Abstract
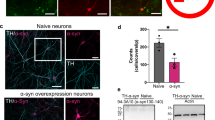
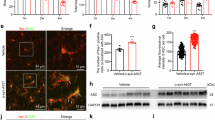
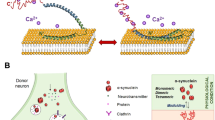
Homozygous (Q789X) DNAJC6 mutation causes PARK19. Q787 of Dnajc6 corresponds to Q789 of DNAJC6. Dnajc6Q787X/Q787X mouse was utilized to elucidate pathomechanisms underlying (Q789X) DNAJC6-induced PARK19. Dnajc6Q787X/Q787X mice displayed PARK19 motor deficits and degeneration of substantia nigra (SN) dopaminergic neurons. (Q787X) Dnajc6 decreased clathrin heavy chain and lysosomal number, leading to downregulation of lysosomal cathepsin D and upregulation of α-synuclein or α-synuclein oligomers in SN dopaminergic neurons. Lysosomal biogenesis activator rapamycin precluded (Q787X) Dnajc6-induced downregulation of cathepsin D, upregulation of α-synuclein, and PARK19 phenotypes. (Q787X) Dnajc6-induced elevation of ER and mitochondrial α-synuclein excited ER stress and mitochondrial pro-apoptotic cascades. (Q787X) Dnajc6-evoked α-synuclein oligomer overexpression activated SN microglia and NLRP3 inflammasome and upregulated IL-1β, IL-18, and TNF-α, which stimulated MKK4-JNK -c-Jun/ATF-2 and RIPK1-RIPK3-MLKL death cascades. Our results suggest that PARK19 (Q789X) DNAJC6 mutation causes lysosomal deficiency and impairs cathepsin D-mediated degradation of α-synuclein, resulting in upregulated α-synuclein-induced neurodegeneration of SN dopaminergic cells.
Similar content being viewed by others
Data availability
All data are available within the published article.
References
Morris, H. R., Spillantini, M. G., Sue, C. M. & Williams-Gray, C. H. The pathogenesis of Parkinson’s disease. Lancet 403, 293–304 (2024).
Coukos, R. & Krainc, D. Key genes and convergent pathogenic mechanisms in Parkinson disease. Nat. Rev. Neurosci. 25, 393–413 (2024).
Trevisan, L. et al. Genetics in Parkinson’s disease, state-of-the-art and future perspectives. Br. Med. Bull. 149, 60–71 (2024).
Edvardson, S. et al. A deleterious mutation in DNAJC6 encoding the neuronal-specific clathrin-uncoating co-chaperone auxilin, is associated with juvenile Parkinsonism. PLoS ONE 7, e36458 (2012).
Olgiati, S. et al. DNAJC6 mutations associated with early-onset Parkinson’s disease. Ann. Neurol. 79, 244–256 (2016).
Ng, J. et al. DNAJC6 mutations disrupt dopamine homeostasis in juvenile Parkinsonism-dystonia. Mov. Disord. 35, 1357–1368 (2020).
Koroglu, C., Baysal, L., Cetinkaya, M., Karasoy, H. & Tolun, A. DNAJC6 is responsible for juvenile Parkinsonism with phenotypic variability. Parkinsonism Relat. Disord. 19, 320–324 (2013).
Elsayed, L. E. O. et al. A novel nonsense mutation in DNAJC6 expands the phenotype of autosomal-recessive juvenile-onset Parkinson’s disease. Ann. Neurol. 79, 335–337 (2016).
Kampinga, H. H. & Craig, E. A. The HSP70 chaperone machinery: J proteins as drivers of functional specificity. Nat. Rev. Mol. Cell Biol. 11, 579–592 (2010).
Rosenzweig, R., Nillegoda, N. B., Mayer, M. P. & Bukau, B. The Hsp70 chaperone network. Nat. Rev. Mol. Cell Biol. 20, 665–680 (2019).
Jiang, J. et al. Structure-function analysis of the auxilin J-domain reveals an extended Hsc70 interaction interface. Biochemistry 42, 5748–5753 (2003).
