Abstract
We investigated the impact of visual states on basal ganglia oscillatory biomarkers, comparing local field potentials (LFPs) dynamics between Parkinson’s disease (PD) and dystonia and developing a decoding model for state identification. Simultaneous LFPs recordings from the subthalamic nucleus (STN) or globus pallidus internus (GPi), and cortex were obtained from 18 PD and 18 dystonia patients. In the eyes-closed state, theta and alpha power increased in the basal ganglia, with stronger coherence to the central cortex, more pronounced in the STN than in the GPi. Machine learning models identified the eyes-closed state with 88% accuracy for STN and 77% for GPi. The sensorimotor STN and GPi were most informative. The present findings provide proof-of-concept that basal ganglia LFPs can reliably predict a physiological state, highlighting the potential influence of physiological oscillatory activity on pathological bands and its relevance for adaptive stimulation paradigms.
Similar content being viewed by others
Introduction
Movement disorders such as Parkinson’s disease (PD), dystonia cannot always be fully treated by medication, with deep brain stimulation (DBS) offering additional hope for patients1. Conventional DBS systems provide continuous stimulation that is not adjusted in real-time, potentially limiting the therapeutic benefits and causing deleterious side effects2. Studies show that adaptive DBS, where stimulation is adjusted in real-time using feedback signals of the patient’s state—provides key benefits, including reduced side effects and improved symptom control3,4. Most current research focuses on using local field potentials (LFPs) as feedback signals for adaptive DBS5.
Current approaches for adaptive DBS generally operate by setting thresholds for power in a given frequency band, changing stimulation amplitude when power exceeds this preset thresholds6. Common oscillatory biomarkers associated with pathological states include theta (4–8 Hz) activity for dystonia7, alpha (9–12 Hz) activity for tremor and dyskinesia8,9, and beta (13–30 Hz) activity for rigidity and bradykinesia10. However, the same feedback signal amplitudes vary with benign physiological context, such as sleep–wake transitions, vigilance/attention, voluntary movement. Thus, a fixed threshold can misclassify normal state changes as pathology and spuriously trigger stimulation changes11,12.
For the development of future adaptive DBS systems, synchronized video monitoring or wearable sensors will be essential to validate signal changes across a variety of everyday scenarios13. Systematically characterizing how candidate frequency-band features evolve across naturally occurring physiological states is therefore critical and motivates the proof-of-concept approach taken here. Eyes-closed state during rest is a natural physiological state, particularly in patients with dystonia and PD, who often experience fatigue14. It is well known that widespread changes in electroencephalogram (EEG) occur during eyes-closed state, a phenomenon referred to as the Berger effect15. However, the effect of eyes-closed on basal ganglia LFPs is still unknown.
To address these issues, we analyzed LFPs from the subthalamic nucleus (STN) and the globus pallidus internus (GPi) during eyes-open and eyes-closed states. We compared changes in dystonia and PD under different states to investigate how physiological state changes affect potential pathological frequency bands. Oscillatory changes induced through eyes-open and -closed conditions had a significant effect on previously described feedback signals for different movement disorders. Because physiological states can modulate the same oscillations used for control, effective adaptive DBS should incorporate state recognition to discount non-pathological fluctuations and withhold stimulation adjustments when changes are not linked to symptoms. Taken together, our data provide proof-of-concept that integrating physiological state recognition is feasible and motivates future efforts to optimize adaptive DBS for real-life use.
Results
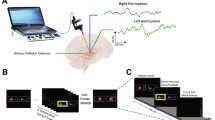
This study involved overall 36 patients undergoing DBS surgery for movement disorders, 18 patients with PD (9 in STN and 9 in GPi) and 18 patients with dystonia (9 in the STN and 9 in the GPi; see detailed demographic information in Tables 1 and 2, respectively). The electrode reconstruction map is illustrated in Fig. 1A. Patients underwent continuous long-term video EEG monitoring during the postoperative external phase (mean duration: 3 ± 2 days), from which recordings were selected for analysis of eyes-open, eyes-closed, and sleep states (Fig. 1B). An example of a time-frequency plot for a subject at the eyes-open and eyes-closed state transit time points is presented in Fig. 1C. We compared LFPs from the STN and GPi, as well as their coupling strength with the cortex across different states. Features were extracted to predict eyes-closed states (Fig. 1D). Finally, we examined the relationship between decoding accuracy and contact locations (Fig. 1E).
A DBS lead location reconstructions show that electrodes were implanted in 18 STN (red) and 18 GPi (blue) targets. B A video-EEG capture system was used to define the eyes-open and eyes-closed states. Six EEG channels (F3, F4; C3, C4; O1, O2) and six basal ganglia recordings (bilateral STN or GPi) were included in the analysis. The green shading highlights the selected signals. C Oscillatory activity in the scalp and basal ganglia was analyzed. In this subject, the time-frequency plots on the left column demonstrate an increase in low-frequency power in the right frontal, central, and posterior scalp regions, as well as the right STN, during the transition from eyes-open to eyes-closed (time point [0] marks eyes-closed time). The right column shows a decrease in low-frequency power in these same regions during the transition from eyes-closed to eyes-open (time point [0] marks eyes-open time). D Machine learning models were developed to classify eyes-open and eyes-closed states. E Decoding accuracy was mapped to the midpoint coordinates of the bipolar contacts on the electrodes. Red dots represent higher decoding accuracy, while blue dots indicate lower accuracy. The highest decoding accuracy was localized to the dorsolateral STN.
