Abstract
Peripheral artery disease (PAD) causes progressive arterial narrowing in the lower limbs and can advance to critical limb ischemia (CLI). Limited revascularization options highlight the need for safer, more effective therapies. Vascular multipotent stem cells (VMSCs) and adipose-derived stem cells (ADSCs) were isolated from adipose tissue, characterized phenotypically, and tested for angiogenic activity in vitro. Their therapeutic efficacy was then examined in a murine critical limb ischemia model through intramuscular transplantation, assessing limb preservation, neovascularization, and cell integration. VMSCs shared mesenchymal stem cell–like features with ADSCs and exhibited robust proliferative capacity, enabling rapid expansion to clinically relevant numbers. VMSCs also demonstrated endothelial-like properties, including CD31, VE-cadherin, and CD141 expression, and formed capillary-like structures in vitro. In contrast, ADSCs displayed perivascular characteristics with α-SMA and Transgelin expression. Co-culture of VMSCs and ADSCs promoted the development of mature tubular networks in vitro. Combined cell transplantation markedly decreased limb loss and promoted both angiogenesis and arteriogenesis in ischemic tissue, with transplanted cells partially integrating into the host vasculature to form hybrid vascular structures. VMSCs and ADSCs show complementary regenerative functions, sustained engraftment, and support for large-vessel formation, underscoring their potential for stem cell–based vascular therapies.
Similar content being viewed by others
Introduction
Peripheral artery disease (PAD) is a common vascular condition involving partial or complete blockage of arteries supplying the lower limbs, resulting in reduced blood flow and impaired oxygen and nutrient delivery. Major risk factors include smoking, hypertension, aging, diabetes, and genetic predisposition1. PAD prevalence increases significantly with age, especially in those over 65 or in individuals over 50 with risk factors such as smoking and diabetes. Critical limb ischemia (CLI) is the most advanced stage, marked by ischemic pain, non-healing ulcers, and gangrene, and is associated with a high risk of amputation and death2. This stage marks a transition from manageable PAD symptoms to life- and limb-threatening complications, highlighting the importance of early detection and prompt therapeutic management.
Therapeutic neovascularization for PAD has been extensively explored in both experimental and clinical studies. Conventional treatments for PAD, such as statin administration and balloon angioplasty, are frequently used to enhance perfusion3,4. Nevertheless, these approaches have notable limitations, often falling short of fully restoring vascular function. Furthermore, a considerable number of patients with CLI are not eligible for revascularization procedures5. To overcome these therapeutic limitations in CLI, stem cell-based approaches have emerged as a promising area of research and clinical interest6,7. Mesenchymal stem cells (MSCs), a prominent type of adult stem cell, have garnered significant attention across multiple medical disciplines8. Both preclinical and clinical studies have demonstrated the therapeutic potential of MSCs in treating vascular diseases9,10,11. In addition to MSCs, endothelial progenitor cells (EPCs) and induced endothelial cells have also been actively investigated for their ability to contribute to vascular endothelium formation12,13,14,15,16. Bone marrow (BM)–derived EPCs, mobilized by granulocyte colony-stimulating factor, have been reported to migrate to ischemic sites and differentiate into endothelial cells, thereby promoting vascular repair17. Additionally, endothelial-like cells induced from stem cells via ETS variant 2 (ETV2) have demonstrated therapeutic potential in mouse models of limb ischemia16.
When stem cells are transplanted into diseased tissue, their survival is limited by inflammation and oxidative stress. Because few cells successfully engraft and differentiate after transplantation, direct tissue integration is rare18,19. Thus, paracrine activity has emerged as the major driver of stem cell–mediated vascular repair. Key secreted factors—such as vascular endothelial growth factor (VEGF), fibroblast growth factor (FGF), hepatocyte growth factor (HGF), and platelet-derived growth factor (PDGF)—stimulate angiogenesis by promoting new capillary growth from existing vessels. These newly formed microvessels improve perfusion and support tissue repair by promoting endothelial cell proliferation, migration, and survival. Therefore, the stem cell secretome mainly supports microvascular network formation necessary for regeneration, rather than directly creating large-caliber vessels.20,21,22. Although this response can modestly improve blood vessel growth, it often remains inadequate to prevent limb loss in CLI patients23,24. This limitation highlights the need for approaches that more effectively drive the formation of mature, perfusable vessels by transplanted stem cells.
To achieve effective neovascularization and restore sufficient blood flow in severe disease states, key factors must be addressed when using stem cell transplantation. First, the development of both large vessels—through arteriogenesis and venogenesis—and small vessels via angiogenesis is crucial for establishing a functional and integrated vascular network.
Arteriogenesis is an essential compensatory mechanism in CLI, where pre-existing collateral arterioles remodel into larger, functional arteries to bypass major arterial obstruction. Despite its clinical importance, most prior studies have centered on angiogenesis rather than arteriolar remodeling. A clear understanding of vascular architecture—including the coordinated roles of endothelial and perivascular cells—is also crucial for stable neovascularization. Notably, analysis of ischemic tissue from an amputated CLI patient showed markedly reduced stable microvessel density compared with non-ischemic tissue25, highlighting the importance of adequate perivascular support. Previous studies have shown that MSCs function primarily as perivascular cells, displaying pericyte-like characteristics both in vivo and in vitro. However, physiological differentiation of MSCs into endothelial cells is rarely observed. Instead, MSCs mainly serve as mesenchymal progenitors that support angiogenesis and contribute to vascular stability26,27. Similarly, transplantation of EPCs has not shown consistent or durable incorporation into host vasculature. This underscores the lack of a stem cell population capable of directly integrating into newly formed vessels. To address this limitation, recent studies have explored combinatorial strategies employing MSCs together with endothelial colony-forming cells (ECFCs). Co-delivery of MSCs and ECFCs in vivo leads to formation of perfused, pericyte-covered blood vessels, which ECFCs alone fail to achieve effectively28,29,30. These strategies are expected to promote the formation of structurally mature and well-organized vasculature, while establishing an ex vivo culture platform for the concurrent preparation of dual stem cells could further expedite their clinical application29.
Recently, novel vasculogenic precursor cells (VPCs) isolated from bone marrow aspirates (BMA) were first identified as highly proliferative, multipotent cells with strong vasculogenic potential7. These VPCs uniquely expressed CD141 (thrombomodulin), a marker absent in BM–derived MSCs. Transplantation of a combination of CD141⁺ VPCs and BM-MSCs promoted limb salvage, enhanced blood flow recovery, and stimulated arteriole-like vessel regeneration in a nude mouse model of CLI. Importantly, dual cell therapy with VPC and BM-MSC from human BM induced the expression of human CD31 and α-smooth muscle actin (αSMA) in murine vessels, demonstrating the direct integration of transplanted cells into the host vasculature.
In this study, adipose tissue (AT) was used as a less invasive alternative to BMA for stem cell isolation, as BMA collection is burdensome for patients undergoing autologous therapy for vascular disease. AT provides an accessible and abundant cell source, from which we successfully isolated and characterized both vascular multipotent stem cells (VMSCs) and adipose-derived stem cells (ADSCs). To our knowledge, the therapeutic potential of these AT-derived dual-cell populations has not previously been evaluated in a CLI model. Therefore, we transplanted a combination of VMSCs and ADSCs into a nude mouse model of vascular dysfunction to assess their capacity to promote therapeutic neovascularization.
Results
Identification and characterization of vascular multipotent stem cells (VMSCs) and adipose-derived stem cells (ADSC) derived from human adipose tissue
AT obtained from human donors was mechanically dissociated and enzymatically digested to isolate the stromal vascular fraction (SVF). The SVF consists of a heterogeneous cell population, including stem cells, adipocytes, endothelial cells, and immune cells, along with diverse growth factors, all of which collectively contribute to tissue repair and regeneration (Fig. 1A). The SVF was then divided into two groups and cultured in distinct media conditions. Cells cultured under endothelial growth conditions are referred to as VMSCs, whereas those maintained in mesenchymal growth medium are termed ADSCs. Both VMSCs and ADSCs exhibited the spindle-shaped morphology characteristic of mesenchymal lineage cells (Fig. 1B), with only moderate differences observed between the two cell types. VMSCs exhibited a population doubling time (PDT) of 1.2 days at passage 1, which progressively increased with subsequent passages. In contrast, ADSCs maintained a consistent PDT of approximately 1 day across all passages (Fig. 1C). Accordingly, the VMSC population expanded from an initial yield of approximately 1 × 106 cells from the SVF to at least 4 × 107 cells by passage 2. (Fig. 1D). In comparison, ADSCs demonstrated a higher proliferative capacity under the same conditions, reaching 100 million cells by passage 2 (Fig. 1E). Given the doubling capacity of VMSCs, further applications of both VMSCs and ADSCs are optimal at passage 2; therefore, cells at this passage were used for subsequent analyses.
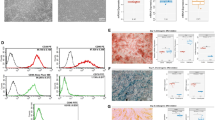
A Isolation scheme of VMSC and ADSC from adipose tissue. B Cell morphology of VMSCs and ADSCs from passages 0 to 3. Scale bar: 200 μm. C Population doubling time of VMSCs and ADSCs from passages 1 to 3. Values are mean ± SD of 6 independent experiments (*P < 0.05, **P < 0.01, Student’s t test). Predicted cumulative cell number of VMSCs (D) and ADSCs (E) from stromal vascular fraction (SVF) to passage 3. F FACS analysis for expression of CD markers on VMSCs and ADSCs. Values are mean ± SD of 3 independent experiments (***P < 0.001, Student’s t test). G Representative images of differentiation assays of VMSCs and ADSCs. Adipogenesis, osteogenesis, and chondrogenesis were analyzed by Oil Red O staining, Alizarin Red S staining, and Alcian Blue staining, respectively. Scale bar: 50 μm. V: VMSC, A: ADSC.
