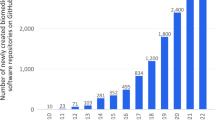
Abstract
Guidelines for managing scientific data have been established under the FAIR principles, requiring that data be Findable, Accessible, Interoperable, and Reusable. In many scientific disciplines, especially computational biology, both data and models are key to progress. For this reason, and recognizing that such models are a very special type of “data”, we argue that computational models, especially mechanistic models prevalent in medicine, physiology and systems biology, deserve a complementary set of guidelines. We propose the CURE principles, emphasizing that models should be Credible, Understandable, Reproducible, and Extensible. We delve into each principle, discussing verification, validation, and uncertainty quantification for model credibility; the clarity of model descriptions and annotations for understandability; adherence to standards and open science practices for reproducibility; and the use of open standards and modular code for extensibility and reuse. We outline recommended and baseline requirements for each aspect of CURE, aiming to enhance the impact and trustworthiness of computational models, particularly in biomedical applications where credibility is paramount. Our perspective underscores the need for a more disciplined approach to modeling, aligning with emerging trends such as Digital Twins and emphasizing the importance of data and modeling standards for interoperability and reuse. Finally, we emphasize that given the non-trivial effort required to implement the guidelines, the community should strive to automate as many of the guidelines as possible.
Similar content being viewed by others
Data availability
No datasets were generated or analyzed during the current study.
References
Wilkinson, M. D. et al. The FAIR guiding principles for scientific data management and stewardship. Sci. Data 3, 1–9 (2016).
Ravi, N. et al. FAIR principles for AI models with a practical application for accelerated high energy diffraction microscopy. Sci. Data 9, 657 (2022).
Shiferaw, K. B., Balaur, I., Welter, D., Waltemath, D. & Zeleke, A. A. Califrame: a proposed method of calibrating reporting guidelines with fair principles to foster reproducibility of ai research in medicine. JAMIA Open 7, ooae105 (2024).
Juarez, M. G., Botti, V. J. & Giret, A. S. Digital twins: review and challenges. J. Comput. Inf. Sci. Eng. 21, 030802 (2021).
Laubenbacher, R. et al. Toward mechanistic medical digital twins: some use cases in immunology. Front. Digit. Health 6, 1349595 (2024).
Michael, C. T., Almohri, S. A., Linderman, J. J. & Kirschner, D. E. A framework for multi-scale intervention modeling: virtual cohorts, virtual clinical trials, and model-to-model comparisons. Front. Syst. Biol. 3, 1283341 (2023).
Dill, K. A. & MacCallum, J. L. The protein-folding problem, 50 years on. Science 338, 1042–1046 (2012).
McCammon, J. A., Gelin, B. R. & Karplus, M. Dynamics of folded proteins. nature 267, 585–590 (1977).
Laubenbacher, R. et al. Building digital twins of the human immune system: toward a roadmap. npj Digit. Med. 5, 64 (2022).
Niarakis, A. et al. Immune digital twins for complex human pathologies: applications, limitations, and challenges. npj Syst. Biol. Appl. 10, 141 (2024).
Karr, J. R. et al. A whole-cell computational model predicts phenotype from genotype. Cell 150, 389–401 (2012).
Macklin, D. N., Ruggero, N. A. & Covert, M. W. The future of whole-cell modeling. Curr. Opin. Biotechnol. 28, 111–115 (2014).
Waltemath, D. et al. Toward community standards and software for whole-cell modeling. IEEE Trans. Biomed. Eng. 63, 2007–2014 (2016).
Waltemath, D. & Wolkenhauer, O. How modeling standards, software, and initiatives support reproducibility in systems biology and systems medicine. IEEE Trans. Biomed. Eng. 63, 1999–2006 (2016).
Tiwari, K. et al. Reproducibility in systems biology modelling. Mol. Syst. Biol. 17, e9982 (2021).
Porubsky, V. L. et al. Best practices for making reproducible biochemical models. Cell Syst. 11, 109–120 (2020).
Shin, J., Porubsky, V., Carothers, J. & Sauro, H. M. Standards, dissemination, and best practices in systems biology. Curr. Opin. Biotechnol. 81, 102922 (2023).
Vieira, L. S. & Laubenbacher, R. C. Computational models in systems biology: standards, dissemination, and best practices. Curr. Opin. Biotechnol. 75, 102702 (2022).