STN and GPi oscillations in eyes-closed state
The average power spectral density of the STN and GPi is illustrated in Fig. 2A, C. In the eyes-closed state, theta, alpha, and low-frequency power are significantly higher compared to the eyes-open and sleep states, while the sleep state shows increased power in the delta band (1–4 Hz). Both eyes-open and eyes-closed states exhibit greater power in the beta and higher frequency bands (>30 Hz) than the sleep state. The distribution of low-frequency peaks in the cortex and the basal ganglia is presented in Fig. 2B, D. Notably, the distribution of GPi peaks significantly differs from EEG peaks (P = 0.02, Mann-Whitney U test), whereas no significant difference was found when comparing with the STN peak distribution (P = 0.14, Mann-Whitney U test).
A Power spectra (mean ± S.E.M.) for eyes-open, eyes-closed, and sleep states in the STN (9 PD and 9 dystonia), with red arrow indicating the group-level peaks in eyes-closed state. B Peak frequency histogram for EEG and STN during the eyes-closed state; no significant distribution differences were observed. C Power spectra (mean ± S.E.M.) for eyes-open, eyes-closed, and sleep states in the GPi (9 PD and 9 dystonia), with red arrow indicating the group-level peaks in eyes-closed state. The differences were less pronounced compared to the STN. D Peak frequency histogram for EEG and GPi during the eyes-closed state, showing a significant shift of GPi peak distribution to lower frequencies compared to EEG (P = 0.02, Mann-Whitney U test). E Comparison of summed band power in the STN reveals significant differences in the low-frequency (4–12 Hz) and alpha (9–12 Hz) ranges, with greater low-frequency and alpha power in the eyes-closed state compared to the eyes-open state. Both eyes-open and eyes-closed states exhibit higher beta power compared to the sleep state. F In the GPi, a similar power trend was observed as in the STN, except that both eyes-closed and sleep states had higher theta (4–8 Hz) power compared to the eyes-open state (* PBonferroni < 0.05, ** PBonferroni < 0.01, *** PBonferroni < 0.001, Kruskal–Wallis test).
Band power was compared across the three behavioral states within each nucleus (STN, GPi) using within-subject Friedman tests conducted separately for each frequency band. To control the familywise error rate, p values were Bonferroni-adjusted across the four bands within a nucleus (m = 4). Where the omnibus test was significant, post hoc pairwise state contrasts were assessed with Wilcoxon signed-rank tests, with multiplicity controlled by Bonferroni within band (m = 3). For the STN, alpha and low-frequency power in the eyes-closed state is significantly higher than in the eyes-open state (PBonferroni < 0.01 and PBonferroni < 0.001 respectively, Friedman test). The beta power in both the eyes-open and eyes-closed states is significantly elevated compared to the sleep state (PBonferroni < 0.001, Friedman test) (Fig. 2E). In the GPi, theta power in the eyes-closed and sleep states is significantly higher than in the eyes-open state (PBonferroni < 0.01, Friedman test). Alpha and low-frequency power during the eyes-closed state is significantly higher than in the eyes-open state (PBonferroni < 0.01 and PBonferroni < 0.05 respectively, Friedman test), while the beta power in both the eyes-open and eyes-closed states is significantly higher than that in the sleep state (PBonferroni < 0.001, Friedman test) (Fig. 2F). Accordingly, eyes-open and eyes-closed states are associated with site- and frequency-specific modulations of oscillatory activity.
Oscillatory changes in dystonia and PD patients after state transition
Given the abundance of previous studies highlighting differential biomarkers in dystonia and PD, we included these two conditions for a subgroup analysis. Within each group, the power transition was assessed with Wilcoxon signed-rank tests, with Bonferroni adjustment across the six a priori frequency bands (m = 6). During the transition from the eyes-closed to the eyes-open state, both the dystonia and PD groups exhibited a significant decrease in low-frequency and alpha band power (PBonferroni < 0.01 and PBonferroni < 0.05 respectively, Wilcoxon signed-rank test) (Fig. 3A, C). In the theta band, however, only the PD group demonstrated a significant reduction in power (PBonferroni < 0.01, Wilcoxon signed-rank test). Between-group differences (PD vs dystonia) were tested within each state (eyes-closed, eyes-open) using Mann–Whitney U tests, with Bonferroni correction across the six a priori frequency bands (m = 6) within each state. Notably, while there were no significant differences in theta band power between the PD and dystonia groups during the eyes-closed state, this difference became significant upon transitioning to the eye-open state (PBonferroni < 0.05, Mann–Whitney U test) (Fig. 3B). This finding suggests that theta power may persist in patients with dystonia when their eyes are opened. No significant differences were observed in other frequency bands between the states or the two patient groups (PBonferroni > 0.05, Mann–Whitney U test) (Fig. 3D, F). Therefore, disease-specific modulations of oscillatory activity were identified for the eyes-open and eyes-closed states.
A Low-frequency (4–12 Hz) power significantly decreased in both PD and dystonia patients during the eyes-open state (**PBonferroni < 0.01, Wilcoxon signed rank test). B Theta (4–8 Hz) power significantly decreased in PD but not in dystonia (** PBonferroni < 0.01, Wilcoxon signed rank test). In the eyes-open state, theta power in dystonia patients was significantly higher than in PD patients (*PBonferroni < 0.05, Mann–Whitney U test). C Alpha (9–12 Hz) power significantly decreased in both PD and dystonia patients (* PBonferroni < 0.05, Wilcoxon signed rank test), with a larger standard deviation observed in PD patients during the eyes-closed state (0.041 vs 0.024). D–F No significant differences in power or change rates were found in the beta (13–30 Hz), low beta (13–20 Hz), or high beta (21–30 Hz) ranges between dystonia and PD patients during the eyes-open and eyes-closed states.
