Abstract
Accumulating evidence suggests that oxidative stress (OS) contributes to the onset and progression of schizophrenia (SCZ). However, the pattern of OS alterations in first-episode, drug-naïve patients and their associations with clinical and cognitive features remain unclear. In this study, 98 first-episode SCZ patients and 96 matched healthy controls were recruited. Plasma levels of total antioxidant capacity (TAC), malondialdehyde (MDA), lipid peroxidation (LPO), NADPH oxidase (NOX), glutathione S-transferase (GST), and glutathione reductase (GR) were measured. Clinical symptoms were assessed using the Positive and Negative Syndrome Scale (PANSS), and cognitive performance was evaluated using the Repeatable Battery for the Assessment of Neuropsychological Status (RBANS). Compared with controls, patients exhibited significantly higher TAC, MDA, and LPO levels but lower NOX levels, while GST and GR showed no significant differences. RBANS total and subscale scores were markedly reduced in patients, indicating generalized cognitive impairment. Correlation analyses revealed that GST was positively correlated with total PANSS scores, GR with negative symptoms, and LPO with overall cognition, attention, and delayed recall. These findings indicate that first-episode SCZ patients display an imbalance between oxidative and antioxidant systems, and specific OS markers are linked to symptom severity and cognitive dysfunction. OS alterations may serve as potential early biomarkers and therapeutic targets for schizophrenia.
Similar content being viewed by others
Introduction
Schizophrenia (SCZ) is a severe mental disorder. Based on large-scale epidemiological studies and systematic reviews, its lifetime prevalence ranges from approximately 0.7% to 1.0%1; some studies report rates exceeding 1%2. The disorder typically begins during adolescence or early adulthood3. SCZ is clinically characterized by positive symptoms, negative symptoms, and widespread cognitive impairment4. Cognitive impairment is considered one of the core deficits of SCZ, often emerging early in the disease course and exerting a lasting impact on social functioning, quality of life, and treatment adherence5,6. Research indicates7 that cognitive impairment is closely associated with symptom severity and disease outcome and serves as a key factor contributing to high recurrence and low recovery rates among patients. Although, current clinical treatments demonstrate clear efficacy in improving positive symptoms, their effectiveness in alleviating cognitive deficits and negative symptoms remains limited, suggesting that the underlying neurobiological mechanisms warrant further investigation8. At the same time, SCZ imposes a heavy disease burden on public health systems, making it imperative to identify novel pathological mechanisms and potential biomarkers.
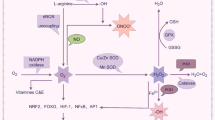
Oxidative stress (OS) refers to a pathological state in which the production of reactive oxygen species (ROS) exceeds the body’s antioxidant defense capacity, thereby disrupting redox homeostasis9. ROS includes superoxide radicals, hydroxyl radicals, and hydrogen peroxide, among others. These are continuously generated during cellular metabolism and, under normal physiological conditions, participate in various functions such as signal transduction, immune regulation, and gene expression10,11. The body eliminates excess ROS through its antioxidant system, in which glutathione reductase (GR) and glutathione S-transferase (GST) play pivotal roles in the glutathione cycle12. Meanwhile, total antioxidant capacity (TAC) is often used as a comprehensive indicator for evaluating overall antioxidant levels13. When the antioxidant system is insufficient to counteract the accumulation of ROS, the body undergoes lipid peroxidation (LPO), generating metabolites such as malondialdehyde (MDA). This process damages in known to dame the cell membrane and compromises neuronal integrity14. Additionally, NADPH oxidase (NOX) serves as a major source of ROS production, within findings indicating that its abnormal activity further exacerbates redox imbalance15. In addition to directly damaging cells, OS is also closely associated with neuroinflammatory responses and impaired neuroplasticity. These mechanisms are thought to potentially contribute to the pathological processes underlying SCZ16.
Building upon the aforementioned oxidative stress mechanisms, previous studies have also investigated alterations in these markers among schizophrenia patients, including chronic cases and first-episode individuals. Overall, multiple studies have found that TAC levels generally decrease in schizophrenia patients17, while MDA and LPO levels predominantly increase18,19,20, suggesting weakened antioxidant capacity and enhanced lipid peroxidation. However, conclusions vary across studies. Some investigations failed to observe significant TAC reduction, and the magnitude of MDA elevation also differed21. Similarly, while one study reported upregulation of NOX activity, potentially reflecting excessive ROS production pathway activation22, another found no notable changes23. Regarding antioxidant enzyme systems, GST and GR results show even greater variability, with reports ranging from decreased activity to no change24,25,26. These inconsistencies may relate to factors such as sample characteristics, disease stage, detection methods, environmental exposures, and medication use. Consequently, current evidence on oxidative stress marker changes across different stages of schizophrenia remains inconclusive, necessitating further systematic research for clarification.
Increasing evidence suggests that alterations in the oxidation/antioxidation system are not only present in chronic SCZ patients but can also be detected in the early stages of the disorder and are associated with the severity of clinical symptoms and cognitive impairment17,27. First-episode SCZ patients, due to their shorter disease duration and often having received no or only short-term antipsychotic medication, exhibit OS levels that better reflect the intrinsic pathological characteristics of the disorder rather than secondary effects from medication or prolonged illness. Previous studies have shown that decreased plasma antioxidant levels in first-episode SCZ patients correlate with negative symptom severity, while elevated LPO products significantly correlate with cognitive impairment28,29. Furthermore, recent studies indicate that inflammation and related oxidative stress pathways exert a more pronounced impact on cognitive function compared to other symptom dimensions, with this association being particularly prominent in patients during the early stages of the disease30. However, inconsistencies persist across different studies, with significant variations observed in the performance of certain indicators such as NOX, GR, and GST31,32. Such inconsistencies suggest that the mechanism by which OS influences SCZ may be more complex than initially perceived. Currently, there is a lack of systematic studies in first-episode SCZ patients to comprehensively explore the relationship between different OS markers and clinical symptoms as well as cognitive impairments.
