Abstract
In algal symbiotic systems (ABSS), bacteria and algae establish mutualistic, commensal, or parasitic relationships, leveraging their respective biosorption and bioaccumulation mechanisms, along with symbiotic effects, to effectively treat heavy metal (HM)-containing wastewater. The HM removal mechanisms by ABSS are modulated by multiple factors, including light intensity, pH, temperature, algal-bacterial ratio, and exhibit distinct patterns for different HMs. Based on a comprehensive literature review, the optimal conditions for ABSS are a light intensity of 60–300 μmol/m2/s, a slightly acidic to neutral pH, a temperature of 23–30 °C, and brown algae being the most effective. Furthermore, the practical applications and limitations of ABSS in different industries producing HM-containing wastewater (such as mining, animal agriculture, urban discharges, and textiles) were discussed, and analyzes its sustainability. This review establishes a robust theoretical framework for ABSS treatment mechanism of HM-containing wastewater, offers practical engineering guidelines, and promotes ABSS research and application.
Similar content being viewed by others
Introduction
With the continuous development of industry and society, human activities such as industrial activities, agricultural farming, and household living all generate wastewater containing heavy metals (HMs). Among various sources, the electroplating industry, the battery industry and leather tanning are major industrial activities that generate HM-containing wastewater with both the largest quantity and usually the highest concentration levels. Tanneries annually discharge over 70 million tons of HM wastewater, while the battery industry’s annual lead output of 10 million tons generates a large volume of lead-containing wastewater1,2. In these industrial wastewaters, the concentration of Cr(III) can reach 11.5–4000 mg/L, the concentration of Pd(II) can reach 2–600 mg/L, and the highest concentration of Ni(II) can reach 53,694.2 mg/L1,2,3. If these wastewaters are discharged into natural water bodies without effective treatment, HMs will accumulate in large quantities in aquatic organisms, seriously affecting their growth, reproduction, and survival, and even leading to the death of the organisms4. Also, HMs can be transferred through the food chain and accumulate in the human body5. This poses significant health risks with potential damage to the nervous, respiratory, renal, and other vital organs4,6. Mercury contamination causes minamata disease and lead contamination results in mental retardation7,8. Hence, effective treatment of HM wastewater is not only of utmost significance in environmental protection but also crucial for human health.
In the field of HM wastewater treatment, physical, chemical and biological methods are the main technological approaches9,10. Among them, biological methods have gradually become a focal point of research due to its advantages, such as low cost, environmental friendliness, and minimal infrastructure requirements11. The organisms used in biological methods include bacteria, fungi, algae, and plants, with bacteria and algae receiving significant attention due to their high efficiency in HM adsorption, transformation, and stabilization12,13. Bacteria possess strong environmental adaptability and immobilize HMs through the secretion of extracellular polymers and intracellular accumulation. Algae efficiently adsorb HMs by virtue of the chelation of polysaccharides and proteins in their cell walls and can obtain energy for operation through photosynthesis. However, the treatment efficiency of single bacteria or algae systems is not satisfactory due to poor environmental adaptability or limited tolerance in practical applications.
To achieve ideal wastewater treatment effects, the algae-bacteria symbiotic system (ABSS), as an emerging technology, has emerged and attracted widespread attention in recent years (Fig. 1a). The research hotspots of this system mainly focus on “biomass growth”, “heavy metals”, and “nutrient removal” (Fig. 1b). Abundant nutrients drive biomass growth, enhancing the system’s metabolic and operational efficiency to more effectively remove HMs from wastewater for high-quality purification. Therefore, the ABSS has multiple advantages in treating HM-containing wastewater. Firstly, it can achieve the simultaneous removal of multiple pollutants. Not only can the ABSS efficiently remove HMs through the synergistic effect of bacteria and algae, but it also has a good removal effect on other pollutants (nutrients, antibiotics, etc.)14,15,16. Secondly, the ABSS is self-sufficient and does not require the input of mechanical aeration or chemical agents17. Thirdly, the algae in the ABSS release oxygen and absorb carbon dioxide (CO2) through photosynthesis, a process that is highly beneficial for reducing the emissions of the greenhouse gas CO218,19. However, ABSS also reveals some deficiencies in practical applications. This system is highly sensitive to environmental conditions, as variations in light intensity and temperature fluctuations can severely disrupt its treatment performance, leading to remarkable discrepancies in sewage treatment outcomes across different seasons and regions20,21. Moreover, its complex composition makes precise operation control challenging, escalating both operational difficulty and treatment result uncertainty.
a A trend chart of the temporal changes in the number of relevant literatures retrieved from the Web of Science and Science Direct scientific research databases using “Algal-Bacterial Symbiotic System” as the keyword. b Using “Algal-Bacterial Symbiotic System” as the keyword, 100 pieces of literature within the recent five years were selected from the two authoritative databases mentioned above to construct a visual map of keyword hotspots.
Despite existing reviews on microalgae-bacteria symbiosis for HM wastewater treatment, understanding of treatment mechanisms and influencing factors remains limited22. Additionally, studies on its applicability and sustainability across various industrial settings are scarce, hindering the widespread engineering application of ABSS. Therefore, this review focuses on the treatment of HM-containing wastewater, aiming to comprehensively analyze the mechanisms, process performance, and applications of ABSS across various industries. By exploring the optimal operating parameter ranges of ABSS and proposing optimization strategies, this review endeavors to provide a robust theoretical foundation and practical guidelines for the engineering implementation of ABSS.
This review adopts a brand-new analytical framework to deeply analyze the internal mechanism of ABSS in treating HM-containing wastewater from three aspects: algae, bacteria, and their synergistic effects. Meanwhile, by extensively integrating literature data, this review systematically summarizes the action laws of key influencing factors and the optimal parameter ranges. In addition, this review explores the treatment effects of ABSS for HM-containing wastewater from different industries, and focuses on analyzing the potential of its resource recycling from the perspective of HM recovery. Finally, this review forecasts the sustainable development of ABSS for treating HM-containing, summarizes the application issues, and proposes targeted solutions.
In this review, relevant articles published in major databases, namely Web of Science (http://www.webofknowledge.com/) and ScienceDirect (http://www.sciencedirect.com/), over the past decade were primarily focused on. Classic older literature was included when necessary. The keywords used for searching journal articles were “Algae-Bacteria Symbiotic System”, “Algae-Bacteria Symbiotic”, “Algae-Bacteria Consortium”, “Algae-Bacteria Co-culture”, “Heavy Metals”, “Algae”, and “Bacteria”.
ABSS and its symbiotic relationships
ABSS
Bacteria and algae represent two fundamental groups of organisms in ecosystems, each with distinct physiological characteristics and specific ecological functions. Through the establishment of a symbiotic relationship, these organisms combine their strengths to form a powerful “biological alliance,” where both contribute essential roles. Algae are able to absorb nutrients and HMs from wastewater and produce oxygen via photosynthesis (Eq. 1), offering support for the metabolism of aerobic bacteria12. Meanwhile, bacteria undertake the task of degrading organic matter and releasing substances like inorganic carbon (Eq. 2), which contributes to the growth of algae19. This intricate biological interaction leads to the establishment of a highly synchronized symbiotic system, referred to as the ABSS. However, the symbiotic relationship between bacteria and algae is not stable and is susceptible to prevailing abiotic factors. Some studies have found that the symbiotic relationship between algae and bacteria depends on temperature and nutrient utilization22. Therefore, an in-depth understanding of the symbiotic relationship between bacteria and algae is crucial for ABSS.
Symbiotic relationship between bacteria and algae
According to the different interactions in ABSS, the symbiotic relationships between bacteria and algae are mainly divided into mutualism, commensalism, and parasitic19. Figure 2 depicts the symbiotic relationship between bacteria and algae and its characteristics.
In mutualism, bacteria and algae have complementary metabolisms and mutually benefit but the relationship is fragile; in commensalism, one party provides energy for the other to promote survival with high dependence; in competition symbiosis, bacteria and algae compete for resources inhibiting growth yet moderate competition can prompt niche differentiation, conducive to maintaining the symbiotic system’s function. This figure is independently drawn by the authors based on citation of the relevant literature.
Mutualistic symbiosis refers to an ecological process in which two species benefit from each other, typically through the exchange of metabolic products. One of the most common forms of mutualism is the production of oxygen by algae through photosynthesis, which serves as an electron acceptor for bacteria degrading organic matter23. In turn, bacteria release CO2 and other compounds, supporting the photosynthetic autotrophic growth of algae. Additionally, mutualistic relationships can involve the complementary exchange of essential nutrients between algae and bacteria, helping each overcome limitations in key trace elements, thereby promoting their growth and reproduction. For example, Chlamydomonas reinhardtii and Saccharomyces cerevisiae can mutually metabolize and exchange available nitrogen and carbon sources24. Research by Coope et al. 25 demonstrated that the bacterium Dinoroseobacter shibae provides vitamin B12 to promote the growth of the green algae Ostreococcus tauri, while the algae reciprocate by supplying fixed carbon. Also, Rawat et al. 26 reported that bacteria release siderophores to help microalgae overcome iron uptake limitations, while the microalgae release amino acids and other organic compounds to support bacterial growth.
In a commensalism relationship, only one species benefits, while the other neither benefits nor suffers23. As a result, there are two possible scenarios of commensalism between bacteria and algae. One scenario is when bacteria benefit, such as when microalgae release organic carbon (OC) to promote bacterial growth, without receiving any return27. The other scenario is when algae benefit, for example, some bacteria provide vitamin B12 to facilitate the growth of Chlamydomonas reinhardtii, while these bacteria cannot utilize the fixed carbon released by the algae28. Chen et al. 18 also reported that bacteria can secrete the signaling molecule indoleacetic acid (IAA) to promote the growth of microalgae, which is not reciprocated by the bacteria.
Parasitism benefits one species while harming the other. Many bacteria produce toxins that inhibit algal growth or kill algae by disrupting cell wall synthesis and blocking the respiratory chain29. For example, Bacillus velezensis V4 can release bacteriocins to lyse cells of Chlorella and Scenedesmus sp30. Similarly, algae can produce microcystins that inhibit or poison bacteria31. Frazzini et al. 32 reported that extracts from Chlorella can inhibit the growth of Escherichia coli O138. Additionally, there exists another form of parasitism that creates competition for nutrients or oxygen33. This can result in the growth rate of a particular species being hindered and eventually surpassed by its competitors after several generations.
In summary, bacteria and algae form the ABSS through various symbiotic relationships. From the mutually beneficial and win-win collaborative symbiosis to the parasitic relationship with a biased interest, these ecological associations constitute the core mechanism of the ABSS for treating HM-containing wastewater. During the treatment of HM-containing wastewater, in addition to the crucial carbon-oxygen exchange between bacteria and algae, the two also form a deep symbiotic relationship through nutrient conversion. Bacteria decompose complex nutrients into small molecular forms that are more easily utilized by algae, enabling the rapid growth of biomass, thus enhancing the purification efficiency of the ABSS for HMs. Organic substances such as polysaccharides and amino acids secreted by algae can serve as carbon sources and nutrient factors for the growth of bacteria. This symbiotic effect endows the ABSS with excellent environmental tolerance, enabling it to exhibit remarkable advantages in treating high-concentration HM-containing wastewater. However, the symbiotic relationship between bacteria and algae is dynamic and highly susceptible to environmental factors like temperature and light, which can in turn affect the performance of ABSS.
Mechanisms of ABSS in HM-containing wastewater treatment
In treating HM-containing wastewater using ABSS, not only do the individual mechanisms of algae and bacteria come into play, but also their synergistic interactions, and ABSS activates specific treatment mechanisms designed for different HMs.