Basal ganglia-cortex coherence analysis between eyes-open and eyes-closed
We examined the impact of the eyes-closed state on the oscillatory coupling between the cortex and the basal ganglia, utilizing the iCOH to minimize volume conduction effects16. Across both eye states, STN–cortex iCOH exceeded phase-shuffled surrogates in the theta, alpha, and beta bands (Fig. 4A). Regional topography in the eyes-closed state was assessed for frontal, central, and occipital cortex using Friedman tests conducted separately for each of the three a priori bands (theta, alpha, beta). Where the omnibus test was significant, the three pairwise contrasts (frontal, central, occipital) were evaluated with Wilcoxon signed-rank tests, with multiplicity controlled by Bonferroni within band (m = 3). Under this scheme, theta coherence was higher for central than occipital; in alpha, frontal and central exceeded occipital; and in beta, central exceeded both occipital and frontal (all PBonferroni < 0.05; Fig. 4B). State effects (eyes-closed vs. eyes-open) were tested within each band using Wilcoxon signed-rank tests and Bonferroni-adjusted across the four bands (m = 4): STN low-frequency and alpha coherence were significantly higher during eyes-closed (PBonferroni < 0.01 and PBonferroni < 0.001, respectively; Fig. 4C).
A Averaged STN-cortex coherence during the eyes-open and eyes-closed states compared with phase-shuffled coherence. B In the eyes-closed state, significantly higher coherence was found between the STN and the central region compared to the occipital region in the theta and alpha ranges. In the beta range, both frontal and central regions showed higher coherence with the STN than the occipital region (*PBonferroni < 0.05, Friedman test). C Comparison of summed coherence in the STN revealed significant differences in the low-frequency (4–12 Hz) and alpha (9–12 Hz) ranges, with greater coherence observed during the eyes-closed state compared to the eyes-open state (**PBonferroni < 0.01, *** PBonferroni < 0.001, Mann-Whitney U test). D Averaged GPi-cortex coherence during the eyes-open and eyes-closed states compared with phase-shuffled coherence. E No significant differences were found in GPi coherence with the frontal, central, or occipital regions. F Summed coherence in the GPi showed a significant difference in the alpha (9–12 Hz) range, with greater coherence during the eyes-closed state compared to the eyes-open state (*PBonferroni < 0.05, Mann-Whitney U test).
For the GPi, iCOH similarly exceeded surrogates in theta, alpha, and beta across both states (Fig. 4D). No regional differences among frontal, central, and occipital cortex were detected in the eyes-closed state after within-band Bonferroni adjustment for the three pairwise contrasts (all PBonferroni > 0.05; Fig. 4E). In the state comparison with Bonferroni across the four bands (m = 4), alpha cortico–pallidal coherence was higher during eyes-closed (PBonferroni < 0.05; Fig. 4F). Throughout, STN and GPi were treated as separate a priori families; As such, site- and frequency-specific modulations in oscillatory network activity were identified for the eyes-open and eyes-closed states.
Decoding the eyes-closed state
To examine whether machine learning-based brain signal decoding could distinguish these states for future adaptive DBS algorithms, we investigated the ability to decode eyes-open and eyes-closed states using real-time-compatible data stream features. We computed the patient -and region-wise classification of eyes open vs eyes closed using a five-fold cross-validation (mean ± std training duration per patient: 427.9 ± 210.2 s, see Supplementary Table 1 for further details). For individual channels with the alpha frequency band feature only we obtained a mean patient-wise best balanced accuracy of 0.67 ± 0.09. We observed a significant improvement when all frequency bands in combination were used (balanced accuracy: 0.82 ± 0.11, P < 0.001, Mann-Whitney U test) (Fig. 5A). We did not observe a significant difference in decoding performance between PD and dystonia (alpha PD balanced accuracy: 0.69 ± 0.11, Dys. 0.65 ± 0.08, PBonferroni > 0.05, Mann-Whitney U Test; all frequency bands PD balanced accuracy: 0.82 ± 0.11, Dys. 0.82 ± 0.11, P > 0.05, Mann-Whitney U test) (Fig. 5B). However, we did find a significant difference in decoding accuracy between the STN and GPi (alpha STN: 0.71 ± 0.1, GPi: 0.62 ± 0.06, PBonferroni < 0.001 Mann-Whitney U test; all frequency bands STN: 0.88 ± 0.08, GPi: 0.77 ± 0.11, PBonferroni < 0.001 Mann-Whitney U test) (Fig. 5C). To investigate if non-linear machine learning methods could outperform the ridge-regularized logistic regression model, we compared performances with the gradient-boosted decision tree classification method CatBoost and observed a significant performance increase (balanced accuracy of all frequency bands logistic regression: 0.82 ± 0.11, CatBoost: 0.86 ± 0.11, PBonferroni < 0.001, Monte Carlo Permutation test, n = 5000) (Fig. 5D). The highest linear regression coefficients were obtained for PD in the STN within the alpha frequency band. STN alpha coefficients were higher than STN-GPi coefficients (Fig. 5E). Furthermore, we obtained high performances of the three-class classification including the sleep class in addition to eyes open and closed conditions (balanced accuracy GPI: 0.70 ± 0.12, STN: 0.81 ± 0.07, chance level: 0.33) (Fig. 5F). Accordingly, eyes-open, eyes-closed, and sleep states could be distinguished, with the incorporation of multiple oscillatory features and non-linear classification models improving decoding accuracy.