Hucka, M. et al. The systems biology markup language (SBML): a medium for representation and exchange of biochemical network models. Bioinformatics 19, 524–531 (2003).
Keating, S. M. et al. SBML Level 3: an extensible format for the exchange and reuse of biological models. Mol. Syst. Biol. 16, e9110 (2020).
Clerx, M. et al. CellML 2.0. J. Integr. Bioinform. 17, 20200021 (2020).
Gleeson, P. et al. NeuroML: a language for describing data driven models of neurons and networks with a high degree of biological detail. PLoS Comput. Biol. 6, e1000815 (2010).
Gu, C., Kim, G. B., Kim, W. J., Kim, H. U. & Lee, S. Y. Current status and applications of genome-scale metabolic models. Genome Biol. 20, 1–18 (2019).
Malik-Sheriff, R. S. et al. BioModels-15 years of sharing computational models in life science. Nucleic Acids Res. 48, D407–D415 (2020).
King, Z. A. et al. BiGG models: A platform for integrating, standardizing and sharing genome-scale models. Nucleic Acids Res. 44, D515–D522 (2016).
Arkin, A. P. et al. KBase: the united states department of energy systems biology knowledgebase. Nat. Biotechnol. 36, 566–569 (2018).
Blinov, M. L. et al. Practical resources for enhancing the reproducibility of mechanistic modeling in systems biology. Curr. Opin. Syst. Biol. 27, 100350 (2021).
Porubsky, V., Smith, L. & Sauro, H. M. Publishing reproducible dynamic kinetic models. Brief. Bioinform. 22, bbaa152 (2021).
Erdem, C. et al. A scalable, open-source implementation of a large-scale mechanistic model for single cell proliferation and death signaling. Nat. Commun. 13, 3555 (2022).
Chaouiya, C. et al. SBML Qualitative Models: a model representation format and infrastructure to foster interactions between qualitative modelling formalisms and tools. BMC Syst. Biol. 7, 135 (2013).
Niarakis, A. et al. Setting the basis of best practices and standards for curation and annotation of logical models in biology-highlights of the [bc]2 2019 colomoto/sysmod workshop. Brief. Bioinform. 22, 1848–1859 (2020).
Blais, E. M., Chavali, A. K. & Papin, J. A. Linking genome-scale metabolic modeling and genome annotation. in Systems Metabolic Engineering: Methods and Protocols (eds. Kim, H. U.) 985, 61–83 (Humana Press, 2013).
Passi, A. et al. Genome-scale metabolic modeling enables in-depth understanding of big data. Metabolites 12, 14 (2021).
Neal, M. L. et al. Harmonizing semantic annotations for computational models in biology. Brief. Bioinform. 20, 540–550 (2018).
Novère, N. L. et al. Minimum information requested in the annotation of biochemical models (miriam). Nat. Biotechnol. 23, 1509–1515 (2005).
Neal, M. L., Galdzicki, M., Gallimore, J. & Sauro, H. M. A C library for retrieving specific reactions from the biomodels database. Bioinformatics 30, 129–130 (2014).
Größler, A. & Milling, P. Inductive and deductive system dynamics modeling. In The 2007 International Conference of the System Dynamics Society (2007).
Ingalls, B. P. Mathematical modeling in systems biology: an introduction (MIT press, 255 Main Street, NE18. Floor 9. Cambridge, MA 02142, 2013).
Sauro, H. M. Systems biology: introduction to pathway modeling (Ambrosius Publishing, Seattle, 2020).
Kim, O. D., Rocha, M. & Maia, P. A review of dynamic modeling approaches and their application in computational strain optimization for metabolic engineering. Front. Microbiol. 9, 378885 (2018).
Peters, M., Eicher, J. J., Van Niekerk, D. D., Waltemath, D. & Snoep, J. L. The jws online simulation database. Bioinformatics 33, 1589–1590 (2017).
McDougal, R. A. et al. Twenty years of ModelDB and beyond: building essential modeling tools for the future of neuroscience. J. Comput. Neurosci. 42, 1–10 (2017).
Choi, J.-S. & Waxman, S. G. Physiological interactions between nav1. 7 and nav1. 8 sodium channels: a computer simulation study. J. Neurophysiol. 106, 3173–3184 (2011).
