeness of the screened literature. For the remaining literature, a more rigorous review was conducted. The following types of studies were mainly excluded: (1) studies with control groups that did not meet the inclusion requirements; (2) studies with only a single trial; and (3) studies that did not utilize a drug-combined rehabilitation approach to intervention. In addition, review articles, conference abstracts, animal studies, research protocols, case reports, retrospective studies, and book chapters were excluded because they often lacked sufficient primary data or scientific validity to provide specific analyses and conclusions about intervention effects. These stringent literature screening criteria were used to ensure that the final included studies could provide high-quality evidence to support this study and further enhance the scientific validity and credibility of the study.
Data items
In this study, data extraction and validation followed a rigorous multi-step process. First, a standardized literature feature matrix was established, capturing study characteristics (e.g., first author, year, sample demographics, intervention details). Data were then independently extracted by two researchers using pre-designed standardized forms. Upon completion, the two independent records were compared. Any discrepancies were resolved through consensus discussion or, if needed, arbitration by a third researcher. For all continuous outcomes, the specific measurement instruments and their scoring directions were explicitly recorded. For scales with opposite directions, means were multiplied by -1 for conversion while keeping standard deviations unchanged. Different versions of scales were harmonized using normalization. Based on the Cochrane Risk of Bias tool, two researchers also independently assessed the risk of bias across key domains (e.g., randomization, blinding, outcome reporting). Outcome data (means and standard deviations) were extracted, with missing data handled via the Hozo formula. This comprehensive workflow was implemented to minimize the impact of data extraction errors and scale inconsistencies on the final results.
To ensure methodological rigor in our network meta-analysis, we implemented standardized procedures for handling complex data structures, particularly multi-arm trials. All multi-arm trials were included, and we decomposed them into independent pairwise comparisons using the “split-unit” approach, which preserves the integrity of the original randomization by treating each pairwise comparison within a multi-arm trial as a separate unit while accounting for the shared control group or overlapping samples. This method avoids inflating statistical power due to non-independent data and maintains consistency with the network synthesis framework.
For studies reporting multiple measurement instruments for the same functional domain, we pre-specified a single primary outcome per domain to avoid double-counting and inflated statistical power, following a pre-defined decision rule: the most frequently reported instrument was prioritized —consistent with clinical practice and methodological rationale in high-impact stroke rehabilitation meta-analyses39; if frequencies were equal, the instrument with better established reliability and validity was selected, with specific instruments detailed in Table S10. For trials reporting multiple time points, we followed our PROSPERO registration with a pre-specified binary extraction protocol: (1) pre-intervention: only baseline data collected immediately before intervention initiation; (2) post-intervention: only data from within 1 week of full intervention completion. Intermediate assessments and long-term follow-up data were explicitly excluded to capture direct acute effects, avoid confounding, and ensure consistency.
Risk of bias in individual studies
Based on the Cochrane 5.1 version of the risk of bias assessment tool (which includes seven domains: random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessor, incomplete data outcome, selective reporting, and other bias)40, 2 researchers (HL.XL.) assessed risk of bias (ROB) for all eligible studies. Risk assessment analyses were performed using Review Manager 5.3 (Nordic Cochrane, Denmark), and each area was assessed as unclear, low risk, or high risk. Based on these assessments, the researchers categorized the overall risk of bias for each study as (1) low ROB: there were no domains assessed as high risk, and there may have been domains assessed as unclear but fewer than or equal to one; (2) medium ROB: there was a domain assessed as high risk but no more than one, or there were no high-risk domains but more than one domain assessed as unclear; (3) high ROB: all but one of the above were classified as high risk. Two researchers (XL and HL) assessed the risk of bias for all eligible studies. In case of disagreement, a third researcher (YH) participated in the discussion to reach consensus.