A Combinations of different frequency band features across the 4–400 Hz range significantly outperformed the use of alpha frequency features alone. B No significant difference in decoding performance was found between Parkinson’s disease and dystonia patients. C STN channels significantly outperformed GPi channels in decoding accuracy (***PBonferroni < 0.001, Mann-Whitney U test). D Non-linear machine learning models (CatBoost) demonstrated better decoding performance compared to linear regression models using all features (***PBonferroni < 0.001, Monte Carlo Permutation test, n = 5000). E Linear regression models indicated that the most contributing frequency bands were within the alpha (8–12 Hz), low-frequency (4–12 Hz), low beta (13–20 Hz), and high beta (20–35 Hz) ranges. F In a three-class classification problem including sleep, eyes-open, and eyes-closed conditions, significant decoding performance was achieved across all targets and disease conditions.
Optimal decoding locations and structural connectivity analysis
We next turned to the spatial specificity of decoding performance. Voxels within the sensorimotor region of the STN were associated with the highest decoding accuracy (peak voxel coordinates X: 13.0, Y: −14.8, Z: −6.0) (Fig. 6A). Additionally, fiber analysis demonstrated that proximity to the hyperdirect pathway was positively associated with decoding accuracy (Fig. 6B). This model was validated through 10-fold cross-validation (sweetspot analysis: R = 0.47, P < 0.001; fiber analysis: R = 0.52, P < 0.001).
A A binarized and thresholded decoding “sweet-spot” was identified in the motor region of the STN, validated through 10-fold cross-validation (R = 0.47, P = 2 × 10⁻⁴). B Fiber tracts analysis showing a significant correlation between R-field magnitude and decoding accuracy, corresponding to the hyperdirect pathway, were validated through 10-fold cross-validation (R = 0.52, P = 2 × 10⁻⁴). C A binarized and thresholded decoding “sweet-spot” was located in the sensorimotor region of the GPi, validated through 10-fold cross-validation (R = 0.58, P = 2 × 10⁻⁴). D Fiber tracts with a significant correlation between R-field magnitude and decoding accuracy, identified as fasciculus fibers connected to the motor region of the STN, were validated through 10-fold cross-validation (R = 0.55, P = 2 × 10⁻⁴). The light-yellow area represents the sensorimotor region of the STN or GPi.
For the GPi, the sensorimotor region was similarly linked to highest decoding accuracy, (peak voxel coordinates at X: 20.9, Y: −8.2, Z: −6.0) (Fig. 6C). Fiber analysis revealed that the subthalamopallidal pathway was related to high decoding accuracy, aligning well with the optimal decoding region in the STN (Fig. 6D). This model was validated through 10-fold cross-validation (sweetspot analysis: R = 0.58, P < 0.001; fiber analysis: R = 0.55, P < 0.001). Therefore, site-specific decoding performance occurred for identification of physiological states.
Discussions
This study provides proof-of-concept evidence that simple physiological activities can substantially modulate basal ganglia oscillatory features, commonly used as candidate feedback signals for adaptive stimulation in movement disorders17,18,19. These findings highlight the risk that adaptive systems relying on fixed thresholds might deliver inappropriate stimulation when confronted with normal physiological state changes. Moreover, we show that movement disorders exhibit distinct LFPs patterns under different physiological conditions. Importantly, the degree of modulation differed across basal ganglia structures: the STN, which is more tightly connected to the cortex, showed stronger effects than the GPi. By integrating multiple features, we achieved more accurate decoding of physiological states, highlighting the potential to incorporate state recognition into future adaptive stimulation algorithms to reduce false triggering.
Our findings demonstrate that eyes-closed, can systematically modulate oscillatory activity within the basal ganglia network, with particularly robust effects in conventionally proposed feedback signals: low-frequency, alpha, and theta. This phenomenon resembles, yet does not fully overlap with, the classical cortical Berger effect15. Interestingly, a previous intraoperative study reported that theta power decreased after eyes-open in patients with PD, rather than the canonical alpha desynchronization observed in cortex20. By expanding the cohort, we confirmed that the principal effect indeed resides in the alpha band, but theta activity in PD also showed significant modulation, though the mechanistic basis remains unresolved. These observations support the notion that top-down cortical inputs can reshape basal ganglia rhythms even at rest. Furthermore, the STN proved more sensitive than the GPi to eyes-closed and exhibited stronger coherence with the cortex, also exhibited similar low-frequency peaks, consistent with its tighter anatomical to cortex. On the other hand, the GPi showed a peak shift to lower frequencies (theta band), which may be related to its indirect connection to the cortex, where lower frequencies tend to preserve21,22. This perspective aligns with previous research on this circuit, which suggests that the STN is more directly influenced by cortical signals, while the GPi processes information modulated by other basal ganglia structures23. Taken together, these findings underscore that eyes-closed should be treated as a critical “background variable” in adaptive DBS algorithms.
Recent investigations into dystonia have increasingly emphasized low-frequency (theta–alpha) activity as a disease-relevant marker24, both in terms of coupling with electromyography and clinical score correlations7,25, and they are suppressed by therapeutic DBS with proportional clinical improvement18,19.One pioneer study already utilized these low-frequency feedback signals for adaptive DBS26. However, it is crucial to recognize that these LFPs feedback signals also have physiological functions which could potentially interfere with the identification of pathological oscillations. For beta oscillations, research has indicated that movements and even festivals like Christmas can affect it11,27. This may explain why previous studies comparing basal ganglia LFPs characteristics between the two disorders have reached inconsistent conclusions28,29,30.