The current study systematically evaluated characteristic alterations in plasma OS and antioxidant defense systems among patients with first-episode SCZ, focusing on indicators such as TAC, MDA, LPO, NOX, GR, and GST levels and comparing these with a healthy controls. To accurately assess the independent associations and interactions among target variables, covariates such as age and educational attainment—which may influence cognitive function or metabolic status—will be incorporated into statistical modeling. This approach aims to eliminate confounding effects, clarify the independence and specificity of associations between oxidative stress markers and clinical/cognitive phenotypes, thereby enhancing model accuracy and the reliability of conclusions33. We hypothesized that first-episode SCZ patients would exhibit a significant imbalance in the oxidative/antioxidant system. This imbalance would not only closely be associated with their clinical symptom spectrum but also linked to cognitive impairment, potentially providing a biological biomarker for developing early intervention strategies and assessing prognosis. Therefore, the objectives of the current study were to (1) systematically analyze the characteristics of alterations in OS and antioxidant defense system markers in the plasma of first-episode SCZ patients and (2) to further investigate the associations between these OS markers and the severity of clinical symptoms as well as cognitive impairment.
Material and methods
Participants
98 first-episode, untreated SCZ patients (patient group) who were hospitalized for the first time in the Department of Psychiatry at Nantong Fourth People’s Hospital between March 2022 and November 2024 were recruited for the current study. Inclusion criteria: (1) meeting the diagnostic criteria for SCZ according to the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5)34 as confirmed by the Structured Clinical Interview for DSM Disorders, Patient Edition(SCID-I/P) Chinese version structured interview; (2) possess an elementary school education or higher and be able to cooperate with cognitive assessment; (3) medication naïve; (4) diagnosed jointly by two psychiatrists; (5) aged 18–65 years; and (6) Voluntarily signed an informed consent form.
Exclusion Criteria: (1) presence of organic brain disorders (e.g., epilepsy, traumatic brain injury, brain tumors); (2) history of psychoactive substance abuse; (3) concurrent major physical illnesses (including endocrine/metabolic disorders, hematological diseases, etc.); (4) pregnant or lactating; (5) receiving modified electroconvulsive therapy; or (6) insufficient study compliance or withdrawal during the trial.
In addition, 96 healthy controls (control group) matched for age, gender, and years of education were recruited concurrently. Inclusion criteria: (1) aged 18–65 years with primary school education or higher; (2) assessed as free of psychiatric disorders using the Chinese version of the Mini-International Neuropsychiatric Interview (MINI); (3) no family history of major mental disorders in two lineages spanning three generations; (4) no history of neurological disorders (including head trauma, epilepsy, cerebrovascular disease, etc.); (5) no major physical illnesses (including metabolic disorders, malignancies, cardiovascular diseases, etc.); (6) no history of psychoactive substance use or drug dependence; (7) non-pregnant or non-lactating females; and (8) voluntary signing of an informed consent form.
All participants or their guardians were fully informed of the study’s purpose and procedures and participated voluntarily. To ensure participant privacy, all data underwent anonymization. This study protocol was approved by the Ethics Committee of Nantong Fourth People’s Hospital (Ethics Approval No.: 2022-K015), and all participants or their guardians signed written informed consent forms.
Clinical and cognitive assessment
Data collection included using a self-designed general demographic form to collect basic clinical information for both the patient group and control group, including age, gender, height, weight, body mass index (BMI), educational level, family history, and smoking status. For the patient group, additional data on age at onset and disease duration were collected. The Positive and Negative Symptom Scale (PANSS) was used to assess psychiatric symptoms35. The scale comprises 33 items covering three dimensions: positive symptoms (7 items), negative symptoms (7 items), and general psychopathology symptoms (16 items). It also includes supplementary items (3 items) to assess aggression risk (not counted in the total score). Symptom severity is rated on a 7-point scale (1 = absent, 2 = very mild, 3 = mild, 4 = moderate, 5 = moderately severe, 6 = severe, 7 = extremely severe). Total scores range from 30 to 210, with higher scores indicating greater symptom severity.
The Repeatable Battery for the Assessment of Neuropsychological Status (RBANS) was employed as the cognitive function assessment tool. This battery was administered immediately upon the patient’s admission and diagnosis by a psychiatrist who had undergone systematic training. It is characterized by its ease of administration, short duration, and ability to be completed by a single examiner. RBANS comprises five dimensions: immediate memory, visual span, verbal function, attention, and delayed memory. It encompasses 12 test items including word learning, story retelling, figure copying, line drawing, picture naming, semantic fluency test, digit span, coding test, word recall, word recognition, story recall, and picture recall. Scores across each dimension show significant positive correlations with individual cognitive function. Higher total scale scores indicate superior cognitive performance. The scale’s reliability and validity within SCZ patient populations have been empirically validated36. Raw data were subsequently converted into scale scores through a standardized scoring system for statistical analysis.
Fasting blood sampling and biochemical analysis
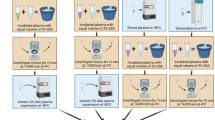
The patient group underwent fasting blood collection on the day following enrollment, whereas the control group completed a single blood draw under fasting conditions during the baseline phase. All blood collection procedures strictly adhered to standardized operating procedures: Blood was drawn during the early morning fasting state (07:00-09:00), with 5 ml of whole blood collected via the antecubital vein. Blood specimens were collected in EDTA anticoagulant tubes, then centrifuged at 3000 r/min for 15 min at 4 °C to separate the plasma layer. Each plasma sample was sequentially numbered and aliquoted into four 0.5 mL cryovials. All samples were ultimately transferred to a − 80°C ultra-low temperature freezer for long-term storage pending subsequent batch analysis.
All OS indicators were measured strictly according to the respective kit instructions (with assistance from Nanjing Jiancheng Biological Engineering Institute). Specifically, MDA levels were detected using the thiobarbituric acid method by measuring the chemical product of MDA and thiobarbituric acid (Catalog No.: A003-1). LPO and NOX levels were detected using spectrophotometry (Catalog Nos.: A106-1, A116-1-1). GST levels were measured using a colorimetric method (Catalog No.: A004-1-1). GR levels were measured using a UV colorimetric method (Catalog No.: A062-2-1). Finally, TAC levels were measured using the ABTS rapid method (Catalog No.: A015-2-1).