The surface marker expression profiles of VMSCs and ADSCs were assessed by FACS (Fig. 1F). Both cell types were positive for CD44, CD73, CD90, CD29, and CD105, and negative for CD34 and CD45, consistent with the classical MSC phenotype. Notably, VMSCs uniquely expressed CD141 (thrombomodulin), a protein known to promote angiogenesis via podosome rosette formation, whereas ADSCs lacked this expression. Therefore, CD141 may serve as a distinguishing marker to separate VMSCs from ADSCs. Both cell types demonstrated trilineage differentiation potential into adipogenic, osteogenic, and chondrogenic lineages (Fig. 1G).
Collectively, these findings demonstrate that a novel population of CD141+ VMSCs can be isolated from adipose tissue and that VMSCs are culture-expandable and exhibit multilineage differentiation potential comparable to ADSCs.
Comparative analysis of the cellular characteristics of VMSCs and ADSCs with respect to their angiogenic potential
In Fig. 1, VMSCs and ADSCs were evaluated based on the established criteria for MSCs. VMSCs demonstrated similar marker expression and differentiation capacity compared to ADSCs, although certain differences were noted. Subsequently, their neovascularization potential was evaluated by comparing the expression levels of angiogenic proteins through Western blot analysis (Fig. 2A). VMSCs exhibited higher expression of angiogenic and endothelial-related markers, including CD141, CD31, VE-cadherin, LYVE-1, FGFR2, and ETV2. In contrast, ADSCs showed elevated expression of perivascular cell markers such as TNAP, α-SMA, and Transgelin. c-Met, the receptor for HGF and a critical mediator of angiogenesis, was expressed in both VMSCs and ADSCs (Fig. 2A and Supplementary Fig. 1). This expression profile indicates that VMSCs exhibit endothelial-like properties, while ADSCs display characteristics more consistent with perivascular cells, despite sharing several common markers.
A, B Western blot analysis for CD141, CD31, VE-cadherin, LYVE1, FGFR2, ETV2, TNAP, αSMA, Transgelin, c-MET, and GAPDH in VMSCs and ADSCs and quantitative densitometry analysis using the NIH ImageJ program. GAPDH was used as a loading control. Values are mean ± SD of 3 independent experiments (*P < 0.05, **P < 0.01, and ***P < 0.001, n.s.: not significant, Student’s t test). Quantification of HGF (C), uPA (D), VEGF (E), and Angiogenin (F) in conditioned medium of VMSCs and ADSCs by ELISA. Values are mean ± SD of 3 independent experiments (*P < 0.05, **P < 0.01, Student’s t test). G Tube formation assay of VMSC, ADSC, and a combination of VMSC and ADSC in a 2:1 ratio in the presence or absence of 500 pg/ml TNF-α. Scale bar: 500 μm. H Quantitative analysis of tube based on number of meshes, number of master segments, total master segments length, and number of isolated segments using NIH ImageJ angiogenesis analyzer. Values are mean ± SD of 4 independent experiments (***P < 0.001; V (-TNF-α) VS. V (+TNF-α), $$$P < 0.001; 2:1 (-TNF-α) VS. 2:1 (+TNF-α), #P < 0.05, ##P < 0.01; n.s.: not significant, One-way ANOVA test followed by Tukey’s multiple comparison test. V: VMSC, A: ADSC.
In addition to cellular marker analysis, angiogenic factor secretion by VMSCs and ADSCs was evaluated using ELISA on their conditioned media. VMSCs predominantly secreted HGF and urokinase-type plasminogen activator (uPA), while ADSCs more abundantly released VEGF and angiogenin (Fig. 2C–F). These findings imply distinct paracrine roles for VMSCs and ADSCs in the regulation of angiogenesis.
Next, the angiogenic potential of VMSCs and ADSCs was assessed in vitro using functional assays performed under inflammatory conditions induced by TNF-α (Supplementary Fig. 2)7. The optimal combination ratio was determined by varying the proportions of VMSCs and ADSCs and evaluating tube formation in vitro (Supplementary Fig. 3). VMSCs, ADSCs, and their combination were seeded onto Matrigel under low-nutrient conditions simulating poor perfusion, and tube formation was assessed after 18 h in the presence or absence of TNF-α. VMSCs formed tubular networks, whereas ADSCs tended to aggregate and were unable to make vascular structures. However, co-culturing VMSCs and ADSCs at a 2:1 ratio significantly enhanced vasculogenic activity, as evidenced by numbers of meshes, master segments, total master segment length, and isolated segments. Under inflammatory stress induced by TNF-α, the tube-forming ability of VMSCs was partially impaired. However, co-culturing VMSCs with ADSCs restored robust tube formation even under TNF-α–mediated inflammatory conditions, indicating that ADSCs provide supportive signals that enhance the vasculogenic activity of VMSCs and promote the formation of compact vascular structures composed of both cell types (Fig. 2G, H and Supplementary Fig. 4). Furthermore, analysis of conditioned medium from VMSC–ADSC co-cultures on Matrigel in the presence of TNF-α showed an increased trend in matrix metalloproteinase (MMP) activity compared with either single-cell condition. These findings further support the synergistic interaction between VMSCs and ADSCs, enhancing cell migration under inflammatory stress (Supplementary Fig. 5).
These results suggest that VMSCs and ADSCs may cooperate in new vessel formation by fulfilling complementary angiogenic roles, with VMSCs contributing endothelial-like functions and ADSCs acting as perivascular support cells in inflammatory environment.
VMSCs and ADSCs synergistically form compact tubular networks by modulating the expression of migratory and angiogenic factors in response to TNF-α in vitro
Upon transplantation into ischemic tissue, stem cells are exposed to a hostile inflammatory environment that severely limits their survival; therefore, their therapeutic efficacy depends on their ability to withstand these conditions and contribute to tissue repair. To investigate the impact of pathological conditions on the angiogenic potential of the dual-cell system in vivo, a strong inflammatory stimulus was mimicked by treating cells with a high concentration of TNF-α for 24 or 48 h. Subsequently, their angiogenic potential and migratory capacity were assessed in vitro (Fig. 3). Although this concentration of TNF-α has been reported to impair the viability of various cell types31,32,33, the viability of both VMSCs and ADSCs remained unaffected (Supplementary Fig. 6). Western blot analysis was conducted to evaluate changes in representative markers of endothelial and perivascular cells. CD141 and CD31, which are predominantly expressed on VMSCs, and α-SMA and Transgelin, which are characteristic of ADSCs, were specifically examined. As shown in Fig. 3A, VMSCs retained CD141 and CD31 expression following TNF-α stimulation, although a reduction in expression intensity was observed. Conversely, TNF-α exposure had minimal impact on α-SMA and Transgelin expression in ADSCs (Fig. 3A–E). These findings indicate that the intrinsic characteristics of each cell type are preserved under severe inflammatory stress without compromising survival, and that these markers may serve as reliable tools for distinguishing VMSCs from ADSCs in vivo.
A Western blot of CD141, CD31, αSMA, Transgelin, and GAPDH expression in VMSCs and ADSCs after treatment of 10 ng/ml TNF-α for 24 or 48 h. Quantitative densitometry analysis of CD141 (B) CD31 (C) αSMA (D) and Transgelin (E) using NIH ImageJ program. GAPDH was used as a loading control. Each value is expressed as a fold change relative to VMSCs without TNF-α treatment (24 h). Values are mean ± SD of 3 independent experiments. Statistical analyses were conducted exclusively against the respective control groups. (*P < 0.05, ***P < 0.001, Student’s t test). F Gelatin zymography to detect activity of pro-MMP-9, pro-MMP-2, and active-MMP-2 in conditioned medium of VMSCs and ADSCs. Quantitative densitometry analysis of pro-MMP-9 (G) pro-MMP-2 (H) and active-MMP-2 (I). Values are mean ± SD of 3 independent experiments. Total protein lysate (μg) of each group was used as loading control. Statistical analyses were conducted exclusively against the respective control groups. (*P < 0.05, **P < 0.01, and ***P < 0.001, Student’s t test). Quantification of HGF (J) and VEGF (K) in conditioned medium in VMSCs and ADSCs by ELISA. Values are mean ± SD of 3 independent experiments. Total protein lysate (μg) of each group was used as loading control. Statistical analyses were conducted exclusively against the respective control groups (**P < 0.01, ***P < 0.001, Student’s t test). L The tube formation assay with VMSC, ADSC and a combination of VMSC (V) and ADSC (A) in a 2:1 ratio was performed after priming of TNF-α for 24 h. Scale bar: 500 μm. M Quantitative analysis of tube architecture was performed based on the number of meshes, the number of master segments, total master segments length, and the number of isolated segments using NIH ImageJ angiogenesis analyzer. Values are mean ± SD of 4 independent experiments (*P < 0.05, **P < 0.01, ***P < 0.001; VMSC (V) TNF-α (-) VS. V TNF-α (+), $P < 0.05, $$P < 0.01; 2:1 TNF-α (-) VS. 2:1 TNF-α (+), #P < 0.05, ##P < 0.01, One-way ANOVA test followed by Tukey’s multiple comparison test). V: VMSC, A: ADSC.
