Abstract
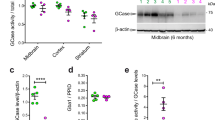
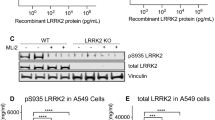
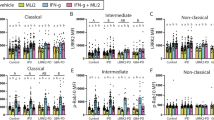
Variants in the LRRK2 and GBA1 genes are among the most common risk factors associated with Parkinson’s disease (PD). Both patients carrying PD-associated variants in GBA1, encoding lysosomal enzyme glucocerebrosidase (GCase), and a subset of non-carrier patients have been shown to have reduced GCase enzymatic activity, suggesting that reduced GCase activity may be a feature of both genetic and a subset of sporadic PD. However, the effect of PD-associated variants in LRRK2, encoding a serine/threonine kinase, on GCase activity remains controversial, with conflicting results in various tissues and cell types. Moreover, rare patients carrying both GBA1 and LRRK2 risk alleles seem to have a more benign disease course than carriers of GBA1 variants alone, suggesting a complex interplay between these two genes in PD. Here, we evaluate the effect of LRRK2 kinase activity on GCase activity in human induced pluripotent stem cell (iPSC)-derived microglia (iMGs), a PD-relevant brain cell type expressing high levels of LRRK2. Using CRISPR editing, isogenic control iPSC lines were generated to match PD patient-derived iPSC lines harbouring the LRRK2 p.G2019S, p.M1646T, or p.N551K-p.R1398H protective haplotype variants. Whereas iMGs harbouring the p.M1646T variant, and the protective haplotype, respectively increased and decreased phosphorylation of canonical LRRK2 substrate, Rab10, GCase protein levels and activity were not altered in any of the LRRK2 variant lines. Additionally, whereas pharmacological inhibition of LRRK2 kinase activity had no impact on GCase activity in iMGs under basal conditions, it attenuated the increase in GCase activity elicited in response to interferon γ (IFNγ) treatment. Moreover, GCase activity induced by IFNγ was reduced in PD risk LRRK2 p.M1646T iMGs and increased in p.N551K-p.R1398H protective haplotype iMGs compared to their isogenic corrected controls, congruent with their respective effects on LRRK2 kinase activity and PD risk. Thus, our data suggest a role for LRRK2 kinase activity in regulation of GCase activity in response to neuroinflammation.
Similar content being viewed by others
Data availability
All data generated or analysed during this study are included in this published article.
References
Kalia, L. V. & Lang, A. E. Parkinson’s disease. Lancet 386, 896–912 (2015).
Senkevich, K., Rudakou, U. & Gan-Or, Z. New therapeutic approaches to Parkinson’s disease targeting GBA, LRRK2 and Parkin. Neuropharmacology 202, 108822–108822 (2022).
Alessi, D. R. & Sammler, E. LRRK2 kinase in Parkinson’s disease. Science 360, 36–37 (2018).
Program, T. G. P. s. G. & Leonard, H. L. Novel Parkinson’s disease genetic risk factors within and across European populations. medRxiv https://doi.org/10.1101/2025.03.14.24319455 (2025).
Ross, O. A. et al. Association of LRRK2 exonic variants with susceptibility to Parkinson’s disease: a case-control study. Lancet Neurol. 10, 898–908 (2011).
Heckman, M. G. et al. Population-specific frequencies for LRRK2 susceptibility variants in the genetic epidemiology of Parkinson’s disease (GEO-PD) consortium. Mov. Disord. 28, 1740–1744 (2013).
Sosero, Y. L. et al. LRRK2 p.M1646T is associated with glucocerebrosidase activity and with Parkinson’s disease. Neurobiol. Aging 103, 142.e141–142.e145 (2021).
Refai, F. S., Ng, S. H. & Tan, E. K. Evaluating LRRK2 genetic variants with unclear pathogenicity. BioMed. Res. Int. 2015, 678701–678701 (2015).
