Abstract
This study aimed to investigate the association between dietary copper intake and muscle mass. Low muscle mass was defined according to the Foundation for the National Institutes of Health (FNIH) consensus. Among 9693 adults (female 50.3%), mean age was 39.5 ± 0.3 years, and mean copper intake was 1.2 ± 0.01 mg/day. Compared to the lowest quintile, participants in the highest quintile of copper intake exhibited a decreased risk of low muscle mass (OR = 0.43; 95% CI: 0.26, 0.72). For per 0.5 mg/day increase in copper intake, the risk of low muscle mass was decreased by 20% (P < 0.05). This association was mediated by serum albumin, bilirubin, and gamma-glutamyl transferase, with mediating proportions of 5.92%, 2.84%, and 2.56% respectively (P < 0.001). In summary, increasing copper intake may serve as a protective factor to prevent muscle loss, and oxidative stress may be crucial to this process.
Similar content being viewed by others
Introduction
Skeletal muscle comprises about 40% of the human body weight, playing a crucial role in maintaining physical functions and physiological health. Low muscle mass is a basic and objective component of sarcopenia, characterized by progressive loss of skeletal muscle mass, occurring in aging and other clinical conditions, with a prevalence as high as 27% among adults1. Growing epidemiological evidence underscores that individuals with low muscle mass consistently face elevated risks of falls, fractures, cardiometabolic diseases, hospitalization, and premature death2,3, contributing to a substantial economic burden on individuals, families, and healthcare systems4,5. To date, low muscle mass remains a largely untreatable condition with no targeted medical therapies5. Therefore, identifying protective or risk factors associated with muscle mass is an imperative need to guide the implementation of early interventions to mitigate adverse outcomes.
Copper is an essential trace element involved in antioxidant defense, inflammation, and immune function. Dietary copper serves as the primary source of copper in the human body6. Current evidence has demonstrated that increased dietary copper intake was associated with lower risks of age-related disease, including metabolic syndrome7, stroke8, myocardial infarction9, Parkinson’s disease10, and telomere length shortening11. Given the strong association between muscle mass and aging2,5, we wondered whether increased dietary copper intake could mitigate low muscle mass risk, but direct evidence was absent. Recent studies showed that serum copper is closely related to muscle mass and function, but relevant evidence remains insufficient and inconsistent12,13,14,15. Additionally, no correlation between serum copper and dietary copper was observed in previous studies16,17, which suggested that serum copper may not well reflect the dietary copper intake levels. Whether dietary copper intake is a protective factor for muscle mass remains unclear.
Oxidative stress is a pathological process characterized by an imbalance between the production of reactive oxygen species (ROS) and antioxidant defenses. Copper is involved in regulating oxidative stress through antioxidant, coenzyme, and signal transduction pathways18. Increasing evidence has shown that oxidative stress is a key factor that triggers skeletal muscle mass loss19,20. Based on the close connection among them, we hypothesize that oxidative stress may be involved in mediating the association of copper and muscle mass.
To fill these knowledge gaps, we sought to investigate the association between dietary copper intake and low muscle mass in the National Health and Nutrition Examination Survey (NHANES) 2011–2018, and further explored the potential mediating effects of oxidative stress-related indicators.
Results
Baseline characteristics
This study included 9693 eligible individuals (mean age 39.5 ± 0.3 years, female 50.3%) with a mean dietary copper intake level of 1.2 ± 0.01 mg/day. Participants with a higher dietary copper intake were characterized as being aged, male, non-Hispanic White, high education level, high family income, non-smokers, drinkers, with exceeding recommended physical activity levels, high total energy intake, high healthy eating index (HEI), low body mass index (BMI), low estimated glomerular filtration rate (eGFR), and without albuminuria. In addition, significant differences in oxidative stress indicators were observed, with participants with an elevated copper intake having the higher levels of serum albumin, bilirubin, and uric acid, and a lower gamma-glutamyl transferase (GGT) concentration (P < 0.001). Muscle mass and relative muscle mass indicators were calculated, and significant differences in them were observed among the groups with different levels of copper intake. Participants in the highest quintile of copper intake exhibited the highest muscle mass and relative muscle mass levels (P < 0.001) (Table 1).
Association of dietary copper intake with low muscle mass
Among 9693 individuals, 837 patients with low muscle mass were identified, with a weighted prevalence rate of 7.2%. Participants in the lowest quintile of dietary copper intake exhibited the highest prevalence rate of low muscle mass, up to 10.6%, while participants in the highest quintile presented the lowest prevalence rate (3.6%). After comprehensive adjustment for confounding factors, participants in the highest quintile of dietary copper intake exhibited a low risk of low muscle mass, with an OR of 0.43 (95% CI, 0.26–0.72), as compared with the lowest quintile (Table 2).