Chiu, C. C. et al. Downregulation of protease cathepsin D and upregulation of pathologic α-synuclein mediate paucity of DNAJC6-induced degeneration of dopaminergic neurons. Int. J. Mol. Sci. 25, 6711 (2024).
Leak, R. K. et al. Current insights and assumptions on α-synuclein in Lewy body disease. Acta Neuropathol. 148, 18 (2024).
Björklund, A. & Mattsson, B. The AAV-α-synuclein model of Parkinson’s disease: an update. J. Parkinsons Dis. 14, 1077–1094 (2024).
Grundemann, J., Schlaudraff, F., Haeckel, O. & Liss, B. Elevated alpha-synuclein mRNA levels in individual UV-laser-microdissected dopaminergic substantia nigra neurons in idiopathic Parkinson’s disease. Nucleic Acids Res. 36, e38 (2008).
Gündner, A. L. et al. Path mediation analysis reveals GBA impacts Lewy body disease status by increasing α-synuclein levels. Neurobiol. Dis. 121, 205–213 (2019).
Fan, L. et al. CHCHD2 p.Thr61Ile knock-in mice exhibit motor defects and neuropathological features of Parkinson’s disease. Brain Pathol. 33, e13124 (2023).
Besemer, A. S. et al. Receptor-mediated endocytosis 8 (RME-8)/DNAJC13 is a novel positive modulator of autophagy and stabilizes cellular protein homeostasis. Cell Mol. Life Sci. 78, 645–660 (2021).
Volpicelli-Daley, L. A. et al. G2019S-LRRK2 expression augments α-synuclein sequestration into inclusions in neurons. J. Neurosci. 36, 7415–7427 (2016).
Fujii, T. et al. Parkinson’s disease-associated ATP13A2/PARK9 functions as a lysosomal H+,K+-ATPase. Nat. Commun. 14, 2174 (2023).
Skou, L. D., Johansen, S. K., Okarmus, J. & Meyer, M. Pathogenesis of DJ-1/PARK7-mediated Parkinson’s disease. Cells 13, 296 (2024).
Hoepken, H. H. et al. Parkinson patient fibroblasts show increased alpha-synuclein expression. Exp. Neurol. 212, 307–313 (2008).
Bellini, G. et al. α-synuclein in Parkinson’s disease: From bench to bedside. Med. Res. Rev. 45, 909–946 (2025).
Park, H., Kam, T. I., Dawson, V. L. & Dawson, T. M. α-Synuclein pathology as a target in neurodegenerative diseases. Nat. Rev. Neurol. 21, 32–47 (2025).
Nixon, R. A. & Rubinsztein, D. C. Mechanisms of autophagy-lysosome dysfunction in neurodegenerative diseases. Nat. Rev. Mol. Cell Biol. 25, 926–946 (2024).
Jiao, F., Meng, L., Du, K. & Li, X. The autophagy-lysosome pathway: a potential target in the chemical and gene therapeutic strategies for Parkinson’s disease. Neural Regen. Res. 20, 139–158 (2025).
Henn, D., Yang, X. & Li, M. Lysosomal quality control. Autophagy 21, 1413–1432 (2025).
Nanayakkara, R. et al. Autophagic lysosome reformation in health and disease. Autophagy 19, 1378–1395 (2023).
Chen, Y. & Yu, L. Development of research into autophagic lysosome reformation. Mol. Cells 41, 45–49 (2018).
Rong, Y. et al. Clathrin and phosphatidylinositol-4,5-bisphosphate regulate autophagic lysosome reformation. Nat. Cell Biol. 14, 924–934 (2012).
McMahon, H. T. & Boucrot, E. Molecular mechanism and physiological functions of clathrin-mediated endocytosis. Nat. Rev. Mol. Cell Biol. 12, 517–533 (2011).
Mijanovic, O. et al. Cathepsin D-managing the delicate balance. Pharmaceutics 13, 837 (2021).