Neovascularization requires cell migration and integration, processes supported by the presence of migratory/angiogenic factors. To assess effect of TNF-α on cellular migration capacity, MMP secretion from both cell types was evaluated using gelatin zymography (Fig. 3F–I). TNF-α exposure tended to increase MMP production in both VMSCs and ADSCs. Notably, MMP (pro MMP-9 and active MMP-2) secretion by VMSCs was progressively enhanced with prolonged TNF-α treatment. Moreover, TNF-α stimulation increased HGF secretion in VMSCs and ADSCs, while VEGF production was elevated in VMSCs but decreased in ADSCs (Fig. 3J, K). These findings suggest that TNF-α exposure modulates the paracrine signaling profile of both VMSCs and ADSCs. Notably, VMSCs exhibited a distinct response to TNF-α compared with ADSCs, suggesting a superior angiogenic potential.
TNF-α-induced changes in VMSCs and ADSCs were evident as early as 24 h, with alterations in marker expression, MMP activity, and growth factor secretion. Next, to determine whether TNF-α-induced paracrine changes can enhance vascular formation by VMSC or ADSC, the tube-forming capacity of VMSCs and ADSCs primed with TNF-α for 24 h was assessed using a Matrigel assay (Fig. 3L and Supplementary Fig. 7). As expected, in the absence of TNF-α priming, co-culture of VMSCs and ADSCs resulted in the formation of more compact tubular structures compared to cultures of VMSCs or ADSCs alone, consistent with the findings shown in Fig. 2G. Interestingly, TNF-α priming did not impair the tube-forming ability of VMSCs; instead, it enhanced it. This effect was further amplified in the co-culture of VMSCs and ADSCs primed with TNF-α, as evidenced by increased mesh formation, number of master segments, and total master segment length, surpassing the tube-forming ability observed in the non-primed co-culture condition (Fig. 3M). This effect may be attributed to a synergistic interaction between VMSCs and ADSCs, which display altered migratory and angiogenic potential in response to TNF-α stimulation.
These results indicate that transplanted VMSCs and ADSCs subjected to intense inflammatory stress not only remain viable but also contribute to establishing a pro-angiogenic and pro-migratory microenvironment that supports vascularization.
Dual cell therapy restores blood flow and prevents limb loss in a CLI mouse
Based on the characteristics of VMSCs and ADSCs derived from AT, this combination therapy is anticipated to exert therapeutic effects in ischemia-induced vascular diseases. To assess the therapeutic potential of dual cell therapy comprising VMSCs and ADSCs, a murine model of critical limb ischemia (CLI) was established through ligation and excision of the femoral artery and then, VMSC and ADSC were intramuscularly transplanted at 2:1 ratio (VMSC: ADSC, 5 sites, 10 μl/ site) (Fig. 4A and Supplementary Fig. 8). The change of mouse limb was monitored for 28 days (Fig. 4B). While non-treated control mouse lost limb within 7 days, dual cell therapy with VMSC and ADSC obviously prevented limb loss. Ischemic necrosis was assessed using a seven-grade scoring system on days 0, 1, 2, 3, 7, 14, and 28 (Fig. 4C). A difference between the two groups became evident from day 3 post-transplantation and was maintained throughout the 4-week observation period. By day 28, the non-treated control group exhibited extensive limb and foot loss, whereas mice receiving dual-cell transplantation retained limb integrity (Fig. 4D).
A Experimental scheme of dual cells transplantation in a mouse CLI model. B Representative images of limb morphological changes on days 7 and 28. Scale bar: 5 mm. C Progression of ischemic necrosis grade over the 4-week period. Values are mean ± SD of 5 independent experiments (**P, and ***P, n.s.: not significant, Two-way repeated measures ANOVA followed by Bonferroni post-tests). D Distribution of grades for ischemic necrosis on day 28. E The injury sites on day 28 (white dashed line: removed femoral artery, yellow arrowhead: newly formed blood vessels and black arrowhead: purulent). F Representative images of laser Doppler analysis on days 7 and 28. G Quantitative analysis of blood flow using laser Doppler analysis over the 4-week period (***P, n.s.: not significant, Two-way repeated measures ANOVA followed by Bonferroni post-tests). H Hematoxylin and Eosin (H&E), Masson’s trichrome (MTC) staining of the gastrocnemius muscle of ischemic limb. Scale bar:100 μm. I Quantitative analysis of collagen deposition area using NIH ImageJ Color Deconvolution 2 analyzer. Values are mean ± SD of 4 independent experiments (***P student’s t test). J Immunohistochemical staining for mouse macrophage (F4/80) of the gastrocnemius muscle of ischemic limb. Scale bar:100 μm. N = 8/group.
Limb preservation should be accompanied by the restoration of blood flow via neovascularization. Prior to sacrifice, the ischemic site was grossly examined with a focus on the vascular architecture (Fig. 4E). Non-treated controls showed absence of arterial structures with inflammation (black arrow head), while dual-cell transplantation induced new large vessel formation (yellow arrow head) near the artery removal site (white dotted line). Blood flow analysis using laser Doppler imaging was also performed (Fig. 4F, G). From day 7 onward, the non-treated control group has maintained blood flow below 20%, whereas dual-cell transplantation significantly increased blood flow, reaching approximately 70% by day 28. To evaluate inflammatory responses at the injury site, histological analyses were performed. In the non-treated control group, the ischemic muscle exhibited marked degeneration accompanied by extensive fibrosis, whereas these pathological changes were substantially alleviated following dual-cell therapy (Fig. 4H, I). In addition, immunohistochemical staining revealed prominent accumulation of F4/80⁺ macrophages around vascular structures in the control group, reflecting heightened inflammatory infiltration. This macrophage enrichment was markedly reduced in mice receiving dual-cell transplantation (Fig. 4J), further supporting the anti-inflammatory and tissue-protective effects of the dual-cell therapy.
Taken together, combined transplantation of VMSCs and ADSCs exerted therapeutic effects in the CLI mouse model by promoting blood vessel regeneration and attenuating inflammation within the ischemic tissue.
Formation of large-caliber vasculature and successful integration with host vessels are accomplished in vivo via dual cell therapy
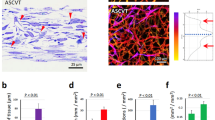
Transplantation of VMSCs and ADSCs could restore blood flow, contributing to limb preservation in CLI mouse (Fig. 4). To elucidate the underlying mechanisms of dual cell therapy efficacy, histological analysis was performed to evaluate vascular formation. Initially, vessel density was quantified by counting CD31+ or Transgelin⁺ vessels exceeding 5 μm in diameter (Fig. 5). In parallel, wheat germ agglutinin (WGA) staining was used to visualize the structural organization of muscle tissue and blood vessels. Compared to the non-treated control, dual cell therapy increased CD31+ vessel density by approximately 56% (Fig. 5A, B). In mice, blood vessels with diameters greater than ~20 μm are typically classified as microvessels, including arterioles and venules, whereas those exceeding ~50 μm are regarded as macrovessels, comprising arteries and veins. This size-based distinction aligns with common vascular classification criteria where arterioles measure roughly 20–50 μm in diameter and serve as resistance vessels controlling blood flow into capillary beds34,35. Analysis of CD31+ vessel distribution by diameter revealed that, in the non-treated control group, 52% of CD31+ vessels had a diameter greater than 20 μm, and 23% exceeded 50 μm. In contrast, dual-cell treatment significantly increased the proportion of CD31+ vessels with diameters greater than 20 μm, comprising 71% of the total CD31+ vessel population, with 34% exhibiting diameters greater than 50 μm (Fig. 5C). Considering the total density of CD31+ vessels in both groups, dual-cell therapy increased the number of vessels with a diameter of 20 μm by approximately 2.1-fold and those with a diameter of 50 μm by approximately 2.3-fold compared with the untreated control. This suggests that dual cell therapy promotes the formation of larger-caliber vessels at the ischemic site, which might be associated with improved blood flow recovery.
A Representative fluorescence images of Human/Mouse CD31+ blood vessels at day 28. Scale bar: 100 μm. B, C Quantification of Human/Mouse CD31+ blood vessels analyzed by NIH image J analyzer. Values are mean ± SD of 5 independent experiments (***P < 0.001, Student’s t test). D Fluorescence images of Human/Mouse Transgelin+ blood vessels at day 28. Scale bar: 100 μm. E, F Quantification of Human/Mouse Transgelin+ blood vessels analyzed by NIH ImageJ analyzer. Values are mean ± SD of 5 independent experiments (***P < 0.001, Student’s t test). G Representative fluorescence images of Human CD31+ blood vessels at day 28 post transplantation. Scale bar: 100 μm. Orange scale bar in high mag. image: 50 μm. H Quantification of diameter distribution of human CD31+ blood vessels by NIH ImageJ analyzer. I Double fluorescence staining for Human CD31 and Human/Mouse CD141. White arrow head: colocalization of CD31 and CD141. Scale bar: 50 μm. J The detection of human nuclear antigen (HN) in vessels in dual cell-transplanted muscle. Yellow arrow head indicates human antigen. Scale bar: 20 μm. N = 8/group.