Dose-response curves showed a decreasing risk of low muscle mass as dietary copper intake increased, and linear dose-response associations were observed in the overall population and the male group (P for overall < 0.01 and P for non-linearity > 0.05). In the overall population, for per 0.5 mg/day increment, the OR of low muscle mass was 0.80 (95% CI, 0.67–0.96). A similar dose-response trend was observed in women, but it was not statistically significant (P for overall > 0.05) (Fig. 1).
All models were adjusted for age (continuous), sex (except in the sex subgroup analyses), race/ethnicity, education, family income, smoking, alcohol intake, physical activity, total energy intake (in quintiles), healthy eating index (in quintiles), BMI (<18.5, 18.5–24.9, 25–29.9, or ≥30 kg/m2), hypertension, dyslipidemia, diabetes, cancer, albuminuria, and eGFR (in quintiles).
Subgroup analyses and sensitivity analyses
In stratified analyses, individuals with an elevated dietary copper intake consistently demonstrated a significant association with a decreased risk of low muscle mass across most subgroups. Notably, significant interactions were observed in groups classified by race/ethnicity (P for interaction < 0.001) and hypertension (P for interaction < 0.05). The ORs of the highest quintile of copper intake (versus quintile 1) and low muscle mass were 0.18 (95% CI, 0.08–0.41) among Non-Hispanic White participants, lower than that of other race/ethnicity participants, and 0.22 (95% CI, 0.09–0.51) among patients with hypertension, lower than that of participants without hypertension. Details are presented in Table S1, Figs. S1 and S2.
In the sensitivity analyses, cotinine was considered as a substitute for smoking status, and the OR of dietary copper intake and low muscle mass was largely unchanged. To identify whether dietary copper intake is an independent protective factor for muscle mass, we further adjusted for other dietary factors, including various macronutrients, vitamins, other trace elements, caffeine, and theobromine, and all yielded consistently significant results (Table S2).
Oxidative stress indicators mediate the association between dietary copper intake and low muscle mass
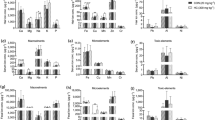
Figure 2A and Table S3 show the associations of dietary copper intake with oxidative stress indicators. We found that an elevated dietary copper intake was associated with an increase in serum albumin and bilirubin levels, and a decrease in GGT concentrations (P for trend < 0.01). Restricted cubic spline (RCS) models showed a nonlinear dose-response association of dietary copper intake with albumin (P for overall < 0.05 and P for non-linearity < 0.05), and linear dose-response associations of dietary copper intake with bilirubin and GGT (P for overall < 0.05 and P for non-linearity > 0.05) (Fig. S3). Additionally, we found no statistically significant association between dietary copper intake and serum uric acid in either the Generalized Linear Model (GLM) or the RCS model.
A Associations of dietary copper intake and oxidative stress indicators. B Associations of oxidative stress indicators and low muscle mass. GGT gamma-glutamyl transferase. All statistical analyses accounted for sample weights, strata, and primary sampling units to reflect the nationally representative estimates. All models were adjusted for age (continuous), sex, race/ethnicity, education, family income, smoking, alcohol intake, physical activity, total energy intake (in quintiles), healthy eating index (in quintiles), BMI (<18.5, 18.5–24.9, 25–29.9, or ≥30 kg/m2), hypertension, dyslipidemia, diabetes, cancer, albuminuria, and eGFR (in quintiles).
Of the four oxidative stress indicators, albumin (P for trend < 0.01) and bilirubin (P for trend = 0.06) showed a negative association with low muscle mass, whereas GGT (P for trend < 0.05) and uric acid (P for trend < 0.01) showed a positive association with low muscle mass (Fig. 2B and Table S4). Subsequently, dose-response curves showed a decreasing risk of low muscle mass as albumin or bilirubin increased (P for overall < 0.01 and P for non-linearity > 0.05), and an increasing risk of low muscle mass as GGT or uric acid increased (P for overall < 0.01 and P for non-linearity < 0.05) (Fig. S4).
Furthermore, mediation analyses were conducted to evaluate the potential mediation effects. Serum albumin, bilirubin, and GGT were all observed to a statistically significant mediated effects on the association of dietary copper intake with low muscle mass, and the proportions of mediation were 5.92%, 2.84%, and 2.56% respectively (P < 0.001), whereas the mediating effect of uric acid was not significant (P = 0.71). Detailed results are shown in Fig. 3.