Mechanisms of algae in HM-containing wastewater treatment
Many studies have shown that algae exhibit a high degree of tolerance and affinity for HMs, endowing them with the ability to remove HMs (Table 1). Sargassum dentifolium efficiently removes Cr(VI) through ion exchange of cell-surface carboxyl groups and complexation of cell-surface hydroxyl groups with Cr(VI). At the optimal conditions of pH 7.0 and 50 °C, 99.68% of 100 mg/L Cr(VI) can be removed by its dry matter within 12 h9. Gracilariacorticata conducts biosorption of cobalt using cell-surface carboxylate and carboxyl groups, attaining an 87.8% removal efficiency of 50 mg/L cobalt within 60 min at pH 5 and 29.85 °C34. Within 3 days at pH 7 and 30 °C, Chlorella vulgaris removes 6 mg/L Zn (II) via biosorption and bioaccumulation, attaining a removal rate of 99%35. Due to the abundant functional groups on their cell surfaces, these algae exhibit a strong biosorption capacity for HMs; however, as the biosorption rate of living algal cells is relatively slow, they are widely processed and utilized as biosorbents. Differing from dead algal cells, living algal cells are capable of removing HMs not only via biosorption but also through bioaccumulation.
The biosorption process depends largely on the composition of the algal cell wall. Algal cell walls contain calcium, magnesium, and sodium ions that can exchange with HM ions. Additionally, the cell walls feature negatively charged functional groups, such as amino and carboxyl groups, which provide numerous binding sites for HM ion adsorption36. These functional groups display specific affinities for different HMs. The nitrogen atom in the amino group has a lone pair of electrons and can form an amino cation upon protonation. This allows for effective adsorption of hexavalent chromium through coordination and electrostatic attraction37. In contrast, the sulfur atom in the thiol group can form extremely stable coordination bonds with Hg(II). This also enables simultaneous biosorption through multiple functional groups.
Part of the HMs that have been biosorbed enters the bioaccumulation process. After entering the algal cytoplasm via transporter proteins (e.g., Natural Resistance-Associated Macrophage Proteins (NRAMP) and Fe-Transporter (FTR) families), HMs bind to phytochelatins (PCs) or metallothioneins (MTs) to form complexes38. With the help of glutathione synthetase (GSH), PC formation starts with -Glu-Cys- binding initiation, which binds HM ions via sulfhydryl groups39. This binding reduces the toxicity of HM ions and promotes their intracellular transport and storage. Additionally, algae utilize enzymatic mechanisms to carry out oxidation and reduction processes, transforming toxic HMs into less harmful or non-toxic forms. These intracellular pathways may achieve compartmentalized storage of HM ions.
Mechanisms of bacteria in HM-containing wastewater treatment
Many bacteria can adapt to environments containing HMs and remove them. Table 2 summarizes the removal effects of most bacterial strains on HMs reported previously. Thermophilic B. cereus SO-16 can achieve 100% removal of 5 mg/L Mn(II) through biosorption and bioaccumulation under the conditions of 16 h, pH 7, and 60 °C40. Pseudomonas cremoricolorata first transports Hg(II) into the cell with the help of merT and merC proteins, and then the merA reductase reduces Hg(II)41. It reaches an 87% removal rate within 24 h at pH 8 and a temperature of 30 °C. Bacillus xiamenensis PbRPSD202 removes 99.19% of 100 mg/L Pb(II) through biosorption, bioaccumulation, and bioprecipitation at pH 6 and a temperature of 35 °C42. Most of these bacteria can remove HMs within a relatively short period of time and possess a rich enzyme system, which enables them to precisely regulate the process of HM removal. The high reproduction rate and environmental adaptability of bacteria enable them to rapidly colonize diverse environments, and through the synergistic actions of biosorption, bioaccumulation, and bioprecipitation, they can further enhance the efficiency of HM removal43,44.
The biosorption exhibited by bacteria is primarily attributed to the peptidoglycan in their cell walls, which is a complex structure composed of linear chains of sugars and peptide chains. The sugar chain consists of the disaccharides N-acetylglucosamine and N-acetylglucuronic acid linked by β1-3 or β1-4 glycosidic bonds. Gram-positive bacteria possess cell walls with multiple layers of peptidoglycan and phospholipid, while Gram-negative bacteria have cell walls containing merely a single layer of peptidoglycan. This structural difference in the cell wall results in Gram-positive bacteria being able to adsorb more HM ions than Gram-negative bacteria40. Biosorption of HMs by bacteria can be classified into physical adsorption, ion exchange, and complexation, which are the mechanisms based on the composition of the cell wall. These mechanisms involve interactions with HMs through van der Waals forces, electrostatic interactions, ion exchange, and the binding of functional groups on the cell wall to form stable complexes. The biosorption of HMs by bacteria may result from these mechanisms acting independently or in combination. Lactobacillus plantarum MF042018 adsorbs cadmium and lead through physical adsorption and complexation44.
The bioaccumulation of bacteria begins with the transmembrane transport of HM ions through transport proteins such as α-helical proteins and β-barrel proteins. Bacteria such as Cornebacterium and Pseudomonas can passively transport arsenic and mercury through α-helical proteins45. In addition, there are transporter proteins that can use ATP hydrolysis to release energy to transport HM ions, such as ABC transport proteins. After entering the bacterial cell, HMs are accumulated in various means, including reduction, methylation, metal chelator production, and compartmentalization. HMs can be methylated via the S-adenosylmethionine, methylcobalamin, and N-methyltetrahydrofolate pathways46. HMs can bind to chelators like metallothionein, ferritin, and glutathione, with all these binding modalities converting HM from highly toxic substances into less toxic products.
Bioprecipitation is a metabolism-dependent process, primarily involving reduction and the formation of sulfides and phosphates. Many anaerobic or partially anaerobic bacteria utilize enzymes or electrons to reduce HMs and form precipitates. For instance, Shewanella oneidensis MR-1 reduces U(VI) to produce a black U(IV) carbonate precipitate47. Sulfate-reducing bacteria convert sulfate to sulfide, which then reacts with HMs to form insoluble metal sulfide precipitates. Phosphatase-producing bacteria convert organic phosphates into inorganic forms, which combine with HMs to form phosphate precipitates.
Synergistic effects of ABSS in HM-containing wastewater treatment
In ABSS, bacteria and algae are interconnected through extracellular polymeric substances (EPS), which enable cooperative interactions such as material exchange and energy transfer. Consequently, the mechanisms underlying HM-containing wastewater treatment in ABSS are intricate and multifaceted. As depicted in Fig. 3, involving both the individual removal mechanisms of single algae or bacteria towards HM and the synergistic effect between them, the removal mechanism of HM by ABSS shows that their synergistic removal effect on HM often occurs in EPS.
Algae remove HMs through biosorption and bioaccumulation; bacteria remove HMs via biosorption, bioprecipitation, and bioaccumulation; in ABSS, the symbiotic effect often stimulates microorganisms to secrete more EPS, thereby facilitating the operation of various HM removal mechanisms. This figure is independently drawn by the authors based on citation of the relevant literature. (AP: Phosphatase; Pi: Inorganic phosphate).
During the biosorption of HMs in ABSS, biosorption occurs on both microbial cell surfaces and within the EPS48. Composed primarily of proteins, polysaccharides, and nucleic acids, EPS possess functional groups similar to those of cell walls, providing binding sites for HM adsorption. This process involves ion exchange and complexation, while the interaction of phosphate, carbonate, and sulfate ions with HM ions in EPS can lead to HM removal via precipitation.
The biosorption occurring on the microbial surface and within the EPS often interact, thereby boosting the overall biosorption capacity of ABSS48. EPS secreted by bacteria can, to some extent, mitigate the inhibitory effects of HMs on algae and increase the specific surface area of algal cells, thus expanding the number of available adsorption sites49. Meanwhile, the EPS secreted by algae also contribute to promoting bacterial growth. The secretion of these EPS collectively enhances the adsorption capacity of microorganisms for HMs in ABSS. Furthermore, the close cooperation between bacteria and algae in ABSS may enhance nutrient availability, thus promoting the growth of algae50. This leads to an increase in the production of EPS, which enhances the effectiveness of the algal biosorption mechanism and ultimately improves the overall adsorption capacity of the ABSS. The ABSS can also create microenvironments conducive to surface complexation, further enhancing the chelation of HM ions. All these effects make the ABSS show excellent biosorption capacity.
ABSS not only capitalizes on the inherent accumulation mechanisms of algae and bacteria but also further bolsters the overall bioaccumulation capacity of the system via siderophores or extracellular polysaccharides generated by specific bacteria. Siderophores are molecules that bind to HM ions and help them to cross the cell membrane, which enhances the accumulation of HM ions by algal cells26. Extracellular polysaccharides promote the accumulation of HM ions in cells by forming a matrix that effectively traps them and enhances cellular uptake51. Moreover, bacteria within the ABSS are capable of generating extracellular enzymes. These enzymes can efficiently facilitate the dissolution and transformation of HM ions, thereby rendering these HM more readily accumulated by cells.
Regulatory mechanisms of influencing factors
The main influencing factors of the ABSS are light intensity, pH, temperature, carbon sources, and HM concentration. Deeply analyzing the influencing factors enables the clarification of the interaction patterns and intrinsic operational rules during the treatment of HM-containing wastewater by ABSS.
Light intensity
In ABSS, photosynthetic algae rely heavily on light intensity for metabolism, generating ATP and NADPH in the light reaction phase, while most non-photoautotrophic bacteria can not directly use light energy. The growth rate of algae increases with light intensity within a certain range, reaching its maximum at the light saturation point, beyond which growth is limited21. At this point, the oxygen production through photosynthesis of algae reaches its optimal state, which can significantly boost the activity of aerobic bacteria, enabling the ABSS to achieve its maximum HM removal capacity.
pH
pH influences both the surface charge of microbial cells and the physicochemical state of HM ions52. At low pH, the microbial surface carries a positive charge, which electrostatically repels HM cations, hindering their adsorption53. As the pH increases, the deprotonation of negatively charged functional groups on the microbial surface exposes active sites, enhancing the adsorption of HMs. This pH increase also promotes the activity of most microbial enzymes, thereby boosting the HM removal mechanisms. However, at high pH, the formation of hydroxides competes with electrostatic interactions between microorganisms and HM anions, leading to a reduced adsorption capacity.
HM concentrations
The increase in the concentration gradient of HMs can enhance the driving force between the liquid phase and microorganisms, thereby surmounting the mass transfer resistance. But after reaching a certain concentration, the removal of HMs by ABSS stops increasing, as further increases in HM concentration saturate the adsorption sites on the microbial surface, preventing any further enhancement of adsorption capacity. Meanwhile, the higher the concentration of HMs, the greater the damage to microorganisms.
Temperature
Temperature variably affects HM-containing wastewater treatment in ABSS. Firstly, changes in temperature can change the growth rate and enzyme activity of bacteria and algae, which has a direct effect on the performance of ABSS54. Secondly, the temperature can affect the stability and solubility of HMs, further influencing the removal effect of HMs by ABSS. Finally, temperature changes regulate biosorption’s thermal effects, shifting the adsorption equilibrium position.
Carbon sources
Concentration and type of carbon source influence microbial metabolism and proliferation, which is critical for treating HM-containing wastewater by ABSS. Either too high or too low a concentration of carbon sources can limit the removal of HMs in this system, as both bacterial and algal life activities depend on carbon sources. Furthermore, the type of carbon source utilized also affects the HM removal capacity of ABSS. Algae absorb inorganic or simple organic carbon sources for photosynthesis, while bacteria utilize complex organic carbon sources for metabolic activities.
Algae/bacteria ratios
Algae/bacteria ratios directly affect the synergistic effect between the two and further influence the removal capacity of ABSS for HMs55. An excessively high inoculation ratio will lead to competition for resources such as nutrients and living space, resulting in oxygen deficiency or nutritional imbalance. Meanwhile, an excessively high inoculation ratio will obstruct the light radiation received by individual algae, limiting the photosynthetic efficiency of algae. An excessively low inoculation ratio prevents sufficient contact between bacteria and algae, leading to insufficient signal exchange, thus restricting the symbiotic effect. Therefore, the optimal ratio of algae to bacteria can balance resource competition and the symbiotic effect, thereby enhancing the removal efficiency of HMs by the ABSS.