Lopez, C. F., Muhlich, J. L., Bachman, J. A. & Sorger, P. K. Programming biological models in python using pysb. Mol. Syst. Biol. 9, 646 (2013).
Brandl, G. Sphinx documentation. http://sphinx-doc.org/sphinx.pdf (2021).
Plank, G. et al. The opencarp simulation environment for cardiac electrophysiology. Comput. Methods Prog. Biomed. 208, 106223 (2021).
Britten, R. D. et al. Fieldml, a proposed open standard for the physiome project for mathematical model representation. Med. Biol. Eng. Comput. 51, 1191–1207 (2013).
Schaff, J. C. et al. SBML level 3 package: spatial processes, version 1, release 1. J. Integr. Bioinform. 20, 20220054 (2023).
Niarakis, A. et al. Addressing barriers in comprehensiveness, accessibility, reusability, interoperability and reproducibility of computational models in systems biology. Brief. Bioinform. 23, bbac212 (2022).
Yilmaz, L. & Liu, B. Model credibility revisited: concepts and considerations for appropriate trust. J. Simul. 16, 312–325 (2022).
Tatka, L. T., Smith, L. P., Hellerstein, J. L. & Sauro, H. M. Adapting modeling and simulation credibility standards to computational systems biology. J. Transl. Med. 21, 501 (2023).
Schlesinger, S. Terminology for model credibility. Simulation 32, 103–104 (1979).
Erdemir, A. et al. Credible practice of modeling and simulation in healthcare: ten rules from a multidisciplinary perspective. J. Transl. Med. 18, 369 (2020).
Musuamba, F., Skottheim Rusten, I., Lesage, R. et al. Scientific and regulatory evaluation of mechanistic in silico drug and disease models in drug development: building model credibility. CPT Pharmacomet. Syst. Pharmacol. 10, 804–825 (2021).
Vadigepalli, R. et al. A rubric for assessing conformance to the ten rules for credible practice of modeling and simulation in healthcare. medRxiv 2024–10 (2024).
Thacker, B. et al. Concepts of model verification and validation (No. LA-14167). Los Alamos National Lab., Los Alamos, NM, USA https://www.osti.gov/servlets/purl/835920 (2004).
Council, N. R. et al. Assessing the reliability of complex models: mathematical and statistical foundations of verification, validation, and uncertainty quantification (National Academies Press, 2012).
Bergmann, F. T. & Sauro, H. M. Comparing simulation results of SBML capable simulators. Bioinformatics 24, 1963–1965 (2008).
Hucka, M., Independent, A. F., He, E. & Keating, S. SBML test suite. Nat. Methods (2022).
Hellerstein, J. L., Gu, S., Choi, K. & Sauro, H. M. Recent advances in biomedical simulations: a manifesto for model engineering. F1000Research 8, 261 (2019).
Mendes, P. & Kell, D. Non-linear optimization of biochemical pathways: applications to metabolic engineering and parameter estimation. Bioinformatics 14, 869–883 (1998).
Marino, S., Hogue, I. B., Ray, C. J. & Kirschner, D. E. A methodology for performing global uncertainty and sensitivity analysis in systems biology. J. Theor. Biol. 254, 178–196 (2008).
Ashyraliyev, M., Fomekong-Nanfack, Y., Kaandorp, J. A. & Blom, J. G. Systems biology: parameter estimation for biochemical models. FEBS J. 276, 886–902 (2009).
Dräger, A. et al. Modeling metabolic networks in C. glutamicum: a comparison of rate laws in combination with various parameter optimization strategies. BMC Syst. Biol. 3, 5 (2009).
Kirk, P., Thorne, T. & Stumpf, M. P. Model selection in systems and synthetic biology. Curr. Opin. Biotechnol. 24, 767–774 (2013).
Liepe, J. et al. A framework for parameter estimation and model selection from experimental data in systems biology using approximate bayesian computation. Nat. Protoc. 9, 439–456 (2014).
Gábor, A. & Banga, J. R. Robust and efficient parameter estimation in dynamic models of biological systems. BMC Syst. Biol. 9, 1–25 (2015).
Mitra, E. D. & Hlavacek, W. S. Parameter estimation and uncertainty quantification for systems biology models. Curr. Opin. Syst. Biol. 18, 9–18 (2019).