GGT, gamma-glutamyl transferase. All models were adjusted for age (continuous), sex, race/ethnicity, education, family income, smoking, alcohol intake, physical activity, total energy intake (in quintiles), healthy eating index (in quintiles), BMI (<18.5, 18.5–24.9, 25–29.9, or ≥30 kg/m2), hypertension, dyslipidemia, diabetes, cancer, albuminuria, and eGFR (in quintiles).
Discussion
Leveraging data from representative populations in the US, we identified a significant negative association between dietary copper intake and low muscle mass. This association remained stable across most subgroups and in sensitivity analyses with adjusting for various other nutrients. Additionally, this association was partially mediated by oxidative stress-related indicators, including albumin, bilirubin, and GGT. These results highlighted that increasing dietary copper intake may serve as a protective factor to mitigate muscle mass loss, and oxidative stress may play a crucial role in this process.
To date, as the most basic components of sarcopenia, low muscle mass remains a largely untreatable condition with no targeted medical therapies, and strengthening dietary nutrition is a major intervention. The role of micronutrients in muscle metabolism and function has recently attracted considerable attention21. As an essential trace element and nutrient component for the human body, copper is closely related to muscle mass and strength. Chen et al.’s12 and Ngu et al.’s13 cross-sectional studies both revealed a significantly inverse association between serum copper concentrations and skeletal muscle mass in adults. However, a case-control study14 conducted in a hospital observed no significant difference in serum copper levels between patients with or without sarcopenia, which is a condition defined as the coexistence of low muscle mass and strength. Sha et al.’s15 Mendelian randomization study among Europeans also observed no causal association between serum copper concentrations and sarcopenia. The current evidence in this field remains insufficient and inconsistent, which may be contributed to by small sample size and differences in muscle mass measurement methods, adjustment of confounders, race/ethnicity, and regions.
Additionally, whether serum copper can well reflect the level of dietary copper intake and copper accumulation in other organs should not be ignored. Previous studies observed no correlation between dietary copper intake and serum copper16, and found that elevated dietary copper dramatically increased liver, kidney, and lung copper levels, but serum copper was not affected17. Our study pre-analyzed this issue and also observed no positive association or linear trend between daily dietary copper intake and serum copper levels. Copper metabolism and balance in the body are affected by physical status. Earlier studies have shown that serum copper increases with age22,23,24 and chronic low-grade inflammation25, which may be accompanied by or prone to serious diseases, cachexia, and sarcopenia. These make it implausible to clarify whether increased serum copper levels and decreased muscle mass are a parallel phenomenon or a causal relationship in cross-sectional studies.
Dietary copper serves as the primary source of copper in the human body6, and using dietary surveys to assess copper exposure levels can partially bypass this limitation, but epidemiologic studies are absent. A pertinent study26 conducted among 786 community-dwelling older adults in Japan observed a significantly lower level of dietary copper intake in the subjects with frailty, which is a geriatric syndrome closely related to low muscle mass27. Bagheri et al.’s28 study conducted among 300 elderly adults in Iran reported that adherence to a copper-rich dietary pattern was inversely associated with low muscle strength, indicating that elevated dietary copper might have a protective effect on muscle. These results both supported our findings. Leveraging data from a large and representative population, we assessed dietary copper intake levels and revealed a negative association with low muscle mass, suggesting that increasing copper intake may be a protective factor for muscle mass. To our knowledge, this is the first study to examine the direct association between dietary copper intake and muscle mass among the general population.
Copper is rich in foods such as nuts, tea, animal livers, some seafood products, and so on29,30. Tea contains a variety of phytochemicals, such as polyphenols, flavonoids, caffeine, and theobromine, which may have strong antioxidant activity31. Animal-based products, especially the internal organs, may contain a large amount of VA32. Therefore, in this study, we further adjusted for HEI and other dietary factors, including various macronutrients, vitamins, some trace elements, caffeine, and theobromine, and all yielded consistently significant results. Nevertheless, due to the complexity of the nutritional profile of foods and the lack of available information, there are still some nutrients that have not been included in models. It is difficult to attribute the observed effects on muscle mass solely to copper. Prospective studies and randomized controlled trials are further required to strengthen the evidence regarding the potential beneficial effects of copper supplementation on muscle health.
After the intake of dietary copper, it is mainly absorbed in the small intestine and excreted through feces and urine. Evidence showed that urine and fecal copper concentrations partially reflected its exposure levels29. After copper is absorbed into the body, it accumulates in organs such as the blood and liver29. Meanwhile, copper serves as an auxiliary factor for many redox enzymes, and ceruloplasmin is the most abundant copper-dependent iron oxidase33. Blood copper and caeruloplasmin are generally used to reflect the copper exposure levels, but their levels may be affected by physical status and illnesses34. Additionally, Cu/Zn superoxide dismutase, diamine oxidase, and skin lysyl oxidase were used as biomarkers of copper exposure levels in some studies30,34. However, the sensitivity and specificity of these indicators remain to be further verified. There is a lack of agreement on the index of copper status, and reliable and sensitive biomarkers are still required in future studies.
