Abstract
Cognitive deficits in schizophrenia significantly hinder functional outcomes and often remain unresponsive to conventional treatments. While initial evidence suggested potential pro-cognitive effects of electrical brain stimulation in schizophrenia, recent meta-analyses have not supported these findings, warranting further investigation on intervention optimization. This sham-controlled crossover study explored cognitive and emotional effects of bilateral dorsolateral prefrontal cortex (DLPFC) anodal transcranial direct current stimulation (tDCS) and high-frequency transcranial random noise stimulation (tRNS) in schizophrenia. Thirty-six male patients with schizophrenia participated in a crossover trial, receiving three sessions (tDCS, tRNS, sham) in counterbalanced order with 1-week intervals. tDCS and tRNS sessions involved 20-min 2 mA anodal stimulation and 2 mA 100–640 Hz random noise stimulation targeting the left and right DLPFCs (F3–F4) with two extracephalic return electrodes. Executive functions (working memory, spatial planning) were assessed during stimulation, and emotional changes were measured with the Positive and Negative Affect Schedule (PANAS) pre- and post-stimulation. Additionally, side effects and blinding efficacy were evaluated. Both bilateral DLPFC anodal tDCS and high-frequency tRNS significantly improved planning performance (mean problems solved, mean number of moves) compared to sham, with tRNS additionally enhancing working memory accuracy and strategy score. Both interventions increased positive affect and reduced negative affect after the intervention, with tRNS showing greater enhancement of positive emotions. Reduced negative affect after tRNS was correlated with improved executive planning. Side effects were minimal, and blinding was effective for the sham condition. Bilateral DLPFC anodal tDCS and high-frequency tRNS show promise as adjunctive treatments for schizophrenia, especially for cognitive deficits, with broader cognitive and emotional benefits observed with tRNS. ClinicalTrials.gov Identifier: NCT06155786 https://clinicaltrials.gov/study/NCT06155786.
Similar content being viewed by others
Introduction
Schizophrenia is a complex and chronic psychiatric disorder characterized by positive symptoms (e.g., hallucinations, delusions), negative symptoms (e.g., amotivation, social withdrawal), and cognitive impairments (e.g., deficits in working memory and executive functions)1,2. In the latest Global Burden of Disease Study, Schizophrenia is among the top 20 diseases in 2021 in disability metrics3. The early term of schizophrenia as dementia praecox4 highlights the significance and relevance of cognitive deficit for the disease’s physio- and psychopathology5. These cognitive deficits significantly impair daily functioning and are often resistant to conventional antipsychotic treatments, and represent one of the main obstacles to clinical and functional recovery in affected individuals6,7. Up to 30% of patients with schizophrenia fail to achieve meaningful improvement with first-line antipsychotics, and even clozapine, a second-line treatment, yields response rates of only 30%–60% 6,8. Consequently, there is a pressing need for novel therapeutic strategies to treat schizophrenia symptoms, especially neurocognitive deficits, which are critical for improving functional outcomes in schizophrenia9.
Novel treatment strategies for schizophrenia are informed by advancing knowledge of its neurobiology, including affected brain structures, functions, and networks. Functional Magnetic Resonance Imaging (fMRI) fMRI markers reveal widespread involvement in cognitive deficits, with the frontoparietal network—key for cognitive control—strongly linked to executive functions like working memory and planning, showing structural and functional abnormalities7. The dorsolateral prefrontal cortex (DLPFC), a key brain region in this network, consistently shows aberrant activation during core cognitive deficits such as working memory tasks, with both hypo- and hyperactivation reported in schizophrenia7. Noninvasive brain stimulation provides safe ways to modify and restore these abnormal activities in cortical and subcortical regions10 and holds promise for patients with schizophrenia8,11,12. Transcranial electrical stimulation (tES), including transcranial direct current stimulation (tDCS) and transcranial random noise stimulation (tRNS), has emerged as a promising neuromodulation tool for psychiatric disorders13,14. tDCS induces sustained membrane potential shifts and ion channel modulation, with polarity-specific effects: anodal enhances excitability, cathodal reduces it15,16. Conversely, tRNS applies random pulses across frequencies and amplitudes, leveraging stochastic resonance to amplify weak neural signals17.
In schizophrenia, tES has been explored for its potential to alleviate positive, negative, and cognitive symptoms18,19, with a particular focus on targeting the DLPFC due to its role in working memory, and emotion regulation20,21,22. The DLPFC is implicated in the pathophysiology of cognitive and negative symptoms in schizophrenia, exhibiting reduced metabolism and altered connectivity7,23. The rationale for using tES in schizophrenia is strengthened by the disorder’s characteristic excitation/inhibition (E/I) imbalance. Studies show that schizophrenia is associated with reduced cortical excitability, reflected in lower E/I balance as measured by EEG24,25. Both tDCS and tRNS can increase E/I balance and enhance cortical excitability26,27, potentially counteracting these deficits. Furthermore, tRNS combined with executive function training has been shown to increase E/I balance in ADHD28, suggesting tRNS may broadly modulate neural dynamics. These findings provide a strong rationale for investigating tES as a therapeutic intervention to restore E/I balance in schizophrenia.
Early tDCS studies suggested cognitive improvements in schizophrenia via frontal connectivity changes29,30, but recent meta-analyses found no robust effects on negative symptoms31 or cognitive outcomes19, highlighting the need for optimized or novel tES protocols. In this line, a large randomized clinical trial demonstrated clinically meaningful reductions in negative symptoms with twice-daily prefrontal tDCS, underscoring that dosing optimization can enhance efficacy32. High-frequency (HF) tRNS (e.g., 100–640 Hz) has showed superior neuromodulatory effects compared to low-frequency tRNS in enhancing neuroplasticity and cognition33. It is shown that it can modulate large-scale functional network connectivity pertaining to negative symptoms of schizophrenia34. Despite its potential, tRNS remains underexplored in schizophrenia, with preliminary case reports suggesting benefits for negative symptoms35,36,37 and a completed multicenter tRNS trial38. Moreover, previous tES studies in schizophrenia have primarily utilized unilateral tDCS montages, such as anodal stimulation over the left DLPFC with a cathode over the temporoparietal cortex, showing mixed results19,39. One such reason could be insufficient modulation of interhemispheric connectivity and bilateral network imbalances in the disorder40. Only one tDCS study applied bilateral DLPFC anodal tDCS (with supraorbital cathodes) and reported reduced negative symptoms and cognitive gains41.
The current tES study has two novel aspects. The use of: (1) bilateral anodal tDCS targeting both the left and right DLPFC, with return electrodes positioned on the shoulders to minimize cortical interference by the return electrodes, and (2) high-frequency tRNS using the same electrode configuration (i.e., bilateral DLPFC). Unlike traditional montages, the extracephalic placement of return electrodes on the shoulders aims to enhance the specificity of DLPFC stimulation by reducing unintended modulation of non-target brain regions42. Furthermore, stimulating both left and right DLPFCs with the same polarity/frequency allows us to see how its modulation, regardless of laterality, which is a critical tES parameter to consider42,43, affects cognitive and emotional performance. We also assessed emotional changes following DLPFC stimulation, given the DLPFC’s critical role in emotion regulation21,22,44,45, which is known to be impaired in schizophrenia46, and monitored emotional instability before the intervention to capture short-term mood changes that may affect cognitive outcomes47.