Transgelin was used to identify vessels surrounded by smooth muscle cells. Dual cell treatment resulted in approximately a 2-fold increase in the density of Transgelin+ vessels compared to the non-treated control (Fig. 5D, E). Analysis of Transgelin+ vessel distribution by diameter revealed an increase in vessels larger than 20 μm following dual-cell therapy. For Transgelin⁺ vessels exceeding 50 μm, both groups initially showed a distribution of approximately 15%; however, dual-cell therapy nearly doubled the total number of Transgelin+ vessel (Fig. 5E), indicating an increased number of large-caliber vessels (>50 μm) in treated mice compared with untreated controls (Fig. 5F).
In the context of vascular diseases, conventional stem cell therapy is largely associated with tissue repair mediated by paracrine effects, primarily through the formation of small-diameter vessels. However, in this study, dual cell therapy with VMSCs and ADSCs promoted the formation of large, arteriole-like vessels at ischemic site. These findings indicates that the therapeutic effects of dual cell therapy may arise not only from paracrine signaling but also from the direct contribution of transplanted cells to vascular regeneration. To identify human-derived vessels at the injection site in the CLI mouse, immunostaining was performed using a human-specific CD31 antibody. Human CD31⁺ vessels were observed exclusively in mice receiving dual cell transplantation and were absent in non-treated controls (Fig. 5G). Immunofluorescence staining with anti-human CD31 and anti-human/mouse CD31 antibodies confirmed the presence of both host mouse vasculature and vessels containing transplanted human cells in ischemic muscle (Supplementary Fig. 9). Quantitative assessment showed that approximately 59% of human CD31⁺ vessels exceeded 20 μm in diameter, indicating their involvement in the formation of arteriole-like structures (Fig. 5H and Supplementary Fig. 10).
Molecular analysis in Fig. 2 revealed that CD141 and CD31 was primarily expressed in VMSCs. Therefore, it was examined whether the human CD31+ vessels also expressed CD141 (Fig. 5I). Analysis of serial-sectioned samples revealed colocalization of CD31 and CD141 on large-diameter vessels (white arrow), supporting the potential involvement of transplanted VMSC in vascularization. To repeatedly confirm the localization of human stem cells, immunofluorescent double staining was performed using antibodies against human nuclear antigen (HN) (Fig. 5J and Supplementary Fig. 11). HN-positive cells (yellow arrow head) were detected within vasculature, indicating the integration of human stem cells into the vascular endothelium with perivascular structures.
These findings indicate that limb preservation following dual cell therapy with VMSCs and ADSCs is associated with the formation of arteriole-like vessels, which are at least partially comprised of transplanted cells.
Analysis of 3D whole-mount imaging for ischemic tissue following dual cell therapy in a murine CLI model
Transplantation of dual cells led to increased vascular density and was accompanied by their direct integration into the host vasculature (Fig. 5). Subsequently, whole mount three-dimensional analysis of the entire muscle tissue was performed to assess overall vessel density and spatial distribution of the vasculature within the ischemic zone, likely comprising various vascular structures such as veins, arterioles, and capillaries. Immunofluorescence staining for CD31 demonstrated that, compared to normal muscle, the non-treated control predominantly exhibited thin and underdeveloped vessels. In contrast, muscle tissue treated with dual cell therapy exhibited vascular quality and robust vascular architecture comparable to that of normal, non-ischemic muscle (Fig. 6A). Quantification of total CD31+ vessels in muscle tissue was performed by analyzing the number of branches and junctions of all CD31+ vessels, irrespective of diameter. As shown in Fig. 6B, C, the total number of CD31+ vessels was higher in non-treated mice compared with those receiving dual-cell therapy. However, when CD31+ vessels excluding capillaries were analyzed, vessel density was greater in dual cell–treated mice (Fig. 5B). These findings imply that the untreated group may contain a vascular network enriched in small-diameter vessels, such as capillaries.
3D Analysis of vascular networks in ischemic limb muscle after dual-cell transplantation was carried out. A whole-mount fluorescence images of Human/Mouse CD31+ blood vessels. Scale bar: 1 mm. High magnification view of yellow, blue and red arrow head. Orange scale bar: 100 μm. Quantification of CD31+ vessel of branches (B) and junctional number (C) (**P < 0.01, ***P < 0.001, One-way ANOVA test followed by Tukey’s multiple comparison test). Values are mean ± SD. D The average diameter of CD31+ vessel. (***P < 0.001, One-way ANOVA test followed by Tukey’s multiple comparison test). Values are mean ± SE. E Vessel distribution by diameter. F Whole-mount fluorescence images of Human/Mouse α-SMA+ blood vessels. Scale bar: 1 mm. G Assessment of α-SMA+ vessel density (human and mouse origin) within whole ischemic muscle. Density of α-SMA+ vessels was shown as area per mm2 (*P < 0.05, ***P < 0.001, One-way ANOVA test followed by Tukey’s multiple comparison test). Values are mean ± SD. H Representative fluorescence images of Human/Mouse CD31+ and α-SMA + vascular network. Scale bar: 100 μm. I Quantification of α-SMA+ cells-encircled CD31+ vessels (**P < 0.01, ***P < 0.001, One-way ANOVA test followed by Tukey’s multiple comparison test). Values are mean ± SD. N = 8/group.
Analysis of vascular diameter across groups revealed that normal vessels had an average diameter of approximately 40 μm, whereas vessels in the non-treated group averaged 10 μm, and those in the dual cell-treated group averaged 26 μm (Fig.6D). More specifically, vascular distribution analysis revealed that the majority of vessels in the non-treated control group (93.1%) had diameters less than 20 μm. In contrast, 48.7% of vessels in dual-cell–treated mice exhibited diameters greater than 20 μm, and 19.4% exceeded 50 μm (Fig. 6E).
Next, vessels surrounded by perivascular cells were quantified by counting α-SMA+ vessels. Normal muscle exhibited compact vessels surrounded by α-SMA+ cells. In contrast, α-SMA+ vessels were rarely observed in the non-treated control group, whereas dual cell–treated tissue exhibited a 1.8-fold higher density of α-SMA+ vessels, indicative of more mature and well-organized vasculature at the treatment site (Fig. 6F, G).
To assess the proportion of vessels encircled by perivascular cells among total vessels in the injured muscle, immunofluorescence double staining for CD31 and α-SMA was performed. Normal muscle contained well-organized, thick arteries positive for both CD31 and α-SMA, whereas the non-treated control showed numerous CD31+ vessels with minimal α-SMA coverage despite a high abundance of CD31+ vessels. In contrast, dual cell treatment promoted the formation of arteriole-like thick vessels with α-SMA+ coverage, alongside the development of smaller, thin capillary-like structures (Fig. 6H). Quantitative analysis of vessel structure revealed that the non-treated control group exhibited high vessel density but only 4.3% α-SMA+ perivascular cell coverage. In contrast, the dual cell-treated group showed α-SMA+ coverage in 34% of vessels, indicating enhanced vascular maturation (Fig. 6I).
Considering both vascular density and vessel quality, the presence of CD31+ vessels alone does not appear to be a decisive determinant of therapeutic efficacy, as demonstrated by the complete limb and foot loss in the non-treated control group. Instead, successful limb preservation in CLI requires the formation of large-caliber vessels stabilized by perivascular cells, resulting in an organized vascular architecture—a therapeutic outcome effectively achieved through dual-cell therapy.
The formation of perfusable vessels induced by dual-cell therapy drives vascular network regeneration
Dual cell therapy promoted the formation of compact, large-caliber vasculature with perivascular cell coverage at the ischemic site, leading to improved blood flow restoration in CLI mouse. These results suggest that dual cell therapy may encourage the formation of functional, perfusable vasculature. Thus, the perfusability of newly formed vessels in dual cell–treated CLI mice was assessed using the endothelial-binding properties of LEL-lectin. LEL-lectin was intravenously administered prior to sacrifice. Subsequently, the density and diameter distribution of LEL-lectin+ vessels were analyzed (Fig. 7). Whole-mount fluorescence imaging revealed that normal, non-ischemic muscle exhibited a dense and widespread distribution of LEL-lectin+ vessels, whereas the non-treated control group displayed sparse perfusable vasculature. In contrast, dual cell treatment preserved a significantly larger area of LEL-lectin+ vessels, exhibiting a 4.4-fold increase compared to the non-treated control, as confirmed by vessel quantification (Fig. 7A, B). These findings indicate that, although numerous CD31+ vessels were observed in the non-treated control (Fig. 6), they exhibit only partial functionality. Furthermore, analysis of the diameter distribution of LEL-lectin+ vessels confirmed a higher proportion of larger-diameter, perfusable vessels in the dual cell–treated group compared with the non-treated control. Notably, vessels exceeding 50 μm in diameter comprised 18% of total perfusable vessels in dual cell–treated mice, versus only 2% in non-treated controls (Fig. 7C).