Statistical analysis
All statistical analyses were performed using SPSS 26.0 statistical software (SPSS, Chicago, IL, USA). Sample size was estimated using G*Power 3.1.9.7 software (http://www.ats.ucla.edu/stat/gpower/). The test type “Mean: Independent Means Comparison” was selected, with a power level of 90%, significance level of 0.10, and effect size of 0.8. Variable normality was assessed using the Kolmogorov–Smirnov test. Continuous variables meeting normality requirements were expressed as mean ± standard deviation, with intergroup comparisons performed using Student’s t-tests. Nonnormally distributed continuous variables were presented as median (25th and 75th percentiles). Categorical variables were analyzed using chi-square tests. To satisfy the normality assumption for parametric tests, non-normally distributed plasma MDA, LPO, NOX, GST, and GR levels underwent natural logarithmic transformation. Power analysis was completed during the study design phase to ensure sufficient sample size for detecting large effect differences (Cohen’s d ≥ 0.8). Cohen’s d was used to report effect sizes, with the following criteria: 0.2 indicates a small effect, 0.5 indicates a moderate effect, and 0.8 indicates a large effect. A p < 0.05 was considered statistically significant.
First, multivariate analysis of covariance (MANCOVA) was employed to examine intergroup differences. OS parameters (MDA, LPO, NOX, GST, GR, TAC) served as dependent variables, with diagnostic group (patients vs. healthy controls) as a fixed factor. Age, gender, education level, BMI, and smoking status were adjusted as covariates. Subsequently, univariate analysis of covariance (ANCOVA) was performed to individually examine differences in each dependent variable between groups, with diagnostic grouping as the independent variable. Adjustments were made for age, gender, education level, BMI, and smoking status. Multiple comparisons were corrected using the Bonferroni method. For variable correlation analysis, Pearson correlation analysis was applied to normally distributed data, while Spearman correlation analysis was used for non-normally distributed data. To control for multiple comparisons, the correlations between each oxidative stress marker and clinical or cognitive scores were adjusted using Bonferroni correction. The adjusted significance thresholds were set as follows: p < 0.0125 for comparisons with the four PANSS scores (0.05/4) and p < 0.0083 for comparisons with the six RBANS scores (0.05/6). Finally, exploratory multiple regression analysis was employed to examine the relationship between levels of OS parameters (MDA, LPO, NOX, GST, GR, TAC) and total scores on the PANSS and RBANS scales, as well as their respective subscale scores, while controlling for demographic covariates.
Results
Comparison between two sets of general clinical data
Table 1 presents demographic information and clinical data between the patient and healthy control groups. Results indicate no statistically significant differences between the groups in age, gender, educational attainment, BMI, or smoking status (all p > 0.05). The mean disease duration for the patient group was 6.57 ± 9.17 months. Positive symptom scores averaged 21.57 ± 4.37 points, negative symptom scores averaged 19.51 ± 4.88 points, general psychopathology scores averaged 44.40 ± 3.57 points, and the PANSS total score averaged 85.48 ± 5.79 points.
OS parameter levels between first episode SCZ patients and healthy controls
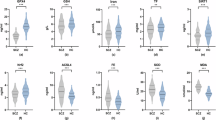
MANCOVA analysis revealed an overall significant diagnostic effect (F = 118.622, p < 0.001). ANCOVA results indicated differences in plasma OS parameter levels between the patient and control groups after controlling for age, gender, education level, BMI, and smoking status (see Table 2). The patient group exhibited significantly higher levels of TAC (F = 40.047, p < 0.001), MDA (F = 10.087, p = 0.002), and LPO (F = 96.906, p < 0.001) than the control group, while that of NOX (F = 591.101, p < 0.001) was significantly lower. No statistically significant differences were observed between the two groups with regard to GST (p = 0.173) and GR (p = 0.984) levels. After Bonferroni correction, differences in MDA, LPO, NOX, and TAC levels remained statistically significant (all p < 0.05).
Comparison of cognitive functions between first episode SCZ patients and healthy controls
RBANS total and subscale scores between the patient and control groups are shown in Table 3. Results indicated that the patient group had significantly lower RBANS total and subscale scores than the control group (both p < 0.01, Bonferroni-corrected, p < 0.01). After controlling for age, education level, BMI, and smoking status, the RBANS total score (F = 307.355, p < 0.001), immediate memory (F = 46.760, p < 0.001), visual field span (F = 287.567, p < 0.001), verbal function (F = 11.062, p < 0.01), attention (F = 519.093, p < 0.001), and delayed memory (F = 215.981, p < 0.001) remained significant (all p < 0.01).
Correlation between plasma OS marker levels and psychiatric symptoms in first episode SCZ patients
Correlation analyses
Correlation analysis was conducted between plasma OS marker levels and PANSS total and subscale scores in the patient group, after which Bonferroni correction was applied. GST levels showed a significant positive correlation with PANSS total scores (r = 0.226, p = 0.026, p corrected = 0.104). GR levels showed a significant positive correlation with PANSS negative symptom scores (r = 0.214, p = 0.034, p corrected = 0.136). Finally, TAC levels exhibited a significant negative correlation with PANSS positive symptoms (r = −0.213, p = 0.036, p corrected = 0.144)(Fig. 1), Preliminary correlation analyses indicated that GST, GR, and TAC showed no significant correlations with other PANSS total scores or subscales (see Figs. S1–S3).
Correlations between log GST levels and the PANSS total score, the PANSS negative subscale scores,the PANSS positive subscale scores.
Regression analyses controlling for confounding factors
Stepwise multiple regression analysis, controlling for age, gender, education level, BMI, smoking status, and disease duration as independent variables and potential confounders revealed a significant correlation between GST levels and the PANSS total score (beta = 0.225, t = 2.258, p = 0.026) (Fig. 2). GR levels were found to be significantly correlated with PANSS negative symptom scores (beta = 0.252, t = 2.716, p = 0.008; Fig. 3).
Correlations between log GST levels and the PANSS total score.
Correlations between log GR levels and the PANSS negative subscale scores.