Tan, E. K. et al. Multiple LRRK2 variants modulate risk of Parkinson's disease: a Chinese multicenter study. Hum. Mutat. 31, 561–568 (2010).
Wang, X. et al. Understanding LRRK2 kinase activity in preclinical models and human subjects through quantitative analysis of LRRK2 and pT73 Rab10. Sci. Rep. 11, 1–17 (2021).
Steger, M. et al. Phosphoproteomics reveals that Parkinson’s disease kinase LRRK2 regulates a subset of Rab GTPases. eLife 5, e12813–e12813 (2016).
Steger, M. et al. Systematic proteomic analysis of LRRK2-mediated Rab GTPase phosphorylation establishes a connection to ciliogenesis. eLife 6, e31012–e31012 (2017).
Waschbüsch, D. et al. Structural basis for Rab8a recruitment of RILPL2 via LRRK2 phosphorylation of Switch 2. Structure 28, 406–417.e406 (2020).
Herbst, S. et al. LRRK2 activation controls the repair of damaged endomembranes in macrophages. EMBO J. 39, https://doi.org/10.15252/embj.2020104494 (2020).
Eguchi, T. et al. LRRK2 and its substrate Rab GTPases are sequentially targeted onto stressed lysosomes and maintain their homeostasis. Proc. Natl. Acad. Sci. USA 115, E9115–E9124 (2018).
Eguchi, T. et al. The V-ATPase–ATG16L1 axis recruits LRRK2 to facilitate the lysosomal stress response. J. Cell Biol. 223, https://doi.org/10.1083/JCB.202302067 (2024).
Henry, A. G. et al. Pathogenic LRRK2 mutations, through increased kinase activity, produce enlarged lysosomes with reduced degradative capacity and increase ATP13A2 expression. Hum. Mol. Genet. 24, 6013–6028 (2015).
Yadavalli, N. & Ferguson, S. M. LRRK2 suppresses lysosome degradative activity in macrophages and microglia through MiT-TFE transcription factor inhibition. Proc. Natl. Acad. Sci. USA 120, e2303789120–e2303789120 (2023).
Madureira, M., Connor-Robson, N. & Wade-Martins, R. “LRRK2: autophagy and lysosomal activity”. Front. Neurosci. 14, https://doi.org/10.3389/FNINS.2020.00498 (2020).
Kuwahara, T. & Iwatsubo, T. The emerging functions of LRRK2 and Rab GTPases in the endolysosomal system. Front. Neurosci. 14, 528270–528270 (2020).
Cook, D. A. et al. LRRK2 levels in immune cells are increased in Parkinson’s disease. npj Parkinson Dis. 3, 1–12 (2017).
Gardet, A. et al. LRRK2 is involved in the IFN-γ Response and Host Response to Pathogens. J. Immunol. 185, 5577–5585 (2010).
Moehle, M. S. et al. LRRK2 inhibition attenuates microglial inflammatory responses. J. Neurosci. Off. J. Soc. Neurosci. 32, 1602–1611 (2012).
Ahmadi Rastegar, D. et al. Effect of LRRK2 protein and activity on stimulated cytokines in human monocytes and macrophages. npj Parkinson Dis. 8, 1–11 (2022).
Panagiotakopoulou, V. et al. Interferon-γ signaling synergizes with LRRK2 in neurons and microglia derived from human induced pluripotent stem cells. Nat. Commun. 11, 5163–5163 (2020).
Deretic, V. Autophagy in inflammation, infection, and immunometabolism. Immunity 54, 437–437 (2021).
Cao, M., Luo, X., Wu, K. & He, X. Targeting lysosomes in human disease: from basic research to clinical applications. Signal Transduct. Target. Ther. 6, 1–28 (2021).
Langston, R. G. et al. Association of a common genetic variant with Parkinson’s disease is mediated by microglia. Sci. Transl. Med. 14, eabp8869-eabp8869 (2022).