In stratified analyses, we observed a stronger protective effect of increased copper intake on muscle mass in Caucasian individuals compared to their counterparts of other racial/ethnic backgrounds. While prior studies have indicated a lower incidence of muscle mass loss among non-Hispanic White individuals35, and demonstrated the racial difference in copper concentrations among the American population36, the interplay among race, copper, and muscle mass remains largely unknown. These suggest that race should be considered in future research and further extending the findings to diverse populations is necessary. Additionally, a stronger protective effect of dietary copper on muscle mass was also observed in individuals with hypertension. A study regarding micronutrients also found a significant association between nutrient Vitamin D and muscle mass in patients with hypertension, but this association was not significant in those without hypertension37, which suggested that the hypertensive condition may play a crucial role between nutritional supplementation and muscle health. The relationship between hypertension and muscle mass is most likely bidirectional, sharing common factors such as inflammation, insulin resistance, and oxidative stress38. Hypertension contributes to progressive loss of muscle mass, while individuals with low muscle mass are prone to develop hypertension39,40. This vicious cycle may be reversed through nutritional supplementation, and it may yield an additional benefit, as shown in our study.
Although copper is recognized as an essential trace element, uncertainties remain regarding copper reference values for humans. The literature indicated that, for adults, the reference copper intake is 2 mg/day in France, 1.7 mg/day in Australia, 1.2 mg/day in the UK, and 0.9 mg/day in the Nordic countries30. The Chinese dietary guidelines indicated that the recommended nutrient intake (RNI) of copper should be 0.8 mg/day, with the tolerable upper intake level (UL) being 8.0 mg/day for adults. However, the dietary guidelines for Americans did not offer the RNI and UL values of copper. Taylor, Alicia A et al. summarized the scientific bases of the key reference values for copper intake for Americans and offered a longstanding oral reference dose for copper, which was 0.04 mg/kg bw/day, and a UL value of 0.14 mg/kg bw/day41. Based on the above values, for an adult weighing 60 kg, the RNI of copper should be 2.4 mg/day, with the UL being 8.4 mg/day. In our study, the mean dietary copper intake level was 1.2 mg/day, which is far lower than 2.4 mg/day, indicating that some individuals may be deficient in copper. In RCS models, within the range of 0–3.5 mg/day, the protective effect on muscle mass increased as dietary copper intake increased. In the future, the formulation and update of dietary guidelines regarding dietary copper are necessary, and our research may provide some references.
The underlying mechanisms of muscle mass loss were complicated and poorly understood. Increased oxidative stress and inflammation were considered important upstream signals of skeletal muscle loss42. For the ongoing renewal of muscle proteins, the processes of synthesis and degradation are balanced in muscle tissue43,44. Previous studies have shown that ROS controlled the redox signaling pathway in muscle fibers, and an increase in ROS will reduce skeletal muscle protein synthesis and promote protein hydrolysis, which may contribute to muscle atrophy45. These findings suggested that improving oxidative stress may mitigate muscle mass loss. Copper has been widely demonstrated to be involved in antioxidant defense through antioxidant, coenzyme, and signal transduction pathways18, which makes us wonder whether oxidative stress plays a role in the association between copper and muscle mass. Albumin, bilirubin, GGT, and uric acid are common clinical biochemical indicators that can partly reflect oxidative stress in the human body46,47. Our results confirmed the significant mediating effect of them on the association between dietary copper intake and low muscle mass risk, which provides clues regarding the underlying mechanisms. In addition, decreased serum albumin and bilirubin, and increased GGT and uric acid levels may also serve as potential blood markers for low muscle mass risk in clinical screening.
Age is a key factor contributing to muscle mass loss, and copper may delay aging by engaging in oxidative stress and inflammatory pathways, thereby protecting muscle health. Mitochondria are generally considered drivers of muscle loss. With aging, mitochondrial function declines, which leads to a decrease in mitochondrial respiratory capacity and an increase in the production of harmful ROS48. Oxidative damage contributes to the loss of muscle regenerative potential48. Copper serves as a cofactor for several enzymes, such as Cn/Zn-SOD. Cn/Zn-SOD serves as the first protector against damage from ROS and superoxide anion radical (O2•−) radicals in both the cytosol and mitochondrial intermembrane space49. Additionally, aging is a proinflammatory process, and many inflammatory mediators are associated with muscle loss48. Copper can enhance anti-inflammatory cytokine secretion and inhibit inflammatory responses50, which may be beneficial for muscle health.