Accordingly, the primary aim of this study was to investigate the comparable cognitive and emotional effects of bilateral DLPFC excitatory stimulation using anodal tDCS and HF-tRNS in patients with schizophrenia. We hypothesized that both anodal tDCS and HF-tRNS of bilateral DLPFCs enhance working memory and executive function, compared to sham stimulation. Furthermore, we anticipated improvements in positive affect and reductions in negative affect post-stimulation. To our knowledge, this is the first tES study that compares the effects of upregulating the bilateral DLPFC with anodal tDCS and HF-tRNS on cognitive deficits and emotional functioning in schizophrenia, addressing the need for optimized/novel neurostimulation interventions for the disease8. We followed recent consensus recommendations for transparent tES reporting (RATES checklist)48 to enhance reproducibility and comparability across studies.
Methods
Study design and participants
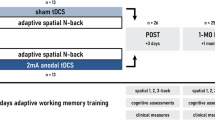
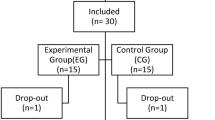
This study used a randomized crossover design. The order of the three stimulation conditions (anodal tDCS, HF-tRNS, sham) was counterbalanced across participants using a Latin square design to control for order effects. Condition sequences were generated using a computer-based random sequence generator (www.random.org) by an independent researcher not involved in data collection or analysis (Fig. 1). The sample size was calculated using a power analysis, and the average sample size in previous studies19,49. For a repeated measures ANOVA with 3 measurements, a power of 0.90, alpha of 0.05, and a medium effect size (partial eta square of 0.06 equal to f = 0.25), a minimum of 36 participants was required. Thirty-six male patients with schizophrenia (mean age = 46.11 ± 5.36) were recruited from the inpatient schizophrenia wards in the Daroshafa Hospital for Psychiatric and Chronic Diseases (manages by the National Welfare Organization, Ardabil, Iran) (see Table 1 for demographics). Inclusion criteria were: (1) Clinician-Rated Dimensions of Psychosis Symptom Severity50,51 (schizophrenia diagnosis was based on the DSM-5 criteria with no minimum symptom severity threshold applied), (2) being 18–50 years of age, (3) stable doses of antipsychotics [defined as a change of not more than 50% of the dose of antipsychotics) and all CNS-active medications for 4–6 weeks before the experiment, and (4) the ability to provide informed written consent. All participants had to meet the safety guidelines for tDCS/tRNS interventions and be fluent in their native language. Exclusion criteria included alcohol or substance dependence, history of seizures, neurological disorders, head injury, or contraindicated implants. Nicotine consumption was not excluded due to its high prevalence among patients with schizophrenia; however, intake was controlled prior to the experimental sessions. This was a registered clinical trial (ClinicalTrials.gov Identifier: NCT06155786) approved by the Ethics Committee of Payame Noor University (Ethics code: IR.PNU.REC.1401.446). Participants and their guardians gave their written informed consent before participation. Patients were free to withdraw from the study upon request.
CONSORT flow diagram of study inclusion.
Measurements
Core executive functions (working memory, spatial planning) were assessed using two subtests of the CANTAB schizophrenia battery52: the Spatial Working Memory task for working memory and the Stockings of Cambridge task for spatial planning. Emotional changes were assessed with the Positive and Negative Affect Schedule53, and tES side effects were monitored via a standard survey54. These executive functions—working memory and planning—were prioritized because they are mediated by the DLPFC, significantly impaired in schizophrenia, and critical to functional outcomes. Other cognitive deficits, such as attentional functioning and social cognition, which are also critical within the neurocognitive domains of schizophrenia9,55, were not included in this study.
Executive functions tasks
The Spatial Working Memory (SWM) and Stockings of Cambridge (SOC) tasks assess core executive functions, specifically working memory and spatial planning. The SWM task involves a computerized setup where participants locate a yellow ‘token’ hidden in colored boxes through a process of elimination, aiming to fill an empty column. The task’s difficulty increases with up to 12 boxes that vary in color and position across trials. Outcome measures used in this study include strategy scores (where lower scores indicate more effective strategy use) and total errors (defined as selecting incorrect boxes). Conversely, the SOC task, derived from the Tower of London test56, evaluates spatial executive planning and frontal lobe function by requiring participants to rearrange colored balls to match a target pattern in the fewest moves possible, progressing from simple one-move to complex multi-move problems. Outcome measures include the mean number of problems solved in minimum moves (higher scores better) and mean number of moves used at each difficulty (2–5‑move problems) level (lower scores better). Both tasks are sensitive to cognitive impairments in conditions like dementia, ADHD, and schizophrenia, providing valuable insights into executive function capabilities57 (see Supplementary Materials for details).
Positive and Negative Affect Schedule (PANAS)
The PANAS53 was used to measure emotional states. It consists of 20 items: 10 for positive affect (e.g., “enthusiastic,” “inspired”) and 10 for negative affect (e.g., “afraid,” “irritable”). Participants rate each item on a 5-point Likert scale (1 = not at all, 5 = very much) based on their current or recent feelings. Scores for positive and negative affect are calculated separately, ranging from 10 to 50. The PANAS is recognized for its high reliability, validity, and sensitivity to short-term mood changes, making it ideal for assessing baseline emotional states and changes before and after interventions, such as tDCS, especially when targeting brain regions involved in emotional regulation (e.g., prefrontal cortex)47. It also provides information about whether and how subjective estimates of affect can influence performance.
Transcranial electrical stimulation
Participants underwent three sessions of tES—anodal tDCS, high-frequency tRNS, and sham stimulation—administered in counterbalanced order with a 1-week interval. Two mA stimulation was delivered using a 4-electrode device (Neurostim2, MedinaTeb Co., Iran) via 0.9% saline-soaked sponge electrodes (7 × 5 cm- current density of 0.06 mA/cm2) positioned on target regions according to the 10-20 EEG international system. In the 2 mA tDCS protocol, bilateral anodal electrodes were placed on the left DLPFC (F3) and right DLPFC (F4), with cathodes over the right and left shoulders. The tRNS protocol used the same electrode placement with target electrodes over the left and right DLPFCs (F3–F4) and two return electrodes over the left and right shoulders, an intensity of 2 mA base to peak (without DC offset), a frequency of 100–640 Hz, and stimulation duration of 20 min. The frequency range was selected based on the results of previous studies that showed neuromodulatory effects of 100–640 Hz tRNS compared to 0–100 Hz tRNS58, although induced electrical fields are independent of stimulation frequency in tRNS33. For sham stimulation, the same electrode configuration was used, with a 30-real stimulation with a 30-s ramp-up at the start and a 30-s ramp-down at the end. Half of the sham sessions involved the application of direct current, while the other half used random noise to simulate both stimulation modalities, evenly distributed across the patients. For the remaining 18.5 min, the device was turned off without the knowledge of the patients. In all sessions, both electrodes were positioned longitudinally along the anteroposterior axis of the target regions to ensure a 6 cm distance between electrodes and were fixed with headbands. Five minutes after starting the stimulation, participants performed the SWM and SOC tasks online. After each stimulation session, participants completed a side-effect checklist54 and were asked to guess about the stimulation type (active or sham) for assessing blinding efficacy. The Report Approval for Transcranial Electrical Stimulation (RATES) checklist48 of the intervention and study design is presented in Table 2.