Differences in treatment mechanisms for varying HMs
ABSS demonstrates diversified HM removal mechanisms that are highly contingent upon the species of HMs. Marked discrepancies exist in the removal processes of Cr(VI), Cd(II), and Pb(II), encompassing distinctive extracellular and intracellular interactions, as well as variations in transporter-mediated uptake (Fig. 4).
This figure illustrates the removal mechanisms of Cr(VI), Cd(II), and Pb(II) by the ABSS, respectively. The commonality is that all rely on biosorption and intracellular accumulation. The differences lie in that the reduction mechanism of Cr(VI) is remarkable, Cd(II) preferentially reacts with sulfur ligands, and the biosedimentation mechanism of Pb(II) is prominent. This figure is independently drawn by the authors based on citation of the relevant literature. (Glu: Glutamic acid, Cys: Cysteine, GCL: γ-Glutamylcysteine ligase, γ-Glu-Cys: γ-Glutamylcysteine; Gly: Glycine, GSy: Glutathione synthetase, GST: Glutathione S-transferase, L-Cys: L-Cysteine, L-Ala: L-Alanine, PS: Phosphate sulfurylase, RSS: Reduced sulfur species, OPC: Organic phosphorus compound).
The extracellular removal mechanisms of ABSS for different HMs vary
Positively charged Cd(II) and Pb(II) undergo electrostatic attraction and complexation with hydroxyl and carboxyl groups on the ABSS surface56,57. Pb(II) and Cr(VI) participate in ion-exchange reactions with sodium and magnesium ions on the ABSS surface56,58. Under acidic conditions, protonated amino groups on ABSS bind to negatively charged Cr(VI) via electrostatic attraction. Moreover, the strong oxidizing Cr(VI) is readily reduced to Cr(III) extracellularly59. Subsequently, Cr(III), Cd(II), and Pb(II) combine with anions to form precipitates, facilitating their removal.
Significant disparities exist in the transport proteins employed by different HMs
Cr(VI), owing to its structural similarity to sulfate anions, enters cells via the sulfate transporter protein SULTR260. For the cellular uptake of Cd(II), a diverse array of transporters, such as IRT1, NRAMP1, CrMTP4, and ABC transporters, are utilized, collectively mediating its entry into the cell61,62. As for Pb(II), transmembrane transport is predominantly mediated by the PbrT and PbrA transporter proteins56,63.
The intracellular mechanisms of different HMs showed significant differences
Upon entry into the cell, Cr (VI) undergoes various metabolic pathways: some of it is reduced to Cr (III) by chromate reductase, cytosolic thiols, or GSH; some of it is bound to the Alr0975 protein in the PC or chelated with GSH; and some of it is immobilized in the membrane by binding to the LCI1 channel of the membrane proteins or through ion-exchange with the membrane of the vesicle-like organisms60,64.
Upon entering cells, Cd(II) preferentially reacts with sulfur ligands. With the assistance of thioredoxin A (TrxA) and reduced nicotinamide adenine dinucleotide phosphate (NADPH), cysteine desulfurization generates reactive sulfur species that precipitate as CdS with Cd(II)65. Cysteine’s sulfhydryl groups also form thiol salts with Cd(II) for excretion. Iron-sulfur clusters (iscS) supply sulfur for Cd(II) binding66. Phosphate forms compartmentalized complexes with Cd(II), while flavonoids chelate it, reducing its toxicity62.
After entering cells, Pb(II) is removed primarily through precipitation and chelation. Phosphatase PbrB hydrolyzes organophosphorus compounds, releasing inorganic phosphate that forms insoluble lead phosphate with Pb(II)63. Meanwhile, phytochelatins, metallothioneins bmtA and smtAB chelate Pb(II) for intracellular sequestration67. Iron carriers from non-ribosomal peptide synthetases (NRPS) or polyketide synthases (PKS) further bind Pb(II), preventing its entry and reducing free ion concentration68.
Treatment process of ABSS
In practical applications, to optimize HM-containing wastewater treatment, an appropriate ABSS treatment process should be chosen based on factors such as wastewater characteristics and effluent standards. Depending on the growth states of microorganisms, ABSS can be categorized into suspended growth and attached growth processes19, as illustrated in Fig. 5. These processes for treating wastewater containing HMs have different reaction conditions and treatment effects respectively (Table 3).
a Process structure diagrams and real-life photos of the High Rate Algal Pond (HRAP); b Process structure diagrams and real-life photos of the flat-panel and stirred-tank Photobioreactors (PBRs); c: Process structure diagrams and real-life photos of the Horizontal Bubble (HB) Photobioreactor; d: Process structure diagrams and real-life photos of the Algal Turf Scrubber (ATS); e: Capabilities of the above four processes120,121,122,123,124. The photograph in a is used with permission from Young et al. 120 (2017) (License Number: 6004710217057); the photographs in b are used with permission from Koller121 (2015) (License Number: 6004760277089) and under the Creative Commons Attribution International License (CC BY 4.0) from Occhipinti et al. 122 (2023); the photograph in c is used with permission from Wollmann et al. 123 (2019) (License Number: 6004740250248); the photograph in d is used under the Creative Commons Attribution International License (CC BY 4.0) from Zettler et al. 124 (2024).
Suspended growth process
In suspended growth processes, bacteria and algae remain in a suspended state, typically involving both open and closed bioreactors. The high-rate algal pond (HRAP) is currently the most commonly used open bioreactor69. and its brief process and actual diagram are shown in Fig. 5a. HRAP is a complex ecosystem constructed on the basis of traditional biologically stabilized ponds by adding algae and bacteria, designed as a channel for raceway ponds. Algae in HRAP produce abundant dissolved oxygen, which both promotes organic matter degradation by aerobic bacteria and serves as aeration. So HRAP is much easier to operate, and its energy and equipment costs are much lower than traditional treatment processes70. However, it is difficult to separate the biomass in HRAP, and an additional harvesting device needs to be connected after its treatment71. Besides, HRAP is easily limited by environmental factors, including temperature fluctuation, heterotrophic contamination, and low light utilization, etc. Tangahu et al. 20 showed that the chromium removal efficacy of the ABSS operated in HRAP is susceptible to pH, temperature, and agitation speed.
Closed bioreactors can rigorously control and monitor cultivation conditions, significantly increasing microbial concentrations. This feature not only allows them to overcome the sensitivity of open bioreactors to external environmental influences but also far surpasses open bioreactors in terms of treatment efficiency and biomass productivity72. Photobioreactor (PBR) is a typical closed bioreactor that provides controlled and suitable environmental conditions such as light, nutrients, and temperature73. Common photobioreactors are flat-panel and stirred-tank reactors as shown in Fig. 5b. The flat-plate PBR features a large illuminated surface area, with its entire plane accessible to light irradiation. This design results in high light utilization and low mechanical requirements for flat-plate PBRs. However, the flat-panel PBR also has some problems, such as a large footprint, high construction cost, and light attenuation caused by internal organisms74. The stirred-tank PBR is equipped with stirring devices and internal or external lighting systems, creating a highly controllable mixing environment for microorganisms. This design features excellent heat and mass transfer properties, but there are significant variations in local shear forces75. Muñoz et al. 76 indicated that the ABSS achieved a maximum adsorption of 8.5 ± 0.4 mg/g for copper in a stirred tank PBR.
Attached growth processes
Compared with the suspended growth process, the attached growth process has better wastewater treatment, higher contaminant tolerance, higher biomass productivity, and easier biomass harvesting77. In attached growth processes, algae and bacteria adhere to carriers with rough surfaces or porous structures for growth, primarily including hybrid biofilm (HB) photobioreactors and algal turf scrubbers (ATS). The HB photobioreactor is constructed by the algal-bacterial biofilm formed on the carrier (Fig. 5c). The HB photobioreactor exhibits strong shock load resistance, superior contaminant removal efficiency, and low energy consumption, offering enhanced nutrient removal and biomass recycling capabilities compared to traditional biofilm reactors78. Orandi et al. 79 used a similar hybrid biological membrane photoreactor, achieving a removal rate of 50% for both copper and nickel from synthetic acidic mine wastewater. Similarly, Zhao et al. 80 employed a comparable hybrid membrane photoreactor to remove all chromium and nickel from sludge supernatant, along with 80% of total phosphorus and 87% of total nitrogen, achieving a biomass production rate of 7.0 g/m²/d within 1.3 days.
ATS system employs solid substrates within shallow flow channels to provide an optimized environment for the growth and harvesting of mixed-community biofilms, primarily composed of algae and bacteria (Fig. 5d). This system effectively removes dissolved pollutants and collects biomass while leveraging the synergistic effects of periphytic plants to improve water quality69. Compared to other processes, the ATS technology, which involves periodic scraping and collection of algal biofilms into designated containers, offers significant advantages in biomass harvesting81. It not only greatly enhances the convenience and efficiency of biomass recovery but also features low energy consumption and construction costs82. Furthermore, more frequent harvesting operations with ATS can substantially increase biomass yield. Sutherland et al.83 observed that weekly harvesting increased denitrification and biomass productivity by about 60% compared to biweekly harvesting and strip harvesting.
As illustrated in Fig. 5e, the HB photobioreactor achieves superior HM removal while simultaneously harvesting the highest biomass in a single operation. Operated under highly controlled conditions, HB photobioreactors optimize both algal growth and pollutant removal more effectively than ATS. Additionally, their attached-growth biofilm design enhances the efficiency and convenience of biomass harvesting compared to HRAP and PBR systems.
Other emerging processes
The combination of ABSS with other processes or technologies can obviously improve the performance of wastewater treatment. The membrane aerated biofilm reactor (MABR) is a wastewater treatment technology that integrates biofilm processes with membrane separation. When combined with ABSS, the resulting membrane aerated algal-bacterial biofilm reactor (MABAR) considerably enhances the removal efficiency of organic matter and nutrients84. In addition, MABAR is energy-efficient and can effectively address the challenges of high aeration requirements and low nitrogen and phosphorus removal rates85.
The sequential batch ABSS integrates the characteristics of the sequential batch reactor (SBR) with algal growth systems (AGS). This reactor effectively addresses challenges such as sludge bulking, low biomass concentrations, and the simultaneous removal of both nitrogen and phosphorus86. By efficiently adjusting processes such as nitrification, phosphorus removal, and carbon compound elimination, it achieves high water quality within short aeration periods while reducing operating costs87. Additionally, this reactor oxygenates, averts sludge sedimentation via thorough mixing and aeration, and efficiently eliminates diverse pollutants. Wang et al. 88 demonstrated that the sequential batch ABSS removed over 80% of Cd(II).
Algal-bacterial electrochemical systems integrate microbial fuel cells (MFCs) with ABSS, addressing inherent limitations of MFCs, such as high energy consumption. In these systems, algal photosynthesis supplies oxygen to the cathode while also serving as an electron acceptor and supporting the growth of anodic bacteria89. This integration reduces energy consumption, enhances energy cycling within the system, and enables net positive energy recovery.
Combining advanced oxidation processes (AOPs) with ABSS can effectively remove persistent contaminants that are difficult to degrade. During the operation of the process, AOPs can convert recalcitrant compounds into intermediates that can be mineralized by ABSS. Therefore, this process not only reduces the toxicity of incomplete oxidation products that may arise from the standalone application of AOPs but also enhances the overall decontamination effectiveness. Tripathy and Kumar90 combined microwave oxidation technology and columnar PBR to treat diluted leachate, achieving a total iron removal rate of 86–95% and a Zn(II) removal rate of 28%.
Analysis of parameters for ABSS treating HM-containing wastewater
As shown in Fig. 6a, light intensity, pH, temperature, nutrients, HM concentration, and the inoculation ratio of algae and bacteria are the key parameters for the treatment of HM-containing wastewater by ABSS. By analyzing the influence of these parameters on the treatment effect, the operating conditions of the system can be optimized, thereby enhancing the removal efficiency of HMs. The influence of key parameters on the removal efficiency of HMs is summarized in Table 4 for reference.