Credibility assessment
This study used the CINeMA tool covering six dimensions to assess the credibility of NMA results, including within-study bias, reporting bias, indirectness, imprecision, heterogeneity, and inconsistency. The comparative evidence for medications combined with 20 rehabilitation therapies versus positive controls was assessed for credibility and was rated as not problematic (no downgrading), somewhat problematic (one level of downgrading), and majorly problematic (two levels of downgrading) based on the severity of bias. Finally, for each outcome indicator, CINeMA categorized the credibility of the evidence as high, moderate, low, or very low.
Statistical analysis
This network meta-analysis (NMA) was performed within a frequentist framework using STATA 17.0 (StataCorp LLC, Texas, USA) with the network package. Random-effects models were fitted to account for the anticipated heterogeneity across studies. When integrating baseline-to-endpoint change values, the correlation between pre- and post-intervention measurements was adjusted using a covariance-based method. The standardized mean difference (SMD) was calculated as the effect size measure, with Hedges’ g applied to correct for small-sample bias. A total of 20 rehabilitation interventions were included to systematically evaluate the multidimensional effects of these therapies, combined with pharmacological treatments, on physical function, neuropsychological status, and quality of life in patients with Parkinson’s disease.
A non-informative prior distribution was set based on a random effects model. Effect estimates were presented as means and 95% confidence intervals (CrI), and the baseline significance level was set at α = 0.05. Local inconsistency was assessed by node-splitting, and global heterogeneity was quantified by the I² parameter. The I² statistic was derived from Cochran’s Q statistic, which is the weighted sum of squared differences between individual study effects and the overall pooled effect. I² values of 25%, 50%, and 75% typically indicate low, moderate, and high heterogeneity, respectively.
Intervention networks were mapped using STATA’s network package, with node area proportional to sample size and connecting line thickness reflecting the number of studies directly compared. Publication bias was assessed by correcting the comparison funnel plots, and the probability of the intervention being the optimal solution was assessed by the area under the cumulative ranked probability curve (SUCRA), with values ranging from 0% (worst) to 100% (optimal). To explore sources of heterogeneity, meta-regression models were used to incorporate both country (categorical variable), mean age of subjects (continuous variable), and intervention duration (weeks, continuous variable), and regression coefficients were visualized in bubble plots, with the bubble area indicating variable weights.
The validity of this network meta-analysis relies on the assumptions of transitivity and consistency. To uphold transitivity, we enforced uniform inclusion criteria: all studies involved patients diagnosed with Parkinson’s disease, evaluated interventions following the “medication plus rehabilitation” paradigm, and used a common control (medication plus clinical nursing therapy). The shared goal of all therapies to improve core PD symptoms justifies their comparability. Potential effect modifiers were explored via meta-regression.
The rationale for grouping specific rehabilitation techniques into the 14 network nodes was based on a hierarchical and pre-specified framework to ensure transitivity and clinical validity. The paramount criterion was a shared core mechanism of action. Interventions operating through a distinct, primary biological or functional pathway were consolidated into a single node. Secondly, interventions within a node were required to demonstrate clinical and methodological homogeneity, meaning they were comparable in their application modality and primary therapeutic goal. Finally, we deliberately preserved distinct nodes for interventions that, despite potential conceptual overlap, represent fundamentally different and clinically meaningful choices. This structured approach ensured that each node represents a unique, coherent, and replicable therapeutic strategy.
For consistency, we statistically evaluated the agreement between direct and indirect evidence. This was done using the node-splitting method for local inconsistency and a global design-by-treatment interaction model.
Data availability
All data are presented in the manuscript and supplementary information. Manuscript-related data are available in https://doi.org/10.6084/m9.figshare.30667034. For any other inquiries, please contact the corresponding author.
References
Hayes, M. T. Parkinson’s disease and Parkinsonism. Am. J. Med. 132, 802–807 (2019).
Chu, Y. et al. Nigrostriatal tau pathology in parkinsonism and Parkinson’s disease. Brain 147, 444–457 (2024).
Pickrell, A. M. & Youle, R. J. The roles of PINK1, parkin, and mitochondrial fidelity in Parkinson’s disease. Neuron 85, 257–273 (2015).
