Abstract
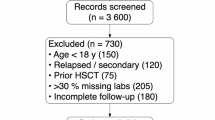
Chemotherapy-induced myelosuppression in acute myeloid leukemia (AML) frequently leads to life-threatening complications, yet current assessment standards lack the specificity required for personalized risk prediction. We present MM-AI-AML, a two-stage framework merging mechanistic mathematical modeling (MM) with artificial intelligence (AI) to predict myelosuppression severity using pre-treatment clinical data. Initially, a dynamic model simulating post-chemotherapy kinetics across four blood cell lineages was developed to derive a quantitative severity indicator, providing objective labels for 479 AML patients and 900 virtual cases. Subsequently, a TabNet deep learning classifier was trained on 51 clinical features to predict risk. MM-AI-AML demonstrated robust performance, achieving AUCs of 0.85 and 0.78 in internal and external validation cohorts, respectively, significantly outperforming traditional classifiers. Key predictive features included serum albumin, A/G ratio, and lactate dehydrogenase. High-risk stratification by the model was significantly associated with reduced in-hospital survival. By bridging mechanistic insights with interpretable machine learning, MM-AI-AML enables precise, personalized clinical decision-making for managing chemotherapy-related complications in AML.
Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding authors with a signed data access agreement. The source code supporting the conclusions of this article is available in the AML-DynamicPredictor repository (https://github.com/oolongice/AML-DynamicPredictor).
Code availability
The source code supporting the conclusions of this article is available in the AML-DynamicPredictor repository, (https://github.com/oolongice/AML-DynamicPredictor).
References
Jan, M. et al. Clonal evolution of preleukemic hematopoietic stem cells precedes human acute myeloid leukemia. Sci. Transl. Med. 4, 149ra118–149ra118 (2012).
Venugopal, K. et al. Dnmt3a harboring leukemia-associated mutations directs sensitivity to DNA damage at replication forks. Clin. Cancer Res. 28, 756–769 (2022).
Rochet, N. et al. Phase II study evaluating consolidation whole abdominal intensity-modulated radiotherapy (IMRT) in patients with advanced ovarian cancer stage FIGO III-the OVAR-IMRT-02 study. BMC Cancer 11, 1–7 (2011).
Wessels, H. et al. Pharmacodynamics, safety, and immunogenicity of pelmeg®, a pegfilgrastim biosimilar in healthy subjects. Pharmacol. Res. Perspect. 7, e00507 (2019).
Epstein, R. S. et al. Patient burden and real-world management of chemotherapy-induced myelosuppression: results from an online survey of patients with solid tumors. Adv. Ther. 37, 3606–3618 (2020).
Repetto, L. et al. Incidence and clinical impact of chemotherapy induced myelotoxicity in cancer patients: an observational retrospective survey. Crit. Rev. Oncol. Hematol. 72, 170–179 (2009).
Dueck, A. C. et al. Validity and reliability of the Us National Cancer Institute’s patient-reported outcomes version of the common terminology criteria for adverse events (PRO-CTCAE). JAMA Oncol. 1, 1051–1059 (2015).
Thanarajasingam, G., Hubbard, J. M., Sloan, J. A. & Grothey, A. The imperative for a new approach to toxicity analysis in oncology clinical trials. J. Natl. Cancer Inst. 107, djv216 (2015).
Thanarajasingam, G. et al. Longitudinal adverse event assessment in oncology clinical trials: the toxicity over time (toxt) analysis of alliance trials ncctg n9741 and 979254. Lancet Oncol. 17, 663–670 (2016).
Thanarajasingam, G. et al. Beyond maximum grade: modernising the assessment and reporting of adverse events in haematological malignancies. Lancet Haematol. 5, e563–e598 (2018).
Harkos, C. et al. Using mathematical modelling and ai to improve delivery and efficacy of therapies in cancer. Nat. Rev. Cancer 25, 324–340 (2025).
Reynolds, D. A. et al. Gaussian mixture models. In: Li, S. Z. & Jain, A. K. (eds) Encyclopedia of Biometrics. 741–742 (Springer, Boston, MA, 2009).