Read, M. N., Alden, K., Timmis, J. & Andrews, P. S. Strategies for calibrating models of biology. Brief. Bioinforma. 21, 24–35 (2020).
Wieland, F.-G., Hauber, A. L., Rosenblatt, M., Tönsing, C. & Timmer, J. On structural and practical identifiability. Curr. Opin. Syst. Biol. 25, 60–69 (2021).
Colebank, M. J. et al. Guidelines for mechanistic modeling and analysis in cardiovascular research. Am. J. Physiol. Heart Circ. Physiol. 327, H473–H503 (2024).
Villaverde, A. F., Pathirana, D., Fröhlich, F., Hasenauer, J. & Banga, J. R. A protocol for dynamic model calibration. Brief. Bioinform. 23, bbab387 (2021).
Touré, V., Flobak, Å, Niarakis, A., Vercruysse, S. & Kuiper, M. The status of causality in biological databases: data resources and data retrieval possibilities to support logical modeling. Brief. Bioinform. 22, bbaa390 (2020).
Hall, B. A. & Niarakis, A. Data integration in logic-based models of biological mechanisms. Curr. Opin. Syst. Biol. 28, 100386 (2021).
Viceconti, M. et al. In silico trials: verification, validation and uncertainty quantification of predictive models used in the regulatory evaluation of biomedical products. Methods 185, 120–127 (2021).
Booker, J. M. & Ross, T. J. An evolution of uncertainty assessment and quantification. Sci. Iran. 18, 669–676 (2011).
Shaikh, B. et al. BioSimulators: a central registry of simulation engines and services for recommending specific tools. Nucleic Acids Res. 50, W108–W114 (2022).
unittest - unit testing framework. https://docs.python.org/3/library/unittest.html.
Bartley, B. et al. Building an open representation for biological protocols. ACM J. Emerg. Technol. Comput. Syst. 19, 1–21 (2023).
Hoops, S. et al. COPASI—a complex pathway simulator. Bioinformatics 22, 3067–3074 (2006).
Lieven, C. et al. MEMOTE for standardized genome-scale metabolic model testing. Nat. Biotechnol. 38, 272–276 (2020).
Fried, E. I. Theories and models: What they are, what they are for, and what they are about. Psychol. Inq. 31, 336–344 (2020).
Regt, D. & W, H. Understanding scientific understanding (Oxford University Press, Oxford, 2017).
Nicolis, G. & Prigogine, I. Exploring complexity: an introduction (1989).
Le Novère, N. Model storage, exchange and integration. BMC Neurosci. 7, 1–9 (2006).
Courtot, M. et al. Controlled vocabularies and semantics in systems biology. Mol. Syst. Biol. 7, 543 (2011).
Carey, M. A., Dräger, A., Beber, M. E., Papin, J. A. & Yurkovich, J. T. Community standards to facilitate development and address challenges in metabolic modeling. Mol. Syst. Biol. 16, e9235 (2020).
Zhukova, A., Waltemath, D., Juty, N., Laibe, C. & Le Novère, N. Kinetic simulation algorithm ontology. Nature Precedings 1–1 (2011).
Kholodenko, B. N. Cell-signalling dynamics in time and space. Nat. Rev. Mol. Cell Biol. 7, 165–176 (2006).
Reijenga, K. A. et al. Yeast glycolytic oscillations that are not controlled by a single oscillophore: a new definition of oscillophore strength. J. Theor. Biol. 232, 385–398 (2005).
Csete, M. E. & Doyle, J. C. Reverse engineering of biological complexity. Science 295, 1664–1669 (2002).
Andrews, S. S., Wiley, H. S. & Sauro, H. M. Design patterns of biological cells. BioEssays 2300188 (2024).
Baker, M. 1,500 scientists lift the lid on reproducibility. Nature 533, 452–454 (2016).
Plesser, H. E. Reproducibility vs. replicability: a brief history of a confused terminology. Front. Neuroinform. 11, 76 (2018).
Mendes, P. Reproducible research using biomodels. Bull. Math. Biol. 80, 3081–3087 (2018).
Höpfl, S., Pleiss, J. & Radde, N. E. Bayesian estimation reveals that reproducible models in systems biology get more citations. Sci. Rep. 13, 2695 (2023).