Tinaz, S. Functional connectome in Parkinson’s disease and Parkinsonism. Curr. Neurol. Neurosci. Rep. 21, 24 (2021).
Lin, G., Wang, L., Marcogliese, P. C. & Bellen, H. J. Sphingolipids in the pathogenesis of Parkinson’s disease and Parkinsonism. Trends Endocrinol. Metab. 30, 106–117 (2019).
Jankovic, J. Parkinson’s disease: Clinical features and diagnosis. J. Neurol. Neurosurg. Psychiatry 79, 368–376 (2008).
Tolosa, E., Wenning, G. & Poewe, W. The diagnosis of Parkinson’s disease. Lancet Neurol. 5, 75–86 (2006).
Finsterer, J. Parkinson’s syndrome and Parkinson’s disease in mitochondrial disorders. Mov. Disord. J. Mov. Disord. Soc. 26, 784–791 (2011).
Langston, J. W. The Parkinson’s complex: parkinsonism is just the tip of the iceberg. Ann. Neurol. 59, 591–596 (2006).
Moreau, C., Barton, B. R. & Devos, D. Virtually reducing fall risk in Parkinson disease. Neurology 89, 1762–1763 (2017).
Sherrington, C. et al. Exercise to prevent falls in older adults: an updated systematic review and meta-analysis. Br. J. sports Med. 51, 1750–1758 (2017).
Shen, X., Wong-Yu, I. S. & Mak, M. K. Effects of exercise on falls, balance, and gait ability in Parkinson’s disease: A meta-analysis. Neurorehab. Neural Repair 30, 512–527 (2016).
Marsh, L. Depression and Parkinson’s disease: Current knowledge. Curr. Neurol. Neurosci. Rep. 13, 409 (2013).
Lewis, S. J. G. & Ricciardi, L. Social cognition in Parkinson’s disease. Parkinsonism Relat. Disord. 85, 122–123 (2021).
Egan, S. J., Laidlaw, K. & Starkstein, S. Cognitive behaviour therapy for depression and anxiety in Parkinson’s disease. J. Parkinson’s. Dis. 5, 443–451 (2015).
Johansson, H., Folkerts, A. K., Hammarström, I., Kalbe, E. & Leavy, B. Effects of motor-cognitive training on dual-task performance in people with Parkinson’s disease: a systematic review and meta-analysis. J. Neurol. 270, 2890–2907 (2023).
Haider, A. et al. Translational molecular imaging and drug development in Parkinson’s disease. Mol. Neurodegeneration 18, 11 (2023).
Carroll, C. B., Wyse, R. K. H. & Grosset, D. G. Statins and Parkinson’s: A complex interaction. Mov. Disord. J. Mov. Disord. Soc. 34, 934–935 (2019).
Church, F. C. Treatment options for motor and non-motor symptoms of Parkinson’s disease. Biomolecules 11, 612 (2021).
Tomlinson, C. L. et al. Physiotherapy intervention in Parkinson’s disease: Systematic review and meta-analysis. BMJ (Clin. Res. ed.) 345, e5004 (2012).
Thompson, A. J. & Playford, E. D. Rehabilitation for patients with Parkinson’s disease. Lancet (Lond., Engl.) 357, 410–411 (2001).
van der Marck, M. A. et al. Integrated multidisciplinary care in Parkinson’s disease: a non-randomised, controlled trial (IMPACT). Lancet Neurol. 12, 947–956 (2013).
Koganemaru, S. et al. Task-specific brain reorganization in motor recovery induced by a hybrid-rehabilitation combining training with brain stimulation after stroke. Neurosci. Res. 92, 29–38 (2015).
Nuic, D. et al. Home-based exergaming to treat gait and balance disorders in patients with Parkinson’s disease: A phase II randomized controlled trial. Eur. J. Neurol. 31, e16055 (2024).