Our study contributes by examining how oscillatory activity varies across disorders during physiological state transitions. At the disease level, theta power in dystonia remained elevated after eyes-open, and differences between PD and dystonia became more apparent in the eyes-open condition. At the site level, the sensorimotor subregions of the STN and GPi exhibited superior decoding accuracy for eye-state classification, consistent with prior evidence that disease-related feedback signals accumulate in these territories7,31. These observations point to a dynamic interaction between physiological and pathological rhythms and reinforce the principle that both disease phenotype and network location shape the expression and susceptibility of oscillatory markers32. Consequently, adaptive DBS strategies should incorporate differential weighting across nuclei, rather than assuming uniform feedback signals validity.
Our data show that eyes-open and eyes-closed modulate low-frequency basal ganglia activity, producing baseline drifts under normal physiology. This exposes a key vulnerability of single-threshold adaptive DBS triggers: band-limited feedback signals can shift with benign physiological events and thereby spuriously trigger or attenuate stimulation12. As an illustrative example distinct from our low-frequency findings, when STN beta band amplitude is used as a trigger, voluntary movement suppresses beta power and can reduce the responsiveness of a closed-loop algorithm precisely when motor demands are highest11. More sophisticated dual-threshold/hysteresis controllers—such as the “ramp” strategy that gradually increases or decreases stimulation when power crosses upper/lower setpoints—improve stability and reduce rapid ON–OFF cycling; in chronic use, patients often preferred the smoother dual-threshold mode at night33,34. Nevertheless, even these improved schemes do not explicitly recognize physiological state.
Building on this, our results indicate that state recognition is both feasible and useful. Compared with a single frequency-band feature, integrating multiband features better identifies sleep stage and supports movement prediction35,36, and in our cohort yielded higher accuracy for decoding eyes-closed with peak performance localized to sensorimotor STN strongly connected to motor cortex. Looking forward, future systems should extend beyond eye-state transitions to encompass a broader set of physiological domains (e.g., sleep–wake cycles, arousal/attentional states, and active motor behavior) and should dynamically calibrate their thresholds or feature weights across both disease phenotypes and anatomical site. We propose a three-dimensional adaptive scheme (disease × site × state): (i) context recognition, (ii) site- and disease-conditioned symptom decoding, and (iii) stimulation decision. Prioritizing context recognition upstream, with downstream decoding explicitly conditioned on disease and target site, is expected to preserve therapeutic efficacy while reducing overstimulation and false triggers arising from normal physiological variability.
This study has three primary limitations. First, although the overall cohort was modest, it represents one of the largest datasets comparing eyes-open and eyes-closed states in PD and dystonia across both STN and GPi. Small subgroup sizes limited independent disease–target comparisons, so we combined data across groups in some analyses to reduce bias. These findings require validation in larger cohorts, and we have therefore de-emphasized disease-specific claims and focused the discussion on implications for adaptive DBS strategies. Second, data were collected in the acute postoperative period and may have been influenced by microlesion effects and short-term medication withdrawal. Our analyses primarily focused on eyes-open versus eyes-closed states and we have reduced claims of direct clinical relevance, longer-term chronic sensing will be necessary to confirm stability and generalizability over days to months. Third, we deliberately employed a simple physiological manipulation (eyes open vs. closed at rest) as a proof-of-concept. While this reductionist design clearly demonstrated state-dependent neural changes, it remains uncertain whether the same principles extend to more complex, behaviorally relevant conditions. Future studies should therefore examine cognitive tasks or varying levels of attention to determine whether state recognition can enhance adaptive DBS in real-world settings. Addressing these questions will be essential for developing more robust and generalizable closed-loop algorithms.
The eyes-closed state enhances low-frequency oscillations in the basal ganglia, with distinct patterns in PD and dystonia. Our findings provide proof-of-concept evidence that physiological activities can modulate candidate biomarkers for adaptive DBS, underscoring the need for state recognition to avoid false triggering and improve clinical reliability.
Methods
Patients and surgery
We recruited 18 idiopathic PD patients and 18 isolated dystonia patients at Beijing Tiantan Hospital. The study was approved by the local ethics committee (KY2022-006-02) and adhered to the standards set by the Declaration of Helsinki. Written informed consent was obtained from all patients before their inclusion in the study. All symptom assessments were performed by an experienced movement disorders specialist as part of the clinical routine. See Tables 1 and 2 for details.
All patients underwent bilateral STN or GPi electrodes implantation. Four-contact PINS L301 (L302) (PINS, Beijing, China) or Medtronic 3389 (3387) (Medtronic, Minneapolis, USA) leads were implanted into the STN or GPi, with a contact length of 1.5 mm and contact spacing of 0.5 mm or 1.5 mm, respectively. The diameter of contacts was 1.27 mm. Electrode contact positions were confirmed through the fused postoperative CT with preoperative MRI. DBS electrodes were visualized using the advanced processing pipeline in Lead-DBS37.