Coveney, P. V., Groen, D. & Hoekstra, A. G. Reliability and reproducibility in computational science: implementing validation, verification and uncertainty quantification in silico. Philos. Trans. A Math Phys. Eng. Sci. 79, 20200409 (2021).
Moraru, I. I. et al. Virtual cell modelling and simulation software environment. IET Syst. Biol. 2, 352–362 (2008).
Choi, K. et al. Tellurium: an extensible python-based modeling environment for systems and synthetic biology. Biosystems 171, 74–79 (2018).
Medley, J. K. et al. Tellurium notebooks-an environment for reproducible dynamical modeling in systems biology. PLoS Comput. Biol. 14, e1006220 (2018).
Shichkova, P. et al. Breakdown and repair of metabolism in the aging brain. Front. Sci. 3, 1441297 (2025).
Neal, M. L. et al. A reappraisal of how to build modular, reusable models of biological systems. PLoS Comput. Biol. 10, e1003849 (2014).
Smith, L. P., Bergmann, F. T., Chandran, D. & Sauro, H. M. Antimony: a modular model definition language. Bioinformatics 25, 2452–2454 (2009).
Mattsson, S. E., Elmqvist, H. & Otter, M. Physical system modeling with modelica. Control Eng. Pract. 6, 501–510 (1998).
Dabney, J. B. & Harman, T. L. Mastering simulink, vol. 230 (Pearson/Prentice Hall Upper Saddle River, New Jersey, 2004).
Food and Drug Administration. Assessing the credibility of computational modeling and simulation in medical device submissions. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/assessing-credibility-computational-modeling-and-simulation-medical-device-submissions (2021).
Verma, A., Antony, A. N., Ogunnaike, B. A., Hoek, J. B. & Vadigepalli, R. Causality analysis and cell network modeling of spatial calcium signaling patterns in liver lobules. Front. Physiol. 9, 1377 (2018).
Gee, M. M., Lenhoff, A. M., Schwaber, J. S., Ogunnaike, B. A. & Vadigepalli, R. Closed-loop modeling of central and intrinsic cardiac nervous system circuits underlying cardiovascular control. AIChE J. 69, e18033 (2023).
National Academies of Sciences, Engineering and Medicine. Foundational research gaps and future directions for digital twins. https://nap.nationalacademies.org/catalog/26894/foundational-research-gaps-and-future-directions-for-digital-twins (2023).
Bhagirath, P., Strocchi, M., Bishop, M. J., Boyle, P. M. & Plank, G. From bits to bedside: Entering the age of digital twins in cardiac electrophysiology. Europace 26, euae295 (2024).
Karr, J. et al. Model integration in computational biology: the role of reproducibility, credibility and utility. front. Syst. Biol. 2, 822606 (2022).
Schmiester, L. et al. Petab-interoperable specification of parameter estimation problems in systems biology. PLoS Comput. Biol. 17, e1008646 (2021).
Novère, N. L. et al. The systems biology graphical notation. Nat. Biotechnol. 27, 735–741 (2009).
Gauges, R., Rost, U., Sahle, S. & Wegner, K. A model diagram layout extension for sbml. Bioinformatics 22, 1879–1885 (2006).
Bergmann, F. T., Keating, S. M., Gauges, R., Sahle, S. & Wengler, K. Sbml level 3 package: render, version 1, release 1. J. Integr. Bioinform. 15, 20170078 (2018).