LEL-FITC was intravenously injected to assess vessel perfusability and visualize vascular structures in three dimensions. A Whole-mount fluorescence images of LEL-lectin+ vessels. Scale bar:1 mm. B Quantification of LEL-lectin+ vessels density in whole ischemic muscle. Density was measured per mm2 (***P < 0.001, One-way ANOVA test followed by Tukey’s multiple comparison test). Values are mean ± SD. C The classification of LEL-lectin+ vessels diameter distribution. D Fluorescence images of LEL-lectin+ and α-SMA+ vessels in ischemic muscle. Scale bar: 100 μm. Quantification of LEL-lectin+ vessel based on branches (E) and junctional number (F) (*P < 0.05, **P < 0.01, ***P < 0.001, n.s.: not significant, One-way ANOVA test followed by Tukey’s multiple comparison test). Values are mean ± SD. G Measurement of LEL-lectin+ vessels diameters in each group (***P < 0.001, One-way ANOVA test followed by Tukey’s multiple comparison test). Values are mean ± SE. H Quantification of α-SMA+ cell coverage of LEL-lectin+ vessels (**P < 0.01, One-way ANOVA test followed by Tukey’s multiple comparison test). Values are mean ± SD. I Representative fluorescence images of LEL-lectin+ and PDGFR+ vessels. Scale bar: 100 μm. High magnification view of yellow and blue box areas, respectively. Orange scale bar: 50 μm. N = 8/group.
High-magnification imaging was subsequently used to evaluate the quality of perfusable vessels by identifying LEL-lectin+ microvessels and their association with perivascular cells within the perfused vascularized regions. α-SMA, a marker of smooth muscle cells, was used to identify perivascular coverage. This was followed by quantitative analyses of vessel branching, junctions, diameters, and perivascular cell coverage. The non-treated control group exhibited irregular LEL-lectin+ vessels with small diameters, whereas both the normal and dual cell–treated groups displayed compact, thick, and perfusable vessels (Fig. 7D). The number of branches and junctions of LEL-lectin+ vessels was significantly higher in the non-treated group compared to both the normal and dual cell-treated groups (Fig. 7E, F). However, vessel diameter analysis revealed that the non-treated control predominantly contained small-caliber vessels, while dual cell-treated CLI mice developed vessels with significantly larger diameters, indicative of more mature and functional vasculature (Fig. 7G). Moreover, the proportion of α-SMA+ vessels among LEL-lectin+ vessels was approximately 20% in the non-treated control, but increased to 52% in dual cell–treated mice, a level comparable to that in normal muscle (Fig. 7H and Supplementary Fig. 12). Detection of human CD31 in LEL-lectin+ vessels confirmed that human CD31+ cells were found to be partially colocalized with LEL-lectin+ vessels, indicating their integration into the functional vasculature (Supplementary Fig. 13).
Pericytes play a crucial role in maintaining the structural integrity and function of small blood vessels, such as capillaries. PDGFR (PDGF receptor) was used to assess pericyte attachment. In normal muscle, PDGFR+ pericytes were densely associated with LEL-lectin+ vessels, whereas the non-treated control exhibited a high density of thin LEL-lectin+ vessels lacking PDGFR+ pericyte coverage. However, dual cell therapy reestablished an environment in which LEL-lectin+ vessels exhibited proper association with pericytes, forming thicker and more structured vasculature (Fig. 7I).
These data corroborate that CLI induction results in a low proportion of perfusable small vessels relative to total vascular density, often lacking perivascular cell coverage, whereas dual cell treatment effectively restores large-caliber perfusable vessels accompanied by perivascular cells at the injury site. This difference in vascular architecture is likely a key factor contributing to the enhanced therapeutic outcomes.
Discussion
Stem cell therapy is being extensively evaluated as a treatment option for incurable patients with inflammatory and vascular diseases, owing to its immunomodulatory and angiogenic properties. To achieve effective tissue regeneration through cell therapy, transplanted cells must not only survive in the hostile microenvironment of the injured tissue but also successfully integrate and function within the host tissue. However, a key challenge limiting clinical success is the typically low survival rate and rapid loss of transplanted cells shortly after administration. This limited persistence is due to factors including immune rejection, ischemic conditions, inflammation, and a lack of suitable trophic support in the damaged tissue microenvironment. Ongoing strategies to enhance the survival and integration of transplanted cells include co-delivery with supportive cell populations, preconditioning or genetic modification to increase tolerance to ischemia and apoptosis, encapsulation within protective biomaterials, and modulation of the host microenvironment to facilitate tissue regeneration36,37.
The ultimate treatment of vascular diseases requires neovascularization, which can be achieved either through the sprouting of new vessels from existing vasculature or via de novo vessel formation. A single type of adult stem cell, such as MSCs or EPCs, has been commonly administered to patients with CLI to enhance perfusion. However, their therapeutic benefits are primarily attributed to paracrine mechanisms rather than direct engraftment or integration into host tissue. These paracrine effects involve the secretion of a broad range of growth factors, cytokines, and extracellular vesicles that promote angiogenesis, tissue regeneration, immunomodulation, and recruitment of endogenous repair cells. This paracrine activity is considered the principal mode of action driving the regenerative effects seen in CLI models and clinical trials9,38. In the context of using two different cell types as a therapeutic strategy for vascular disease, several studies have reported synergistic effects. For example, combination stem cell therapy using dental pulp stem cells and human umbilical vein endothelial cells has demonstrated superior therapeutic efficacy in CLI compared with either single-cell treatment alone.39 Likewise, enhanced angiogenic potency has been observed with MSCs and mast cells in a CLI model.40 However, in most of these studies, the therapeutic benefits have been attributed predominantly to paracrine-mediated angiogenesis rather than to the direct structural incorporation of transplanted cells into newly formed vessels. In addition, many of these approaches rely on cell sources that are not autologously available, which may limit their translational feasibility for CLI patients.
Thus, the rationale for this study was that, although extensive research and numerous attempts have been made at therapeutic neovascularization for the treatment of PAD, the use of stem cell from same source without genetic modification was not explored and more importantly, functional limb salvage has not been achieved through the direct participation of stem cells in arteriole formation41.
Our group were the first to identify CD141+ VPCs in the BM and demonstrated that combined transplantation of CD141+ VPCs and BM-MSCs in a mouse model of CLI facilitated the integration of transplanted cells into the host vasculature, resulting in restored blood perfusion and, ultimately, limb salvage7. These findings underscore the importance of employing dual, rather than single, cell types to establish new vasculature in damaged tissue.
We aimed to develop a novel therapeutic strategy to induce neovascularization in ischemic limbs by transplanting a combination of stem cell populations derived from AT, as an alternative to BM–derived cells, despite the reported superior efficacy of BMSCs compared to ADSCs42. In the present study, we report, to our knowledge for the first time, the identification of a VMSC population derived from AT. Importantly, while prior studies have focused on the therapeutic potential of CD31+ endothelial cells from human AT, VMSCs represent a phenotypically and functionally distinct population with superior capacity for vascular regeneration. VMSCs exhibited characteristics comparable to those of ADSCs with respect to MSC-specific marker expression and multipotent differentiation capacity. However, angiogenic protein profiling revealed that endothelial markers-including CD141, CD31, VE-cadherin, LYVE-1, ETV-2, and FGFR2—were expressed at significantly higher levels in VMSCs compared to ADSCs (Fig. 2). In contrast, ADSCs predominantly expressed perivascular markers such as α-SMA, Transgelin and TNAP. Moreover, the two cell types exhibited distinct growth factor secretion profiles, and tube formation capacity was observed exclusively in VMSCs. These findings indicate that VMSCs possess distinct cellular characteristics that differentiate them from ADSCs, supporting the notion that VMSCs may function as endothelial-like cells, whereas ADSCs primarily serve as perivascular support cells.
Tube formation assays with fluorescently labeled VMSCs and ADSCs revealed their distinct spatial roles in vascular structures. VMSCs formed elongated tubular frameworks, while ADSCs adhered to and supported the tubular structures generated by VMSCs (Supplementary Fig. 4).
VMSCs exhibit key characteristics comparable to BM–derived VPCs, including expression of markers such as CD141, multipotency, robust survival, and tube-forming ability, while differing in population doubling time and growth factor profiles in vitro7. These attributes position VMSCs as a potent and clinically promising alternative to VPCs for vascular regeneration.
Interestingly, exposure to high concentrations of TNF-α, simulating a severe inflammatory environment in vivo, did not impair viability and vascular potential. Instead, it appeared to establish a microenvironment conducive to cell migration and angiogenesis, likely through modulation of protease activity and growth factor secretion (Fig. 3). Notably, the co-existence of VMSCs and ADSCs preconditioned with TNF-α promoted the formation of compact vascular structures in vitro. The coordinated action of VMSCs and ADSCs in vitro (Fig. 3) demonstrates their synergistic effects on vascular formation and paracrine signaling. Accordingly, we hypothesized that co-transplantation of VMSCs and ADSCs would enhance neovascularization in CLI, thereby improving tissue perfusion and preserving limb function in vivo. Consistent with this hypothesis, intramuscular delivery of the dual-cell population markedly improved blood flow to the ischemic limb, prevented limb loss, and attenuated fibrosis in the CLI mouse model. These findings suggest that the coordinated actions of endothelial-like VMSCs and perivascular ADSCs are essential for establishing stable arteriolar networks capable of restoring perfusion under ischemic stress. Importantly, therapeutic benefits were observed only with the dual-cell therapy, whereas either VMSCs or ADSCs alone failed to produce meaningful efficacy (Supplementary Fig. 14). These highlight the synergistic contribution of endothelial-like and perivascular cell populations and underscore the potential of autologous AT-derived dual-cell therapy as a clinically feasible strategy for vascular regeneration in patients with CLI.