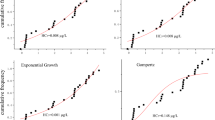
a Key parameters for the removal of HMs by ABSS; b The light saturation points of different algae and the effects of the light saturation points on ABSS; c The removal amounts of HMs by ABSS at different pH values; d The removal capacities of ABSS under different initial concentrations and types of HMs; e The selection frequencies and ranges of the optimal temperature for ABSS; f The effects of the optimal carbon source on ABSS; g The removal amounts of Cd, Cr, and Pd by different algae, respectively16,20,55,57,58,59,63,76,79,91,95,96,100–104,107,108,125,137,171,172,174. This figure is independently drawn by the authors based on citation of the relevant literature.
Light intensity
The ABSS exhibits the strongest treatment capacity for HM-containing wastewater at this light saturation point (Fig. 6b), Figure 6b summarizes the light saturation points of some algae, and it is found that the light saturation points of most algae are within the range of 60–300 μmol/m2/s. Since algae play an important role in the ABSS, it is reasonable to infer that the optimal light intensity for most ABSSs also falls within this range. Tangahu et al. 20 revealed that the removal of Cr(VI) by ABSS consisting of Azotobacter S8 and Chlorella vulgaris could be up to 18.68% at light intensities ranging from 102 to119μmol/m2/s. The ABSS composed of Chlorella vulgaris and Enterobacter sp. MN17 also achieved a maximum removal rate of 79% for Cr(VI) within the range of 100–120 μmol/m2/s91.
pH
The optimal pH range for algal growth is between 7.0–9.0 and for bacterial growth is between 6.5–8.5, while the ABSS shows a different optimal pH range. Figure 6c shows that the optimum pH of ABSS in treating HM-containing wastewater is neutral to acidic. At pH 7, the ABSS composed of C. sorokiniana 211/8k and R. basilensis achieved a maximum removal of 79% of 20 mg/L Cu(II)76. Yang et al. 59 discovered that ABSS consisting of algal-bacterial granular sludge reached the highest adsorption of Cr to 0.9 mg/g at pH = 6. Similarly, Wang et al. 63 used fresh algal-bacterial aerobic granular sludge to achieve a maximum adsorption of 12.30 ± 0.39 mg/g of Pb(II) at pH = 6.
HM concentrations and types
As illustrated in Fig. 6d, while the amount of HMs removed by ABSS increases with the rise in the HM initial concentration, the removal efficiency of HMs by ABSS decreases accordingly. Muñoz et al. 76 observed that the ABSS of C. sorokiniana and R. basilensis increased its biosorption capacity of Cu(II) as the concentration of Cu(II) in solution increased. The maximum biosorption capacity of the system was 8.5 ± 0.4 mg/g when the initial Cu(II) concentration was 20 mg/L. Furthermore, the symbiotic system shows varying removal efficiency for different HMs (Fig. 6d). The ABSS consisting of Scenedesmus almeriensis and an activated sludge studied by Antolín et al. 92 was found to achieve 78% and 96% removal of Cu(II) and Zn(II) at the same concentration, respectively.
Temperature
The suitable temperature is necessary for ABSS to show the best performance. The ABSS consisting of Chlorella vulgaris and Aspergillus niger exhibites the highest biological activity, the largest biomass, and the highest adsorption efficiency at 25 °C93. The optimum temperature of the ABSS for HM-containing wastewater treatment is in the range of 23–30 °C (Fig. 6e). Among them, most of the ABSS have an optimum temperature at 25 °C and shows the best removal of HMs. Tangahu et al. 20 observed that the ABSS attained the highest removal of chromium of 18.68% in the range of 25–27 °C.
Carbon sources
The ABSS can show the highest removal rate only when the optimal carbon source is utilized for HM-containing wastewater treatment (Fig. 6f). The ABSS using fructose as a carbon source shows better Cr(VI) removal effectiveness than when using glucose and sucrose59. The ABSS composed of Scenedesmus almeriensis and the activated sludge removed 96% of Zn(II) when UTEA was used as the carbon source92. Apart from carbon sources, nitrogen and phosphorus sources also influence treatment of HM-containing wastewater by ABSS16.
Algae species
Algae are crucial for HM-containing wastewater treatment in ABSS. Common algae species used for HM adsorption include Chlorophyta (green algae), Phaeophyta (brown algae), Rhodophyta (red algae), and Cyanophyta (cyanobacteria) (Fig. 7). These algae vary in cell wall composition, structure, and metabolites, resulting in significant differences in their capacity for HM removal. As shown in Fig. 6g, brown algae are more prominent in the removal of HMs. This result is similar to that of Romera et al. 94, who found that among other red and green algae, the brown alga Ascophyllum nodosum had the best adsorption capacity for HMs, which was twice as high as that of the other algae in the study. Chandrashekharaiah et al. 95 reported the differences in the removal rates of Pb(II) by Scenedesmus acutus and Chlorella pyrenoidosa in the symbiotic system, which were 52.2% and 32.2% respectively.
This phylogenetic tree presents the evolutionary relationships among numerous algal species capable of removing HMs, among which the species of Chlorophyta dominate in number. This provides crucial phylogenetic evidence for exploring the evolution of the HM removal characteristics of algae and their applications in bioremediation. This figure is independently drawn by the authors based on citation of the relevant literature.
Algae/bacteria ratios
There are significant differences between algae and bacteria in terms of physiological characteristics and pollutant removal ability, resulting in different optimal ratios for different types of wastewater. The ABSS composed of Chlorella vulgaris-BH1 and Exiguobacterium profundum-BH2 achieved the highest removal rate of 78.7% for 100 ppm of Cu(II) when the inoculation ratio was 3:3, and reached the highest removal rate of 80.0% for 100 ppm of Ni at an inoculation ratio of 3:296. The ABSS composed of Chlorella salina and Bacillus subtilis showed a maximum removal rate of 51.66% for 1 mg/L of Cd(II) under an inoculation ratio of 1:455. This is because a higher proportion of Bacillus subtilis can promote the accumulation and detoxification of Cd(II) by the ABSS.
Others
The selection of appropriate bacterial species in ABSS is crucial, as it can optimize the symbiotic relationship and improve the efficiency of HM-containing wastewater treatment. Ri et al. 57 compared the removal effects of lead by four symbiotic systems respectively composed of the cyanobacterium Leptolyngbya sp. XZ1 and four different heterotrophic bacterial strains. It was found that the symbiotic system composed of Leptolyngbya sp. XZ1 and Bacillus sp. S1 not only had a high removal rate of lead up to 71.8%, but also exhibited the largest biomass. In addition, salinity can also affect the treatment effect of ABSS on HM-containing wastewater. Yang et al. 59 found that the removal of Cr (VI) by ABSS decreased drastically when NaCl was increased to 5 mg/L. Meng et al. 97 demonstrated that high salinity is conducive to the formation of a favorable symbiotic relationship in the ABSS composed of Halomonas and Nitzschia, achieving rapid biomass growth and a relatively high removal efficiency of nutrients.
Application of ABSS to HM-containing wastewater from different industries
Industrial emissions, mining activities, agricultural practices, and urban discharges can all generate HM-containing wastewater, which poses significant risks to human health and the ecological environment. As an emerging bioremediation technology, the ABSS offers an environmentally friendly and sustainable solution for effectively removing HMs from wastewater. Table 5 shows the treatment effect of ABSS on HM-containing wastewater from different industries.
Treatment of mine wastewater
HMs in mine wastewater primarily result from the mining and processing of ore, where sulfide minerals like pyrite react with water and oxygen to produce sulfuric acid, leading to the formation of Acid Mine Drainage (AMD)98,99. Sulfate-reducing bacteria (SRB) utilize sulfate as an electron acceptor, reducing it to sulfide in anoxic conditions. These sulfides react with HMs to form insoluble metal sulfides, removing HMs from AMD100. However, the acidic environment, high HM concentrations, and limited carbon sources in AMD can hinder SRB growth and activity, leading to elevated chemical oxygen demand (COD) in the treated effluent.
The integration of SRB and algae in ABSS enhances AMD treatment by protecting SRB and improving efficiency. Algal photosynthesis absorbs CO2, reducing water acidity and promoting HM precipitation, while producing organic carbon (DOC) to support SRB. Moreover, EPS produced by algae helps protect SRB in AMD, while both algae and EPS possess the ability to adsorb HMs. Therefore, ABSS presents significant potential for AMD treatment applications. Sahoo et al. 100 discovered that the immobilized SRB-Chlorella symbiosis system could remove more than 95–99% of HMs in AMD. Li et al. 101 also observed that the immobilized SRB-Chlorella symbiosis system could remove 72.4–74.4% of sulfate and more than 91.7% of Cu(II) from AMD. Mining wastewater typically contains a variety of HMs, yet existing ABSS does not provide optimal removal efficacy for all types of HMs.
Treatment of animal agriculture wastewater
Animal agriculture wastewater, primarily originating from animal excreta, feed residues, and wash water, is characterized by high concentrations of ammonia nitrogen, HMs, and organic compounds, posing significant environmental risks. Conventional aquaculture wastewater treatment methods are energy-intensive and inefficient in nutrient utilization, leading to the underuse of biodegradable organic matter and nutrients in animal agriculture wastewater. Based on these concerns, the ABSS are gradually being considered as a cost-effective alternative to traditional treatment technologies for animal agriculture wastewater.
The ABSS can absorb nutrients to produce biomass while removing HMs from animal agriculture wastewater, making resource recycling possible. García et al. 102 discovered that ABSS for treating piggery wastewater achieved 94% total carbon, 100% total phosphorus, and 83% zinc removal, and increased biomass to 1328 mg TSS/L. In another of his studies, it was also observed that ABSS could remove 86%, 82%, 90%, and 98% of total organic carbon, total nitrogen, total phosphorus, and zinc from piggery wastewater, respectively, and achieved a biomass productivity of 7.8 ± 0.3 g/m²/d103. In addition, to enhance the cost-effectiveness of ABSS for treating animal agriculture wastewater, some researchers introduced photosynthetic bacteria into ABSS and obtained 87% removal of total nitrogen, 91% removal of total phosphorus, 98% removal of zinc, and biomass productivity of up to 18.4 g/m²/d for the piggery wastewater104. Along with HMs, animal agriculture wastewater contains pollutants such as antibiotics. However, the research on removing these two contaminants effectively with ABSS is insufficient.
Treatment of municipal wastewater
Municipal wastewater, originating from domestic, commercial, and service effluents, can increase HM levels and is typically treated in wastewater treatment plants before being discharged to meet regulatory standards. Most municipal wastewater treatment plants apply physico-chemical methods like UV, ozone, and chlorine for wastewater disinfection. These disinfection techniques pose risks of generating disinfection by-products, which may have adverse effects on human health105. Additionally, aeration in municipal wastewater treatment plants accounts for 50% of the total energy consumption of the facility106. Therefore, municipal wastewater treatment plants especially need a wastewater treatment technology that can avoid disinfection by-products and save energy.
ABSS generates oxygen, minimizing aeration requirements and energy consumption, while reducing the need for additional chlorination and effectively eliminating pathogens. Additionally, ABSS facilitates resource recycling, offering potential economic benefits for municipal wastewater treatment plants. Kumar et al. 107 showed that an ABSS consisting of microalgae and bacteria from municipal wastewater not only removes HMs and organic contaminants in treating municipal wastewater, but also completely reduces the total bacterial and total coliform counts while achieving 1.27 g/L biomass. Abdel-Razek et al. 108 found that the ABSS removed both 88% of cadmium, 95% of nickel, and 89% of lead, and 99% of the organophosphorus pesticide malathion, when treating a mixed wastewater consisting of municipal wastewater.