Procedure
Before the experiment, participants completed a brief questionnaire to evaluate their suitability for brain stimulation. All participants received 3 sessions of tES with 1-week intervals between the sessions. To avoid confounding effects of the intervention at circadian non-preferred time and sleep pressure, which can significantly affect neuroplasticity induction and cognitive performance59,60, all stimulation sessions took place at a specific time of day between 2:00 and 5:00 p.m. Cognitive measures were evaluated during each 20-min stimulation session (online), started 5 min after the beginning of the stimulation and took about 15 min to complete. This timing allowed the tasks to be performed while cortical excitability was actively modulated, enabling the assessment of state-dependent effects of tES on cognitive performance, rather than post-stimulation aftereffects, and is in line with previous studies61,62,63. To assess changes in affective states during the experimental procedure, participants completed the PANAS twice throughout each session, before and after each stimulation. This was also done to control for baseline emotional variability47.
All patients had normal or corrected-to-normal eyesight. Patients were instructed about the tasks before the beginning of the experiment. Stimulus presentation in all computerized tasks was conducted by a 15.6” screen, at a viewing distance of approximately 50 cm, and the response box was adjustable for left and right-handed individuals. To mitigate potential practice effects and stabilize performance, stimuli were presented in a randomized order for each run, and a practice session familiarized participants with the task before the baseline. Practice session data were excluded from the analysis. None of the patients received any type of psychotherapy during the course of study. All sessions were conducted in a dedicated space for research with similar environmental conditions in all sessions. Participants were blind to the study hypotheses and stimulation conditions. To maintain a double-blind design, a separate investigator prepared the device and administered the stimulation, while the experimenter who evaluated emotional changes (PANAS) and conducted the outcome measures during stimulation was blinded to the tES conditions (Fig. 2).
This study had a randomized, double-blind, crossover design. Participants were randomly assigned to three tES sessions with at least a 1-week interval: bilateral DLPFC anodal tDCS, bilateral DLPFC HF-tRNS, and sham tES. In all tES sessions, 4 electrodes were used with two active electrodes applied to the left DLPFC (F3) and right DLPFC (F4), and two return electrodes over the right and left shoulders. Participants underwent pre- and post-intervention assessments of emotional states. Additionally, executive functions (working memory and planning) were measured with the Cambridge Neuropsychological Test Automated Battery (CANTAB) during each stimulation session. After each stimulation session, participants completed a side-effect checklist and were asked to guess about the stimulation type for assessing blinding efficacy. Cognitive measures were evaluated during each 20-min stimulation session (online), started 5 min after the beginning of the stimulation and took about 15 min to complete. tES transcranial electrical stimulation, tDCS transcranial direct current stimulation, tRNS transcranial random noise stimulation, HF high frequency (100–640 Hz), DLPFC dorsolateral prefrontal cortex, F3/F4 left/right DLPFC, LS/RS left/right shoulder, PANAS Positive and Negative Affect Schedule, SWM spatial working memory, SOC Stockings of Cambridge, M mean, SD standard deviation, mA milliampere, min minute, s second.
Statistical analysis
Data analyses were conducted with the statistical package SPSS, version 26.0 (IBM, SPSS, Inc., Chicago, IL), and GraphPad Prism 8.2.1 (GraphPad Software, San Diego, California). The normality and homogeneity of data distribution and variance were confirmed by Shapiro–Wilk and Levene tests, respectively. To explore the effect of tES conditions on cognitive performance (working memory, planning), a repeated-measure ANOVA was conducted with “stimulation” (Sham, tDCS, tRNS) as the within-subject factor. Mauchly’s Test of Sphericity was employed to assess the sphericity of the data, and adjustments to degrees of freedom were made using the Greenhouse-Geisser method when necessary. In the event of significant findings in the ANOVAs, post hoc analyses were executed using Bonferroni-corrected t-tests to account for multiple comparisons. The scores on PANAS before and after each stimulation condition were analyzed with a 3 (stimulation) × 2 (emotion) × 2 (time) factorial ANOVA, and baseline PANAS scores were analyzed with a one-way ANOVA. Two-sided Pearson correlations examined relationships between tES effects on task performance and mood changes (PANAS) only when Bonferroni-corrected pre-post PANAS differences were significant, limiting tests to hypothesis-relevant cases and minimizing Type I error. These exploratory correlations between cognitive performance and mood changes were not further corrected because they were conditional on significant pre-post PANAS differences and were few in number. Side effects were analyzed with the same one-way ANOVAs for each domain. Blinding efficacy was examined with the chi-squared test for Independence on participants’ guesses of blinding (0, 1) and Bang’s Blinding Index (−1, 1)64, with negative values suggesting that participants frequently guessed the opposite of their actual treatment. The critical level of significance was 0.05 for all statistical analyses.
Results
Safety outcomes and blinding
The stimulation was well-tolerated without major side effects in all participants, and none dropped out of the study. Minor side effects were mild (mean ratings <1.0 on a 0–4 scale) and did not differ significantly between conditions (Table 3). One-way ANOVAs revealed non-significant effect of stimulation on pain (F2,70 = 0.761, p = 0.470), itching (F2,70 = 2.369, p = 0.099), fatigue (F2,70 = 0.583, p = 0.560), burning sensation (F2,70 = 0.957, p = 0.387), skin redness (F2,70 = 1.467, p = 0.235), and problems in concentration (F2,70 = 0.062, p = 0.940), indicating that side effects did not differ between tES conditions (Table 3). Regarding blinding efficacy, four patients during sham tES and one patient during tDCS and tRNS guessed no stimulation (chose 0). The results of the Chi-Square test (χ2 = 3.176; p = 0.204) and Bang’s Blinding Index (tDCS and tRNS, BBI = 0.94 each, sham BBI = −0.78) indicated that blinding for the sham condition was successful, while blinding for active tDCS and tRNS was not (negative BBI reflects effective sham credibility)65, though participants’ awareness of active stimulation did not significantly influence their perception of side effects.
tES effects on executive functions
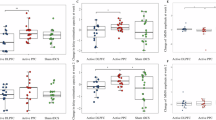
A repeated-measure ANOVA was conducted to investigate the effect of bilateral DLPFC excitatory stimulation on cognitive performance across outcome measures. For working memory, a significant main effect of stimulation (F2,70 = 3.479, p = 0.036, ηp2 = 0.090) was observed on SWM strategy score (lower strategy score = efficient performance). Bonferroni-corrected post hoc t-tests revealed a lower strategy score during HF-tRNS when compared with anodal tDCS (MD = 1.944, p = 0.022) but not with the sham condition, despite a numerically larger score, Fig. 3B. For SWM error, the analysis revealed a significant main effect of stimulation (F2,70 = 4.060, p = 0.021, ηp2 = 0.104). Bonferroni-corrected post-hoc t-tests yielded significantly fewer errors only during HF-tRNS compared with sham (MD = 7.944, p = 0.028) and not during anodal tDCS, Fig. 3A.