Sidransky, E. et al. Multicenter analysis of glucocerebrosidase mutations in Parkinson’s disease. N. Engl. J. Med. 361, 1651–1661 (2009).
Mitsui, J. et al. Mutations for Gaucher disease confer high susceptibility to Parkinson's disease. Arch. Neurol. 66, 571–576 (2009).
Nalls, M. A. et al. Identification of novel risk loci, causal insights, and heritable risk for Parkinson’s disease: a meta-genome wide association study. Lancet Neurol. 18, 1091–1091 (2019).
Gan-Or, Z. et al. Differential effects of severe vs mild GBA mutations on Parkinson's disease. Neurology 84, 880–887 (2015).
Alcalay, R. N. et al. Glucocerebrosidase activity in Parkinson’s disease with and without GBA mutations. https://doi.org/10.1093/brain/awv179 (2015).
Gegg, M. E. et al. Glucocerebrosidase deficiency in substantia nigra of parkinson disease brains. Ann. Neurol. 72, 455–463 (2012).
Atashrazm, F. et al. Reduced glucocerebrosidase activity in monocytes from patients with Parkinson’s disease. Sci. Rep. 8, 1–12 (2018).
Hughes, L. P. et al. Glucocerebrosidase activity is reduced in cryopreserved parkinson’s disease patient monocytes and inversely correlates with motor severity. J. Parkinson Dis. 11, 1157–1165 (2021).
Ysselstein, D. et al. LRRK2 kinase activity regulates lysosomal glucocerebrosidase in neurons derived from Parkinson’s disease patients. Nat. Commun. 10, 5570–5570 (2019).
Chedid, J. et al. Dopamine and cortical neurons with different Parkinsonian mutations show variation in lysosomal and mitochondrial dysfunction. npj Parkinson Dis. 11, 177–177 (2025).
Ferrazza, R. et al. LRRK2 deficiency impacts ceramide metabolism in brain. Biochem. Biophys. Res. Commun. 478, 1141–1146 (2016).
Sanyal, A. et al. Lysosome and inflammatory defects in GBA1-mutant astrocytes are normalized by LRRK2 iInhibition. Mov. Disord. 35, 760–773 (2020).
Serebryany-Piavsky, V. et al. The modifying effect of mutant LRRK2 on mutant GBA1-associated Parkinson disease. Hum. Mol. Genet. 34, 1184–1203 (2025).
Wallings, R. L. et al. WHOPPA enables parallel assessment of leucine-rich repeat kinase 2 and glucocerebrosidase enzymatic activity in Parkinson’s disease monocytes. Front. Cell. Neurosci. 16, https://doi.org/10.3389/FNCEL.2022.892899/FULL (2022).
Kedariti, M. et al. LRRK2 kinase activity regulates GCase level and enzymatic activity differently depending on cell type in Parkinson’s disease. npj Parkinson Dis. 8, 1–14 (2022).
Omer, N. et al. A possible modifying effect of the G2019S mutation in the LRRK2 gene on GBA Parkinson’s disease. Mov. Disord. 35, 1249–1253 (2020).
Ortega, R. A. et al. Association of dual LRRK2 G2019S and GBA variations with Parkinson disease progression. JAMA Netw. Open 4, e215845–e215845 (2021).
Yahalom, G. et al. Carriers of both GBA and LRRK2 mutations, compared to carriers of either, in Parkinson’s disease: risk estimates and genotype-phenotype correlations. Parkinsonism Relat. Disord. 62, 179–184 (2019).
Kluss, J. H., Lewis, P. A. & Greggio, E. Leucine-rich repeat kinase 2 (LRRK2): an update on the potential therapeutic target for Parkinson’s disease. Expert Opin. Ther. Targets 26, 537–546 (2022).
Menozzi, E., Toffoli, M. & Schapira, A. H. V. Targeting the GBA1 pathway to slow Parkinson disease: insights into clinical aspects, pathogenic mechanisms and new therapeutic avenues. Pharmacol. Ther. 246, 108419–108419 (2023).