Our study possessed two noteworthy strengths. First, leveraging data from representative populations in the US and considering sample weights in statistical analyses, we furnished the first scientific and persuasive evidence regarding dietary copper intake and low muscle mass. Furthermore, utilizing the high-quality data, we were able to consider numerous potential confounders, including lifestyle, various dietary factors, and common long-term conditions, which allowed us to identify that increasing dietary copper intake may serve as a potentially protective factor for muscle mass.
However, several limitations warrant consideration. First, given the observational nature of our research utilized, direct causality inference should be cautious. Second, the statistical power is restricted in detecting weak or moderate differences in the subgroup analyses, necessitating cautious interpretation of the results. Third, although a 24-h dietary recall is relatively detailed in determining nutrient intake of individuals and has been widely used in previous epidemiology studies, dietary assessments reflect exposure levels at the time, and long-term exposure is largely uncertain. Finally, due to the complexity of the nutritional components in foods, although we have attempted to adjust for various dietary factors, it remains difficult to attribute the observed effects on muscle mass solely to copper.
In conclusion, increasing dietary copper intake may serve as a protective factor to prevent and mitigate muscle loss, and oxidative stress may play a role in this process.
Methods
Study population
The NHANES is a large-scale and nationally representative program conducted among the U.S. civilians, providing comprehensive information on demography, sociology, lifestyle, physical examination, and laboratory biochemical indicators51. All procedures adhered to the principles outlined in the Declaration of Helsinki. The program was approved by the National Center for Health Statistics Ethics Review Board (Protocols 98-12, 2005-06, 2011-17, and 2018-01). All participants signed informed consent forms.
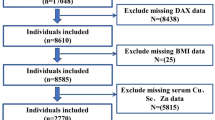
In this study, we utilized the data from NHANES 2011–2018, during which muscle mass was assessed using dual-energy X-ray absorptiometry (DXA) scans. After excluding participants aged <20 years, and those without complete information on dietary survey, DXA, BMI, demographic data, or oxidative stress indicators, a total of 9693 participants were finally included in analyses. The flow chart is exhibited in Fig. S5.
Dietary and serum copper assessment
All participants were eligible for two 24-h dietary recall interviews. The first dietary recall interview was conducted at Mobile Examination Centers, and the second interview was conducted using a telephone. Dietary nutrients and food components were estimated based on the Food and Nutrient Database for Dietary Studies from the United States Department of Agriculture51. In this study, the daily intake of copper and other dietary nutrients was calculated as the average of two reliable interviews, or a single interview when only one was deemed reliable.
Among some participants in NHANES, serum copper concentrations were detected using inductively coupled plasma dynamic reaction cell mass spectrometry. The NHANES Laboratory Procedures Manual elaborates on the detailed instructions for sample collection, preparation, and laboratory method51.
We pre-analyzed the association between dietary copper intake and serum copper concentrations, and found no positive associations or linear trends, which suggested that serum copper may not truly reflect the dietary copper intake levels. Detailed results are presented in Tables S5–S7 and Fig. S6.
Low muscle mass ascertainment
DXA method and appendicular lean mass (ALM) index were used to identify low muscle mass because they were standardized, valid, reliable, accurate, and cost-effective52. ALM was calculated as the sum of the muscle mass of both legs and arms measured by DXA. Considering that the study population was citizens in the U.S., low muscle mass was defined based on the guidelines established by the Foundation for the National Institutes of Health53 and was characterized by ALM adjusted by BMI (ALM/BMI) < 0.512 for women and <0.789 for men, as reported in previous studies in NHANES46,54,55.
Oxidative stress indicators detection
Albumin, bilirubin, GGT, and uric acid were selected as oxidative stress indicators based on previous studies in NHANES46,47. Blood samples were obtained during the in-person examination and then processed, stored, and shipped to the Collaborative Laboratory Services for analysis. Serum specimens were used to determine concentrations of the above four indicators. Detailed methodologies for sample handling, processing, and laboratory analyses were delineated in the NHANES Laboratory/Medical Technologists Procedures Manual51.
Covariates
Sociodemographic characteristics encompassed age, sex, race/ethnicity, education, and family income. Behavioral factors and physical examination indicators included smoking, alcohol consumption, physical activity (classified according to the 2018 Physical Activity Guidelines for Americans56), total energy intake, HEI, and BMI. HEI was calculated using HEI-2015, with higher scores indicating a better-quality diet57,58. Previous studies have demonstrated the associations of long-term conditions (especially cancer) with chronic inflammation, muscle mass loss, and sarcopenia2,59, so we considered several common morbidities as potential confounding factors, including hypertension, dyslipidemia, diabetes, and cancer. Details are provided in Supplementary Methods for covariates definitions. We further considered albuminuria (defined as a urinary albumin to creatinine ratio of >30 mg/g) and eGFR (calculated using the Chronic Kidney Disease Epidemiology Collaboration equation)60.