Correlation between plasma OS marker levels and cognitive function in first episode SCZ patients
Correlation analyses
Correlation analysis was conducted between plasma OS marker levels and the RBANS total and subscale scores in the patient group, after which Bonferroni correction was applied. Results showed that TAC levels negatively correlated with attention (r = −0.212, p = 0.036, p corrected = 0.216) and delayed memory (r = −0.209, p = 0.039, p corrected = 0.234) scores(Fig. 4). LPO levels positively correlated with the RBANS total (r = 0.286, p = 0.004, p corrected = 0.024), immediate memory (r = 0.255, p = 0.011, p corrected = 0.066), and visual field score (r = 0.325, p = 0.001, p corrected = 0.006) scores. Furthermore, LPO levels positively correlated with attention (r = 0.307, p = 0.002, p corrected = 0.012), and delayed recall (r = 0.322, p = 0.001, p corrected = 0.006) scores (Fig.5), Preliminary correlation analyses indicated that TAC, and LPO showed no significant correlations with other RBANS total scores or subscales (see Figs. S4–5).
Correlations between TAC levels and the attention subscale scores,delayed memory subscale scores.
Correlations between LogLPO levels and the RBANS total score, the Immediate memory subscale scores,the Visuospatial/constructional subscale scores,the attention subscale scores,the delayed memory subscale scores.
Regression analyses controlling for confounding factors
Stepwise multiple regression analysis, controlling for age, gender, education level, BMI, smoking status, and disease duration as independent variables and potential confounders revealed a significant correlation between LPO levels and the RBANS total score (beta = 0.235, t = 2.657, p = 0.009) (Fig. 6). In addition, LPO levels were significantly correlated with attention (beta = 0.299, t = 3.332, p = 0.001; Fig. 7) and delayed recall (beta = 0.205, t = 2.195, p = 0.031; Fig. 8) scores.
Correlations between log LPO levels and the RBANS total score.
Correlations between log LPO levels and the attention subscale scores.
Correlations between log LPO levels and the delayed memory subscale scores.
Discussion
We found that plasma TAC, MDA, and LPO levels were significantly higher in first-episode SCZ patients than in health controls, while NOX levels were significantly lower. Overall, no significant differences were observed in GST and GR levels between groups. In addition, the RBANS total and subscale scores were significantly lower in first-episode SCZ patients than in healthy controls. Correlation analysis further revealed that there was a significant positive correlation between GST levels and the PANSS total score, while GR levels were significantly correlated with PANSS negative symptom scores. Finally, we showed that LPO levels showed significant positive correlations with the RBANS total, attention function, and delayed recall scores.
Furthermore, we confirmed that plasma OS marker levels in first-episode SCZ patients exhibited significant abnormalities, suggesting an imbalance in the oxidation/antioxidation system during the early stages of the disorder. This finding supports the critical role of OS in the onset and progression of SCZ. We also found significantly elevated levels of MDA and LPO in first episode SCZ patients compared with healthy controls, consistent with previous research37. The increase in these markers indicates enhanced LPO reactions, suggesting that cell membrane structures have sustained significant damage38.LPO products not only weaken membrane stability but may also interfere with neuronal signaling, thereby exacerbating neurotoxic effects14. Second, elevated TAC levels reflects the body’s defensive response to OS. Unlike the TAC level reduction reported in some chronic or medication-treated patients, the increase we observed may represent a compensatory mechanism in the early stages of SCZ. When free radical production increases, the body maintains homeostasis by enhancing overall antioxidant capacity39. However, as SCZ progresses, antioxidant reserves may gradually become depleted, ultimately manifesting as a decline in TAC levels. These previous findings may explain the discrepancies observed across different studies and suggests that TAC exhibits stage-dependent dynamic characteristics40. The previous study also found decreased NOX levels, yet it remained a significant source of ROS41. However, the reduction in NOX levels appears to be at odds with the overall increase in oxidative stress. Research indicates42,43,44. Indeed, under prolonged stress conditions, the body downregulates NOX levels to reduce ROS production, thereby exerting a protective effect. However, mitochondrial dysfunction, glutamate excitotoxicity, and inflammation-related pathways may still sustain elevated ROS levels, indicating that OS associated with SCZ is likely not driven by a single pathway but rather results from the combined effects of multiple mechanisms. Furthermore, oxidative stress and neuroinflammation may influence schizophrenia symptoms and cognitive performance through broader neurobiological mechanisms. Recent studies emphasize that activated microglia play a pivotal role in neurodevelopmental abnormalities of schizophrenia. Dysregulation of microglia can lead to abnormal synaptic pruning, thereby disrupting the integration efficiency of neural networks45. Concurrently, microglia-mediated inflammatory responses correlate with reduced white matter integrity, and white matter damage further impairs inter-brain region information transmission45. Collectively, these processes constitute key pathological links affecting cognitive impairment and other symptom dimensions. However, no significant differences in GST and GR observed were observed in the current study. The glutathione system plays a central role in antioxidant defense, and alterations in its activity may be associated with disorder stage and patient characteristics, as well as external factors such as diet and smoking46. We hypothesize that the index patients have not yet experienced significant depletion of the glutathione system; however, this conclusion requires further validation in larger sample sizes and longitudinal studies. Compared with previous studies, the findings of the current study show both consistencies and differences. Most studies agree that patients with SCZ exhibit elevated levels of OS17, which is consistent with the elevated MDA and LPO levels we observed. However, regarding TAC, some studies have reported decreased levels or no change, with variations potentially related to disorder stage and drug exposure, as well as methodological differences47. Our findings support the hypothesis that TAC levels exhibit a dynamic pattern of “early elevation and late decline” during SCZ progression. Regarding NOX, previous studies have predominantly reported enhanced levels48; however, we observed the opposite, suggesting that underlying mechanisms associated with OS are complex, and not solely dependent on NOX but also involving mitochondrial and inflammatory pathways. These findings have significant clinical implications. First, elevated MDA and LPO levels indicate membrane structural damage in the early stages of SCZ. These markers therefore show promise as potential peripheral biomarkers for early risk assessment and auxiliary diagnosis. Second, increased TAC levels suggest a possible intervention window during stages of the disorder. During the early stages of SCZ, enhancing defenses through exogenous antioxidants or lifestyle interventions may potentially slow symptom progression. Likewise, the decrease in NOX levels suggest that future therapeutic strategies require careful design. Simply inhibiting NOX may be insufficient to alleviate overall OS; greater attention should be paid to mitochondrial function and other ROS sources. In summary, first-episode SCZ patients exhibit a characteristic OS profile indicative of enhanced LPO levels, compensatory elevation of antioxidant defenses, and reduced NOX levels. These findings not only deepen our understanding of the early molecular pathophysiology of SCZ but also provide new insights for early identification and personalized intervention.