Recordings and eyes-closed state definition
Subjects underwent long-time video EEG recordings during lead externalization using the same protocol as for epilepsy monitoring. We included data segments where eyes-open or eyes-closed was sustained for at least 10 s, from which random samples were taken. The duration of spontaneous eyes blinks averaged between 100–150 ms, which is considered part of the eyes-open state. Eyes-closed state recordings were not selected if they occurred right before sleep, and only segments where eyes-open followed the eyes-closed state were included. After examining the synchronized video, data were selected from periods when patients remained at rest, lying comfortably in bed with eyes-open and eyes-closed states. To confirm that the eyes-closed state was not related to sleep and to compare their differential neural activity patterns, sleep data (Non-Rapid Eye Movement sleep) as confirmed by polysomnography were additionally analyzed as a third condition. At least 5 min of artifact-free data was annotated in each condition. All medication was discontinued on the day prior to recording. Surface EEG was recorded (frontal: F3, F4; central: C3, C4, occipital: O1, O2 by the 10–20 layout). LFPs were recorded in a bipolar configuration from adjacent contacts using a JE-212 amplifier (Nihon Kohden, Tokyo, Japan). The amplification gain was standardized at 195, and the bandpass hardware filter was set between 0.08 and 300 Hz. The signals were digitized at a sampling rate of either 1000 Hz or 2000 Hz.
Power analysis
Offline signal analysis was performed using MNE-Python38, SciPy39, and py_neuromodulation40. Five minutes of artifact-free eyes-open/closed and sleep stage data were selected for analysis. To minimize selection bias, all possible contact pairs were included in this analysis. Signals were down-sampled to a common sampling rate of 1000 Hz, 50 Hz line noise and harmonics were filtered using a finite positive response (FIR) notch-filter, and then a band-pass filter applied between 1 and 100 Hz. Spectral analysis included a total of 108 STN and 108 GPi adjacent contact pairs from 72 DBS electrodes implanted in the 36 patients. Additionally, 216 EEG contact pairs in the same patients were analyzed. All continuous data were segmented into epochs of 1.024 s length (1024 samples). We normalized the epoch data using Z-scoring. Power spectral densities of the epoch data were computed using Welch’s periodogram with a fast Fourier transform method. To align with previous research reports, power was averaged across the following six canonical frequency bands: theta (4–8 Hz), alpha (9–12 Hz), low-frequency (4–12 Hz), low beta (13–20 Hz), high beta (21–30 Hz) and beta (13–30 Hz). We employed the find_peaks algorithm from SciPy17 and visually inspected the spectral peaks in the STN, GPi and EEG for descriptive statistics in the eyes-closed state within the low frequency range. As we matched the number of subjects in each group, we combined the STN and GPi data for comparison between dystonia and PD. For comparison between STN and GPi, we combined the data from both PD and dystonia groups. We compared power across each frequency band. The power change rate was calculated using the formula:
To visualize the low frequency power change at the eyes-open or eyes-closed transit time point in individual patients, we computed the time-frequency representation of the epoched data using the Morlet wavelet transform, with 6 cycles per frequency. For visualization, we applied Gaussian smoothing to the data in the frequency domain of 2 Hz, and in the time domain of 0.25 s.
Coherence analysis
Coherence analysis was conducted between each STN/GPi channel and the ipsilateral EEG to investigate their functional connectivity. Coherence provides a frequency-domain measure of correlation between signals, revealing spectrally specific functional connectivity between neuronal populations. To mitigate volume conduction effects, we utilized the imaginary part of coherence (iCOH)16. For this analysis, spectral information was computed using Welch’s method. The coherence of the original data was compared to that of surrogate data, where the phase alignment of signals was shuffled. In alignment with the power analysis, coherence was compared across six canonical frequency bands after averaging all channel coherence within one hemisphere and subsequently across hemispheres. Additionally, coherence with the frontal, middle, and occipital regions was also compared.
Eyes-closed state decoding
We applied the real-time compatible feature-estimation and decoding toolbox py-neuromodulation40,41. In this toolbox we defined a data-stream with a feature sampling rate of 1 Hz to compute non-overlapping epochs, followed by re-sampling to a common sampling rate of 1 kHz and notch-filtering at the 50 Hz line noise frequency. We defined the epoch window size as 1 s, and in combination with the 1 Hz feature sampling rate, this guaranteed that adjacent epochs did not overlap in the underlying raw data. We then computed FFT features in the following frequency ranges: theta (4–8 Hz), alpha (8–12 Hz), low-frequency activity (4–12 Hz), low beta (13–20 Hz), high beta (20–35 Hz), low gamma (60–80 Hz), high gamma (90–200 Hz), and high-frequency activity (200–400 Hz). We tested for decoding performances of either a ridge-regularized logistic regression model42 (scikit-learn implementation, class-weight balanced, solver: L-BFGS, iterations: 100) or gradient-boosted decision trees using CatBoost43 (max. trees: 500, depth: 6, L2-regularization: 3, loss-function: log-loss) to decode manually annotated eyes-open versus eyes-closed and sleep states within a shuffled and stratified five-fold cross-validation to report model performance, where each fold of samples served as test-set within repeated model train and test splits. To account for class imbalances, we resampled training data to uniform sample sizes. We reported performances by the balanced accuracy metric and investigated linear model coefficients.
Decoding accuracy and contact localization analysis
To explore the correlation between decoding accuracy and the spatial distribution in the STN and GPi, electrode contacts were localized using each patient’s pre-operative MRI and post-operative CT (co-registered in native space). The reconstructed electrode trajectories were then non-linearly normalized to the MNI152 template space, and all contact coordinates are reported in MNI space. Each patient’s decoding accuracy was assigned to the Euclidean midpoint between the contacts of the bipolar pairs. These midpoints were utilized to create pseudo recording-fields (R-fields) for 1 V amplitude, which represent a similar concept of electric-fields (E-fields)36. It is important to note that R-fields from the left hemisphere were nonlinearly mirrored to the right hemisphere, under the assumption of no asymmetric effects. This process resulted in a total of 108 R-fields across the patient cohort. These steps facilitated the visualization and analysis of the simulated R-fields using the Lead-Group toolbox, along with DBS Sweet Spot and Fiber Filtering in Lead-DBS37.