Hearst, M. A., Dumais, S. T., Osuna, E., Platt, J. & Scholkopf, B. Support vector machines. IEEE Intell. Syst. Appl. 13, 18–28 (1998).
Rumelhart, D. E., Hinton, G. E. & Williams, R. J. Learning representations by back-propagating errors. Nature 323, 533–536 (1986).
Cox, D. R. The regression analysis of binary sequences. J. R. Stat. Soc. Ser. B Stat. Methodol. 20, 215–232 (1958).
Breiman, L. Random forests. Mach. Learn. 45, 5–32 (2001).
Chen, T. Xgboost: A Scalable Tree Boosting System (Cornell University, 2016).
Schauer, R. & Kamerling, J. P. Sialic acids in cancer biology and immunity. ChemBioChem 12, 2246–2264 (2011).
Varki, A. Multiple changes in sialic acid biology during human evolution. Glycoconj. J. 26, 231–245 (2009).
Zhang, Q. et al. Serum sialic acid level as a novel biomarker for disease activity in systemic lupus erythematosus. Lupus 28, 1319–1326 (2019).
Zhou, X., Sun, X. & Zhao, W. Prognostic value of lactate dehydrogenase in hematological malignancies. Medicine 96, e8502 (2017).
Liu, D. et al. Procalcitonin as a biomarker for severe sepsis and septic shock patients: a meta-analysis. Crit. Care 19, 1–9 (2015).
Wang, K.-W. et al. Coagulation parameters and risk of progressive hemorrhagic injury after traumatic brain injury: a systematic review and meta-analysis. BioMed. Res. Int. 2020, 2020 (2020).
Ye, X.-L., Zhang, J.-M. & Liu, H.-C. Serum amyloid a as a marker of disease activity and treatment response in acute leukemia. World J. Clin. Cases 4, 47 (2016).
Ali, R. et al. Glucose metabolism in cancer and ischemia: possible therapeutic consequences of metabolic reprogramming. Curr. Pharm. Des. 22, 5392–5400 (2016).
Sproston, N. R. & Ashworth, J. J. Role of c-reactive protein at sites of inflammation and infection. Front. Immunol. 9, 754 (2018).
Okuno, S. et al. Serum β2-microglobulin level is a significant predictor of mortality in maintenance haemodialysis patients. Nephrol. Dial. Transplant. 24, 571–577 (2009).
Wang, J. et al. Globulin as a predictor for treatment-free survival in chronic myeloid leukemia. J. Cancer 9, 2335–2341 (2018).
Azab, B. et al. The prognostic value of the albumin/globulin ratio in cancer patients. Int. J. Surg. 11, 882–886 (2013).
Li, S. et al. Albumin and neutrophil-lymphocyte ratio (nlr) predict survival in patients with pancreatic adenocarcinoma treated with sbrt. Radiat. Oncol. 13, 1–8 (2018).
Yang, X.-H. et al. Cholinesterase and mortality risk in acute leukemia patients. Hematology 24, 343–348 (2019).
Chen, Z. et al. Complement factor b production in renal tubular cells and its role in sodium transporter expression. Circulation 137, 2423–2442 (2018).
Park, J. H. et al. Platelet to lymphocyte ratio as a prognostic indicator in patients with acute myeloid leukemia. Blood Res. 51, 227–232 (2016).
Batool, R., Butt, M. S., Sultan, M. T., Saeed, F. & Naz, R. Protein–energy malnutrition: a risk factor for various ailments. Crit. Rev. Food Sci. Nutr. 55, 242–253 (2015).
Gerbing, R. B. et al. Shorter remission telomere length predicts delayed neutrophil recovery after acute myeloid leukemia therapy: a report from the children’s oncology group. J. Clin. Oncol. 34, 3766–3772 (2016).
Arik, S. Ö & Pfister, T. Tabnet: attentive interpretable tabular learning. Proc. AAAI Conf. Artif. Intell. 35, 6679–6687 (2021).
Buhmann, M. D. Radial basis functions. Acta Numer. 9, 1–38 (2000).
Pezoulas, V. C. et al. Generation of virtual patient data for in-silico cardiomyopathies drug development using tree ensembles: a comparative study. In 2020 42nd Annual International Conference of the IEEE Engineering in Medicine & Biology Society (EMBC) (eds Akay, M., Weiland, J. & Spandidos, D. A.) 5343–5346 (IEEE, 2020).