We also analyzed the relationship between plasma OS marker levels and PANSS total and subscale scores in first-episode SCZ patients. Results demonstrated that GST and GR levels exhibited specific correlations across different symptom dimensions. GST, as a key component of the glutathione-related enzyme system, participates in peroxide clearance and maintenance of intracellular redox balance49. We found that GST levels showed a significant positive correlation with PANSS total scores, suggesting that antioxidant enzymes may, to some extent, reflect overall SCZ symptom severity. Research indicates that enhanced OS may occur in the early stages of SCZ, leading to GST upregulation to counteract free radicals. However, this compensatory mechanism may not fully mitigate neurotoxicity, implying that elevated GST levels may correlate positively with symptom burden26. Indeed, it has been shown that there is a consistent trend between elevated plasma GST levels in SCZ patients and the severity of clinical symptoms50. This finding supports the role of OS markers in disease phenotypes and suggests that GST holds potential as an auxiliary indicator for assessing the overall symptom burden in first-episode SCZ patients. Similarly, GR, which also part of the glutathione system, maintains stable intracellular levels of reduced glutathione, protecting cells from oxidative damage51. We found that GR levels were positively correlated with PANSS negative symptom scores, suggesting that the glutathione system may be closely involved in the development or maintenance of negative symptoms. Previous research has also indicated that GSH metabolic abnormalities in SCZ patients are associated with functional deficits in the prefrontal cortex and hippocampus, brain regions that precisely implicated in the emergence of negative symptoms52. Negative symptoms such as emotional blunting and social withdrawal often emerge in the early stages of the disorder and are associated with neuronal dysfunction and reduced synaptic plasticity53. The upregulation of GR may reflect a compensatory response to OS, but its positive correlation with negative symptoms also suggests that this compensation is insufficient to fully protect neural function, potentially indicating underlying neuropathological processes in SCZ. Overall, these findings further underscore the differential role of OS systems in SCZ symptom classification. Specifically, GST levels primarily correlate with overall symptom severity, whereas GR is more strongly associated with negative symptoms. This discovery aligns with previous research suggesting that OS-related markers may not only serve as potential biomarkers of symptom progression but also hold specific clinical guidance value across different symptom dimensions46,54,55. Future research may further explore whether dynamic changes in the glutathione system can predict symptom progression and treatment response, thereby providing a basis for personalized interventions in SCZ.
In addition our results indicated that first-episode SCZ patients scored significantly lower than healthy controls on both the total RBANS score and all subscales scores. This difference persisted even after controlling for confounding factors such as age, education level, BMI, and smoking status. These results suggest that cognitive impairment is not merely a consequence of disorder progression or medication use, but may represent one of the core features of SCZ, present even in the early stages of the illness56. Overall, in terms of overall cognitive functioning, the patient group exhibited a significant decline in total RBANS scores, indicating that cognitive deficits are widespread rather than confined to a single domain. This aligns with growing evidence that cognitive impairment in SCZ is cross-domain and pervasive, significantly impacting patients’ social functioning and prognosis53. Multiple studies have indicated that the relationship between cognitive impairment and functional recovery is more closely linked than that with clinical symptom improvement57. The results from the subscale scores indicate that the most significant differences were observed in attention and Visuospatial/constructional. This finding holds clinical significance, as attention deficits are considered one of the most core and stable cognitive impairments in SCZ patients. These deficits often precede the emergence of clinical symptoms and are closely associated with information processing speed53,58. Some scholars have proposed that attention processing deficits may represent one of the “endophenotypes” of SCZ, serving as an indicator of susceptibility to the disorder59. A marked decline in visual span suggests deficits in spatial processing and immediate visual memory, consistent with previous findings of impaired performance in working memory and visuospatial tasks60. These deficits may directly impact their ability to engage in daily learning and adapt to their environment. Furthermore, significant impairments in both immediate and delayed memory suggest that patients may experience difficulties across all stages of encoding, retaining, and retrieving new information. This not only limits their learning capacity but may also exacerbate communication barriers in social interactions. Research indicates that impaired memory function is closely associated with structural abnormalities in the hippocampus, which is considered one of the key pieces of evidence supporting the neurodevelopmental hypothesis of SCZ61. It is noteworthy that while speech function also exhibits significant decline, the degree of difference is relatively minor62. This may be related to the fact that language abilities remain partially preserved in some patients. However, existing research indicates that persistent impairment in speech function may be associated with cognitive deficits and clinical positive symptoms63.
In summary, our findings support the hypothesis that SCZ is characterized by widespread cognitive impairment. Given that the current study involved a population of first-episode SCZ patients, the findings suggest that cognitive deficits may stem from the intrinsic pathological mechanisms associated with SCZ rather than being the result of long-term medication, side effects, or social withdrawal. This discovery further underscores the necessity of early cognitive intervention.