Acknowledgements
This work was supported by NIH Biomedical Imaging and Bioengineering award P41 EB023912 through HMS at the Center for Reproducible Biomedical Modeling (https://reproduciblebiomodels.org/). The content expressed here is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health, or the University of Washington. H.M.S. wishes to thank Eric Johnson Chavarria for suggesting the CURE acronym at the 2023 IMAG meeting in Bethesda, MD. H.M.S. also wishes to thank Hunter Robbins for assistance in collating the author names and addresses. T.E.G. was supported by a Royal Society University Research Fellowship (URF\R\221008) and the UKRI-BBSRC Engineering Biology Mission Award CYBER (BB/Y007638/1). S.F. was supported by the Predictive Phenomics Initiative under the Laboratory Directed Research and Development Program at Pacific Northwest National Laboratory, operated by Battelle for the U.S. Department of Energy under Contract No. DE-AC05-76RL01830. J.F. was supported by DARPA through the Automating Scientific Knowledge Extraction and Modeling (ASKEM) program, Agreement No. HR0011262087; NSF awards IIS-2106888, CMMI-2146306, and OAC-2411221. The views, opinions, and findings expressed are those of the authors and should not be interpreted as representing the official views or policies of the Department of Defense, the U.S. Government, or NSF. M.K. was supported by the BMBF within ATLAS by grant number 031L0304B and by the German Research Foundation (DFG) within QuaLiPerF by grant number 436883643 and by grant number 465194077 (Priority Programme SPP 2311, Subproject SimLivA). H.M.S. acknowledges research reported in this publication was supported by NIBIB of the National Institutes of Health under award number NIH grant number P41EB023912. R.L. acknowledges funding from the following awards: NIH 1 R01 HL169974-01, U.S. DoD DARPA HR00112220038, NIH 1 R011AI135128-01, NIH 1 R01 HL169974-01. R.V. acknowledges funding from the following awards: National Institute on Alcohol Abuse and Alcoholism R01 AA018873, National Heart, Lung, and Blood Institute R01 HL161696. The funding sponsors had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, and in the decision to publish the results. N.R. was funded by Deutsche Forschungsgemeinschaft (DFG, German Research Foundation) under Germany's Excellence Strategy - EXC 2075 – 390740016. G.D.B acknowledges work was supported by NRNB (U.S. National Institutes of Health, National Center for Research Resources grant number P41 GM103504). J.H.G acknowledges research reported in this publication was supported by NIBIB of the National Institutes of Health under award number NIH grant number P41EB023912. L.M.L acknowledges work was supported by NIH grant R24 GM137787 from the National Institute of General Medical Sciences. J.L.S acknowledges funding from the following award: DST/NRF SARCHI-82813. D.v.N. acknowledges funding from the following award: DST/NRF SRUG2204173612. I.I.M. acknowledges research reported in this publication was supported by NIBIB of the National Institutes of Health under award number NIH grant number P41EB023912 and by NIGMS of the National Institutes of Health under award number NIH grant number R24GM137787 P.M. acknowledges work was supported by NIH grant R24 GM137787 from the National Institute of General Medical Sciences. F.F. acknowledges support by the Francis Crick Institute, which receives its core funding from Cancer Research UK (CC2242), the UK Medical Research Council (CC2242), and the Wellcome Trust (CC2242). J.R.F acknowledges support from NIH grants P41GM10371 and R01GM115805. T.J.S. acknowledges funding from NSF grant 2000281. H.S.W. acknowledges support from NIH Grant 5U01-CA227544. J.M.R. acknowledges funding from the following award: NRF grant number SRUG2204295377. F.S. acknowledges funding from the Deutsche Forschungsgemeinschaft (DFG), under Germany’s Excellence Strategy--EXC 2117--422037984 and DFG project ID 251654672--TRR 161. D.W. acknowledges funding from the European Open Science Cloud (EOSC) Future program. M.L.B. acknowledges funding from NIH grants R24 GM137787 from the National Institute of General Medical Sciences and P41 EB023912 from the National Institute of Biomedical Imaging and Bioengineering.
Author information
Authors and Affiliations
Contributions
H.M.S. conceived the study. H.M.S., A.D., A.M., B.E.J., C.F.L., C.J.M., E.M., G.D.B., H.M.S., I.I.M., J.F., J.H., J.H.G., P.H., P.M., P.M.B., R.V., T.E.G. and W.W.L., wrote and edited the manuscript. The remaining authors A.A.P., A.H., A.N., B.G.O., D.D.v.N., D.P.N., D.W., E.A., E.M.Q., F.F., F.S., F.T.B., H.H., J.A.G., J.C.S., J.G.M., J.L.H., J.L.S., J.M.R., J.R.F., J.S., L.M., L.M.L., L.P.S., M.K., M.L.B., N.R., P.I.I., R.L., R.S.M., S.F., S.H., S.M.K., S.S., T.H. and T.J.S., read and approved the manuscript content.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sauro, H.M., Agmon, E., Blinov, M.L. et al. From FAIR to CURE: guidelines for computational models of biological systems. npj Syst Biol Appl (2026). https://doi.org/10.1038/s41540-026-00651-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41540-026-00651-0