For the sweet-spot analysis, in order to avoid spurious model fitting, a 100 V/m threshold was applied. A voxel was analyzed if it was exposed to the fields above this threshold in at least 20% of stimulations. The Fiber filtering analysis was based on Basal Ganglia Pathway Atlas44. Similar to the approach used in the sweet-spot analysis, each fiber was assigned the maximum recording field magnitude along its trajectory, and only fibers with at least 20% of the R-field with the magnitude above 100 V/m were considered. This approach aimed to address variability in fibers involved across R-fields and to avoid biases and model overfitting arising from voxels/fibers residing only in low magnitude fields.
The sweet-spots and fiber analysis employed Spearman’s correlations. As the sweet-spots and fiber were computed using a mass-univariate design, the robustness and generalizability of the identified tract models were assessed through cross-validation. We implemented ten-fold cross-validation with ten re-shuffles for the R value in fiber filtering analysis. For the sweet-spot analysis, a ten-fold cross-validation was performed to evaluate the model.
Statistical analysis
All statistical analyses were conducted using SciPy39. The nonparametric Friedman, Wilcoxon sign rank, Mann–Whitney U, Kruskal–Wallis, and Monte Carlo permutation tests were used wherever applicable. For correlation analyses, Spearman’s rho was reported to account for potential influence by extreme values. Multiple comparisons were controlled using the Bonferroni correction. P values less than 0.05 were considered statistically significant.
Data availability
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
Code availability
Code is available on Github: (https://github.com/neuromodulation/py_neuromodulation/tree/eyes).
References
Larson, P. S. Deep brain stimulation for movement disorders. Neurotherapeutics 11, 465–474 (2014).
Guidetti, M. et al. Clinical perspectives of adaptive deep brain stimulation. Brain Stimul. 14, 1238–1247 (2021).
An, Q. et al. Adaptive deep brain stimulation for Parkinson’s disease: looking back at the past decade on motor outcomes. J. Neurol. 270, 1371–1387 (2023).
Oehrn, C. R. et al. Chronic adaptive deep brain stimulation versus conventional stimulation in Parkinson’s disease: a blinded randomized feasibility trial. Nat. Med. https://doi.org/10.1038/s41591-024-03196-z (2024).
Priori, A., Foffani, G., Rossi, L. & Marceglia, S. Adaptive deep brain stimulation (aDBS) controlled by local field potential oscillations. Exp. Neurol. 245, 77–86 (2013).
Arlotti, M. et al. Eight-hours adaptive deep brain stimulation in patients with Parkinson disease. Neurology 90, e971–e976 (2018).
Neumann, W. J. et al. A localized pallidal physiomarker in cervical dystonia. Ann. Neurol. 82, 912–924 (2017).
Alonso-Frech, F. et al. Slow oscillatory activity and levodopa-induced dyskinesias in Parkinson’s disease. Brain 129, 1748–1757 (2006).
Foffani, G. & Alegre, M. Brain oscillations and Parkinson disease. Handb. Clin. Neurol. 184, 259–271 (2022).
Thompson, J. A., Lanctin, D., Ince, N. F. & Abosch, A. Clinical implications of local field potentials for understanding and treating movement disorders. Stereotact. Funct. Neurosurg. 92, 251–263 (2014).
Busch, J. L. et al. Single threshold adaptive deep brain stimulation in Parkinson’s disease depends on parameter selection, movement state and controllability of subthalamic beta activity. Brain Stimul. 17, 125–133 (2024).
Gilron, R. E. et al. Sleep-aware adaptive deep brain stimulation control: chronic use at home with dual independent linear discriminate detectors. Front. Neurosci. 15, 732499 (2021).
Gilron, R. E. et al. Long-term wireless streaming of neural recordings for circuit discovery and adaptive stimulation in individuals with Parkinson’s disease. Nat. Biotechnol. 39, 1078–1085 (2021).
Di Vico, I. A. et al. Fatigue in hypokinetic, hyperkinetic, and functional movement disorders. Park. Relat. Disord. 86, 114–123 (2021).
Kirschfeld, K. The physical basis of alpha waves in the electroencephalogram and the origin of the “Berger effect”. Biol. Cybern. 92, 177–185 (2005).
Nolte, G. et al. Identifying true brain interaction from EEG data using the imaginary part of coherency. Clin. Neurophysiol. 115, 2292–2307 (2004).
Neumann, W. J., Gilron, R., Little, S. & Tinkhauser, G. Adaptive deep brain stimulation: from experimental evidence toward practical implementation. Mov. Disord. 38, 937–948 (2023).
Barow, E. et al. Deep brain stimulation suppresses pallidal low frequency activity in patients with phasic dystonic movements. Brain 137, 3012–3024 (2014).
Piña-Fuentes, D. et al. Low-frequency oscillation suppression in dystonia: implications for adaptive deep brain stimulation. Park. Relat. Disord. 79, 105–109 (2020).
Chockalingam, A. et al. Effect of eye opening on single-unit activity and local field potentials in the subthalamic nucleus. Neuromodulation 20, 471–477 (2017).
Moran, L. V. & Hong, L. E. High vs low frequency neural oscillations in schizophrenia. Schizophr. Bull. 37, 659–663 (2011).
Ziaeemehr, A. & Valizadeh, A. Frequency-resolved functional connectivity: role of delay and the strength of connections. Front. neural circuits 15, 608655 (2021).