Finally, we found that plasma LPO levels in first-episode SCZ patients were positively correlated with RBANS total scores and multiple cognitive dimensions including immediate memory, visual span, attention, and delayed recall. Multivariate regression analysis further confirmed that LPO levels independently predicted overall cognitive function, attention, and delayed recall. LPO is the end product of lipid peroxidation reactions and is commonly regarded as a marker of oxidative damage64. However, this study observed that elevated LPO levels were associated with better cognitive performance. This seemingly contradictory finding suggests that a more complex relationship may exist between OS and cognitive decline. One possibility is that elevated LPO levels partially reflect an adaptive response to early OS. Specifically, during the initial stages of SCZ, moderate oxidative signaling may activate intracellular stress response pathways and release neurotrophic factors, thereby exerting a compensatory effect on neural plasticity and cognitive maintenance in the short term. Research suggests that low-dose oxidative stimulation can induce antioxidant gene expression and enhance synaptic plasticity, mirroring the phenomenon where moderate stress promotes functional benefits65. Animal studies have revealed that mild elevations in LPO levels can enhance learning and memory performance, suggesting that OS in early stages is not solely a detrimental factor66,67. Another interpretation suggests that elevated LPO levels may correlate with patients’ metabolic status, inflammatory responses, or characteristics prior to drug treatment. In the early stages of SCZ, the body remains in a compensatory phase where certain metabolic and antioxidant pathways may not yet be suppressed by long-term pathological damage. Thus, increased LPO levels may more accurately reflect an active metabolic state associated with relatively well-preserved cognitive function. Studies indicate that in untreated first-episode SCZ patients, certain OS markers show a positive correlation with cognitive function. This relationship reverses following long-term medication use or symptom progression68,69. Additionally, some scholars have proposed that an interaction between OS and inflammatory responses exists, suggesting that mild oxidative stimulation may promote the maintenance of immune homeostasis, thereby indirectly supporting neural function70. Research indicates that as the disorder progresses, sustained elevated levels of LPO ultimately cause cumulative damage to the nervous system, leading to more severe cognitive deficits71. Others have found that persistently elevated levels of LPO in long-term SCZ patients are significantly associated with impairments in working memory and executive function72. Therefore, the findings of our study may reflect a dynamic window in the early stages of SCZ, where a nonlinear relationship between OS and cognitive function may emerge. At mild or moderate levels, LPO may exert compensatory or protective effects, but when accumulated over time and excessively activated, it may transition into pathological impairment. This finding offers a new perspective on understanding the early pathological processes of SCZ and suggests that future research should incorporate longitudinal follow-up to explore the dynamic trajectory of OS and its causal relationship with the progression of cognitive impairment.
The current study has several limitations that warrant attention. First, its cross-sectional design precludes establishing a causal relationship between changes in OS marker levels and clinical symptoms of SCZ or cognitive impairment. Prospective follow-up studies will be needed for further validation. Second, the sample size was relatively small. Although rigorous statistical adjustments were applied, the results may still be subject to sample bias. Third, the cognitive assessment in this study was conducted immediately after patients were admitted and diagnosed, aiming to avoid the influence of medication. However, patients were often in the acute phase or in an unstable condition at this time, and positive symptoms might interfere with their attention and comprehension abilities, thereby affecting the reliability of the cognitive assessment results. Fourth, OS marker levels are influenced by multiple physiological and environmental factors (e.g., diet, lifestyle, medication use). Although we controlled for several confounding variables (age, gender, education level, BMI, smoking, and disease duration), interference from other potential factors cannot be entirely ruled out. Finally, we assessed only a limited number of plasma OS markers, failing to encompass the entire molecular network of the oxidative/antioxidant system. Therefore, further research is needed to expand our understanding of mechanisms associated with SCZ symptom progression.
Conclusion
In summary, the findings suggest that OS imbalance may play a significant role in the clinical symptoms and cognitive impairment associated with SCZ. OS-related markers hold promise as potential biomarkers, offering valuable insights for early disorder identification and intervention. Future research should involve large-scale, longitudinal, and multicenter studies, integrating additional molecular markers and neuroimaging techniques to elucidate the underlying mechanisms of OS in the pathogenesis and progression of SCZ.
Data availability
The data supporting the results of this study are available upon request from the corresponding author.
References
Owen, M. J., Sawa, A. & Mortensen, P. B. Schizophrenia. Lancet 388, 86–97 (2016).
Simeone, J. C., Ward, A. J., Rotella, P., Collins, J. & Windisch, R. An evaluation of variation in published estimates of schizophrenia prevalence from 1990─2013: a systematic literature review. BMC Psychiatry 15, 193 (2015).
G. B. D. 2019 Mental Disorders Collaborators &. Global, regional, and national burden of 12 mental disorders in 204 countries and territories, 1990–2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet Psychiatry 9, 137–150 (2022).
Cai, R. et al. Relationship between negative symptoms, cognitive function and social function in schizophrenia: new insight from a network analysis. Front Psychiatry 16, 1623147 (2025).
Harvey, P. D. et al. Cognitive dysfunction in schizophrenia: an expert group paper on the current state of the art. Schizophr. Res. Cogn. 29, 100249 (2022).
Calzavara-Pinton, I. et al. Treatment of cognitive impairment associated with schizophrenia spectrum disorders: new evidence, challenges, and future perspectives. Brain Sci. 14, 791 (2024).
Gebreegziabhere, Y., Habatmu, K., Mihretu, A., Cella, M. & Alem, A. Cognitive impairment in people with schizophrenia: an umbrella review. Eur. Arch. Psychiatry Clin. Neurosci. 272, 1139–1155 (2022).
Tsapakis, E. M. et al. Pharmacological treatments of negative symptoms in schizophrenia-an update. J. Clin. Med 13, 5637 (2024).
Nitti, M. et al. Hormesis and oxidative distress: pathophysiology of reactive oxygen species and the open question of antioxidant modulation and supplementation. Antioxidants (Basel) 11, 1613 (2022).
Helmut, S. & Dean, P. J. Reactive oxygen species (ROS) as pleiotropic physiological signalling agents. Nat. Rev. Mol. Cell Biol. 21, 363–383 (2020).
Abdur, R. et al. Reactive oxygen species in biological systems: Pathways, associated diseases, and potential inhibitors-a review. Food Sci. Nutr 12, 675–693 (2024).
Morgenstern, R. Kinetic behavior of glutathione transferases: understanding cellular protection from reactive intermediates. Biomolecules 14, 641 (2024).
Silvestrini, A., Meucci, E., Ricerca, B. M. & Mancini, A. Total antioxidant capacity: biochemical aspects and clinical significance. Int J. Mol. Sci. 24, 10978 (2023).
Tortajada-Pérez, J. et al. Lipid oxidation at the crossroads: oxidative stress and neurodegeneration explored in caenorhabditis elegans. Antioxidants (Basel) 14, 78 (2025).
Qian, X. et al. The role of reactive oxygen species derived from different NADPH oxidase isoforms and mitochondria in oxalate-induced oxidative stress and cell injury. Urolithiasis 50, 149–158 (2022).
De Simone, G. et al. Schizophrenia synaptic pathology and antipsychotic treatment in the framework of oxidative and mitochondrial dysfunction: translational highlights for the clinics and treatment. Antioxidants (Basel) 12, 975 (2023).
Rambaud, V., Marzo, A. & Chaumette, B. Oxidative stress and emergence of psychosis. Antioxidants (Basel) 11, 1870 (2022).