Treatment of textile wastewater
The composition of textile wastewater is highly complex, consisting of various contaminants such as organic and inorganic dyes, HMs, and nutrient salts. Traditional biological methods are often inadequate in treating textile wastewater. ABSS can remove dyes, HMs, and nutrients from textile wastewater due to its remarkable integrated decontamination capacity and resistance to unfavorable environments. Certain bacteria can decompose dyes and HMs, while algae can do photosynthesis and absorb nutrients. Another situation is that bacteria release growth factors to promote algal growth, while algae achieve decolorization and purification of textile wastewater through a series of biochemical processes. These processes include the assimilation of chromophores, the adsorption of dissolved dye molecules, and the mineralization of colored organic matter. In ABSS constructed by Mubashar et al. 91, Enterobacter sp. MN17 promoted C.vulgaris growth, which enabled this system to achieve 71.5% decolorization and remove 79%, 93%, 79%, and 74% of chromium, cadmium, lead, and COD, respectively, from 5% dilution of textile wastewater. Bashir et al. 109 constructed an ABSS that achieved both 85.7% decolourisation and 81.8% COD removal, while removing 98.2% of manganese, 94% of copper, 97.7% of chromium, 91.6% of cadmium, and 92.8% of cobalt.
HM recovery from biomass
During the treatment of HM-containing wastewater, the ABSS generates substantial biomass enriched with HMs. Without proper treatment, this may lead to solid waste pollution. Although traditional methods such as incineration and landfill disposal can be used to process this biomass, they result in resource waste and pose additional environmental problems. Therefore, the recovery of HMs from biomass not only helps reduce environmental pollution but also facilitates resource recycling. The recovery of HMs from biomass is primarily carried out through chemical leaching techniques, which involve the use of chemical reagents to treat the biomass and extract HM ions.
Various chemical reagents have been developed for the recovery of HMs from biomass, including acids, bases, chelating agents, and salts, such as sodium hydroxide (NaOH), hydrochloric acid (HCl), ethylenediaminetetraacetic acid (EDTA), and potassium chloride (KCl). NaOH forms metal hydroxides through its strong alkalinity, thereby releasing HMs. HCl uses an acidic environment to disrupt algal cell walls, facilitating the dissolution of HM ions for recovery. EDTA, as a chelating agent, forms stable complexes with HM ions, significantly enhancing desorption efficiency. KCl employs ion exchange to desorb metal ions from the surface of algae, making it suitable for the recovery of positively charged HM ions.
Sathvika et al. 110 demonstrated that 1.5 mol/L NaOH can recover 95% of Cr(VI) from biomass. Katsara et al. 111 employed a mixed reagent of HNO3 and H2O2 to recover Zn from algal biomass, achieving a recovery rate of 178 g/100 g. Yang et al. 52 evaluated the application of six different chemical reagents (including KCl, NaHCO3, Na2CO3, NaOH, HCl, and H2SO4) for Cr(VI) recovery from biomass, finding that H2SO4 provided the highest recovery rate of 33.5%. These reagents each have distinct characteristics, and the choice of recovery agent should be based on the type of HM and the properties of the biomass. However, the reactivation of microorganisms and the adsorption capacity for HMs after desorption have not been fully validated and require further investigation.
Sustainability of ABSS for HM-containing wastewater
The sustainability of ABSS for HM-containing wastewater is manifested in several key aspects: resource recycling, low cost and energy consumption, and contribution to carbon neutrality.
In terms of resource recycling, ABSS can recover HMs from biomass for industries such as metal refining, battery manufacturing, and electroplating. The HM-containing wastewater generated by these industries can then flow back to ABSS for treatment (Fig. 8). Meanwhile, biomass can be converted into green products like biofuels, biofertilizers, and biochar. Integrating relevant production plants downstream of ABSS can effectively reduce the overall treatment cost and achieve resource recycling.
This figure is an original hand-drawn illustration created based on the data and concepts from the relevant cited literature.
Regarding cost and energy consumption, ABSS is light-driven and does not require chemical reagents, thus reducing the costs associated with reagent procurement and hazardous chemicals. It has a low demand for mechanical aeration, and the proportion of aeration energy consumption is much lower than that of traditional wastewater treatment plants. Aeration usually accounts for 40%-60% of the total energy demand in traditional plants, while the power consumption of ABSS is approximately 22% lower than that of the traditional activated sludge process17,70. Moreover, the symbiotic relationship between algae and bacteria improves the energy utilization efficiency. Bacteria assist algae in nutrient cycling and decomposition, reducing the energy consumption for algae to obtain nutrients.
In terms of contributing to carbon neutrality, algae in ABSS can efficiently absorb the CO2 generated within the system and convert it into biomass, reducing greenhouse gas emissions, whereas approximately half of the carbon is released into the atmosphere as CO2 during the wastewater treatment by traditional biological methods112. In addition, the greenhouse gas emissions throughout the entire life cycle of biodiesel produced from algal biomass are only one-sixth of those of traditional diesel113.
Current challenges and future prospects
Complexity of actual wastewater
The actual wastewater is of diverse origin and complex composition, often containing not only HMs but also microplastics, antibiotics, and pathogenic microorganisms. These diverse pollutants impact microbial activity through different mechanisms, modifying the toxicity of HMs and reducing the effectiveness of ABSSs in HM removal. Moreover, the functional group variability on the surfaces of different microorganisms imparts selectivity to ABSS, making it less effective in removing a broad spectrum of HMs. Additionally, high concentrations of HMs in wastewater can suppress microbial activity, further reducing the ABSS’s capacity for metal removal16. Such wastewater often requires dilution or other pretreatment processes prior to entering ABSSs, which inevitably increases treatment costs.
To effectively address the complexity of actual HM-containing wastewater, the algae and bacterial strains in the ABSS can be optimized by using domestication and genetic engineering techniques to enhance their capacity to tolerate high concentrations of HMs and remove diverse contaminants. Simultaneously, in-depth studies are required to elucidate the mechanisms of multi-contaminant removal in complex HM-containing wastewater, revealing the intrinsic principles of their synergistic effects and providing theoretical support for system design and optimization.
Stability and efficiency of the system
The hydraulic retention time required for the remediation of HM-containing wastewater by the ABSS typically ranges from 2 to 10 days, which is longer than many other wastewater treatment technologies, indicating potential for efficiency improvement. Moreover, the ABSS’s performance in treating HM-containing wastewater is influenced by various factors, including hydraulic disturbances, and light intensity.
Optimizing system parameters is crucial for improving the efficiency and stability of HM-containing wastewater treatment in ABSS. Artificial intelligence (AI) demonstrates significant advantages in modeling complex nonlinear relationships by enabling real-time monitoring of system parameters, thereby uncovering intricate interactions among system components. This capability facilitates parameter optimization, reactor design, and internal configuration refinement. Mathematical models, as effective tools, can simulate and predict the impacts of various factors on pollutant removal pathways. Furthermore, they can model the dynamic changes in functional microbial communities within the ABSS, providing a theoretical foundation for enhancing overall system performance114. Additionally, the introduction of external substances or the optimization of system components can enhance performance. Adding exogenous EPS or promoting microbial EPS secretion can effectively increase EPS content, thereby reducing the operational time of the system115. The incorporation of light-conducting materials can provide efficient illumination for algal photosynthesis, further improving the overall system efficiency.
Biomass Harvesting and Processing
The biomass harvesting process faces the problem of high investment costs, which account for a relatively large proportion of the entire upstream processing costs, ranging from 20% to 30%116. Currently, filtration and centrifugation are the most common methods for biomass harvesting. However, for biomass intended for the production of low-value products, these methods are too costly. Although a relatively inexpensive biological flocculation method has emerged in recent years for biomass harvesting, the application scope of this method is rather limited. It is only applicable to the production of biofuels and cannot be used in the production of food-related products.
Regarding the biomass generated by the ABSS system, since it contains both algae and bacteria, the separation of these two types of biomass is extremely difficult. This, in turn, makes it hard to achieve the industrial production of products that rely on the separated biomass. In addition, after the desorption of HMs in biomass during the HM recovery process, the regenerative activity of microorganisms and their re-adsorption capacity for HMs have not been fully and effectively verified117. In order to further promote the application of ABSS technology in a wider range of fields, in the future, it is necessary to conduct in-depth research and exploration on different types of biomass, combine various treatment methods, and across multiple disciplinary fields.
Conclusions
ABSS is a sustainable technology for HM (HM) wastewater treatment, offering the dual benefits of HM removal and resource recovery. By harnessing the symbiotic interactions between bacteria and algae, ABSS utilizes biosorption and bioaccumulation mechanisms to effectively remediate HM-containing wastewater. This system operates through suspension or attached growth processes, and understanding the key influencing factors can enhance treatment efficacy. Despite its potential for treating HM-contaminated wastewater across various industries, ABSS faces challenges in practical applications. Future research should focus on improving ABSS adaptability in complex wastewater environments, ensuring stability under varying conditions, and enhancing treatment efficiency. Additionally, exploring HM desorption and biomass conversion technologies can help address by-product disposal and increase resource recovery potential.
Data availability
No datasets were generated or analyzed during the current study.
References
Chen, C. et al. Electro-peroxone enables efficient Cr removal and recovery from Cr(III) complexes and inhibits intermediate Cr(VI) generation in wastewater: Performance and mechanism. Water Res 218, 118502 (2022).
Xie, Y. et al. Stable and antibacterial tannic acid-based covalent polymeric hydrogel for highly selective Pb2+. recovery from lead-acid battery industrial wastewater. J. Hazard. Mater. 479, 135654 (2024).
Li, S., Dai, M., Ali, I., Bian, H. & Peng, C. Recovery of nickel from actual electroplating wastewater by integrated electrodeposition with adsorption pretreatment technique. Process Saf. Environ. Prot. 172, 417–424 (2023).
Moiseenko, T. I. & Gashkina, N. A. Distribution and bioaccumulation of heavy metals (Hg, Cd and Pb) in fish: influence of the aquatic environment and climate. Environ. Res. Lett. 15, 115013 (2020).
Gao, S., Zhang, R., Zhang, H. & Zhang, S. The seasonal variation in heavy metal accumulation in the food web in the coastal waters of Jiangsu based on carbon and nitrogen isotope technology. Environ. Pollut. 297, 118649 (2022).
Döndü, M., Özdemir, N., Demirak, A., Keskin, F. & Zeynalova, N. Bioaccumulation and human health risk assessment of some heavy metals in sediments, Sparus aurata and Salicornia europaea in Güllük Lagoon, the south of Aegean Sea. Environ. Sci. Pollut. Res. 30, 18227–18243 (2023).
Hirai, T. et al. Brain structural changes in patients with chronic methylmercury poisoning in Minamata. Brain Res 1805, 148278 (2023).
Słota, M., Wąsik, M., Stołtny, T., Machoń-Grecka, A. & Kasperczyk, S. Effects of environmental and occupational lead toxicity and its association with iron metabolism. Toxicol. Appl. Pharmacol. 434, 115794 (2022).
Husien, S., Labena, A., El-Belely, E. F., Mahmoud, H. M. & Hamouda, A. S. Adsorption studies of hexavalent chromium [Cr (VI)] on micro-scale biomass of Sargassum dentifolium, seaweed. J. Environ. Chem. Eng. 7, 103444 (2019).
El Sayed, M. T. & El-Sayed, A. S. A. Tolerance and mycoremediation of silver ions by Fusarium solani. Heliyon 6, e03866 (2020).
Ciobanu, A.-A., Lucaci, A.-R. & Bulgariu, L. Efficient metal ions biosorption on red and green algae biomass: Isotherm, kinetic and thermodynamic study. J. Appl. Phycol. 36, 3809–3827 (2024).
Li, Z. et al. Algal-bacterial granular sludge for simultaneous denitrification and yttrium removal. J. Environ. Chem. Eng. 11, 111316 (2023).
Wang, D. et al. Greenhouse gas emissions from municipal wastewater treatment facilities in China from 2006 to 2019. Sci. Data 9, 317 (2022).
Raza, N., Rizwan, M. & Mujtaba, G. Bioremediation of real textile wastewater with a microalgal-bacterial consortium: an eco-friendly strategy. Biomass-.-. Convers. Biorefin. 14, 7359–7371 (2024).
Eheneden, I., Wang, R. & Zhao, J. Antibiotic removal by microalgae-bacteria consortium: Metabolic pathways and microbial responses. Sci. Total Environ. 891, 164489 (2023).
Tang, C.-C. et al. Role of phosphate in microalgal-bacterial symbiosis system treating wastewater containing heavy metals. Environ. Pollut. 349, 123951 (2024).