Analysis of vascular density and quality in injured tissue revealed that CLI induced numerous irregular, thin vessels lacking perivascular cell coverage. In contrast, dual cell therapy promoted the formation of perfusable, large-diameter vessels consistently surrounded by perivascular cells, despite a relatively lower overall CD31+ vessel density. These findings indicate that ischemia-induced excessive and disorganized vessel formation is insufficient for restoring blood perfusion and is likely inadequate to prevent limb loss. Instead, the presence of structurally mature blood vessels, including arterioles accompanied by perivascular cells, appears to be essential for effective perfusion recovery in CLI—a condition that can be achieved through dual cell therapy.
It is widely known that the majority of transplanted stem cells are rapidly lost within several days after administration, reinforcing the notion that their therapeutic benefits are predominantly mediated by paracrine signaling rather than long-term engraftment. However, dual cells were obviously detected within functional large-caliber vessels at 28 days post-transplantation (Fig. 5), representing a distinctive observation that underscores the unique therapeutic advantage of the dual cell approach, distinguishing it from conventional stem cell therapy. Moreover, approximately 50% of the human cell–derived vessels exhibited a large diameter with perivascular cell coverage, indicating that dual cell therapy directly contributes to arteriole-like vessel formation.
Although this study primarily focused on smooth muscle cell-covered vasculature, such as arterioles, pericytes also represent key perivascular cells that modulate the function of small-sized vessels, including capillaries. As shown in Fig. 7I, CLI seldom generated a vascular environment with substantial pericyte coverage; rather, it was characterized by a disorganized network of small-caliber vessels, even within limited vascularized regions. In contrast, dual cell therapy markedly promoted the formation of PDGFR+ pericyte-associated vessels with significantly larger diameters compared to the non-treated control. These findings consistently highlight the critical role of perivascular cell coverage and vessel caliber in achieving effective perfusion recovery.
Our study demonstrates the therapeutic potential of dual-cell therapy for CLI and highlights its clinical applicability. A key consideration for successful translation is the feasibility of obtaining both stem cell populations from patients and implementing an autologous dual-cell approach in the clinical setting. Autologous cell therapies may face challenges related to patient-specific factors, including age, comorbidities, and variability in stem-cell function. In addition, surgical procedures could impose additional burden on patients with advanced peripheral vascular disease. Nevertheless, adipose tissue harvesting is relatively minimally invasive compared with BMA or vascular tissue biopsy, thereby providing a practical and patient-friendly cell source. Moreover, establishment of an optimized culture protocol that enables efficient expansion and functional recovery of cells with initially reduced biological activity allows consistent quality of therapeutic cells across variable donor conditions. These findings and translational preparations support the clinical applicability of autologous adipose-derived VMSCs and ADSCs for patients with CLI.
Although this study clearly corroborated the therapeutic efficacy of dual-cell therapy in CLI, several limitations should be acknowledged. First, the follow-up period was relatively short, and longer-term investigations will be necessary to determine the durability of neovascularization, the stability of newly formed vessels, and any potential late-onset adverse effects. Second, all in vivo experiments were performed in immunodeficient mice, which may not fully recapitulate the host immune responses encountered in clinical settings and could influence the interpretation of therapeutic outcomes. All in vivo experiments were conducted using young male mice to minimize biological variability and ensure stable baseline conditions. Female mice are less commonly used in CLI models because estrous cycle–associated hormonal fluctuations can influence inflammation, vascular remodeling, and neovascularization, potentially confounding therapeutic outcomes. With respect to age, although aged animals may better reflect clinical settings, evaluation of human stem cell therapies is typically performed in immunodeficient nude mice, in which aging substantially increases susceptibility to infection and health instability, limiting their suitability for long-term ischemia studies. Therefore, young male mice were selected to allow reliable longitudinal assessment under controlled conditions. Nevertheless, we acknowledge that sex- and age-related factors may affect therapeutic responses, and future studies incorporating female and aged models will be important to assess the broader applicability of our findings. These limitations should be considered when extrapolating our findings to future clinical applications.
Collectively, these findings highlight the therapeutic potential of autologous adipose-tissue–derived dual-cell therapy as a clinically translatable strategy to enhance vascular regeneration and preserve limb function in patients with CLI. Future studies will be required to refine dosing, assess long-term safety, and validate efficacy in clinically relevant settings, but the present results provide a strong foundation for advancing dual-cell therapy toward clinical application.
Methods
Cell culture
Human adipose tissues were obtained from 13 donors (Supplementary Table 1). For harvesting stromal vascular fraction (SVF), the adipose tissues were chopped and enzymatically digested with collagen type I (Nordmark Pharma GmbH, Uetersen, Germany) at 37 °C for 1 h. The SVF was neutralized with an equal volume of 10% fetal bovine serum (FBS). The SVF and FBS mixture was passed through a 100 µm strainer and centrifuged at 500 g at 4 °C for 5 min. For VMSCs culture, SVFs were plated on Humatein-coated flask (ROKIT Healthcare, Seoul, Korea) in FBS-depleted Endothelial growth medium-2 (EGM-2; Lonza, Basel, Switzerland) supplemented with 2% human platelet lysate (hPL; PL BioScience, Aachen, Germany) and 2 IU/ml heparin (Sigma-Aldrich, St. Louis, MO, USA). For ADSCs culture, SVFs were plated on flask in StemMACS MSC Expansion medium (Miltenyi Biotec, Bergisch Gladbach, Germany). The cells were incubated at 37 °C in 5% CO2. The medium was changed once every 2 days. Subculture was performed when cells reached 80–90% confluency. The cells were observed by inverted research microscope Eclipse TS100 (Nikon, Tokyo, Japan). The estimated cumulative cell number and population doubling time were calculated based on cell counts.
Flow cytometry
VMSCs and ADSCs were blocked with a human FcR blocking reagent (Miltenyi Biotec), then centrifuged at 500 g at 4 °C for 5 min. The cells were incubated with allophycocyanin (APC)-conjugated antibodies against CD29, CD34, CD44, CD45, CD73, CD90, CD105, and CD141 (Miltenyi Biotec). An APC-conjugated isotype control was used as a negative control. The cells were analyzed by NovoCyte 3000 flow cytometer (Agilent Technologies, Santa Clara, CA, USA) and NovoExpress software (Agilent Technologies).
Adipogenic, osteogenic, and chondrogenic differentiation
To examine adipogenic and osteogenic differentiation potential, VMSCs and ADSCs were seeded on 6-well plates and treated with the StemPro adipogenesis differentiation media (Thermo Fisher Scientific, Waltham, MA, USA) and StemPro osteogenic differentiation media (Thermo Fisher Scientific), respectively. Differentiation media were refreshed every three days. Adipogenic differentiation was terminated and fixed on day 14 using 3.7% formaldehyde (Sigma-Aldrich), whereas osteogenic differentiation was fixed on day 21. Adipogenic differentiation was assessed by Oil Red O staining to visualize intracellular lipid droplets. Osteogenic differentiation was confirmed using Alizarin Red S staining, which detects calcium deposition. To induce chondrogenic differentiation, pellets of VMSCs and ADSCs were cultured in 15 mL conical tubes using StemPro™ chondrogenic differentiation medium (Thermo Fisher Scientific). The medium was changed every 3 days and cell pellets was fixed on day 28 using 3.7% formaldehyde (Sigma-Aldrich). The pellets were processed, embedded in paraffin, and sectioned at a thickness of 5 μm. The sections were then deparaffinized, rehydrated, and stained using an Alcian Blue staining kit (Abcam, Cambridge, UK). The stained samples were observed by Eclipse TS100 microscope (Nikon).
Western blot
Protein lysates from VMSCs and ADSCs were prepared using cell lysis buffer (Cell Signaling Technology, Danvers, MA, USA) supplemented with 2 mM phenylmethylsulfonyl fluoride (PMSF; Sigma-Aldrich) and benzonase (Sigma-Aldrich). Protein concentrations were determined using a bicinchoninic acid (BCA) assay kit (Thermo Fisher Scientific). Protein lysates were separated by SDS-PAGE and transferred onto polyvinylidene difluoride (PVDF) membranes. Membranes were blocked with 5% skim milk in TBS-T and incubated with primary antibodies against CD141, CD31, Lymphatic Vessel Endothelial Receptor 1(LYVE1), Fibroblast growth factor receptor 2 (FGFR 2), ETS variant transcription factor 2 (ETV 2), alpha smooth muscle actin (α-SMA), Transgelin and GAPDH (Abcam), c-MET and vascular endothelial-cadherin (VE-cadherin, Cell Signaling Technology), and Tissue-nonspecific alkaline phosphatase (TNAP, GeneTex, Irvine, CA, USA). Detection was performed using horseradish peroxidase (HRP)-conjugated secondary antibodies (BD, San Jose, CA, USA). The membranes were developed using chemiluminescent substrate (CYANAGEN, Bologna, Italy). The signals were detected using chemiluminator (Vilver, Marne-la-Vallée, France). Band intensities were quantified using NIH ImageJ software.
Tube formation assay
Ten microliters of Matrigel (Corning, Corning, NY, USA) was plated onto the μ-slide (ibidi, Gräfelfing, Germany) and incubated at 37 °C for 30 min to allow gelation. Tube formation assays were performed using 7.5 × 103 cells, including VMSCs, ADSCs (monocultures), and a VMSC: ADSC co-culture at a 2:1 ratio. The assay was conducted in α-MEM (Thermo Fisher Scientific) supplemented with 0.2% hPL, with or without 500 pg/ml tumor necrosis factor-alpha (TNF-α). After 18 h, vessel-like structures were visualized using an inverted research microscope (Eclipse TS100, Nikon). Quantitative analysis of mesh numbers, master segments, isolated segments, and total master segment length was performed using the Angiogenesis Analyzer plugin in NIH ImageJ.