Goh, X. X., Tang, P. Y. & Tee, S. F. Blood-based oxidation markers in medicated and unmedicated schizophrenia patients: a meta-analysis. Asian J. Psychiatr. 67, 102932 (2022).
Saidane, L. et al. Peripheral oxidative s,tress markers in schizophrenia: insights from erythrocyte glutathione peroxidase, plasma malondialdehyde, and trace element ratios in algerian patients. Biol. Trace Elem. Res. (2025).
Śmierciak, N. et al. Benefits and Meaning of Lipids Profile in Relation to Oxidative Balance and Brain Morphology in Schizophrenia. Int J. Mol. Sci. 24, 11375 (2023).
Sawa, A. & Sedlak, T. W. Oxidative stress and inflammation in schizophrenia. Schizophr. Res 176, 1–2 (2016).
Zhu, X. et al. Revealing the Oxidative Stress-Related Molecular Characteristics and Potential Therapeutic Targets of Schizophrenia through Integrated Gene Expression Data Analysis. Mol. Neurobiol. 62, 10484–10498 (2025).
Dwir, D. et al. Redox and Immune Signaling in Schizophrenia: New Therapeutic Potential. Int J. Neuropsychopharmacol. 26, 309–321 (2023).
Tsugawa, S. et al. Glutathione levels and activities of glutathione metabolism enzymes in patients with schizophrenia: A systematic review and meta-analysis. J. Psychopharmacol. 33, 1199–1214 (2019).
Liu, H., Xu, Y. & Peng, J. Glutathione S-Transferase M1/T1 Polymorphisms and Schizophrenia Risk: A New Method for Quality Assessment and a Systematic Review. Neuropsychiatr. Dis. Treat. 19, 97–107 (2023).
Carletti, B., Banaj, N., Piras, F. & Bossù, P. Schizophrenia and Glutathione: A Challenging Story. J. Pers. Med 13, 1526 (2023).
Li, J., Li, D., Guo, J., Wang, D. & Zhang, X. Age of Onset Moderates the Association between Total Antioxidant Capacity and Cognitive Deficits in Patients with Drug-Naïve Schizophrenia. Antioxid. (Basel) 12, 1259 (2023).
Xie, T. et al. Plasma total antioxidant status and cognitive impairments in first-episode drug-naïve patients with schizophrenia. Cogn. Neurodyn 13, 357–365 (2019).
Wang, D. M., Chen, D. C., Wang, L. & Zhang, X. Y. Sex differences in the association between symptoms and superoxide dismutase in patients with never-treated first-episode schizophrenia. World J. Biol. Psychiatry 22, 325–334 (2021).
Sapienza, J. et al. Neuroinflammation and kynurenines in schizophrenia: Impact on cognition depending on cognitive functioning and modulatory properties in relation to cognitive remediation and aerobic exercise. Schizophr. Res Cogn. 38, 100328 (2024).
Kaur, P., Khan, H., Grewal, A. K., Dua, K. & Singh, T. G. Therapeutic potential of NOX inhibitors in neuropsychiatric disorders. Psychopharmacol. (Berl.) 240, 1825–1840 (2023).
Rong, S. L. et al. Glutathione S-Transferase M1 and T1 polymorphisms and hypertension risk: an updated meta-analysis. J. Hum. Hypertens. 33, 454–465 (2019).
Bosia, M. et al. Cognition in Schizophrenia: Modeling the Interplay between Interleukin-1β C-511T Polymorphism, Metabolic Syndrome, and Sex. Neuropsychobiology 80, 321–332 (2021).
First, M. B. Diagnostic and statistical manual of mental disorders, 5th edition, and clinical utility. J. Nerv. Ment. Dis. 201, 727–729 (2013).
S R, K., A, F. & L A, O. The positive and negative syndrome scale (PANSS) for schizophrenia. Schizophr Bull, (1987).
Shura, R. D. et al. RBANS Validity Indices: a Systematic Review and Meta-Analysis. Neuropsychol. Rev. 28, 269–284 (2018).
Fraguas, D. et al. Oxidative Stress and Inflammation in First-Episode Psychosis: A Systematic Review and Meta-analysis. Schizophr. Bull. 45, 742–751 (2019).
Hurşitoğlu, O. et al. Diagnostic Performance of Increased Malondialdehyde Level and Oxidative Stress in Patients with Schizophrenia. Noro Psikiyatr Ars 58, 184–188 (2021).
Silvestrini, A. & Mancini, A. The Double-Edged Sword of Total Antioxidant Capacity: Clinical Significance and Personal Experience. Antioxid. (Basel) 13, 933 (2024).
Ruiz-Litago, F. et al. Adaptive response in the antioxidant defence system in the course and outcome in first-episode schizophrenia patients: a 12-months follow-up study. Psychiatry Res 200, 218–222 (2012).
Rada, B. & Leto, T. L. Oxidative innate immune defenses by Nox/Duox family NADPH oxidases. Contrib. Microbiol 15, 164–187 (2008).
Pecchillo Cimmino, T., Ammendola, R., Cattaneo, F. & Esposito, G. NOX Dependent ROS Generation and Cell Metabolism. Int J. Mol. Sci. 24, 2086 (2023).
Fizíková, I., Dragašek, J. & Račay, P. Mitochondrial Dysfunction, Altered Mitochondrial Oxygen, and Energy Metabolism Associated with the Pathogenesis of Schizophrenia. Int J. Mol. Sci. 24, 7991 (2023).
S S, P. & M A, M. [Oxidative stress in schizophrenia as a promising target for psychopharmacotherapy]. Zh Nevrol Psikhiatr Im S S Korsakova 121:131-138, (2021).
Sapienza, J., Pacchioni, F., Spangaro, M. & Bosia, M. Dysconnection in schizophrenia: Filling the dots from old to new evidence. Clin. Neurophysiol. 162, 226–228 (2024).
Poladian, N., Navasardyan, I., Narinyan, W., Orujyan, D. & Venketaraman, V. Potential Role of Glutathione Antioxidant Pathways in the Pathophysiology and Adjunct Treatment of Psychiatric Disorders. Clin. Pr. 13, 768–779 (2023).
Yao, J. K., Reddy, R., McElhinny, L. G. & van Kammen, D. P. Reduced status of plasma total antioxidant capacity in schizophrenia. Schizophr. Res 32, 1–8 (1998).