Statistical analysis
All statistical analyses accounted for sample weights, strata, and primary sampling units to reflect the nationally representative estimates. Baseline characteristics were presented as mean (SE) and percentages (%). The participants were stratified into quintiles 1–5 (Q1–Q5) according to the levels of dietary copper intake or oxidative stress indicators. Multivariate logistic regression models were performed to explore the associations of dietary copper intake and oxidative stress indicators with low muscle mass. GLM was used to observe the associations of dietary copper intake with serum oxidative stress indicators. In each regression above, three models were fitted. Model 1 was adjusted for age (continuous), sex, and race/ethnicity. Model 2 was further adjusted for education, family income, smoking, alcohol consumption, physical activity, total energy intake (in quintiles), HEI (in quintiles), and BMI (<18.5, 18.5–24.9, 25–29.9, or ≥30 kg/m2). Model 3 was further adjusted for hypertension, dyslipidemia, diabetes, cancer, albuminuria, and eGFR (in quintiles). Stratified analyses and sensitivity analyses were further performed to test the robustness of the results. SAS version 9.4 (SAS Institute, USA) was used for these statistical analyses.
RCS regression models were performed to visualize the dose-response associations among dietary copper intake, oxidative stress indicators, and low muscle mass in men, women, and the overall population. Causal mediation analyses were performed to calculate the mediating effects and proportions of oxidative stress indicators on the association between copper intake and low muscle mass. Due to the lack of available RCS and mediation models for a complex, multistage sampling survey, weights were not considered. R 4.4.2, R-Studio, and R packages (“rms” and “mediation”) were used for these analyses.
Data availability
The datasets used to support the findings of this study are available from the corresponding author upon reasonable request.
Code availability
The code generated during the current study is available from the corresponding author on reasonable request.
References
Petermann-Rocha, F. et al. Global prevalence of sarcopenia and severe sarcopenia: a systematic review and meta-analysis. J. Cachexia Sarcopenia Muscle 13, 86–99 (2022).
Yuan, S. & Larsson, S. C. Epidemiology of sarcopenia: prevalence, risk factors, and consequences. Metabolism 144, 155533 (2023).
Zhou, H.-H. et al. Association of muscle wasting with mortality risk among adults: a systematic review and meta-analysis of prospective studies. J. Cachexia Sarcopenia Muscle 14, 1596–1612 (2023).
Papadopoulou, S. K. Sarcopenia: a contemporary health problem among older adult populations. Nutrients 12, 1293 (2020).
Sayer, A. A. et al. Sarcopenia. Nat. Rev. Dis. Prim. 10, 68 (2024).
Ruiz, L. M., Libedinsky, A. & Elorza, A. A. Role of copper on mitochondrial function and metabolism. Front. Mol. Biosci. 8, 711227 (2021).
Ding, J. et al. Associations of the dietary iron, copper, and selenium level with metabolic syndrome: a meta-analysis of observational studies. Front. Nutr. 8, 810494 (2021).
Yang, L., Chen, X., Cheng, H. & Zhang, L. Dietary copper intake and risk of stroke in adults: a case-control study based on National Health and Nutrition Examination Survey 2013-2018. Nutrients 14, 409 (2022).
Wen, H. et al. Dietary copper intake and risk of myocardial infarction in US adults: a propensity score-matched analysis. Front. Cardiovasc. Med. 9, 942000 (2022).
Zeng, Z. et al. Dietary Copper Intake and Risk of Parkinson’s Disease: a cross-sectional Study. Biol. Trace Elem. Res. 202, 955–964 (2024).
Gong, H. et al. The relationship between dietary copper intake and telomere length in hypertension. J. Nutr. Health Aging 26, 510–514 (2022).
Chen, Z. et al. Association between serum copper levels and muscle mass: results from NHANES 2011-2016. Environ. Sci. Pollut. Res. Int. 31, 6847–6856 (2024).
Ngu, Y. J. et al. Association between essential and non-essential metals, body composition, and metabolic syndrome in adults. Biol. Trace Elem. Res. 200, 4903–4915 (2022).
Xu, B. et al. Factors affecting sarcopenia in older patients with chronic diseases. Ann. Palliat. Med. 11, 972–983 (2022).
Sha, T. et al. Causal relationship of genetically predicted serum micronutrients levels with sarcopenia: a mendelian randomization study. Front. Nutr. 9, 913155 (2022).
Gonoodi, K. et al. Serum and dietary zinc and copper in Iranian girls. Clin. Biochem. 54, 25–31 (2018).