Enzyme-linked immunosorbent assay (ELISA)
To measure the secretion of human angiogenic growth factors, the conditioned media of VMSCs and ADSCs were measured by the human VEGF, HGF, Angiogenin, and uPA ELISA Kit (R&D system) according to the manufacturer’s instructions. Absorbance was measured at 450 nm using an ELISA microplate reader (Molecular Devices, Sunnyvale, CA, USA).
Gelatin zymography
The conditioned media of VMSCs and ADSCs were separated by SDS-PAGE containing 0.1% gelatin under non-reducing conditions. The separated proteins in the gel were renatured for 30 min. Subsequently, the gels were incubated with developing buffer at 37 °C overnight and then stained with Coomassie Brilliant Blue R-250 solution (Bio-Rad, Hercules, CA, USA) for 2 h. Stained gels were destained until clear zones of proteolysis became visible against the blue background, indicating gelatin digestion. The digested areas were quantified using NIH ImageJ software.
Induction of CLI experimental model
All animal experiments were approved and performed according to the Ethics Committee for Experimental Animals of Kyung Hee University Hospital (Approval number: KHMC-IACUC 20-008). Five-week-old male BALB/C nude mice (18–20 g) for xenograft experiments were purchased from DBL (Daehan Bio Link, Seoul, Korea). Mice were housed in a specific pathogen-free animal room under controlled conditions at 55–60% humidity, 12/12-h light dark cycle, 21 °C, and ad libitum access to food and water. Mice were anesthetized by intraperitoneal injection of Ketamine (75 mg/kg; Yuhan, Seoul, Korea) and Rompun (1.2 mg/kg; Bayer Healthcare, Gyeonggi-do, Korea). To achieve complete sedation, anesthesia was induced with 5% isoflurane (JW Pharmaceutical Corporation, Gyeonggi-do, Korea). Unilateral femoral artery ligation and excision were performed in mice as previously described7. Briefly, an incision was made along the medial aspect of the right hindlimb to expose the common and superficial femoral arteries. The proximal portion of the common femoral artery and the mid-to-distal segments of the superficial femoral artery were then ligated using 6–0 silk sutures. Subsequently, the arterial segment between the ligation points in the superficial femoral artery was excised. Hindlimb blood flow was assessed using a laser Doppler blood flow imager (OMEGAZONE OZ-2, OMEGAWAVE, Tokyo, Japan). Mice exhibiting hindlimb blood flow below 40% of the baseline were selected for subsequent experiment. For analysis, all mice were euthanized with CO2 (3 L/min), followed by bilateral thoracotomy.
Cell transplantation
A total of 2.4 × 105 cells per mouse, consisting of human adipose-derived passage 2 VMSCs (1.6 × 105 cells) and ADSCs (8.0 × 104 cells), was suspended in 50 μL of Plasma Solution A (HK inno.N Corporation, Chuncheongbuk-do, Korea). The cell suspension was injected intramuscularly into the ischemic gastrocnemius muscle, delivering 10 μl at each of five injection sites. Plasma Solution A without cells was used as a vehicle control.
Evaluation of ischemic score
At 0, 1, 2, 3, 7, 14, and 28 days after cell treatment, limb morphology was visually assessed to evaluate the extent of ischemia-induced damage, as previously described7. Briefly, seven-grades scoring system were used to assess the extent of limb injury. Grade 0, Normal (limb salvage); Grade 1, discoloration, swelling, and nail necrosis; Grade 2, mild necrosis of the toe, Grade 3, severe necrosis of toe (necrosis near the walking pad); Grade 4, foot necrosis (below the ankle joint); Grade 5, knee necrosis (above the ankle joint); Grade 6, limb loss.
Blood flow analysis
Blood flow was assessed using a Laser Doppler blood flow imager (OMEGAZONE OZ-2). Mice were anesthetized, and hindlimb blood flow was assessed by measuring signal intensities generated by laser scattering of red blood cells. The perfusion ratio between the ischemic and contralateral non-ischemic limb was calculated using LIA v4.2 software (OMEGAWAVE).
Histological analysis
Hindlimb muscles were harvested 28 days after cell treatment and fixed in 3.7% formaldehyde (Sigma-Aldrich) for histological analysis. The tissues were then processed, embedded in paraffin, and sectioned at a thickness of 5 μm. For immunofluorescence staining, the sections were deparaffinized by immersing in xylene (Daejung, Gyeonggi-do, Korea) and rehydrated. Antigen retrieval was performed by boiling the sections in 10 mM sodium citrate buffer (Sigma-Aldrich) for 15 min. To reduce tissue autofluorescence, sections were treated with 0.01% sodium borohydride (Sigma-Aldrich) for 1 h, followed by permeabilization with 0.3% Triton X-100 (Sigma-Aldrich) for 15 min. Samples were incubated in Alexa-488 conjugated wheat germ agglutinin (WGA, Thermo Fisher scientific) at 37 °C for 2 h and subsequently treated with 2% normal goat serum (Vector Laboratories, Inc., Newark, CA, USA). Primary antibodies targeting CD31, Transgelin, α-SMA and CD141 were applied, followed by incubation with Cy3-conjugated secondary antibodies (Jackson ImmunoResearch, West Grove, PA, USA). Nuclei were counterstained with 4′,6-diamidino-2-phenylindole (DAPI; Sigma-Aldrich), and sections were cover-slipped using mounting medium (Vector Laboratories, Inc.). Imaging was conducted with a Carl Zeiss confocal microscope (Oberkochen, Germany), and data analysis was performed using ZEN Microscopy software (Carl Zeiss). Quantification of vessel size was restricted to luminal structures with a diameter >5 µm, thereby excluding small-diameter capillaries from the analysis.
To detect F4/80+ macrophages, paraffin-embedded tissue sections were incubated with an anti-F4/80 primary antibody (Abcam), followed by a biotinylated goat secondary antibody for 1 h. The positive signal was visualized using the Vector Blue AP Substrate Kit (Vector Laboratories), and nuclei were counterstained with Fast Red.
For hematoxylin and eosin (H&E) staining, sections were deparaffinized in xylene and rehydrated through graded alcohols. The tissues were then stained with hematoxylin (Sigma-Aldrich) followed by eosin (BBC Biochemical, Mount Vernon, WA, USA). Imaging was performed using a KB-600 Biological Microscope (Korea Lab Tech, Gyeonggi-do, Korea).
For Masson’s Trichrome staining, sections were deparaffinized in xylene and rehydrated. Staining was performed using the NovaUltra™ Masson’s Trichrome Stain Kit (IHC World, Ellicott City, MD, USA) according to the manufacturer’s instructions. Imaging was conducted with a KB-600 Biological Microscope (Korea Lab Tech, Gyeonggi-do, Korea). Quantification of blue-stained collagen fibers was carried out using the Color Deconvolution 2 plugin in NIH ImageJ.
LEL (Lycopersicon Esculentum Lectin) perfusion
LEL (Vector Laboratories, Inc.) was prepared at a concentration of 100 μg/ml. Four weeks following the induction of limb ischemia and transplantation of dual cells, LEL conjugated with FITC was administered intravenously to visualize newly formed blood vessels. After injection, the mice were placed on a heated pad for 20 min to facilitate LEL-FITC perfusion, after which they were sacrificed.
Tissue clearing and 3D immunostaining
The muscles were briefly washed with PBS and then fixed in 4% paraformaldehyde overnight. After fixation, the muscles were incubated in a tissue clearing solution (Binaree Inc., Daegu, Korea) on a shaker for 7 days at 37 °C. Following the clearing process, the muscles were washed three times with cold PBS on a shaker for 30 min each at 4 °C. This was followed by permeabilization/blocking using a buffer containing 0.5% Triton X-100, 15% DMSO, and 5% NGS in PBS for 2 days at 37 °C. Primary antibody incubation was performed at 37 °C on a shaker for 3 days in an antibody diluent solution (0.2% Tween 20, 5% DMSO, and 5% NGS in PBS) using the following antibodies: Anti-CD31 (Abcam), α-SMA conjugated to Alexa Fluor 647 (Abcam), α-SMA (DAKO, Glostrup, Denmark), and Platelet-derived growth factor receptors (PDGFR, Abcam). Afterward, the muscle tissues were washed three times with cold PBS on a shaker (30 min each), then incubated for 4 days at 37 °C with secondary antibodies in antibody diluent solution: FITC (Vector Laboratories, Inc.) and Alexa Fluor 647 (Invitrogen, Waltham, MA, USA). Following antibody incubation, the muscles were washed, and then mounted in mounting solution (Binaree Inc.) at room temperature for 2 days. Whole-mount images were captured using a Zeiss confocal microscope.
Statistical analysis
Data are presented as mean ± standard deviation (SD). Statistical analysis was performed with GraphPad Prism 5 (GraphPad, San Diego, CA, USA). Depending on the experimental design, paired or unpaired t tests, or one-way analysis of variance (ANOVA), or two-way ANOVA were used to determine statistical significance. ANOVA was followed by post hoc tests such as Tukey’s or Bonferroni’s multiple comparison test. P < 0.05 was considered statistically significant. *P < 0.05, **P < 0.01, and ***P < 0.001.