Working memory was evaluated using the CANTAB SWM, with errors (A, where a lower number of errors indicates better performance) and strategy score (B, where a lower strategy score reflects better performance) as outcome measures. Executive function (planning) was evaluated using the CANTAB SOC, with the outcome measures including problems solved in minimum moves (mean) (C) and mean moves used to solve problems in 2-move (D), 3-move (E), 4-move (F), and 5-move (G) problems (lower = more efficient planning). SWM spatial working memory, SOC Stockings of Cambridge, tES transcranial electrical stimulation, tDCS transcranial direct current stimulation, tRNS transcranial random noise stimulation, ns non-significant. * denotes significant differences between active tES conditions (red for anodal tDCS, green for HF-tRNS) versus sham or other conditions, if applicable, at p < 0.05 (Bonferroni corrected) based on pairwise comparison results. Brackets indicate comparisons between active conditions. Error bars, where applicable, represent standard error of the mean (s.e.m.).
For the planning measured by SOC, the ANOVA results for the mean number of problems solved indicated a significant main effect of stimulation (F2,70 = 34.309, p ≤ 0.001, ηp2 = 0.495). Bonferroni-corrected post hoc t-tests yielded a higher number of solved problems on minimum move solutions during both bilateral DLPFC anodal tDCS (MD = 2.806, p ≤ 0.001) and HF-tRNS (MD = 2.917, p ≤ 0.001) stimulations compared to the sham condition, Fig. 3A. Results for 2-moves (F2,70 = 9.15, p = 0.405, ηp2 = 0.025) and 5-moves difficulty (F2,70 = 2.346, p = 0.103, ηp2 = 0.063) showed a non-significant main effect of stimulation. In the 3-moves (F2,70 = 29.919, p ≤ 0.001, ηp2 = 0.461) and 4-moves (F1.55,54.18 = 19.641, p ≤ 0.001, ηp2 = 0.359) difficulty conditions, however, a significant main effect of stimulation was found indicating that patients under bilateral DLPFC anodal tDCS (3 and 4 moves) and HF-tRNS (3-moves) needed lower number of moves to complete the task (i.e., more efficient problem solving). Bonferroni-corrected post hoc t-tests revealed better performance during bilateral DLPFC anodal tDCS (MD3-moves = 1.417, p ≤ 0.001; MD4-moves = 1.661, p ≤ 0.001) and HF-tRNS (MD3-moves = 1.319, p ≤ 0.001; MD4-moves = 0.951, p = 0.005) compared with sham (Fig. 3D–G).
Emotional stability and tES effects on positive/negative affect
PANAS was used to (1) assess patients’ emotional stability before each bilateral DLPFC excitatory stimulation (comparing only pre-intervention scores) and (2) monitor emotional changes post-stimulation (comparing pre- and post-intervention scores). There were no significant differences in pre-intervention PANAS positive (F2,70 = 0.812, p = 0.448) and negative scores (F2,70 = 1.483, p = 0.234) across conditions, indicating a similar emotional state among patients before each stimulation. With respect to stimulation-specific effects, the results of the 3 × 2 × 2 ANOVA showed a significant three-way interaction of stimulation×time×emotion on PANAS scores (F2,70 = 23.053, p < 0.001, ηp2 = 0.397) as well as stimulation × time (F1,35 = 8.153, p ≤ 0.001, ηp2 = 0.189) and time×emotion (F1,35 = 34.492, p < 0.001, ηp2 = 0.496) interactions. The main effect of time (F1,35 = 119.73, p < 0.001, ηp2 = 0.774) was significant too. For within-condition comparisons, the Bonferroni-corrected post hoc t-tests showed that positive emotions significantly increased after both bilateral DLPFC anodal tDCS (p < 0.001) and HF-tRNS (p < 0.001) conditions, but not after sham tES (p = 0.190), while negative emotions significantly decreased after bilateral DLPFC anodal tDCS (p < 0.001) and HF-tRNS (p < 0.001), but not after sham tES (p = 0.146). Post hoc analyses of the between-condition comparisons revealed that both bilateral DLPFC anodal tDCS (p < 0.001) and HF-tRNS (p < 0.001) significantly reduced negative emotions after the intervention compared to the sham condition, whereas only tRNS (p = 0.003) but not tDCS (p = 0.108) significantly enhanced positive emotions post-intervention relative to the sham (Fig. 4A, B).
Both bilateral DLPFC anodal tDCS and bilateral DLPFC HF-tRNS, but not sham tES, significantly increased positive affect and decreased negative affect post-intervention. Only significant correlations are presented. A, B show that emotional stability before the intervention showed no significant differences across stimulation conditions, while both bilateral DLPFC anodal tDCS and HF-tRNS significantly reduced negative affect and also increased positive affect. Filled symbols indicate a significant difference between post- and pre-intervention, while [*] denotes a significant difference compared to sham. C–H present scatterplots of patients’ post-intervention cognitive performance and negative affect, showcasing only significant comparisons. In C, an increase in solved problems after the intervention (represented by darker green nodes) correlates with lower PANAS-negative affect scores (closer to 15), suggesting that improved executive function is associated with reduced negative affect. In D, a lower number of moves required to solve problems (darker green nodes) correlates with lower PANAS-negative affect scores (closer to 15), indicating that better performance in the SOC task may also be linked to reduced negative affect. Scatterplots E, F and G, H depict similar data but show no significant results for the bilateral anodal tDCS and sham conditions.
Correlational analysis
The results of Pearson’s correlational analyses of the association between cognitive performance and mood changes revealed a significant positive correlation between reduced negative affect (i.e., a greater decrease in negative emotions post-intervention, indicating improved emotional state) and the mean value of solved problems during bilateral DLPFC HF-tRNS (r = 0.387, ptwo-sided = 0.020) (Fig. 4C) indicating that higher solved problems was associated with less negative affects. Additionally, a significant negative correlation was found between the mean moves required to solve the problem at level 4 difficulty (lower values better) and decreased negative affect during bilateral DLPFC HF-tRNS (r = −0.363, ptwo-sided = 0.029) (Fig. 4D), indicating that the fewer number of moves for problem solving was associated with lower negative affect. The remaining correlations (Fig. 4E–H) were non-significant.
Discussion
This randomized sham-controlled study explored the cognitive and emotional effects of bilateral DLPFC anodal tDCS and high-frequency tRNS in patients with schizophrenia. Our results show that both active excitatory tES interventions enhanced executive functioning measured via an executive planning task, with tRNS additionally improving working memory performance. Additionally, both anodal tDCS and high-frequency tRNS increased positive affect and decreased negative affect after the intervention, with tRNS showing a more pronounced effect on positive emotions. These findings underscore procognitive effects and have implications regarding the therapeutic potential of bilateral DLPFC excitatory stimulation as an adjunctive treatment for schizophrenia.