Chen, C. X. Q. et al. A multistep workflow to evaluate newly generated iPSCs and their ability to generate different cell types. Methods Protoc. 4, 50–50 (2021).
McQuade, A. et al. Development and validation of a simplified method to generate human microglia from pluripotent stem cells. Mol. Neurodegeneration 13, 67–67 (2018).
West, A. B. et al. Parkinson’s disease-associated mutations in leucine-rich repeat kinase 2 augment kinase activity. Proc. Natl. Acad. Sci. USA 102, 16842–16847 (2005).
Greggio, E. et al. The Parkinson’s disease kinase LRRK2 autophosphorylates its GTPase domain at multiple sites. Biochem. Biophys. Res. Commun. 389, 449–454 (2009).
Kalogeropulou, A. F. et al. Impact of 100 LRRK2 variants linked to Parkinson’s disease on kinase activity and microtubule binding. Biochem. J. 479, 1759–1783 (2022).
Kluss, J. H. et al. Detection of endogenous S1292 LRRK2 autophosphorylation in mouse tissue as a readout for kinase activity. npj Parkinson Dis. 4, 13–13 (2018).
Iannotta, L. et al. Divergent effects of G2019S and R1441C LRRK2 mutations on LRRK2 and Rab10 Phosphorylations in Mouse Tissues. Cells 9, 2344–2344 (2020).
Fan, Y. et al. R1441G but not G2019S mutation enhances LRRK2 mediated Rab10 phosphorylation in human peripheral blood neutrophils. Acta Neuropathol. 142, 475–494 (2021).
Hui, K. Y. et al. Functional variants in the LRRK2 gene confer shared effects on risk for Crohn’s disease and Parkinson’s disease. Sci. Transl. Med. 10, 7795–7795 (2018).
Nixon-Abell, J. et al. Protective LRRK2 R1398H variant enhances GTPase and Wnt signaling activity. Front. Mol. Neurosci. 9, 179717–179717 (2016).
O’Hara, D. M., Pawar, G., Kalia, S. K. & Kalia, L. V. LRRK2 and α-synuclein: distinct or synergistic players in Parkinson’s disease? Front. Neurosci. 14, 541220–541220 (2020).
Mazzulli, J. R. et al. Gaucher disease glucocerebrosidase and α-synuclein form a bidirectional pathogenic loop in synucleinopathies. Cell 146, 37–52 (2011).
Muñoz, S. S., Petersen, D., Marlet, F. R., Kücükköse, E. & Galvagnion, C. The interplay between Glucocerebrosidase, α-synuclein and lipids in human models of Parkinson’s disease. Biophys. Chem. 273, 106534–106534 (2021).
Herzig, M. C. et al. LRRK2 protein levels are determined by kinase function and are crucial for kidney and lung homeostasis in mice. Hum. Mol. Genet. 20 https://doi.org/10.1093/hmg/ddr348 (2011).
Baptista, M. A. S. et al. Loss of leucine-rich repeat kinase 2 (LRRK2) in rats leads to progressive abnormal phenotypes in peripheral organs. PLoS ONE 8, https://doi.org/10.1371/journal.pone.0080705 (2013).
Mogi, M., Kondo, T., Mizuno, Y. & Nagatsu, T. p53 protein, interferon-γ, and NF-κB levels are elevated in the Parkinsonian brain. Neurosci. Lett. 414, 94–97 (2007).
Mogi, M. et al. Interleukin (IL)-1β, IL-2, IL-4, IL-6 and transforming growth factor-α levels are elevated in ventricular cerebrospinal fluid in juvenile Parkinsonism and Parkinson’s disease. Neurosci. Lett. 211, 13–16 (1996).