Suzuki, C. et al. Lack of cathepsin D in the central nervous system results in microglia and astrocyte activation and the accumulation of proteinopathy-related proteins. Sci. Rep. 12, 11662 (2022).
Huarcaya, S. P. et al. Recombinant pro-CTSD (cathepsin D) enhances SNCA/α-Synuclein degradation in α-Synucleinopathy models. Autophagy 18, 1127–1151 (2022).
Hetz, C., Zhang, K. & Kaufman, R. J. Mechanisms, regulation and functions of the unfolded protein response. Nat. Rev. Mol. Cell Biol. 21, 421–436 (2020).
Colla, E. Linking the endoplasmic reticulum to Parkinson’s disease and α-synucleinopathy. Front. Neurosci. 13, 560 (2019).
Hrabos, D. et al. Unfolded protein response markers Grp78 and eIF2alpha are upregulated with increasing alpha-synuclein levels in Lewy body disease. Neuropathol. Appl. Neurobiol. 50, e12999 (2024).
Kim, D. Y. et al. A selective ER-phagy exerts neuroprotective effects via modulation of α-synuclein clearance in Parkinsonian models. Proc. Natl. Acad. Sci. USA 120, e2221929120 (2023).
Henrich, M. T., Oertel, W. H., Surmeier, D. J. & Geibl, F. F. Mitochondrial dysfunction in Parkinson’s disease -a key disease hallmark with therapeutic potential. Mol. Neurodegener. 18, 83 (2023).
Song, N., Mei, S., Wang, X., Hu, G. & Lu, M. Focusing on mitochondria in the brain: from biology to therapeutics. Transl. Neurodegener. 13, 23 (2024).
Toulorge, D., Schapira, A. H. V. & Hajj, R. Molecular changes in the postmortem Parkinsonian brain. J. Neurochem. 139, 27–58 (2016).
Epifane-de-Assunção, M. C., Bispo, A. G., Ribeiro-Dos-Santos, Â & Cavalcante, G. C. Molecular alterations in core subunits of mitochondrial complex I and their relation to Parkinson’s disease. Mol. Neurobiol. 62, 6968–6982 (2025).
Zhang, X. et al. Enhancing mitochondrial proteolysis alleviates alpha-synuclein-mediated cellular toxicity. NPJ Parkinsons Dis. 10, 120 (2024).
Geibl, F. F. et al. α-Synuclein pathology disrupts mitochondrial function in dopaminergic and cholinergic neurons at-risk in Parkinson’s disease. Mol. Neurodegener. 19, 69 (2024).
Mingo, Y. B., Escobar Galvis, M. L. & Henderson, M. X. α-Synuclein pathology and mitochondrial dysfunction: toxic partners in Parkinson’s disease. Neurobiol. Dis. 209, 106889 (2025).
Han, T., Xu, Y., Sun, L., Hashimoto, M. & Wei, J. Microglial response to aging and neuroinflammation in the development of neurodegenerative diseases. Neural Regen. Res. 19, 1241–1248 (2024).
Wang, Q. et al. Inflammasomes in neurodegenerative diseases. Transl. Neurodegener. 13, 65 (2024).
Kuo, G. et al. Emerging targets of α-synuclein spreading in α-synucleinopathies: a review of mechanistic pathways and interventions. Mol. Neurodegener. 20, 10 (2025).
Soraci, L. et al. Toll-like receptors and NLRP3 inflammasome-dependent pathways in Parkinson’s disease: mechanisms and therapeutic implications. J. Neurol. 270, 1346–1360 (2023).
Doudna, J. A. & Charpentier, E. Genome editing. The new frontier of genome engineering with CRISPR-Cas9. Science 346, 1258096 (2014).
Kumar, A. V., Mills, J. & Lapierre, L. R. Selective autophagy receptor p62/SQSTM1, a pivotal player in stress and aging. Front. Cell Dev. Biol. 10, 793328 (2022).
Sharon, R. et al. The formation of highly soluble oligomers of alpha-synuclein is regulated by fatty acids and enhanced in Parkinson’s disease. Neuron 37, 583–595 (2003).