Southern, L. L. & Stewart, T. B. Performance and tissue copper concentrations of control and Ascaris suum-infected pigs fed excess dietary copper. J. Parasitol. 70, 668–670 (1984).
Gromadzka, G., Tarnacka, B., Flaga, A. & Adamczyk, A. Copper dyshomeostasis in neurodegenerative diseases-therapeutic implications. Int. J. Mol. Sci. 21, 9259 (2020).
Zhang, H. et al. Oxidative stress: roles in skeletal muscle atrophy. Biochem. Pharmacol. 214, 115664 (2023).
Fanò-Illic, G. & Fulle, S. Oxidative stress in skeletal muscle. Antioxidants 11, 1299 (2022).
van Dronkelaar, C. et al. Minerals and sarcopenia; the role of calcium, iron, magnesium, phosphorus, potassium, selenium, sodium, and zinc on muscle mass, muscle strength, and physical performance in older adults: a systematic review. J. Am. Med. Dir. Assoc. 19, 6–11.e3 (2018).
Harman, D. The free radical theory of aging: effect of age on serum copper levels. J. Gerontol. 20, 151–153 (1965).
Madarić, A., Ginter, E. & Kadrabová, J. Serum copper, zinc and copper/zinc ratio in males: influence of aging. Physiol. Res. 43, 107–111 (1994).
Lossow, K. et al. Aging affects sex- and organ-specific trace element profiles in mice. Aging 12, 13762–13790 (2020).
Myhre, O., Utkilen, H., Duale, N., Brunborg, G. & Hofer, T. Metal dyshomeostasis and inflammation in Alzheimer’s and Parkinson’s diseases: possible impact of environmental exposures. Oxid. Med. Cell. Longev. 2013, 726954 (2013).
Naito, Y. et al. A cross-sectional study on the relationship between nutrient/food intake and gut microbiota in frailty among older community residents: The Kyotango study. J. Clin. Biochem. Nutr. 75, 161–173 (2024).
Wen, L. et al. Association of frailty and pre-frailty with all-cause and cardiovascular mortality in diabetes: three prospective cohorts and a meta-analysis. Ageing Res. Rev. 106, 102696 (2025).
Bagheri, A., Hashemi, R., Heshmat, R., Motlagh, A. D. & Esmaillzadeh, A. Patterns of Nutrient Intake in Relation to Sarcopenia and Its Components. Front. Nutr. 8, 645072 (2021).
Linder, M. C. Copper homeostasis in mammals, with emphasis on secretion and excretion. A review. Int. J. Mol. Sci. 21, 4932 (2020).
Bost, M. et al. Dietary copper and human health: current evidence and unresolved issues. J. Trace Elem. Med. Biol. 35, 107–115 (2016).
Bag, S., Mondal, A., Majumder, A. & Banik, A. Tea and its phytochemicals: Hidden health benefits & modulation of signaling cascade by phytochemicals. Food Chem. 371, 131098 (2022).
Schweiggert, R. M. & Carle, R. Carotenoid deposition in plant and animal foods and its impact on bioavailability. Crit. Rev. Food Sci. Nutr. 57, 1807–1830 (2017).
Adelstein, S. J., Coombs, T. L. & Vallee, B. L. Metalloenzymes and myocardial infarction. I. The relation between serum copper and ceruloplasmin and its catalytic activity. N. Engl. J. Med. 255, 105–109 (1956).
Uriu-Adams, J. Y., Rucker, R. B., Commisso, J. F. & Keen, C. L. Diabetes and dietary copper alter 67Cu metabolism and oxidant defense in the rat. J. Nutr. Biochem. 16, 312–320 (2005).
Bigman, G. & Ryan, A. S. Implications of race and ethnicity in Sarcopenia US National Prevalence of Sarcopenia by muscle mass, strength, and function indices. Gerontol. Geriatr. Res. 4, 126 (2021).
Glauser, T. A., Titanic-Schefft, M. & Pippenger, C. E. Racial differences in free radical scavenging enzyme activity in children. J. Child Neurol. 14, 382–387 (1999).
Baretić, M., Matovinović, M., Vukić, T. & Ranilović, D. Vitamin D: missing link between hypertension and muscle mass. J. Hum. Hypertens. 32, 1–2 (2018).
Jurdana, M. & Žiberna, L. Sarcopenic obesity and hypertension in elderly patients: a narrative review of pathophysiology and management strategies. Ann. Ist. Super. Sanita 59, 231–239 (2023).
Han, J. M., Lee, M.-Y., Lee, K.-B., Kim, H. & Hyun, Y. Y. Low relative skeletal muscle mass predicts incident hypertension in Korean men: a prospective cohort study. J. Hypertens. 38, 2223–2229 (2020).