Pezoulas, V. C. et al. Medical data quality assessment: on the development of an automated framework for medical data curation. Comput. Biol. Med. 107, 270–283 (2019).
Pezoulas, V. C. et al. Enhancing medical data quality through data curation: a case study in primary sjögren’s syndrome. Clin. Exp. Rheumatol. 118, 90–96 (2019).
Robnik-Sikonja, M. semiArtificial: Generator of Semi-Artificial Data. R package version 2.4.1. https://CRAN.R-project.org/package=semiArtificial (2021).
Teutonico, D. et al. Generating virtual patients by multivariate and discrete re-sampling techniques. Pharm. Res. 32, 3228–3237 (2015).
Friberg, L. E., Henningsson, A., Maas, H., Nguyen, L. & Karlsson, M. O. Model of chemotherapy-induced myelosuppression with parameter consistency across drugs. J. Clin. Oncol. 20, 4713–4721 (2002).
Friberg, L. E. & Karlsson, M. O. Mechanistic models for myelosuppression. Investig. N. Drugs 21, 183–194 (2003).
Fornari, C. et al. Understanding hematological toxicities using mathematical modeling. Clin. Pharmacol. Ther. 104, 644–654 (2018).
Kennedy, J. & Eberhart, R. Particle swarm optimization. In Proceedings of ICNN’95—International Conference on Neural Networks, Vol. 4 (eds Palaniswami, M., II, R. J. M., Fukuda, T. & deSilva, C.) 1942–1948 (IEEE, 1995).
Fornari, C., Pin, C., Yates, J. W., Mettetal, J. T. & Collins, T. A. Importance of stability analysis when using nonlinear semimechanistic models to describe drug-induced hematotoxicity. CPT: Pharmacomet. Syst. Pharmacol. 9, 498–508 (2020).
Marini, F. & Walczak, B. Particle swarm optimization (pso). a tutorial. Chemometr. Intell. Lab. Syst. 149, 153–165 (2015).
Miller, A. B. et al. Reporting results of cancer treatment. Cancer 47, 207–214 (1981).
Razali, N. M. et al. Power comparisons of Shapiro-Wilk, Kolmogorov-Smirnov, Lilliefors and Anderson-Darling tests. J. Stat. Model. Anal. 2, 21–33 (2011).
McKnight, P. E. & Najab, J. Mann-Whitney U test. In: Weiner, I. B. & Craighead, W. E. (eds) The Corsini Encyclopedia of Psychology. (John Wiley & Sons, Hoboken, NJ, 2010).
Moon, T. K. The expectation-maximization algorithm. IEEE Signal Process. Mag. 13, 47–60 (1996).
Berthold, M. & Diamond, J. Boosting the performance of RBF networks with dynamic decay adjustment. Adv. Neural Inform Proc. Syst. 7, 521–528 (1994).
Acknowledgements
This work was supported by the Major Research Plan of the National Natural Science Foundation of China (Grant No. 92374108 to X.Z.), the Key Program of the National Natural Science Foundation of China (Grant No. 12331018 to X.Z. and No. 82370176 to F.Z.), and the Major Research Plan of Hubei Province of China(Grant No. CZKYXM2023036JZ to F.Z.). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
F.Z. and X.Z. conceived and designed the study. Q.W., R.C., H.L., X.M., L.F., H.G., and L.X. acquired the data. Y.X. and Y.Z. analyzed the data. Y.X., C.M., and S.J. developed and trained the model. Y.X., Q.W., and Y.Z. verified the underlying raw data. The first draft of the manuscript was written by Y.X. and Q.W., and revised by Y.Z., F.Z. and X.Z. All authors contributed to manuscript preparation. All authors had full access to all the data in the study and had final responsibility for the decision to submit for publication.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhou, Y., Xiao, Y., Wang, Q. et al. Improving severity grading of chemotherapy-induced myelosuppression in AML via data-driven and model-based deep learning. npj Syst Biol Appl (2026). https://doi.org/10.1038/s41540-026-00687-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41540-026-00687-2