Neumann, M. et al. Misfolded proteinase K-resistant hyperphosphorylated alpha-synuclein in aged transgenic mice with locomotor deterioration and in human alpha-synucleinopathies. J. Clin. Invest. 110, 1429–1439 (2002).
Napolitano, G. & Ballabio, A. TFEB at a glance. J. Cell Sci. 129, 2475–2481 (2016).
Raben, N. & Puertollano, R. TFEB and TFE3: linking lysosomes to cellular adaptation to stress. Annu. Rev. Cell Dev. Biol. 32, 255–278 (2016).
Tan, A., Prasad, R., Lee, C. & Jho, E. H. Past, present, and future perspectives of transcription factor EB (TFEB): mechanisms of regulation and association with disease. Cell Death Differ. 29, 1433–1449 (2022).
Fu, Y. et al. Activating autophagy to eliminate toxic protein aggregates with small molecules in neurodegenerative diseases. Pharmacol. Rev. 77, 100053 (2025).
Alvarsson, A. et al. Emotional memory impairments induced by AAV-mediated overexpression of human α-synuclein in dopaminergic neurons of the ventral tegmental area. Behav. Brain Res. 296, 129–133 (2016).
Viana, R. J. S., Nunes, A. F. & Rodrigues, C. M. P. Endoplasmic reticulum enrollment in Alzheimer’s disease. Mol. Neurobiol. 46, 522–534 (2012).
Vogler, M. et al. The BCL2 family: from apoptosis mechanisms to new advances in targeted therapy. Signal. Transduct. Target Ther. 10, 91 (2025).
Kathiresan, D. S. et al. Role of mitochondrial dysfunctions in neurodegenerative disorders: advances in mitochondrial biology. Mol. Neurobiol. 62, 6827–6855 (2025).
Hoogland, I. C. M., Houbolt, C., van Westerloo, D. J., van Gool, W. A. & van de Beek, D. Systemic inflammation and microglial activation: systematic review of animal experiments. J. Neuroinflamm. 12, 114 (2015).
Isik, S. et al. Microglia mediated neuroinflammation in Parkinson’s disease. Cells 12, 1012 (2023).
Choi, K., Lee, J. & Choi, C. Divergent effect of proteasome inhibition on interleukin-1beta and tumor necrosis factor alpha signaling in human astroglial cells. FEBS Lett. 581, 4691–4696 (2007).
Yasuda, K., Nakanishi, K. & Tsutsui, H. Interleukin-18 in health and disease. Int. J. Mol. Sci. 20, 649 (2019).
Xia, X. G. et al. Gene transfer of the JNK interacting protein-1 protects dopaminergic neurons in the MPTP model of Parkinson’s disease. Proc. Natl. Acad. Sci. USA 98, 10433–10438 (2001).
Chen, C. Y. et al. (G2019S) LRRK2 activates MKK4-JNK pathway and causes degeneration of SN dopaminergic neurons in a transgenic mouse model of PD. Cell Death Differ. 19, 1623–1633 (2012).
Huang, Q. et al. JNK-mediated activation of ATF2 contributes to dopaminergic neurodegeneration in the MPTP mouse model of Parkinson’s disease. Exp. Neurol. 277, 296–304 (2016).
Kang, A. et al. From RIPK1 to necroptosis: pathogenic mechanisms in neurodegenerative diseases. Neurochem. Res. 50, 194 (2025).
Regoni, M., Valtorta, F. & Sassone, J. Dopaminergic neuronal death via necroptosis in Parkinson’s disease: A review of the literature. Eur. J. Neurosci. 59, 1079–1098 (2024).
Yoshida, S. et al. Parkinson’s disease-linked DNAJC13 mutation aggravates alpha-synuclein-induced neurotoxicity through perturbation of endosomal trafficking. Hum. Mol. Genet. 27, 823–836 (2018).
Swords, S. B. et al. A conserved requirement for RME-8/DNAJC13 in neuronal autophagic lysosome reformation. Autophagy 20, 792–808 (2024).