Ji, B., Zhang, M., Gu, J., Ma, Y. & Liu, Y. A self-sustaining synergetic microalgal-bacterial granular sludge process towards energy-efficient and environmentally sustainable municipal wastewater treatment. Water Res 179, 115884 (2020).
Chen, X., Hu, Z., Qi, Y., Song, C. & Chen, G. The interactions of algae-activated sludge symbiotic system and its effects on wastewater treatment and lipid accumulation. Bioresour. Technol. 292, 122017 (2019).
Oruganti, R. K. et al. A comprehensive review on the use of algal-bacterial systems for wastewater treatment with emphasis on nutrient and micropollutant removal. Bioengineered 13, 10412–10453 (2022).
Tangahu, B. V., Berlianto, M. & Kartika, A. A. G. Deconcentration of chromium contained in wastewater using a bacteria and microalgae consortia with a high rate algal reactor system. J. Ecol. Eng. 21, 72–284 (2020).
Patel, A. K., Joun, J. M., Hong, M. E. & Sim, S. J. Effect of light conditions on mixotrophic cultivation of green microalgae. Bioresour. Technol. 282, 245–253 (2019).
Zhao, D. et al. Symbiosis of microalgae and bacteria consortium for heavy metal remediation in wastewater. J. Environ Chem. Eng. 11, 109943 (2023).
Zhang, X. et al. Development of a microalgal (Chlorella)-bacterial (Paracoccus) symbiotic system for pyridine biodegradation under photosynthetic oxygenation.ACS EST. Water 1, 356–365 (2020).
Karitani, Y., Yamada, R., Matsumoto, T. & Ogino, H. Improvement of cell growth in green algae Chlamydomonas reinhardtii through co-cultivation with yeast Saccharomyces cerevisiae. Biotechnol. Lett. 46, 431–441 (2024).
Cooper, M. B. et al. Cross-exchange of B-vitamins underpins a mutualistic interaction between Ostreococcus tauri and Dinoroseobacter shibae. ISME J. 13, 334–345 (2019).
Rawat, D. et al. Iron-dependent mutualism between Chlorella sorokiniana and Ralstonia pickettii forms the basis for a sustainable bioremediation system. ISME Commun. 2, 83 (2022).
Perera, I. A. et al. Extracellular polymeric substances drive symbiotic interactions in bacterial?microalgal consortia. Microb. Ecol. 83, 596–607 (2022).
Ji, X., Luo, X., Zhang, J. & Huang, D. Effects of exogenous vitamin B12 on nutrient removal and protein expression of algal-bacterial consortium. Environ. Sci. Pollut. Res. 28, 15954–15965 (2021).
Zhang, Z. et al. Plastoquinone synthesis inhibition by tetrabromo biphenyldiol as a widespread algicidal mechanism of marine bacteria. ISME J 17, 1979–1992 (2023).
Gao, X.-Y., Xie, W. & Liu, Z.-P. Algae control in oligotrophic surface water under the joint effect of nutritional competition and microbial algae-lytic substances. Environ. Sci.: Water Res. Technol. 8, 375–384 (2022).
Alghazeer, R. et al. Alkaloids rich extracts from brown algae against multidrug-resistant bacteria by distinctive mode of action. Arab. J. Sci. Eng. 47, 179–188 (2022).
Frazzini, S. et al. Antioxidant and antimicrobial activity of algal and cyanobacterial extracts: An in vitro study. Antioxidants 11, 992 (2022).
Li, T. et al. Dissolved organic carbon spurs bacterial-algal competition and phosphorus-paucity adaptation: Boosting Microcystis’ phosphorus uptake capacity. Water Res 255, 121465 (2024).
Raju, C. A. I., Anitha, J., Mahalakshmi Kalyani, R., Satyanandam, K. & Jagadeesh, P. Sorption of cobalt using marine macro seaweed Gracilaria corticata red algae powder. Mater. Today.: Proc. 44, 1816–1827 (2021).
Al-Khiat, S. H., Bukhari, N. A., Ameen, F. & Abdel-Raouf, N. Comparison of the microalgae Phormidium tenue and Chlorella vulgaris as biosorbents of Cd and Zn from aqueous environments. Environ. Res. 235, 116675 (2023).
Sahu, S., Kaur, A., Singh, G. & Arya, S. K. Integrating biosorption and machine learning for efficient remazol red removal by algae-bacteria co-culture and comparative analysis of predicted models. Chemosphere 355, 141791 (2024).
Ren, B. et al. Tracking the molecular-scale mechanistic pathway of trapping-bonding CTAB/Fe3O4-AS for high-performance Cr(VI) adsorption. Chem. Eng. J. 497, 155053 (2024).
Blaby-Haas, C. E. & Merchant, S. S. The ins and outs of algal metal transport. Biochim. et. Biophys. Acta (BBA) - Mol. Cell Res. 1823, 1531–1552 (2012).
Chakravorty, M. et al. Heavy metal tolerance in microalgae: Detoxification mechanisms and applications. Aquat. Toxicol. 260, 106555 (2023).
Ozdemir, S., Turkan, Z., Kilinc, E., Bayat, R. & Sen, F. The removal of heavy metal pollution from wastewaters using thermophilic B. cereus SO-16 bacteria. Chemosphere 311, 136986 (2023).
Zhao, M. M. et al. Bioremediation of wastewater containing mercury using three newly isolated bacterial strains. J. Clean. Prod. 299, 126869 (2021).
Mohapatra, R. K. et al. Active and passive biosorption of Pb(II)using live and dead biomass of marine bacterium Bacillus xiamenensis PbRPSD202: Kinetics and isotherm studies. J. Environ. Manag. 247, 121–134 (2019).
Cui, D. et al. Biosorption mechanism of aqueous Pb2+, Cd2+, and Ni2+ ions on extracellular polymeric substances (EPS). Archaea 2020, 8891543 (2020).
Ameen, F. A., Hamdan, A. M. & El-Naggar, M. Y. Assessment of the heavy metal bioremediation efficiency of the novel marine lactic acid bacterium, Lactobacillus plantarum MF042018. Sci. Rep. 10, 314 (2020).
Diep, P., Mahadevan, R. & Yakunin, A. F. Heavy metal removal by bioaccumulation using genetically engineered microorganisms. Front. Bioeng. Biotechnol. 6, 157 (2018).
Patel, R. J., Mohapatra, S. & Hansda, A. Mycoremediation of metallic pollutants. Eco-Restoration of Polluted Environment 53-65 (CRC Press, 2025).
Vettese, G. F. et al. Multiple Lines of Evidence Identify U(V) as a Key Intermediate during U(VI) Reduction by Shewanella oneidensis MR1. Environ. Sci. Technol. 54, 2268–2276 (2020).
Mohamed, M. S., Hozayen, W. G., Alharbi, R. M. & Ibraheem, I. B. M. Adsorptive recovery of arsenic (III) ions from aqueous solutions using dried Chlamydomonas sp. Heliyon 8, e12398 (2022).
Arce-Inga, M. et al. Bioremediation potential of native Bacillus sp. strains as a sustainable strategy for cadmium accumulation of theobroma cacao in Amazonas region. Microorganisms 10, 2108 (2022).
Wang, Z. et al. Effect of acetochlor on the symbiotic relationship between microalgae and bacteria. J. Hazard. Mater. 463, 132848 (2024).
Contreras, F. et al. Reduction of gold (III) and tellurium (IV) by Enterobacter cloacae MF01 results in nanostructure formation both in aerobic and anaerobic conditions. Front. Microbiol. 9, 3118 (2018).
Yang, X. et al. Cr(VI) bioremediation by active algal-bacterial aerobic granular sludge: Importance of microbial viability, contribution of microalgae and fractionation of loaded Cr. J. Hazard Mater. 418, 126342 (2021).
Li, Y. et al. Enhanced Pb (II) removal by algal-based biosorbent cultivated in high-phosphorus cultures. Chem. Eng. J. 361, 167–179 (2019).
Ren, G. et al. Global environmental change shifts ecological stoichiometry coupling between plant and soil in early-stage invasions. J. Soil Sci. Plant Nutr. 24, 2402–2412 (2024).
Yu, Q. et al. Effects of algal–bacterial ratio on the growth and cadmium accumulation of Chlorella salina–Bacillus subtilis consortia. J. Basic Microbiol. 62, 518–529 (2022).
Wang, Z. et al. Pb(II) bioremediation using fresh algal-bacterial aerobic granular sludge and its underlying mechanisms highlighting the role of extracellular polymeric substances. J. Hazard. Mater. 444, 130452 (2023).
Ri, C. et al. Improvement of the Cd removal efficiency of a filamentous cyanobacterium Leptolyngbya sp. XZ1 through co-culture with Bacillus sp. S1. J. Appl. Phycol. 35, 2935–2944 (2023).
Yang, X. et al. Enhanced biosorption of Cr(VI) from synthetic wastewater using algal-bacterial aerobic granular sludge: Batch experiments, kinetics and mechanisms. Sep. Purif. Technol. 251, 117323 (2020).
Yang, X. et al. Insight into Cr(VI) biosorption onto algal-bacterial granular sludge: Cr(VI) bioreduction and its intracellular accumulation in addition to the effects of environmental factors. J. Hazard. Mater. 414, 125479 (2021).
Tang, Y. et al. Overexpression of the sulfate transporter-encoding SULTR2 increases chromium accumulation in Chlamydomonas reinhardtii. Biotechnol. Bioeng. 120, 1334–1345 (2023).
Nagarajan, D. et al. Microalgae-based wastewater treatment – Microalgae-bacteria consortia, multi-omics approaches and algal stress response. Sci. Total Environ. 845, 157110 (2022).
Zhang, B. et al. Translatomics and physiological analyses of the detoxification mechanism of green alga Chlamydomonas reinhardtii to cadmium toxicity. J. Hazard. Mater. 448, 130990 (2023).
Wang, Z. et al. Pb (II) bioremediation using fresh algal-bacterial aerobic granular sludge and its underlying mechanisms highlighting the role of extracellular polymeric substances. J. Hazard. Mater. 444, 130452 (2023).
Jin, M. et al. Physiological and morphological responses and tolerance mechanisms of Isochrysis galbana to Cr(VI) stress. Bioresour. Technol. 302, 122860 (2020).
Ma, N., Cai, R. & Sun, C. Threonine dehydratase enhances bacterial cadmium resistance via driving cysteine desulfuration and biomineralization of cadmium sulfide nanocrystals. J. Hazard. Mater. 417, 126102 (2021).
Wang, J. et al. Effect of extracellular proteins on Cd(II) adsorption in fungus and algae symbiotic system. J. Environ. Manag. 323, 116173 (2022).
Kumar, A. & Prasad, M. N. V. Plant-lead interactions: Transport, toxicity, tolerance, and detoxification mechanisms. Ecotoxicol. Environ. Saf. 166, 401–418 (2018).
Dror, B., Jurkevitch, E. & Cytryn, E. Global-scale analysis reveals distinct patterns of non-ribosomal peptide and polyketide synthase encoding genes in root and soil bacterial communities. Soil Ecol. Lett. 5, 38–45 (2023).
Leong, Y. K., Huang, C.-Y. & Chang, J.-S. Pollution prevention and waste phycoremediation by algal-based wastewater treatment technologies: The applications of high-rate algal ponds (HRAPs) and algal turf scrubber (ATS). J. Environ. Manag. 296, 113193 (2021).
Kohlheb, N. et al. Assessing the life-cycle sustainability of algae and bacteria-based wastewater treatment systems: High-rate algae pond and sequencing batch reactor. J. Environ. Manag. 264, 110459 (2020).
Rodrigues de Assis, L., Calijuri, M. L., Assemany, P. P., Silva, T. A. & Teixeira, J. S. Innovative hybrid system for wastewater treatment: High-rate algal ponds for effluent treatment and biofilm reactor for biomass production and harvesting. J. Environ. Manag. 274, 111183 (2020).