Brodacki, B. et al. Serum interleukin (IL-2, IL-10, IL-6, IL-4), TNFα, and INFγ concentrations are elevated in patients with atypical and idiopathic Parkinsonism. Neurosci. Lett. 441, 158–162 (2008).
Rentzos, M. et al. Circulating interleukin-10 and interleukin-12 in Parkinson’s disease - PubMed. Acta Neurol. Scandinavica 119, https://doi.org/10.1111/j.1600-0404.2008.01103.x (2009).
Lee, H. W., Choi, J. & Suk, K. Increases of pentraxin 3 plasma levels in patients with Parkinson’s disease. Mov. Disord. 26, 2364–2370 (2011).
Tang, P. et al. Correlation between Serum RANTES levels and the severity of Parkinson’s disease. Oxid. Med. Cell. Longev. 2014, 208408–208408 (2014).
Zhang, X. et al. Association of IL-16 gene polymorphisms with sporadic Parkinson’s disease in a Han Chinese population. Neurosci. Lett. 724, https://doi.org/10.1016/j.neulet.2020.134877 (2020).
Xu, X. et al. Interleukin-18 promoter polymorphisms and risk of Parkinson’s disease in a Han Chinese population. Brain Res. 1381, https://doi.org/10.1016/j.brainres.2011.01.025 (2011).
Aguirre, C. A. et al. Two single nucleotide polymorphisms in IL13 and IL13RA1 from individuals with idiopathic Parkinson’s disease increase cellular susceptibility to oxidative stress. Brain Behav. Immun. 88, https://doi.org/10.1016/j.bbi.2020.04.007 (2020).
Mogi, M. et al. Tumor necrosis factor-α (TNF-α) increases both in the brain and in the cerebrospinal fluid from parkinsonian patients. Neurosci. Lett. 165, 208–210 (1994).
Wu, M. et al. Immunomodulators targeting MARCO expression improve resistance to postinfluenza bacterial pneumonia. Am. J. Physiol. Lung Cell. Mol. Physiol. 313, L138–L153 (2017).
Singh, N. et al. Antimycobacterial effect of IFNG (interferon gamma)-induced autophagy depends on HMOX1 (heme oxygenase 1)-mediated increase in intracellular calcium levels and modulation of PPP3/calcineurin-TFEB (transcription factor EB) axis. Autophagy 14, 972–991 (2018).
Lieberman, R. L. et al. Structure of acid β-glucosidase with pharmacological chaperone provides insight into Gaucher disease. Nat. Chem. Biol. 3, 101–107 (2007).
Offman, M. N., Krol, M., Silman, I., Sussman, J. L. & Futerman, A. H. Molecular basis of reduced glucosylceramidase activity in the most common gaucher disease mutant, N370S. J. Biol. Chem. 285, 42105–42114 (2010).
Karatas, M. et al. Enzyme kinetics and inhibition parameters of human leukocyte glucosylceramidase. Heliyon 6, e05191–e05191 (2020).
Quick, J. D. et al. Lysosomal acidification dysfunction in microglia: an emerging pathogenic mechanism of neuroinflammation and neurodegeneration. J. Neuroinflammation 20, 1–13 (2023).
Alcalay, R. N. et al. Glucocerebrosidase activity in Parkinson’s disease with and without GBA mutations. Brain 138, 2648–2658 (2015).
Ohtonen, S. et al. Human iPSC-derived microglia carrying the LRRK2-G2019S mutation show a Parkinson’s disease related transcriptional profile and function. Sci. Rep. 13, 1–16 (2023).
Deniston, C. K. et al. Structure of LRRK2 in Parkinson’s disease and model for microtubule interaction. Nature 588, 344–349 (2020).
Vides, E. G. et al. A feed-forward pathway drives LRRK2 kinase membrane recruitment and activation. eLife 11, https://doi.org/10.7554/ELIFE.79771 (2022).
Dhekne, H. S. et al. Genome-wide screen reveals Rab12 GTPase as a critical activator of Parkinson’s disease-linked LRRK2 kinase. eLife 12, https://doi.org/10.7554/ELIFE.87098 (2023).