Calcagni', A. et al. Loss of the batten disease protein CLN3 leads to mis-trafficking of M6PR and defective autophagic-lysosomal reformation. Nat. Commun. 14, 3911 (2023).
Serra-Vinardell, J. et al. LYST deficiency impairs autophagic lysosome reformation in neurons and alters lysosome number and size. Cell Mol. Life Sci. 80, 53 (2023).
Wu, L. et al. TMEM175: A lysosomal ion channel associated with neurological diseases. Neurobiol. Dis. 185, 106244 (2023).
Parnetti, L. et al. Cerebrospinal fluid β-glucocerebrosidase activity is reduced in Parkinson’s disease patients. Mov. Disord. 32, 1423–1431 (2017).
Kang, J. et al. Identification of BAG2 and cathepsin D as plasma biomarkers for Parkinson’s disease. Clin. Transl. Sci. 14, 606–616 (2021).
Suire, C. N. et al. Cathepsin D regulates cerebral Aβ42/40 ratios via differential degradation of Aβ42 and Aβ40. Alzheimers Res. Ther. 12, 80 (2020).
Kim, J. W. et al. Identification of cathepsin D as a plasma biomarker for Alzheimer’s disease. Cells 10, 138 (2021).
Urbanelli, L. et al. Cathepsin D expression is decreased in Alzheimer’s disease fibroblasts. Neurobiol. Aging 29, 12–22 (2008).
Valdez, C. et al. Progranulin-mediated deficiency of cathepsin D results in FTD and NCL-like phenotypes in neurons derived from FTD patients. Hum. Mol. Genet. 26, 4861–4872 (2017).
Beel, S. et al. Progranulin functions as a cathepsin D chaperone to stimulate axonal outgrowth in vivo. Hum. Mol. Genet. 26, 2850–2863 (2017).
Álvarez-Luquín, D. D. et al. Neurodegeneration models in Parkinson’s disease: cellular and molecular paths to neuron death. Behav. Brain Funct. 21, 14 (2025).
Chiu, C. C. et al. (D620N) VPS35 causes the impairment of Wnt/β-catenin signaling cascade and mitochondrial dysfunction in a PARK17 knockin mouse model. Cell Death Dis. 11, 1018 (2020).
Chiu, C. C. et al. Absence of Rab39b-induced macroautophagy impairment increases neurotoxic α-synuclein and causes degeneration of substantia nigra dopaminergic neurons in mouse model of X-linked Parkinson’s disease. Life Sci. 337, 123789 (2025).
Acknowledgements
The authors thank the National Laboratory Animal Center (Tainan, Taiwan) for preparing Dnajc6Q787X/+ knockin mice. This study was supported by the National Science and Technology Council, Taiwan (NSTC 113- 2320-B-182-026-MY3 to H.-L.W. and NSTC 114-2314-B-038-061 to T.-H.Y.), Chang Gung Medical Foundation (CMRPD1M0142 to H.-L.W.), and the Healthy Aging Research Center, Chang Gung University (EMRPD1M0451 to H.-L.W.).
Author information
Authors and Affiliations
Contributions
Hung-Li Wang, Ying-Ling Chen, Yi-Hsin Weng, and Tu-Hsueh Yeh conceptualized the study and designed the experiments; Hung-Li Wang, Ying-Ling Chen, Tai-Ju Chiu, Ching-Chi Chiu, Shu-Yu Liu, and Allen Hon-Lun Li performed the experiments and analyzed the data; Hung-Li Wang, Ying-Ling Chen, Yi-Hsin Weng, and Tu-Hsueh Yeh wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wang, HL., Chen, YL., Chiu, TJ. et al. PARK19 truncation mutant Dnajc6 causes lysosomal deficiency-induced upregulation of pathologic α-synuclein and neurodegeneration of substantia nigra dopaminergic cells in PARK19 knockin mice. npj Parkinsons Dis. (2026). https://doi.org/10.1038/s41531-026-01317-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41531-026-01317-8