Magalhães, I. B., Ferreira, J., Castro, J. D. S., Assis, L. R. D. & Calijuri, M. L. Agro-industrial wastewater-grown microalgae: A techno-environmental assessment of open and closed systems. Sci. Total Environ. 834, 155282 (2022).
Carone, M. et al. Design and characterization of a new pressurized flat panel photobioreactor for microalgae cultivation and CO2 bio-fixation. Chemosphere 307, 135755 (2022).
Benner, P. et al. Lab-scale photobioreactor systems: principles, applications, and scalability. Bioprocess Biosyst. Eng. 45, 791–813 (2022).
Wang, H., Duan, X., Feng, X., Mao, Z.-S. & Yang, C. Effect of impeller type and scale-up on spatial distribution of shear rate in a stirred tank. Chin. J. Chem. Eng. 42, 351–363 (2022).
Muñoz, R. et al. Sequential removal of heavy metals ions and organic pollutants using an algal-bacterial consortium. Chemosphere 63, 903–911 (2006).
Gong, X. et al. Effect of different size microplastic particles on the construction of algal-bacterial biofilms and microbial communities. J. Environ. Manag. 343, 118246 (2023).
Sun, L. et al. A novel symbiotic system combining algae and sludge membrane bioreactor technology for wastewater treatment and membrane fouling mitigation: Performance and mechanism. Chem. Eng. J. 344, 246–253 (2018).
Orandi, S., Lewis, D. M. & Moheimani, N. R. Biofilm establishment and heavy metal removal capacity of an indigenous mining algal-microbial consortium in a photo-rotating biological contactor. J. Ind. Microbiol. Biotechnol. 39, 1321–1331 (2012).
Zhao, X., Kumar, K., Gross, M. A., Kunetz, T. E. & Wen, Z. Evaluation of revolving algae biofilm reactors for nutrients and metals removal from sludge thickening supernatant in a municipal wastewater treatment facility. Water Res 143, 467–478 (2018).
de Souza, M. P. et al. Bioproducts characterization of residual periphytic biomass produced in an algal turf scrubber (ATS) bioremediation system. Water Sci. Technol. 82, 1247–1259 (2020).
Gan, X., Klose, H. & Reinecke, D. Stable year-round nutrients removal and recovery from wastewater by technical-scale algal turf scrubber (ATS). Sep. Purif. Technol. 307, 122693 (2023).
Sutherland, D. L., Burke, J. & Ralph, P. J. Increased harvest frequency improves biomass yields and nutrient removal on a filamentous algae nutrient scrubber. Algal Res 51, 102073 (2020).
Ren, H. et al. Removal of sulfamethoxazole in an algal-bacterial membrane aerated biofilm reactor: Microbial responses and antibiotic resistance genes. Water Res 268, 122595 (2025).
Cheng, S. et al. Harnessing diurnal dynamics: Deciphering the interplay of light cycles on algal-bacterial membrane bioreactors. Sci. Total Environ. 913, 169644 (2024).
Du, Y.-D. et al. Wastewater treatment and simultaneous production of algal lipids in sequencing batch reactors containing a microalgal-bacterial consortium. Int. Biodeterior. Biodegrad. 175, 105491 (2022).
Mhedhbi, E., Khelifi, N., Foladori, P. & Smaali, I. Real-time behavior of a microalgae–bacteria consortium treating wastewater in a sequencing batch reactor in response to feeding time and agitation mode. Water 12, 1893 (2020).
Wang, S. et al. Cadmium-effect on performance and symbiotic relationship of microalgal-bacterial granules. J. Clean. Prod. 282, 125383 (2021).
Luo, S., Berges, J. A., He, Z. & Young, E. B. Algal-microbial community collaboration for energy recovery and nutrient remediation from wastewater in integrated photobioelectrochemical systems. Algal Res 24, 527–539 (2017).
Tripathy, B. K. & Kumar, M. Leachate treatment using sequential microwave and algal photo-bioreactor: Effect of pretreatment on reactor performance and biomass productivity. J. Environ. Manag. 311, 114830 (2022).
Mubashar, M. et al. Experimental Investigation of Chlorella vulgaris and Enterobacter sp. MN17 for decolorization and removal of heavy metals from textile wastewater. Water 12, 3034 (2020).
Antolín, B., Torres, A., García, P. A., Bolado, S. & Vega, M. Mechanisms of copper and zinc bioremoval by microalgae and bacteria grown in nutrient rich wastewaters. Chemosphere 355, 141803 (2024).
Li, L., Liu, W., Liang, T. & Ma, F. The adsorption mechanisms of algae-bacteria symbiotic system and its fast formation process. Bioresour. Technol. 315, 123854 (2020).
Romera, E., González, F., Ballester, A., Blázquez, M. L. & Muñoz, J. A. Comparative study of biosorption of heavy metals using different types of algae. Bioresour. Technol. 98, 3344–3353 (2007).
Chandrashekharaiah, P. et al. Algae-bacterial aquaculture can enhance heavy metals (Pb2+ and Cd2+) remediation and water re-use efficiency of synthetic streams. Resour., Conserv. Recycl. 180, 106211 (2022).
Batool, S. et al. Implication of highly metal-resistant microalgal-bacterial co-cultures for the treatment of simulated metal-loaded wastewaters. Int. Microbiol. 22, 41–48 (2019).
Meng, F. et al. Application of aerobic granules-continuous flow reactor for saline wastewater treatment: Granular stability, lipid production and symbiotic relationship between bacteria and algae. Bioresour. Technol. 295, 122291 (2020).
Ayala-Parra, P., Sierra-Alvarez, R. & Field, J. A. Algae as an electron donor promoting sulfate reduction for the bioremediation of acid rock drainage. J. Hazard. Mater. 317, 335–343 (2016).
Yasmin, F., Sakib, T. U., Emon, S. Z. & Bari, L. & Sultana, G. N. N. The physicochemical and microbiological quality assessment of Maddhapara hard rock-mine discharged water in Dinajpur, Bangladesh. Resour., Environ. Sustain. 8, 100061 (2022).
Sahoo, H., Senapati, D., Thakur, I. S. & Naik, U. C. Integrated bacteria-algal bioreactor for removal of toxic metals in acid mine drainage from iron ore mines. Bioresour. Technol. Rep. 11, 100422 (2020).
Li, Y., Yang, X. & Geng, B. Preparation of immobilized sulfate-reducing bacteria-microalgae beads for effective bioremediation of copper-containing wastewater. Water Air Soil Pollut. 229, 54 (2018).
García, D. et al. Comparative evaluation of piggery wastewater treatment in algal-bacterial photobioreactors under indoor and outdoor conditions. Bioresour. Technol. 245, 483–490 (2017).
García, D. et al. Evaluation of the dynamics of microalgae population structure and process performance during piggery wastewater treatment in algal-bacterial photobioreactors. Bioresour. Technol. 248, 120–126 (2018).
García, D. et al. A systematic comparison of the potential of microalgae-bacteria and purple phototrophic bacteria consortia for the treatment of piggery wastewater. Bioresour. Technol. 276, 18–27 (2019).
Werkneh, A. A. & Gebru, S. B. Development of ecological sanitation approaches for integrated recovery of biogas, nutrients and clean water from domestic wastewater. Resour. Environ. Sustain. 11, 100095 (2023).
Drewnowski, J., Remiszewska-Skwarek, A., Duda, S. & Łagód, G. Aeration process in bioreactors as the main energy consumer in a wastewater treatment plant. review of solutions and methods of process optimization. Processes 7, 311 (2019).
Kumar, V. et al. Algae-based sustainable approach for simultaneous removal of micropollutants, and bacteria from urban wastewater and its real-time reuse for aquaculture. Sci. Total Environ. 774, 145556 (2021).
Abdel-Razek, M. A. et al. Bioremediation of a pesticide and selected heavy metals in wastewater from various sources using a consortium of microalgae and cyanobacteria. Slov. Vet. 56, 61–73 (2019).
Bashir, K. et al. Bioremediation of metal-polluted industrial wastewater with algal-bacterial consortia: A sustainable strategy. Sustainability 15, 14056 (2023).
Sathvika, T. et al. A co-operative endeavor by nitrifying bacteria Nitrosomonas and Zirconium based metal organic framework to remove hexavalent chromium. Chem. Eng. J. 360, 879–889 (2019).
Katsara, A. et al. Cultivation of the macrophyte Lemna minor and the microalgae Chlorella sorokiniana in thermal mineral waters: Biomass characteristics, radioisotopes and heavy metals content. Environ. Pollut. 349, 123881 (2024).
Casagli, F., Zuccaro, G., Bernard, O., Steyer, J.-P. & Ficara, E. ALBA: A comprehensive growth model to optimize algae-bacteria wastewater treatment in raceway ponds. Water Res 190, 116734 (2021).
Ankita, J. & Ganti, S. M. Evaluating the potential of renewable diesel production from algae cultured on wastewater: techno-economic analysis and life cycle assessment. AIMS Energy 5, 239–257 (2017).
Sánchez-Zurano, A. et al. ABACO: a new model of microalgae-bacteria consortia for biological treatment of wastewaters. Appl. Sci. 11, 998 (2021).
Liu, X., Ji, B. & Li, A. Enhancing biolipid production and self-flocculation of Chlorella vulgaris by extracellular polymeric substances from granular sludge with CO2 addition: microscopic mechanism of microalgae-bacteria symbiosis. Water Res. 236, 119960 (2023).
Mirakhorli, F., Razavi Bazaz, S., Warkiani, M. E. & Ralph, P. J. Ultra-high throughput microfluidic concentrator for harvesting of Tetraselmis sp. (Chlorodendrophyceae, Chlorophyta). Algal Res. 72, 103145 (2023).
Chilian, A. et al. Extraction of heavy metals and phosphorus from sewage sludge with elimination of antibiotics and biological risks. Chem. Eng. J. 437, 135298 (2022).
Tong, C. Y., Honda, K. & Derek, C. J. C. A review on microalgal-bacterial co-culture: The multifaceted role of beneficial bacteria towards enhancement of microalgal metabolite production. Environ. Res. 228, 115872 (2023).
Hobman, J. L., Julian, D. J. & Brown, N. L. Cysteine coordination of Pb(II) is involved in the PbrR-dependent activation of the lead-resistance promoter, PpbrA, from Cupriavidus metallidurans CH34. BMC Microbiol. 12, 109 (2012).
Young, P., Taylor, M. & Fallowfield, H. J. Mini-review: high rate algal ponds, flexible systems for sustainable wastewater treatment. World J. Microbiol. Biotechnol. 33, 117 (2017).
Koller, M. Design of Closed Photobioreactors for Algal Cultivation. In Algal Biorefineries: Volume 2: Products and Refinery Design (eds. Prokop, A., Bajpai, R.K. & Zappi, M.E.) 133-186 (Springer International Publishing, Cham, 2015).
Occhipinti, P. S. et al. Mixotrophic and heterotrophic growth of Galdieria sulphuraria using buttermilk as a carbon source. J. Appl. Phycol. 35, 2631–2643 (2023).
Wollmann, F. et al. Microalgae wastewater treatment: Biological and technological approaches. Eng. Life Sci. 19, 860–871 (2019).
Zettler, J., Samuels, N., Schachner, C. & Conner, A. Exploring the intersection of art, science, and marine conservation using algal biomass waste. Current 39, 33–42 (2024).
Fouilland, E. et al. Influence of bacteria on the response of microalgae to contaminant mixtures. Chemosphere 211, 449–455 (2018).
Yalçın, S. & Özyürek, M. Biosorption potential of two brown seaweeds in the removal of chromium. Water Sci. Technol. 78, 2564–2576 (2018).
Moino, B. P., Costa, C. S., da Silva, M. G. & Vieira, M. G. Removal of nickel ions on residue of alginate extraction from Sargassum filipendula seaweed in packed bed. Can. J. Chem. Eng. 95, 2120–2128 (2017).
Vieira, B. R., Pintor, A. M., Boaventura, R. A., Botelho, C. M. & Santos, S. C. Arsenic removal from water using iron-coated seaweeds. J. Environ. Manag. 192, 224–233 (2017).