Cognitive effects of bilateral DLPFC tDCS and tRNS
In this study, both bilateral DLPFC anodal tDCS and tRNS improved executive function, as shown by increased problem-solving scores and fewer moves required to achieve the goal in the planning task compared to sham stimulation. Furthermore, tRNS demonstrated additional benefits in working memory by improving both strategy (numerically vs sham, significantly vs tDCS) and errors (significantly vs. sham). In contrast, tDCS exhibited a significantly less efficient strategy compared to tRNS and, numerically, to sham, although it was associated with fewer errors than sham. This cognitive improvement aligns with the DLPFC’s critical role in executive processes and its known dysfunction in schizophrenia7,9,66 and indicates that upregulating bilateral DLPFC activity can enhance core cognitive deficits, consistent with prior evidence of prefrontal hypoactivity contributing to cognitive deficits in schizophrenia7,23,67,68. Additionally, this supports the potential cognitive benefits of tES in schizophrenia, specifically the tRNS, which were not demonstrated in a recent meta-analysis of tDCS trials19. Although these cognitive effects found in our study were robust, the large effect sizes for the executive function task should be interpreted with caution due to the potential for residual practice effects in this crossover design, despite randomization and standard washout procedures.
A key distinction in our study is stimulation of bilateral DLPFC, a method applied in only one tDCS study in Sawfi’s meta-analysis19, which also demonstrated improvements in negative symptoms and cognitive deficits41. These findings support the notion that upregulation of both left and right DLPFCs is more effective than unilateral upregulation, as applied in the majority of tES studies on schizophrenia. This pattern aligns with the recent evidence in major depressive disorder, where larger effects are found for bilateral DLPFC stimulation compared with unilateral approaches69. It also suggests that bilateral DLPFC excitatory tES techniques (i.e., anodal tDCS, high-frequency tRNS) may provide a more effective approach to addressing core cognitive deficits in schizophrenia—a critical therapeutic target that is not targeted by standard treatments6,7. Furthermore, this protocol configuration (with extracranial return electrodes) emphasizes the importance of target specificity in tES studies, which is often compromised by placing the cathode over regions that are essential for both cognitive and emotional functioning (e.g., supraorbital, frontopolar cortex, contralateral prefrontal cortex), yet their role is ignored.
Emotional effects and relevance to DLPFC upregulation
We also found that both bilateral DLPFC anodal tDCS and high-frequency tRNS reduced negative affect after the stimulation compared to the sham, with tRNS also enhancing positive emotions compared to the sham. These effects are consistent with the DLPFC’s role in emotion regulation, where it exerts top-down control over limbic regions to modulate affective responses20,70,22. They are further in line with previous tES studies that have targeted DLPFC and shown mood/emotional enhancement in schizophrenia, including negative symptoms71,72,73, and other emotional disorders with DLPFC abnormalities44,45,74,75,76. The more pronounced emotional effect of tRNS may reflect its greater capacity to enhance prefrontal excitability, potentially amplifying its regulatory influence on emotional processing. Furthermore, our correlational analysis revealed that reduced negative affect during tRNS was associated with better executive function, suggesting an interplay between emotional and cognitive improvements. This finding supports a bidirectional relationship between cognition and emotion in schizophrenia, where reduced negative affect may facilitate executive function by alleviating emotional interference on prefrontal resources, or enhanced cognition may improve emotion regulation via modulated DLPFC activity20,47.
Another point to consider here is that while both interventions reduced negative affect compared to sham, only high-frequency tRNS significantly enhanced positive affect, suggesting tRNS may exert broader emotional benefits. This could be associated with tRNS’s unique stochastic resonance mechanism, which amplifies weak neural signals and enhances prefrontal synchronization more robustly than tDCS’s polarity-dependent excitability shifts17,33. Additionally, bilateral prefrontal tRNS is shown to increase E/I marker in the brain27. By improving E/I balance and neural dynamics disrupted in schizophrenia24,25,40, tRNS might facilitate greater top-down regulation of positive affective states via DLPFC-limbic pathways20. These findings underscore tRNS’s potential as a more versatile tool for addressing the full spectrum of emotional dysregulation in schizophrenia, warranting further mechanistic studies with neuroimaging to delineate these effects.
Mechanisms of tDCS and tRNS
The distinct mechanisms of tDCS and tRNS, although not yet fully understood, might explain the partially differing effects between active interventions. tDCS delivers a constant current that shifts neuronal resting membrane potentials, with anodal stimulation increasing excitability10,16. In schizophrenia, anodal tDCS over the DLPFC is thought to counteract hypofrontality, enhancing prefrontal activity to support cognitive and emotional functions18,31. However, anodal tDCS effects are related to excitability enhancement and do not affect brain oscillatory activities, and may be less effective for tasks requiring rapid neural adaptability, such as working memory77. Conversely, tRNS applies random oscillatory currents within a high-frequency range (100–640 Hz), enhancing both excitability and synchronization, promoting stochastic resonance—a process where noise enhances weak signals in neural systems33,78. This mechanism may improve signal-to-noise ratios and neuronal synchronization, which are disrupted in schizophrenia40,79. For example, bilateral high-frequency tRNS over the visual cortex has been shown to induce stochastic resonance, enhancing evidence accumulation during perceptual decision-making for subthreshold stimuli by increasing drift rates80. This suggests that tRNS amplifies weak neural signals by recruiting voltage-gated sodium channels and pushing neurons above spiking thresholds, potentially extending to prefrontal regions to benefit executive planning and working in schizophrenia.
Additionally, this mechanism may further modulate E/I balance by improving signal-to-noise ratios and neuronal synchronization, which are disrupted in schizophrenia24,25. EEG studies suggest that tRNS increases E/I balance, potentially more effectively than tDCS, by enhancing cortical excitability and facilitating neuroplasticity, which aligns with its observed benefits in conditions like ADHD when combined with cognitive training28. High-frequency tRNS has been shown to increase cortical excitability and facilitate neuroplasticity more effectively than tDCS58, potentially explaining its broader cognitive benefits, particularly in working memory. These mechanistic differences, particularly the modulation of E/I balance as a core mechanism for both tDCS and tRNS, highlight tRNS as a promising tool for modulating the complex neural dynamics of schizophrenia.
Implications for novel tES treatment in schizophrenia
Our findings have implications for advancing excitatory tES as a novel treatment for schizophrenia. The use of bilateral DLPFC excitatory stimulation (via anodal tDCS or high-frequency tRNS) with extracephalic return electrodes could be introduced as an innovative tES montage that enhances target region specificity by minimizing unintended effects on non-target regions42 although it should be replicated in trials with a larger sample size. This approach may optimize modulation of the frontoparietal network, crucial for cognitive control7,77. Although preliminary, these findings warrant confirmation in larger, multi-site studies to establish their potential contribution toward standardizing tES protocols in schizophrenia. The pro-cognitive and emotional effects of bilateral DLPFC excitatory tDCS and tRNS, specifically tRNS, may suggest their application as an adjunctive therapy, particularly for cognitive deficits in schizophrenia. Its emotional benefits, if proven in other trials, further broaden its therapeutic scope, addressing affective symptoms often neglected in current treatments46.