Morris, R. et al. Overview of the cholinergic contribution to gait, balance and falls in Parkinson’s disease. Parkinsonism Relat. Disord. 63, 20–30 (2019).
Kwok, J. Y. Y. et al. Managing freezing of gait in Parkinson’s disease: a systematic review and network meta-analysis. J. Neurol. 269, 3310–3324 (2022).
Hvingelby, V. S. et al. Interventions to improve gait in Parkinson’s disease: a systematic review of randomized controlled trials and network meta-analysis. J. Neurol. 269, 4068–4079 (2022).
Petzinger, G. M. et al. Exercise-enhanced neuroplasticity targeting motor and cognitive circuitry in Parkinson’s disease. Lancet Neurol. 12, 716–726 (2013).
Intzandt, B., Beck, E. N. & Silveira, C. R. A. The effects of exercise on cognition and gait in Parkinson’s disease: A scoping review. Neurosci. Biobehav. Rev. 95, 136–169 (2018).
Folkerts, A. K. et al. Can physical exercise be considered as a promising enhancer of global cognition in people with Parkinson’s Disease? Results of a systematic review and meta-analysis. J. Parkinson’s. Dis. 14, S115–S133 (2024).
Sokolov, A. A., Miall, R. C. & Ivry, R. B. The cerebellum: Adaptive prediction for movement and cognition. Trends Cogn. Sci. 21, 313–332 (2017).
Tomporowski, P. D. & Pesce, C. Exercise, sports, and performance arts benefit cognition via a common process. Psychol. Bull. 145, 929–951 (2019).
Piefke, M. et al. Neurofunctional modulation of brain regions by distinct forms of motor cognition and movement features. Hum. Brain Mapp. 30, 432–451 (2009).
Cramer, H., Lauche, R., Langhorst, J. & Dobos, G. Yoga for depression: A systematic review and meta-analysis. Depress. Anxiety 30, 1068–1083 (2013).
van der Kolk, B. A. et al. Yoga as an adjunctive treatment for posttraumatic stress disorder: a randomized controlled trial. J. Clin. psychiatry 75, e559–e565 (2014).
Saeed, S. A., Cunningham, K. & Bloch, R. M. Depression and anxiety disorders: Benefits of exercise, yoga, and meditation. Am. Fam. physician 99, 620–627 (2019).
Goyal, M. et al. Meditation programs for psychological stress and well-being: a systematic review and meta-analysis. JAMA Intern. Med. 174, 357–368 (2014).
Kuhlman, G. D., Flanigan, J. L., Sperling, S. A. & Barrett, M. J. Predictors of health-related quality of life in Parkinson’s disease. Parkinsonism Relat. Disord. 65, 86–90 (2019).
Lin, X., Li, H., Chen, N. & Wu, X. Network meta-analysis of 4 rehabilitation methods with rTMS on upper limb function and daily activities in patients with stroke. Stroke 56, 2644–2657 (2025).
Higgins, J. P. et al. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ (Clin. Res. ed.) 343, d5928 (2011).
Acknowledgements
This study was supported by the Science and Technology Development Fund of Shanghai Pudong New Area (Grant No. KJW2024-Y18) and the Medical Engineering Cross Innovation Special Project of Shanghai Seventh People’s Hospital (Grant No. QYYGZ0202). Shanghai University of Sport “sports and fitness technology” key laboratory of the ministry of education and provincial government joint construction open fund grant.
Author information
Authors and Affiliations
Contributions
X.W. and S.H.: Study conception and design. H.L., X.L., and R.H.: Data collection and analysis. H.L. and X.L.: Drafting the manuscript. X.W. and S.H.: Critical revision of the manuscript. All authors reviewed and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Li, H., Lin, X., Huang, R. et al. Comparative effects of medication combined with twenty rehabilitation therapies: core outcomes in 8202 parkinson’s patients. npj Parkinsons Dis. 12, 52 (2026). https://doi.org/10.1038/s41531-026-01266-2
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41531-026-01266-2