Bai, T. et al. Sarcopenia is associated with hypertension in older adults: a systematic review and meta-analysis. BMC Geriatr. 20, 279 (2020).
Taylor, A. A., Tsuji, J. S., McArdle, M. E., Adams, W. J. & Goodfellow, W. L. Recommended reference values for risk assessment of oral exposure to copper. Risk Anal. 43, 211–218 (2023).
Shen, Y. et al. Isoquercitrin delays denervated soleus muscle atrophy by inhibiting oxidative stress and inflammation. Front. Physiol. 11, 988 (2020).
Choi, D. H., Yang, J. & Kim, Y. S. Rapamycin suppresses postnatal muscle hypertrophy induced by myostatin-inhibition accompanied by transcriptional suppression of the Akt/mTOR pathway. Biochem. Biophys. Rep. 17, 182–190 (2019).
Yin, L. et al. Skeletal muscle atrophy: from mechanisms to treatments. Pharmacol. Res. 172, 105807 (2021).
Powers, S. K. Can antioxidants protect against disuse muscle atrophy? Sports Med. 44, S155–S165 (2014).
Yang, J. et al. The association between the triglyceride-glucose index and sarcopenia: data from the NHANES 2011-2018. Lipids Health Dis. 23, 219 (2024).
Song, L., Li, H., Fu, X., Cen, M. & Wu, J. Association of the oxidative balance score and cognitive function and the mediating role of oxidative stress: evidence from the National Health and Nutrition Examination Survey (NHANES) 2011-2014. J. Nutr. 153, 1974–1983 (2023).
Csete, M. E. Basic science of frailty-biological mechanisms of age-related sarcopenia. Anesth. Analg. 132, 293–304 (2021).
Chen, L. et al. Homeostasis and metabolism of iron and other metal ions in neurodegenerative diseases. Signal Transduct. Target. Ther. 10, 31 (2025).
Li, G., Cheng, T. & Yu, X. The impact of trace elements on osteoarthritis. Front. Med. 8, 771297 (2021).
Centers for Disease Control and Prevention. National Health and Nutrition Examination Survey: NHANES Questionnaires, Datasets, and Related Documentation. https://www.cdc.gov/nchs/nhanes/index.htm.
Buckinx, F. et al. Pitfalls in the measurement of muscle mass: a need for a reference standard. J. Cachexia Sarcopenia Muscle 9, 269–278 (2018).
McLean, R. R. et al. Criteria for clinically relevant weakness and low lean mass and their longitudinal association with incident mobility impairment and mortality: the foundation for the National Institutes of Health (FNIH) sarcopenia project. J. Gerontol. A Biol. Sci. Med. Sci. 69, 576–583 (2014).
Batsis, J. A., Mackenzie, T. A., Jones, J. D., Lopez-Jimenez, F. & Bartels, S. J. Sarcopenia, sarcopenic obesity and inflammation: results from the 1999-2004 National Health and Nutrition Examination Survey. Clin. Nutr. 35, 1472–1483 (2016).
Huang, Q. et al. Association between manganese exposure in heavy metals mixtures and the prevalence of sarcopenia in US adults from NHANES 2011-2018. J. Hazard. Mater. 464, 133005 (2024).
Piercy, K. L. et al. The physical activity guidelines for Americans. JAMA 320, 2020–2028 (2018).
Reedy, J. et al. Evaluation of the Healthy Eating Index-2015. J. Acad. Nutr. Diet. 118, 1622–1633 (2018).
Shan, Z. et al. Healthy eating patterns and risk of total and cause-specific mortality. JAMA Intern. Med. 183, 142–153 (2023).
Dent, E., Wright, O. R. L., Woo, J. & Hoogendijk, E. O. Malnutrition in older adults. Lancet 401, 951–966 (2023).
Levey, A. S. et al. A new equation to estimate glomerular filtration rate. Ann. Intern. Med. 150, 604–612 (2009).
Acknowledgements
This research was supported by the National Natural Science Foundation of China (grant numbers: NSFC82304153) and the Fundamental Research Funds for the Central Universities (grant numbers: YCJJ20251404).
Author information
Authors and Affiliations
Contributions
L.L. and J.W. designed the study. L.W. and X.X. analyzed the data. J.G., N.Z. and L.C. assisted in data analysis. L.W. wrote the manuscript. Y.L., L.L. and J.W. commented on drafts and edited the manuscript. L.L. and J.W. had primary responsibility for final content. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wen, L., Xu, X., Guo, J. et al. Oxidative stress indicators mediated the association of dietary copper intake with low muscle mass: a cross-sectional study. npj Sci Food 9, 285 (2025). https://doi.org/10.1038/s41538-025-00645-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41538-025-00645-y