Limitations, strengths, and future directions
This study’s limitations include the absence of neuroimaging or neurophysiological data, which could have offered more detailed insights into the intervention’s neurophysiological effects. Additional cognitive assessments, which are central to neurocognitive domains in schizophrenia (e.g., attentional functioning and social cognition9,55) and were not among out assessment, are needed for future studies. These assessments are recommended to be conducted after the intervention as well (not just during), to capture the effects of tES. Second, the study included only male participants, which may limit the generalizability of the results81. While randomized stimuli, performance stabilization training, and inter-session intervals minimized practice effects, these remain potential confounds. Future studies should also consider unified sham protocols, as splitting DC and random noise could subtly influence blinding. Finally, emotional changes were solely assessed via self-report PANAS. Future research should include physiological or implicit measures to validate affective modulation. Despite these limitations, the study’s strengths include its innovative use of bilateral DLPFC stimulation with extracephalic return electrodes, enhancing targeting specificity, and its sham-controlled, crossover design, which minimized individual differences and ensured robust comparisons between tDCS, tRNS, and sham conditions and finally, the inclusion of blinding efficacy, which is missed in the majority of tES studies in schizophrenia. Future studies should investigate the durability of these effects with repeated sessions and optimize stimulation parameters (e.g., intensity, duration) to maximize outcomes82. Additionally, integrating neuroimaging or electrophysiological measures could clarify the neural mechanisms of tES, paving the way for personalized treatment strategies in schizophrenia.
Data availability
The data that support the findings are publicly available at OSF: https://doi.org/10.17605/OSF.IO/B9JT8 and are accessible after the publication of the study.
References
American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders (DSM-5®) (American Psychiatric Pub, 2013).
McCutcheon, R. A., Reis Marques, T. & Howes, O. D. Schizophrenia—an overview. JAMA Psychiatry 77, 201–210 (2020).
Ferrari, A. J. et al. Global incidence, prevalence, years lived with disability (YLDs), disability-adjusted life-years (DALYs), and healthy life expectancy (HALE) for 371 diseases and injuries in 204 countries and territories and 811 subnational locations, 1990–2021: a systematic analysis for the Global Burden of Disease Study 2021. Lancet 403, 2133–2161 (2024).
Adityanjee, Aderibigbe, Y. A., Theodoridis, D. & Vieweg, W. V. R. Dementia praecox to schizophrenia: the first 100 years. Psychiatry Clin. Neurosci. 53, 437–448 (1999).
Green, M. F. & Harvey, P. D. Cognition in schizophrenia: past, present, and future. Schizophr. Res. Cogn. 1, e1–e9 (2014).
Marder, S. R. & Cannon, T. D. Schizophrenia. N. Engl. J. Med. 381, 1753–1761 (2019).
Voineskos, A. N. et al. Functional magnetic resonance imaging in schizophrenia: current evidence, methodological advances, limitations and future directions. World Psychiatry 23, 26–51 (2024).
Neufeld, N. H. & Blumberger, D. M. An update on the use of neuromodulation strategies in the treatment of schizophrenia. Am. J. Psychiatry 182, 332–340 (2025).
Halverson, T. F. et al. Pathways to functional outcomes in schizophrenia spectrum disorders: meta-analysis of social cognitive and neurocognitive predictors. Neurosci. Biobehav Rev. 105, 212–219 (2019).
Polanía, R., Nitsche, M. A. & Ruff, C. C. Studying and modifying brain function with non-invasive brain stimulation. Nat. Neurosci. 21, 174–187 (2018).
Hallajian, A. H. et al. Neurocognitive effects of 3 mA prefrontal electrical stimulation in schizophrenia: a randomized sham-controlled tDCS-fMRI study protocol. PLoS ONE 19, e0306422 (2024).
Tseng, P.-T. et al. Assessment of Noninvasive Brain Stimulation Interventions for Negative Symptoms of Schizophrenia. JAMA Psychiatry. 79, 770–779 (2022).
Philip, N. S. et al. Low-intensity transcranial current stimulation in psychiatry. Am. J. Psychiatry 174, 628–639 (2017).
Fregni, F. et al. Evidence-based guidelines and secondary meta-analysis for the use of transcranial direct current stimulation in neurological and psychiatric disorders. Int. J. Neuropsychopharmacol. 24, 256–313 (2020).
Bikson, M., Paulus, W., Esmaeilpour, Z., Kronberg, G. & Nitsche, M. A. Mechanisms of acute and after effects of transcranial direct current stimulation. in Practical Guide to Transcranial Direct Current Stimulation: Principles, Procedures and Applications (eds Knotkova, H., Nitsche, M. A., Bikson, M. & Woods A. J.) 81–113 (Springer International Publishing, 2019).
Sánchez-León, C. A., Reyes-Velasquez, P. A., van Thriel, C. & Nitsche, M. A. An in vivo model for transcranial direct current stimulation of the motor cortex in awake mice. Brain Stimulation 18, 1978–1989 (2025).
Carvalho, S., Leite, J. & Fregni, F. Transcranial alternating current stimulation and transcranial random noise stimulation. in Neuromodulation 2nd edn (eds Krames, E. S., Peckham, P. H. & Rezai, A. R.) 1611–1617 (Academic Press, 2018).
Aleman, A., Enriquez-Geppert, S., Knegtering, H. & Dlabac-de Lange, J. J. Moderate effects of noninvasive brain stimulation of the frontal cortex for improving negative symptoms in schizophrenia: meta-analysis of controlled trials. Neurosci. Biobehav Rev. 89, 111–118 (2018).
Safwi, S. R. et al. Transcranial direct current stimulation and its effect on cognitive symptoms of schizophrenia: an updated review. Schizophr. Res. Cogn. 39, 100335 (2025).
Nejati, V., Majdi, R., Salehinejad, M. A. & Nitsche, M. A. The role of dorsolateral and ventromedial prefrontal cortex in the processing of emotional dimensions. Sci. Rep. 11, 1971 (2021).
Salehinejad, M. A., Ghanavati, E., Rashid, M. H. A. & Nitsche, M. A. Hot and cold executive functions in the brain: a prefrontal-cingular network. Brain Neurosci. Adv. 5, 23982128211007769 (2021).
Kohn, N. et al. Neural network of cognitive emotion regulation — An ALE meta-analysis and MACM analysis. NeuroImage. 87, 87345–87355 (2014).
Andreasen, N. C. et al. Hypofrontality in schizophrenia: distributed dysfunctional circuits in neuroleptic-naïve patients. Lancet 349, 1730–1734 (1997).
Peterson, E. J., Rosen, B. Q., Belger, A., Voytek, B. & Campbell, A. M. Aperiodic neural activity is a better predictor of schizophrenia than neural oscillations. Clin. EEG Neurosci. 54, 434–445 (2023).