Irazoqui, J. E. Key roles of MiT transcription factors in innate immunity and inflammation. Trends Immunol. 41, 157–171 (2020).
Kuwahara, T. et al. LRRK2 and RAB7L1 coordinately regulate axonal morphology and lysosome integrity in diverse cellular contexts. Sci. Rep. 6, 1–12 (2016).
Gosselin, D. et al. An environment-dependent transcriptional network specifies human microglia identity. Science 356, 1248–1259 (2017).
Ginhoux, F. et al. Fate mapping analysis reveals that adult microglia derive from primitive macrophages. Science 330, 841–845 (2010).
Kierdorf, K. et al. Microglia emerge from erythromyeloid precursors via Pu.1- and Irf8-dependent pathways. Nat. Neurosci. 16, 273–280 (2013).
Wallings, R. L., Herrick, M. K. & Tansey, M. G. Frontiers | LRRK2 at the interface between peripheral and central immune function in Parkinson’s. Front. Neurosci. 14, https://doi.org/10.3389/fnins.2020.00443 (2020).
Tansey, M. G. et al. Inflammation and immune dysfunction in Parkinson disease. Nat. Rev. Immunol. 22, 657–673 (2022).
Boddupalli, C. S. et al. Neuroinflammation in neuronopathic Gaucher disease: role of microglia and NK cells, biomarkers, and response to substrate reduction therapy. eLife 11, https://doi.org/10.7554/ELIFE.79830 (2022).
Wang, A. et al. Innate immune sensing of lysosomal dysfunction drives multiple lysosomal storage disorders. Nat. Cell Biol. 26, 219–234 (2024).
Acknowledgements
Thanks to Wolfgang Reintsch and Julien Sirois for training and support with high content imaging and flow cytometry experiments, respectively. Thanks to Marie-France Dorion for training and advice on differentiation and characterization of iPSC-derived microglia. Thanks to Jace Jones-Tabah, Roxanne Larivière, and Andrea Krahn for establishing and optimizing live-cell GCase, lysosomal proteolytic, and pH assays. E.J.M. has been supported by a Fonds de Recherche du Québec-Santé Doctoral Fellowship and Jeanne Timmins Costello Fellowship awarded by the Integrated program in Neuroscience at McGill University. This work was funded by grants from The Michael J. Fox Foundation for Parkinson’s Research (MJFF-019045) and from the Canadian Institutes of Health Research (FDN-154301). G-Can (GBA1 Canada) Initiative, an open-science collaborative initiative aimed at addressing GBA1 associated neurodegeneration, has contributed to this research. G-Can is supported by The Hilary & Galen Weston Foundation, J. Sebastian van Berkom, Ghislaine Saucier and Silverstein Foundation. E.A.F. is supported by a Canada Research Chair (Tier 1) in Parkinson’s disease.
Author information
Authors and Affiliations
Contributions
E.J.M. generated p.M1646T and protective haplotype isogenic correction cell lines, differentiated iMGs, designed and performed experiments, analysed data, and prepared the manuscript. C.X-.Q.C. and N.A. performed quality control experiments on iPSC lines used in this study. E.D. designed CRISPR editing strategies and ddPCR screening strategies to generate isogenic correction lines, and generated the p.G2019S isogenic correction line. Z.Y. designed CRISPR KO strategy and generated the LKO line. D.K. performed xMAP experiments and analysed data. T.M.D., K.S., and Z.G-.O. designed experiments. E.A.F. designed experiments, supervised the project, and prepared the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
MacDougall, E.J., Chen, C.XQ., Deneault, E. et al. LRRK2 kinase mediates increased GCase activity in microglia in response to IFNγ-induced proinflammatory stimulation. npj Parkinsons Dis. (2026). https://doi.org/10.1038/s41531-026-01310-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41531-026-01310-1