Oswal, A. et al. Neural signatures of hyperdirect pathway activity in Parkinson’s disease. Nat. Commun. 12, 5185 (2021).
Piña-Fuentes, D. et al. Direct comparison of oscillatory activity in the motor system of Parkinson’s disease and dystonia: a review of the literature and meta-analysis. Clin. Neurophysiol. 130, 917–924 (2019).
Piña-Fuentes, D. et al. The characteristics of pallidal low-frequency and beta bursts could help implementing adaptive brain stimulation in the Parkinsonian and dystonic internal globus pallidus. Neurobiol. Dis. 121, 47–57 (2019).
Piña-Fuentes, D. et al. Toward adaptive deep brain stimulation for dystonia. Neurosurg. Focus 45, E3 (2018).
Feldmann, L. K. et al. Christmas-related reduction in beta activity in Parkinson’s disease. Mov. Disord. 38, 692–697 (2023).
Geng, X. et al. Comparison of oscillatory activity in subthalamic nucleus in Parkinson’s disease and dystonia. Neurobiol. Dis. 98, 100–107 (2017).
Silberstein, P. et al. Patterning of globus pallidus local field potentials differs between Parkinson’s disease and dystonia. Brain 126, 2597–2608 (2003).
Wang, D. D. et al. Subthalamic local field potentials in Parkinson’s disease and isolated dystonia: an evaluation of potential biomarkers. Neurobiol. Dis. 89, 213–222 (2016).
Zaidel, A., Spivak, A., Grieb, B., Bergman, H. & Israel, Z. Subthalamic span of β oscillations predicts deep brain stimulation efficacy for patients with Parkinson’s disease. Brain 133, 2007–2021 (2010).
Horn, A. & Neumann, W.-J. From adaptive deep brain stimulation to adaptive circuit targeting. Nat. Rev. Neurol. 21, 556–566 (2025).
Busch, J. L. et al. Chronic adaptive deep brain stimulation for Parkinson’s disease: clinical outcomes and programming strategies. npj Parkinson’s. Dis. 11, 264 (2025).
Wagle Shukla, A., Bange, M. & Muthuraman, M. Patient, target, device, and program selection for DBS in Parkinson’s disease: advancing toward precision care. npj Parkinson’s. Dis. 11, 195 (2025).
Yin, Z. et al. Generalized sleep decoding with basal ganglia signals in multiple movement disorders. npj Digit. Med. 7, 122 (2024).
Merk, T. et al. Electrocorticography is superior to subthalamic local field potentials for movement decoding in Parkinson’s disease. eLife 11, e75126 (2022).
Neudorfer, C. et al. Lead-DBS v3.0: mapping deep brain stimulation effects to local anatomy and global networks. NeuroImage 268, 119862 (2023).
Gramfort, A. et al. MEG and EEG data analysis with MNE-Python. Front. Neurosci. 7, 267 (2013).
Virtanen, P. et al. SciPy 1.0: fundamental algorithms for scientific computing in Python. Nat. Methods 17, 261–272 (2020).
Merk, T. et al. Invasive neurophysiology and whole brain connectomics for neural decoding in patients with brain implants. Nat. Biomed. Eng. 1–18 (2025). https://doi.org/10.1038/s41551-025-01467-9
Merk, T. et al. py_neuromodulation: signal processing and decoding for neural electrophysiological recordings. J. Open Source Softw. 10, 8258 (2025).
Pedregosa, F. et al. Scikit-learn: machine learning in Python. J. Mach. Learn. Res. 12, 2825–2830 (2011).
Dorogush, A. V., Ershov, V. & Gulin, A. CatBoost: gradient boosting with categorical features support. arXiv preprint. https://doi.org/10.48550/arXiv.1810.11363 (2018).
Petersen, M. V. et al. Holographic reconstruction of axonal pathways in the human brain. Neuron 104, 1056–1064.e1053 (2019).
Acknowledgements
This study was support by National Nature Science Foundation of China (No. 82201634), Yong Elite Scientists Sponsorship Program by BAST(BYESS2023393). W.J.N. received funding from the European Union (ERC, ReinforceBG, project 101077060), Deutsche Forschungsgemeinschaft (DFG, German Research Foundation)—Project-ID 424778381—TRR 295 and the Bundesministerium für Bildung und Forschung (BMBF, project FKZ01GQ1802).
Author information
Authors and Affiliations
Contributions
G.Z.: conceptualization, data curation, formal analysis, investigation, methodology, and writing the original draft; T.M., K.B., and Z.Y.: data curation, formal analysis, and investigation; Y.C.: data curation and formal analysis; N.L.: methodology support; T.B.: writing the original draft; R.M., T.D., Y.L., and H.X.: data curation; L.S., A.Y., and F.M.: supervised and validated the work; A.A.K., J.Z., and W.J.N.: supervised and reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
A.A.K. has served on advisory boards for Medtronic and has received honoraria and travel support from Medtronic, Boston Scientific, Ipsen Pharma, and Teva, unrelated to this work. W.J.N. received honoraria for consulting from InBrain—Neuroelectronics that is a neurotechnology company and honoraria for talks from Medtronic that is a manufacturer of deep brain stimulation devices unrelated to this manuscript. The remaining authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhu, GY., Merk, T., Butenko, K. et al. Decoding the impact of visual states on adaptive deep brain stimulation feedback signals in movement disorders. npj Parkinsons Dis. 12, 61 (2026). https://doi.org/10.1038/s41531-026-01273-3
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41531-026-01273-3