Ethics approval information
Human cell donors provided informed consent prior to sample collection. This study was approved by Kyung Hee University Hospital (Approval number: KHUH 2021-01-011-013) and it followed the principles outlined in the Declaration of Helsinki. Written informed consent was obtained from all human participants.
Data availability
All data generated or analyzed during this study will be shared by the lead contact upon request.
References
Golledge, J. Update on the pathophysiology and medical treatment of peripheral artery disease. Nat. Rev. Cardiol. 19, 456–474 (2022).
Qadura, M., Terenzi, D. C., Verma, S., Al-Omran, M. & Hess, D. A. Concise review: cell therapy for critical limb ischemia: an integrated review of preclinical and clinical studies. Stem Cells 36, 161–171 (2018).
Jansen-Chaparro, S. et al. Statins and peripheral arterial disease: a narrative review. Front. Cardiovasc. Med. 8, 777016 (2021).
Dormandy, J. A. & Rutherford, R. B. Management of peripheral arterial disease (PAD). TASC Working Group. TransAtlantic Inter-Society Consensus (TASC). J. Vasc. Surg. 31, S1–296 (2000).
Gupta, R. & Losordo, D. W. Cell therapy for critical limb ischemia: moving forward one step at a time. Circ. Cardiovasc. Interv. 4, 2–5 (2011).
Shirbaghaee, Z., Hassani, M., Heidari Keshel, S. & Soleimani, M. Emerging roles of mesenchymal stem cell therapy in patients with critical limb ischemia. Stem Cell Res. Ther. 13, 462 (2022).
Park, G. et al. Identification of CD141+vasculogenic precursor cells from human bone marrow and their endothelial engagement in the arteriogenesis by co-transplantation with mesenchymal stem cells. Stem Cell Res. Ther. 15, 388 (2024).
Kolf, C. M., Cho, E. & Tuan, R. S. Mesenchymal stromal cells. Biology of adult mesenchymal stem cells: regulation of niche, self-renewal and differentiation. Arthritis Res. Ther. 9, 204 (2007).
Lozano Navarro, L. V. et al. Mesenchymal stem cells for critical limb ischemia: their function, mechanism, and therapeutic potential. Stem Cell Res. Ther. 13, 345 (2022).
Bagno, L., Hatzistergos, K. E., Balkan, W. & Hare, J. M. Mesenchymal stem cell-based therapy for cardiovascular disease: Progress and challenges. Mol. Ther. 26, 1610–1623 (2018).
Guo, Y., Peng, Y., Zeng, H. & Chen, G. Progress in mesenchymal stem cell therapy for ischemic stroke. Stem Cells Int. 2021, 9923566 (2021).
Yang, Q. et al. Combined transplantation of adipose tissue-derived stem cells and endothelial progenitor cells improve diabetic erectile dysfunction in a rat model. Stem Cells Int. 2020, 2154053 (2020).
Kim, D. Y. et al. Platelet-derived growth factor-BB priming enhances vasculogenic capacity of bone marrow-derived endothelial precursor like cells. Tissue Eng. Regen. Med. 20, 695–704 (2023).
Alev, C., Ii, M. & Asahara, T. Endothelial progenitor cells: a novel tool for the therapy of ischemic diseases. Antioxid. Redox Signal. 15, 949–965 (2011).
MacAskill, M. G. et al. Robust revascularization in models of limb ischemia using a clinically translatable human stem cell-derived endothelial cell product. Mol. Ther. 26, 1669–1684 (2018).
Cheng, F. et al. Conversion of human adipose-derived stem cells into functional and expandable endothelial-like cells for cell-based therapies. Stem Cell Res. Ther. 9, 350 (2018).
Minamino, K. et al. Macrophage colony-stimulating factor (M-CSF), as well as granulocyte colony-stimulating factor (G-CSF), accelerates neovascularization. Stem Cells 23, 347–354 (2005).
Popescu, S. et al. Dual stem cell therapy improves the myocardial recovery post-infarction through reciprocal modulation of cell functions. Int. J. Mol. Sci. 22, 5631 (2021).
Kuo, T. K. et al. Stem cell therapy for liver disease: parameters governing the success of using bone marrow mesenchymal stem cells. Gastroenterology 134, 2111–2121 (2008).
Daneshmandi, L. et al. Emergence of the stem cell secretome in regenerative engineering. Trends Biotechnol. 38, 1373–1384 (2020).
Kwon, J. W. et al. Mesenchymal stem cell-derived secretomes-enriched alginate/ extracellular matrix hydrogel patch accelerates skin wound healing. Biomater. Res. 27, 107 (2023).
Park, S. R. et al. Stem cell secretome and its effect on cellular mechanisms relevant to wound healing. Mol. Ther. 26, 606–617 (2018).
Compagna, R. et al. Cell therapy in patients with critical limb ischemia. Stem Cells Int. 2015, 931420 (2015).
Hussein, E. A. Stem cell therapy for vascular disorders. Vasc. Endovasc. Rev. 1, 17-21 (2018).
Cartland S. P. et al. The generation of stable microvessels in ischemia is mediated by endothelial cell derived TRAIL. Sci. Adv. 10, eadn8760 (2024).
Pittenger, M. F. et al. Mesenchymal stem cell perspective: cell biology to clinical progress. Npj Regen. Med. 4, 22 (2019).
Benabid, A. & Peduto, L. Mesenchymal perivascular cells in immunity and disease. Curr. Opin. Immunol. 64, 50–55 (2020).
Lee, H. Huh, Y. H. & Kang, K. T. Mesenchymal stem cells potentiate the vasculogenic capacity of endothelial colony-forming cells under hyperglycemic conditions. Life 12, 469 (2022).
Iqbal, F. et al. Combination human umbilical cord perivascular and endothelial colony forming cell therapy for ischemic cardiac injury. Npj Regen. Med. 8, 45 (2023).
Rojas-Torres, M. et al. Unraveling the differential mechanisms of revascularization promoted by MSCs & ECFCs from adipose tissue or umbilical cord in a murine model of critical limb-threatening ischemia. J. Biomed. Sci. 31, 71 (2024).
Barros Ferreira, L. et al. Effects of tumor necrosis factor-α and interleukin-1β on human retinal endothelial cells. Cytokine 173, 156407 (2024).
Kim, D. Y. et al. Substance P ameliorates TNF-α-mediated impairment of human aortic vascular cells in vitro. Clin. Exp. Pharmacol. Physiol. 48, 1288–1297 (2021).
González-Flores, D., Rodríguez, A. B. & Pariente, J. A. TNFα-induced apoptosis in human myeloid cell lines HL-60 and K562 is dependent of intracellular ROS generation. Mol. Cell Biochem. 390, 281–287 (2014).
Honkura, N. et al. Intravital imaging-based analysis tools for vessel identification and assessment of concurrent dynamic vascular events. Nat. Commun. 9, 2746 (2018).
Thurston, G. et al. Angiogenesis in mice with chronic airway inflammation: strain-dependent differences. Am. J. Pathol. 153, 1099–1112 (1998).
Zama, N. & Toda, S. Designer cell therapy for tissue regeneration. Inflamm. Regen. 44, 15 (2024).
Iismaa, S. E. et al. Comparative regenerative mechanisms across different mammalian tissues. Npj Regen. Med. 3, 6 (2018).
Sanz-Nogués, C. & O’Brien, T. MSCs isolated from patients with ischemic vascular disease have normal angiogenic potential. Mol. Ther. 22, 1888–1889 (2014).
Kim, C. K. et al. Combination stem cell therapy using dental pulp stem cells and human umbilical vein endothelial cells for critical hindlimb ischemia. BMB Rep. 55, 336–341 (2022).
Amani, S. et al. Angiogenic effects of cell therapy within a biomaterial scaffold in a rat hind limb ischemia model. Sci. Rep. 11, 20545 (2021).
Kim, J. J. et al. Vascular regeneration and skeletal muscle repair induced by long-term exposure to SDF-1α derived from engineered mesenchymal stem cells after hindlimb ischemia. Exp. Mol. Med. 55, 2248–2259 (2023).
Nammian, P. et al. Comparative analysis of mouse bone marrow and adipose tissue mesenchymal stem cells for critical limb ischemia cell therapy. Stem Cell Res. Ther. 12, 58 (2021).
Acknowledgements
This study was supported by Korean Fund for Regenerative Medicine (KFRM) grant funded by the Korea government (the Ministry of Science and ICT, the Ministry of Health & Welfare) (23C0110L1, 25A0205L1).
Author information
Authors and Affiliations
Contributions
D.Y.K. performed experiments, analyzed the data (in vivo and vitro) and wrote the manuscript draft; D.Y.H. performed experiments, analyzed the data (in vivo) and wrote the manuscript draft; G.P. performed experiments and analyzed the data (in vitro); Y.J.S. performed experiments (in vivo); J.K. performed experiments and analyzed the data (in vitro); S.V.Y., H.S.H., and Y.S. designed these studies, coordinated the project, analyzed data and provided financial support. H.S.H. performed experiments, analyzed the data (in vivo), supervised the study and finalized the manuscript. All authors read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Kim, D.Y., Hwang, D.Y., Park, G. et al. Adipose-derived dual cell therapy enhances arteriogenesis and limb preservation through vascular integration in critical limb ischemia. npj Regen Med 11, 13 (2026). https://doi.org/10.1038/s41536-026-00458-x
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41536-026-00458-x