Ungureanu, G., Santos, S., Boaventura, R. & Botelho, C. Biosorption of antimony by brown algae S. muticum and A. nodosum. Environ. Eng. Manage. J. 14, 455–463 (2015).
Fabre, E. et al. Negligible effect of potentially toxic elements and rare earth elements on mercury removal from contaminated waters by green, brown and red living marine macroalgae. Sci. Total Environ. 724, 138133 (2020).
Pham, B. N., Kang, J.-K., Lee, C.-G. & Park, S.-J. Removal of heavy metals (Cd2+, Cu2+, Ni2+, Pb2+) from aqueous solution using Hizikia fusiformis as an algae-based bioadsorbent. Appl. Sci. 11, 8604 (2021).
Roy, A. S., Hazarika, J., Manikandan, N. A., Pakshirajan, K. & Syiem, M. B. Heavy metal removal from multicomponent system by the cyanobacterium Nostoc muscorum: Kinetics and Interaction Study. Appl. Biochem. Biotechnol. 175, 3863–3874 (2015).
Geng, W. et al. Response and tolerance ability of Chlorella vulgaris to cadmium pollution stress. Environ. Technol. 43, 4391–4401 (2022).
Danouche, M., El Ghachtouli, N., El Baouchi, A. & El Arroussi, H. Heavy metals phycoremediation using tolerant green microalgae: Enzymatic and non-enzymatic antioxidant systems for the management of oxidative stress. J. Environ. Chem. Eng. 8, 104460 (2020).
Ahad, R. I. A. & Syiem, M. B. Influence of calcium on cadmium uptake and toxicity to the cyanobacterium Nostoc muscorum Meg 1. Biotechnol. Res. Innov. 3, 231–241 (2019).
Sayadi, M. H., Rashki, O. & Shahri, E. Application of modified Spirulina platensis and Chlorella vulgaris powder on the adsorption of heavy metals from aqueous solutions. J. Environ. Chem. Eng. 7, 103169 (2019).
Dirbaz, M. & Roosta, A. Adsorption, kinetic and thermodynamic studies for the biosorption of cadmium onto microalgae Parachlorella sp. J. Environ. Chem. Eng. 6, 2302–2309 (2018).
Dulla, J. B., Sumalatha, B., King, P. & Yekula, P. K. Investigation on biosorption of Cd (II) onto Gelidiella acerosa (brown algae): Optimization (using RSM & ANN) and mechanistic studies. Desalin. Water Treat. 107, 195–206 (2018).
Jayakumar, V., Govindaradjane, S., Rajamohan, N. & Rajasimman, M. Biosorption potential of brown algae, Sargassum polycystum, for the removal of toxic metals, cadmium and zinc. Environ. Sci. Pollut. Res. 29, 41909–41922 (2022).
Kharel, H. L., Tan, M., Jha, L. & Selvaratnam, T. Removal of cadmium (II), lead (II), nickel (II), and zinc (II) from synthetic medium by extremophile red alga Galdieria sulphuraria: Investigating single and mixed metal systems. Algal Res 83, 103699 (2024).
Ye, J. et al. Bioremediation of heavy metal contaminated aqueous solution by using red algae Porphyra leucosticta. Water Sci. Technol. 72, 1662–1666 (2015).
Sayyaf, H., Mostafaii, G. R., Iranshahi, L., Mosavi, G. A. & Aseman, E. Removal of Cr(VI) from synthetic aqueous solutions by filamentous green algae Spirogyra porticalis. Desalin. Water Treat. 57, 18438–18445 (2016).
de Souza, P. O. et al. Bioremediation of chromium and lead in wastewater from chemistry laboratories promotes by cyanobacteria. Bioresour. Technol. Rep. 19, 101161 (2022).
Kumar, N., Hans, S. & Srivastava, A. Chromium bioremediation from river Yamuna using cyanobacteria at white and red illumination wavelengths. J. Water Process Eng 44, 102416 (2021).
El Nemr, A., El-Sikaily, A., Khaled, A. & Abdelwahab, O. Removal of toxic chromium from aqueous solution, wastewater and saline water by marine red alga Pterocladia capillacea and its activated carbon. Arab. J. Chem. 8, 105–117 (2015).
Zhao, D. et al. Symbiosis of microalgae and bacteria consortium for heavy metal remediation in wastewater. J. Environ. Chem. Eng. 11, 109943 (2023).
Nishikawa, E., da Silva, M. G. C. & Vieira, M. G. A. Cadmium biosorption by alginate extraction waste and process overview in life cycle assessment context. J. Clean. Prod. 178, 166–175 (2018).
Al-Dhabi, N. A. & Arasu, M. V. Biosorption of hazardous waste from the municipal wastewater by marine algal biomass. Environ. Res. 204, 112115 (2022).
El-Naggar, N. E.-A., Hamouda, R. A., Mousa, I. E., Abdel-Hamid, M. S. & Rabei, N. H. Biosorption optimization, characterization, immobilization and application of Gelidium amansii biomass for complete Pb2+ removal from aqueous solutions. Sci. Rep. 8, 13456 (2018).
Vafajoo, L., Cheraghi, R., Dabbagh, R. & McKay, G. Removal of cobalt (II) ions from aqueous solutions utilizing the pre-treated 2-Hypnea Valentiae algae: Equilibrium, thermodynamic, and dynamic studies. Chem. Eng. J. 331, 39–47 (2018).
Gunasundari, E. & Kumar, P. S. Adsorption isotherm, kinetics and thermodynamic analysis of Cu(II) ions onto the dried algal biomass (Spirulina platensis). J. Ind. Eng. Chem. 56, 129–144 (2017).
Nath, A., Sharma, A., Singh, S. K. & Sundaram, S. Assessing the Impact of Hexavalent Chromium (Cr VI) at varied concentrations on spirulina platensis for growth, metal sorption, and photosynthetic responses. Curr. Microbiol. 81, 231 (2024).
Abdel -Aty, A. M., Ammar, N. S., Abdel Ghafar, H. H. & Ali, R. K. Biosorption of cadmium and lead from aqueous solution by fresh water alga Anabaena sphaerica biomass. J. Adv. Res. 4, 367–374 (2013).
Senthilkumar, R., Prasad, D. R., Govindarajan, L., Saravanakumar, K. & Prasad, B. N. Green alga-mediated treatment process for removal of zinc from synthetic solution and industrial effluent. Environ. Technol. 40, 1262–1270 (2019).
Kücüker, M. A., Nadal, J.-B. & Kuchta, K. Comparison between batch and continuous reactor systems for biosorption of neodymium (Nd) using microalgae. Int. J. Plant, Anim. Environ. Sci. 6, 197–203 (2016).
Fawzy, M. A. Biosorption of copper ions from aqueous solution by Codium vermilara: Optimization, kinetic, isotherm and thermodynamic studies. Adv. Powder Technol. 31, 3724–3735 (2020).
Abbas, S. Z., Yee, C. J., Hossain, K., Ahmad, A. & Rafatullah, M. Isolation and characterization of mercury-resistant bacteria from industrial wastewater. Desalin. Water Treat. 138, 128–133 (2019).
Bravo, G., Vega-Celedón, P., Gentina, J. C. & Seeger, M. Bioremediation by Cupriavidus metallidurans Strain MSR33 of mercury-polluted agricultural soil in a rotary drum bioreactor and its effects on nitrogen cycle microorganisms. Microorganisms 8, 1952 (2020).
Jobby, R., Jha, P., Gupta, A., Gupte, A. & Desai, N. Biotransformation of chromium by root nodule bacteria Sinorhizobium sp. SAR1. PloS one 14, e0219387 (2019).
Li, K. et al. Compost-derived humic and fulvic acid coupling with Shewanella oneidensis MR-1 for the bioreduction of Cr(VI). J. Environ. Manag. 345, 118596 (2023).
Qiao, S. et al. Multiple heavy metals immobilization based on microbially induced carbonate precipitation by ureolytic bacteria and the precipitation patterns exploration. Chemosphere 274, 129661 (2021).
Altowayti, W. A. H., Algaifi, H. A., Bakar, S. A. & Shahir, S. The adsorptive removal of As (III) using biomass of arsenic resistant Bacillus thuringiensis strain WS3: Characteristics and modelling studies. Ecotoxicol. Environ. Saf. 172, 176–185 (2019).
Jebelli, M. A. et al. Isolation and identification of indigenous prokaryotic bacteria from arsenic-contaminated water resources and their impact on arsenic transformation. Ecotoxicol. Environ. Saf. 140, 170–176 (2017).
Das, P., Sinha, S. & Mukherjee, S. K. Nickel bioremediation potential of Bacillus thuringiensis KUNi1 and some environmental factors in nickel removal. Bioremediation J. 18, 169–177 (2014).
Chirizzi, D. et al. Nickel ion extracellular uptake by the phototrophic bacterium Rhodobacter sphaeroides: new insights from Langmuir modelling and X-ray photoelectron spectroscopic analysis. Appl. Surf. Sci. 593, 153385 (2022).
Mathivanan, K., Rajaram, R. & Annadurai, G. Biosorption potential of Lysinibacillus fusiformis KMNTT-10 biomass in removing lead (II) from aqueous solutions. Sep. Sci. Technol. 53, 1991–2003 (2018).
Mwandira, W. et al. Biosorption of Pb (II) and Zn (II) from aqueous solution by Oceanobacillus profundus isolated from an abandoned mine. Sci. Rep. 10, 21189 (2020).
Dabai, A. I. & Mohammed, K. Biosorption and desorption of chromium using hybrid microalgae-activated sludge treatment system. Appl. Water Sci. 14, 16 (2024).
Santas, P., Danielidis, D. & Santas, R. Removal of heavy metals from industrial and municipal waste waters using an algal turf scrubber. Proc. 1st Intern. Expos. & Conference on Env. Techn. HELECO. 3, 178–183 (1993).
Cheenakula, D. et al. Anaerobic digestion of algal–bacterial biomass of an Algal Turf Scrubber system. Biomass Convers. Biorefin. 13, 13605–13619 (2023).
Collao, J., García-Encina, P. A., Blanco, S., Bolado-Rodríguez, S. & Fernandez-Gonzalez, N. Current Concentrations of Zn, Cu, and As in piggery wastewater compromise nutrient removals in microalgae–bacteria photobioreactors due to altered microbial communities. Biology 11, 1176 (2022).
Rose, P. et al. An integrated algal sulphate reducing high rate ponding process for the treatment of acid mine drainage wastewaters. Biodegradation 9, 247–257 (1998).
Cardoso, L. G. et al. Spirulina sp. LEB 18 cultivation in outdoor pilot scale using aquaculture wastewater: High biomass, carotenoid, lipid and carbohydrate production. Aquaculture 525, 735272 (2020).
Sharma, J. et al. Microalgal consortia for municipal wastewater treatment – Lipid augmentation and fatty acid profiling for biodiesel production. J. Photochem. Photobiol. B: Biol. 202, 111638 (2020).
Bellucci, M. et al. Assessment of anammox, microalgae and white-rot fungi-based processes for the treatment of textile wastewater. Plos one 16, e0247452 (2021).
Acknowledgements
This work was supported by the National Natural Science Foundation of China (No.42161144003).
Author information
Authors and Affiliations
Contributions
Y. H. was responsible for the original draft writing, visualization, conceptualization, and formal analysis of the paper. J. Y. participated in the review and editing of the paper, conducted investigation research, and obtained funding support. X. H. was involved in the review and editing of the original draft as well as the conceptualization of the paper. J. S. was mainly in charge of the review and editing of the paper and data curation. W. T. participated in formal analysis and data curation work. M. W. was tasked with formal analysis and data curation. G. Y. was involved in work related to formal analysis and data curation. Y. F. was responsible for formal analysis and data curation. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Huang, Y., Yu, J., Huang, X. et al. Algal-bacterial symbiotic system for treatment of heavy metal containing wastewater: performance, mechanisms and applications. npj Clean Water 8, 86 (2025). https://doi.org/10.1038/s41545-025-00475-1
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41545-025-00475-1