Wang, X., Pinto-Duarte, A., Sejnowski, T. J. & Behrens, M. M. How Nox2-containing NADPH oxidase affects cortical circuits in the NMDA receptor antagonist model of schizophrenia. Antioxid. Redox Signal 18, 1444–1462 (2013).
Hayes, J. D., Flanagan, J. U. & Jowsey, I. R. Glutathione transferases. Annu Rev. Pharm. Toxicol. 45, 51–88 (2005).
Spalletta, G. et al. Glutathione S-transferase alpha 1 risk polymorphism and increased bilateral thalamus mean diffusivity in schizophrenia. Psychiatry Res 203, 180–183 (2012).
Couto, N., Wood, J. & Barber, J. The role of glutathione reductase and related enzymes on cellular redox homoeostasis network. Free Radic. Biol. Med 95, 27–42 (2016).
Matsuzawa, D. et al. Negative correlation between brain glutathione level and negative symptoms in schizophrenia: a 3T 1H-MRS study. PLoS One 3, e1944 (2008).
Cullen, K. et al. Trajectories of social withdrawal and cognitive decline in the schizophrenia prodrome. Clin. Schizophr. Relat. Psychoses 4, 229–238 (2011).
Mednova, I. A. et al. The Role of GST Gene Polymorphic Variants in Antipsychotic-Induced Metabolic Disorders in Schizophrenia: A Pilot Study. Pharm. (Basel) 18, 941 (2025).
Nucifora, L. G. et al. Reduction of plasma glutathione in psychosis associated with schizophrenia and bipolar disorder in translational psychiatry. Transl. Psychiatry 7, e1215 (2017).
Kathryn, C. et al. Trajectories of social withdrawal and cognitive decline in the schizophrenia prodrome. Clin Schizophr Relat Psychoses, (2010).
Kharawala, S. et al. The relationship between cognition and functioning in schizophrenia: A semi-systematic review. Schizophr. Res Cogn. 27, 100217 (2022).
Vita, A. et al. Definition, assessment and treatment of cognitive impairment associated with schizophrenia: expert opinion and practical recommendations. Front Psychiatry 15, 1451832 (2024).
Cheng, P. et al. Decreased integration of default-mode network during a working memory task in schizophrenia with severe attention deficits. Front Cell Neurosci. 16, 1006797 (2022).
Liu, Y. et al. Face and object visual working memory deficits in first-episode schizophrenia correlate with multiple neurocognitive performances. Gen. Psychiatr. 34, e100338 (2021).
Dong, Y. et al. Abnormal hippocampal neurogenesis and impaired social recognition memory in two neurodevelopmental models of schizophrenia. FASEB J. 38, e70138 (2024).
Tyburski, E., Sokołowski, A., Chęć, M., Pełka-Wysiecka, J. & Samochowiec, A. Neuropsychological characteristics of verbal and non-verbal fluency in schizophrenia patients. Arch. Psychiatr. Nurs. 29, 33–38 (2015).
Jung, S., Lee, A., Bang, M. & Lee, S. H. Gray matter abnormalities in language processing areas and their associations with verbal ability and positive symptoms in first-episode patients with schizophrenia spectrum psychosis. Neuroimage Clin. 24, 102022 (2019).
Niki, E. Lipid peroxidation: physiological levels and dual biological effects. Free Radic. Biol. Med 47, 469–484 (2009).
Noriko, N. Role of oxidative stress in adaptive responses in special reference to atherogenesis. J. Clin. Biochem Nutr. 43, 131–138 (2008).
Farimah, B. et al. PPARγ activation improved learning and memory and attenuated oxidative stress in the hippocampus and cortex of aged rats. Physiological Rep. 10, e15538 (2022).
Antonio, V. et al. Oxidative Stress in the Early Stage of Psychosis. Curr. Top. Med Chem. 21, 1457–1470 (2021).
Mi, Y. et al. Comparative Analysis of the Pre- and Post-Medication Effects of Antipsychotic Agents on the Blood-Based Oxidative Stress Biomarkers in Patients with Schizophrenia: A Meta-Analysis. Curr Neuropharmacol, (2022).
Giuseppe, C. et al. Antioxidant Properties of Second-Generation Antipsychotics: Focus on Microglia. Pharm. (Basel) 13, 457 (2020).
Vallée, A. Neuroinflammation in Schizophrenia: The Key Role of the WNT/β-Catenin Pathway. Int J. Mol. Sci. 23, 2810 (2022).
Wang, Y. et al. Long-term exposure to advanced lipid peroxidation end products impairs cognitive function through microbiota-gut-brain axis. Food Chem. 461, 140864 (2024).
Morén, C. et al. Mitochondrial Oxidative Phosphorylation System Dysfunction in Schizophrenia. Int J. Mol. Sci. 26, 4415 (2025).
Acknowledgements
We sincerely appreciate everyone who participated in the research, both the patients and healthy controls. The study was financially supported by the Suzhou Clinical Medical Center for Mood Disorders (grant no. Szlcyxzx202109), Suzhou Key Laboratory (grant no. SZS2024016), Suzhou Multicenter Clinical Research Project on Major Diseases (grant no. DZXYJ202413, MR-32-25-054378), Suzhou Municipal Key Project for Applied Basic Research in Medical and Health Sciences(SYW2025022), Science Foundation of Kangda College of Nanjing Medical University (grant no. KD2024KYJJ306), Nantong Municipal Health Commission Research Project (Grant No. MS2024076, MS2024074, QN2023040). The study’s funding sources had no influence on the study’splanning, data gathering, analysis, publication choice, or paper preparation.
Author information
Authors and Affiliations
Contributions
X.Z., F.J., Q.T., and T.J. were responsible for study design and manuscript preparation. Q.Y. and P.W. performed statistical analysis. L.J., X.M., and C.Z. handled patient recruitment, clinical scoring, and sample collection. All authors contributed to the final manuscript and approved it for publication.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Jiang, F., Jin, T., Yang, Q. et al. Abnormal plasma oxidative stress markers in first-episode schizophrenia and associations with clinical symptoms and cognitive function. Schizophr 12, 20 (2026). https://doi.org/10.1038/s41537-025-00726-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41537-025-00726-7