Zhang, K. et al. The aperiodic and periodic activities of EEG dynamically relate with cognitive performance in schizophrenia. Schizophr. Res Cogn. 42, 100383 (2025).
Krause, B., Márquez-Ruiz, J. & Cohen Kadosh, R. The effect of transcranial direct current stimulation: a role for cortical excitation/inhibition balance? Front. Hum. Neurosci. 7, 602 (2013).
van Bueren, N. E. R., van der Ven, S. H. G., Hochman, S., Sella, F. & Cohen Kadosh, R. Human neuronal excitation/inhibition balance explains and predicts neurostimulation induced learning benefits. PLoS Biol. 21, e3002193 (2023).
Dakwar-Kawar, O. et al. Transcranial random noise stimulation combined with cognitive training for treating ADHD: a randomized, sham-controlled clinical trial. Transl. Psychiatry 13, 271 (2023).
Orlov, N. D. et al. Stimulating thought: a functional MRI study of transcranial direct current stimulation in schizophrenia. Brain 140, 2490–2497 (2017).
Martin, D. M. et al. Can transcranial direct current stimulation enhance outcomes from cognitive training? A randomized controlled trial in healthy participants. Int. J. Neuropsychopharmacol. 16, 1927–1936 (2013).
Yu, L. et al. Efficacy of transcranial direct current stimulation in ameliorating negative symptoms and cognitive impairments in schizophrenia: a systematic review and meta-analysis. Schizophr. Res. 224, 2–10 (2020).
Valiengo, L. D. C. L. et al. Efficacy and Safety of Transcranial Direct Current Stimulation for Treating Negative Symptoms in Schizophrenia. JAMA Psychiatry. 77, 121–129 (2020).
van der Groen, O. et al. Using noise for the better: the effects of transcranial random noise stimulation on the brain and behavior. Neurosci. Biobehav. Rev. 138, 104702 (2022).
Yeh, T.-C. et al. High-Frequency Transcranial Random Noise Stimulation Modulates Gamma-Band EEG Source-Based Large-Scale Functional Network Connectivity in Patients with Schizophrenia: A Randomized Double-Blind Sham-Controlled Clinical Trial. J. Pers. Med. 12, 1617 (2022).
Chang, C. C., Lin, Y. Y., Tzeng, N. S., Kao, Y. C. & Chang, H. A. Adjunct high-frequency transcranial random noise stimulation over the lateral prefrontal cortex improves negative symptoms of schizophrenia: a randomized, double-blind, sham-controlled pilot study. J. Psychiatr. Res. 132, 151–160 (2021).
Mondino, M., Janin, D., Galvao, F. & Brunelin, J. High-frequency transcranial random noise stimulation for auditory hallucinations of schizophrenia: a case series. Biomedicines 10, 2698 (2022).
Haesebaert, F., Mondino, M., Saoud, M., Poulet, E. & Brunelin, J. Efficacy and safety of fronto-temporal transcranial random noise stimulation (tRNS) in drug-free patients with schizophrenia: a case study. Schizophr. Res. 159, 251–252 (2014).
Brunelin J. et al. Examining transcranial random noise stimulation as an add-on treatment for persistent symptoms in schizophrenia (STIM’Zo): a study protocol for a multicentre double-blind randomized sham-controlled clinical trial. Trials. 22, 964 (2021).
Adam, O. et al. Efficacy of transcranial direct current stimulation to improve insight in patients with schizophrenia: a systematic review and meta-analysis of randomized controlled trials. Schizophr. Bull. 48, 1284–1294 (2022).
Uhlhaas, P. J. & Singer, W. Abnormal neural oscillations and synchrony in schizophrenia. Nat. Rev. Neurosci. 11, 100–113 (2010).
Chang, C.-C., Kao, Y.-C., Chao, C.-Y., Tzeng, N.-S. & Chang, H.-A. Examining bi-anodal transcranial direct current stimulation (tDCS) over bilateral dorsolateral prefrontal cortex coupled with bilateral extracephalic references as a treatment for negative symptoms in non-acute schizophrenia patients: a randomized, double-blind, sham-controlled trial. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 96, 109715 (2020).
Nejati, V., Salehinejad, M. A. & Nitsche, M. A. Interaction of the left dorsolateral prefrontal cortex (l-DLPFC) and right orbitofrontal cortex (OFC) in hot and cold executive functions: evidence from transcranial direct current stimulation (tDCS). Neuroscience 369, 109–123 (2018).
Glinski B. et al. Phase-synchroni zed 40 Hz tACS and iTBS effects on gamma oscillations. Imaging Neurosci (Camb). 3, https://doi.org/10.1162/IMAG.a.140 (2025).
Molavi, P. et al. Repeated transcranial direct current stimulation of dorsolateral-prefrontal cortex improves executive functions, cognitive reappraisal emotion regulation, and control over emotional processing in borderline personality disorder: a randomized, sham-controlled, parallel-group study. J. Affect. Disord. 274, 93–102 (2020).
Zemestani, M., Hoseinpanahi, O., Salehinejad, M. A. & Nitsche, M. A. The impact of prefrontal transcranial direct current stimulation (tDCS) on theory of mind, emotion regulation and emotional-behavioral functions in children with autism disorder: a randomized, sham-controlled, and parallel-group study. Autism Res. 15, 1985–200 (2022).
Kring, A. M. & Elis, O. Emotion deficits in people with schizophrenia. Annu. Rev. Clin. Psychol. 9, 409–433 (2013).
Wiegand, A., Sommer, A., Nieratschker, V. & Plewnia, C. Improvement of cognitive control and stabilization of affect by prefrontal transcranial direct current stimulation (tDCS). Sci. Rep. 9, 6797 (2019).
Nejati, V. et al. Report Approval for Transcranial Electrical Stimulation (RATES): expert recommendation based on a Delphi consensus study. Nat. Protoc. https://doi.org/10.1038/s41596-025-01259-0 (2025).
Sloan, N. P., Byrne, L. K., Enticott, P. G. & Lum, J. A. G. Non-invasive brain stimulation does not improve working memory in schizophrenia: a meta-analysis of randomised controlled trials. Neuropsychol. Rev. 31, 115–138 (2020).
Liemburg, E., Nienhuis, F. & Veling, W. DSM-5 clinician-rated dimensions of psychosis symptom severity: psychometric properties. Schizophr. Bull. 46, S170–S171 (2020).
Amini, H. et al. Validity of the Iranian version of the Structured Clinical Interview for DSM-IV (SCID-I) in the diagnosis of psychiatric disorders. Payesh Health Monit. J. 7, 49–57 (2008).
Fray, P. J., Robbins, T. W. & Sahakian, B. J. Neuorpsychiatyric applications of CANTAB. Int. J. Geriatr. Psychiatry 11, 329–336 (1996).
Watson, D., Clark, L. A. & Tellegen, A. Development and validation of brief measures of positive and negative affect: the PANAS scales. J. Personal. Soc. Psychol. 54, 1063 (1988).
Brunoni, A. R. et al. A systematic review on reporting and assessment of adverse effects associated with transcranial direct current stimulation. Int. J. Neuropsychopharmacol. 14, 1133–1145 (2011).
Green, M. F., Horan, W. P. & Lee, J. Social cognition in schizophrenia. Nat. Rev. Neurosci. 16, 620–631 (2015).
Langley, C., Sahakian, B. J. & Robbins, T. W. Cambridge Neuropsychological Test Automated Battery (CANTAB). The SAGE Handbook of Clinical Neuropsychology: Clinical Neuropsychological Assessment and Diagnosis 435 (SAGE Publications Ltd, 2023).
Owen, A. M., Downes, J. J., Sahakian, B. J., Polkey, C. E. & Robbins, T. W. Planning and spatial working memory following frontal lobe lesions in man. Neuropsychologia 28, 1021–1034 (1990).
Fertonani, A., Pirulli, C. & Miniussi, C. Random noise stimulation improves neuroplasticity in perceptual learning. J. Neurosci. 31, 15416–15423 (2011).
Salehinejad, M. A. et al. Sleep-dependent upscaled excitability, saturated neuroplasticity, and modulated cognition in the human brain. eLife 11, e69308 (2022).
Salehinejad, M. A. et al. Cognitive functions and underlying parameters of human brain physiology are associated with chronotype. Nat. Commun. 12, 4672 (2021).
Hill, A. T., Fitzgerald, P. B. & Hoy, K. E. Effects of anodal transcranial direct current stimulation on working memory: a systematic review and meta-analysis of findings from healthy and neuropsychiatric populations. Brain Stimul. 9, 197–208 (2016).
Ghanavati, E., Salehinejad, M. A., Nejati, V. & Nitsche, M. A. Differential role of prefrontal, temporal and parietal cortices in verbal and figural fluency: implications for the supramodal contribution of executive functions. Sci. Rep. 9, 3700 (2019).
Ghanavati, E., Nejati, V. & Salehinejad, M. A. Transcranial direct current stimulation over the posterior parietal cortex (PPC) enhances figural fluency: implications for creative cognition. J. Cogn. Enhanc. 2, 88–96 (2018).
Bang, H., Ni, L. & Davis, C. E. Assessment of blinding in clinical trials. Control Clin. Trials 25, 143–156 (2004).
Turner, C., Jackson, C. & Learmonth, G. Is the “end-of-study guess” a valid measure of sham blinding during transcranial direct current stimulation? Eur. J. Neurosci. 53, 1592–1604 (2021).
Kerns, J. G., Nuechterlein, K. H., Braver, T. S. & Barch, D. M. Executive functioning component mechanisms and schizophrenia. Biol. Psychiatry 64, 26–33 (2008).
Minzenberg, M. J., Laird, A. R., Thelen, S., Carter, C. S. & Glahn, D. C. Meta-analysis of 41 functional neuroimaging studies of executive function in schizophrenia. Arch. Gen. Psychiatry 66, 811–822 (2009).
Li, Y. T. et al. Neuroimaging features of cognitive impairments in schizophrenia and major depressive disorder. Ther. Adv. Psychopharmacol. 14, 20451253241243290 (2024).
Nejati, V. et al. The effectiveness of non-invasive brain stimulation in treatment of major depressive disorder (MDD): a systematic review and transfer analysis. J. Neural. Transm. 132, 369–385 (2025).
Ochsner, K. N. & Gross, J. J. The cognitive control of emotion. Trends Cogn. Sci. 9, 242–249 (2005).
Gupta, T. & Mittal, V. A. Transcranial direct current stimulation and emotion processing deficits in psychosis and depression. Eur. Arch. Psychiatry Clin. Neurosci. 271, 69–84 (2021).
Valiengo, L.dC. L. et al. Efficacy and safety of transcranial direct current stimulation for treating negative symptoms in schizophrenia: a randomized clinical trial. JAMA Psychiatry 77, 121–129 (2020).
Cheng, P. W. C. et al. The effects of transcranial direct current stimulation (tDCS) on clinical symptoms in schizophrenia: a systematic review and meta-analysis. Asian J. Psychiatry 53, 102392 (2020).
Nikolin, S. et al. Time-course of the tDCS antidepressant effect: an individual participant data meta-analysis. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 125, 110752 (2023).
Vicario, C. M., Salehinejad, M. A., Felmingham, K., Martino, G. & Nitsche, M. A. A systematic review on the therapeutic effectiveness of non-invasive brain stimulation for the treatment of anxiety disorders. Neurosci. Biobehav. Rev. 96, 219–231 (2019).
Zakibakhsh, N. et al. Repeated prefrontal tDCS for improving mental health and cognitive deficits in multiple sclerosis: a randomized, double-blind, parallel-group study. J. Transl. Med. 22, 843 (2024).
Barch, D. M. & Ceaser, A. Cognition in schizophrenia: core psychological and neural mechanisms. Trends Cogn. Sci. 16, 27–34 (2012).
Van Der Groen, O. & Wenderoth, N. Transcranial random noise stimulation of visual cortex: stochastic resonance enhances central mechanisms of perception. J. Neurosci. 36, 5289–5298 (2016).
Winterer, G. & Weinberger, D. R. Genes, dopamine and cortical signal-to-noise ratio in schizophrenia. Trends Neurosci. 27, 683–690 (2004).
van der Groen, O., Tang, M. F., Wenderoth, N. & Mattingley, J. B. Stochastic resonance enhances the rate of evidence accumulation during combined brain stimulation and perceptual decision-making. PLOS Comput. Biol. 14, e1006301 (2018).
Moniem, I. & Kafetzopoulos, V. Sex differences in schizophrenia: symptomatology, treatment efficacy and adverse effects. Front. Psychiatry 16, 1594334 (2025).
Cohen Kadosh, R., Ciobotaru, D., Karstens, M. I. & Nguyen, V. Personalized home based neurostimulation via AI optimization augments sustained attention. NPJ Digit. Med. 8, 463 (2025).
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
E.J.: investigation, project administration, data curation, and validation. A.M.: resources. Z.V.: formal analysis (lead), writing—original draft (support), and visualization. A.A.: software, investigation (cognitive assessment), and project administration (patient diagnosis and recruitment). A.F.J.: visualization. R.C.K.: writing—review & editing. M.A.N.: writing—review & editing. D.M.B.: writing—review & editing. M.A.S.: conceptualization, methodology, supervision, formal analysis (support), writing—original draft, and writing—review and editing.
Corresponding authors
Ethics declarations
Competing interests
M.A.N. is a member of the Scientific Advisory Boards of Neuroelectrics and Precisis. R.C.K. serves on the scientific advisory boards of Neuroelectrics Inc. and Tech InnoSphere Engineering Ltd. and is a founder, director, and shareholder of Cognite Neurotechnology Ltd. All other authors report no biomedical financial interests or potential conflicts of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Jafari, E., Moghadamzadeh, A., Vaziri, Z. et al. Cognitive and emotional effects of bilateral prefrontal anodal tDCS and high-frequency tRNS in schizophrenia: a randomized sham-controlled study. Schizophr 12, 28 (2026). https://doi.org/10.1038/s41537-025-00720-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41537-025-00720-z